Electrochemically enabled nickel-catalyzed controllable synthesis of monoaryl or diaryl amines from aryl halides and trimethylsilyl azides†
Jiawei
Huang
,
Xiaoman
Li
,
Xue
Zhao
,
Yu
Wei
 * and
Liang
Xu
* and
Liang
Xu
 *
*
School of Chemistry and Chemical Engineering/State Key Laboratory Incubation Base for Green Processing of Chemical Engineering, Shihezi University, Shihezi, China. E-mail: yuweichem@shzu.edu.cn; xuliang4423@shzu.edu.cn
First published on 2nd April 2025
Abstract
Aryl amines serve as fundamental building blocks in the production of many pharmaceuticals, agrochemicals, and functional materials, underscoring their preparation in synthetic chemistry. This work presents an approach that combines electrolysis with nickel catalysis to facilitate the C–N cross-coupling between aryl halides and trimethylsilyl azides (TMSN3), marking a pioneering advancement in the direct synthesis of aryl amines from aryl halides via electrochemically enabled nickel catalysis. Furthermore, by adjusting the reaction conditions, this strategy could deliver monoaryl or diaryl amines chemoselectively. The approach exhibits broad substrate scope and robust functional group compatibility, allowing for the practical and versatile late-stage modification of complex pharmaceutical molecules.
Green foundation1. The quest for direct and environmentally benign methods for preparing primary amines has emerged as a prominent research frontier. This nickel-catalyzed electrocatalytic method provides an additional robust protocol for the green synthesis of aromatic amines. This approach employs TMSN3 as a nitrogen source for the construction of C–N bonds from aryl halides, overcoming the following challenges: (1) the industrially prevalent method for synthesizing aromatic amines generally involves the nitration of aromatics using HNO3 and H2SO4, followed by the hydrogenation reduction of the nitro groups, a process that generates substantial waste and causes severe environmental pollution; (2) although there have been significant advancements in converting aryl halides into primary amines through light-driven nickel catalysis in recent years, the high-energy radiation required for this photo-driven process may pose considerable environmental risks.2. This strategy boasts an extremely broad applicability, not only applicable to bromides and iodides but also to chlorides. Furthermore, by modulating the reaction conditions, a unified platform is established for the selective synthesis of either mono- or diarylamines. Additionally, this method is also applicable to brominated drugs, offering a powerful tool for late-stage modifications of drug molecules. 3. To enhance the atomic utilization efficiency of the reaction, we intend to delve deeper into exploring alternative nitrogen sources to achieve the amination of aryl halides, thereby improving atomic economy and presenting a more environmentally friendly methodology. |
Introduction
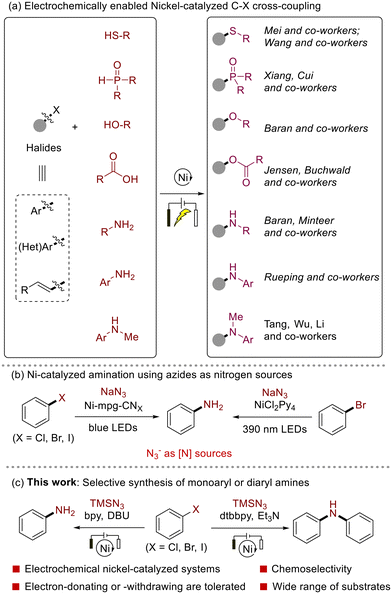
Electrochemical organic synthesis offers an efficient and controllable method for conducting chemical transformations, and overcoming previously challenging transformations.1 It leverages the inherent anodic and cathodic processes of electrochemistry, enabling the engagement of reactants in the redox processes through the direct electron transfer. Consequently, this approach avoids the requirement for stoichiometric traditional chemical oxidants or reductants, thereby reducing chemical consumption and waste production.2 On the other hand, in recent years, nickel-catalyzed methodologies have garnered considerable attention from the synthetic community, due to the abundance, cost-effectiveness, variable valences and the resulting versatile reactivity of the nickel complexes.3The combination of these two research focal points, namely electrochemically enabled nickel catalysis, has merged their advantages and advanced significantly recently, particularly in the realm of C–C and C–heteroatom cross-coupling reactions. The established protocols enable the synthesis of a spectrum of valuable architectures in more efficient and sustainable pathways, showcasing the versatility of the electrolysis-enabled nickel-catalyzed strategy.3b,4
As illustrated in Scheme 1a, substantial contributions to this area demonstrated the use of electrochemical nickel catalysis to couple C–heteroatom bonds between aryl halides and X–H (X = N, S, O, P) species. These approaches usually effectively circumvented the need for high temperatures or specialized ligands, which are often required when employing traditional palladium or copper catalysts to overcome the high energy barriers of the reductive elimination step in Pd-catalyzed cases or the oxidative addition step in Cu-catalyzed cases.5 For example, in 2019, Mei group,6 and Wang group7 independently reported the use of nickel catalysis under electrochemical conditions for the sulfurization of aryl halides, elegantly sidestepping the harsh conditions such as high temperatures or strong bases, which are typically associated with such transformations. In the same year, Xiang and Cui groups,8 and Rueping group9 detailed the phosphonylation reactions utilizing electrochemical nickel catalysis. Jensen and Buchwald groups,10 and Baran group11 further expanded the synthetic repertoire in this domain to include the esterification and etherification reactions, respectively.
Due to the ubiquity and importance of the aryl amine species, metal-catalyzed C–N cross-coupling reactions have consistently been a pivotal area of synthetic interest,12 leading to the establishment and widespread application of Ullmann-Ma13 and Buchwald–Hartwig amination reactions. As for the electrochemically enabled nickel-catalyzed amination reactions, Baran group pioneered a pathway for the amination of aryl halides with aliphatic amines,14 and further expanded its applicability to include nucleoside oligopeptides and amino acid esters, underscoring the versatility of this method.15 Subsequently, Rueping group16 accomplished the construction of C–N bonds between aryl electrophiles and a variety of weak nucleophiles, such as anilines, sulfonamides, sulfinimides, carbamates, benzylamines, and imines.17 This versatile method highlighted the adaptability of electrochemical nickel catalysis in various synthetic contexts. More recently, Tang, Wu and Li groups18 introduced a nickel-catalyzed photoelectrochemical (PEC) amination strategy of aryl bromides. Leveraging the power of light, this PEC approach not only achieved high yields in C–N coupling but also operated at remarkably low potentials.
Despite these achievements, the direct synthesis of primary aryl amines (monoaryl amines) with the –NH2 moiety, which represented a fundamental building block in the synthesis of a myriad of biologically active molecules and functional materials,19 has not yet been realized through the electrochemical nickel-catalyzed approach. Recently, Reisner group,20 and Rueping group21 have independently reported the use of sodium azides as nitrogen sources in photochemical nickel-catalyzed systems to synthesize monoaryl amines from aryl halides (Scheme 1b). Given our expertise in electrochemical transformations22 and the demand for enriching the synthetic methods of monoaryl amines, it was thus questioned whether azide species could be utilized in electrochemical nickel-catalyzed amination reactions to obtain aryl amines. Herein, this manuscript details the results of this hypothesis (Scheme 1c). Under electrochemical conditions, nickel-catalyzed reactions between aryl halides and TMSN3 directly produces monoaryl amines. Intriguingly, by fine-tuning the reaction parameters, the electrochemical avenue enables double C–N cross-coupling in the same vessel to deliver diaryl amines compounds.
In recent years, the synthesis of monoaryl amines and diaryl amines from aryl halides has conventionally been achieved through photocatalytic20,21,23 or metal-catalytic23d,24 methodologies. Although effective, these approaches frequently require the use of high-energy radiation sources or operate under elevated temperatures, resulting in energy-intensive processes that can impose a substantial environmental impact. In sharp contrast, the advent of electrochemical nickel catalysis offers a highly promising alternative, not only overcoming these limitations but also more faithfully embodying the principles of green chemistry.
Results and discussion
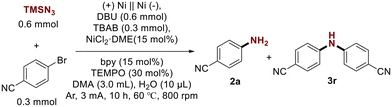
Initially, 4-bromobenzonitrile was chosen as the model substrate to demonstrate the amination reaction of aryl halides. In preliminary attempts, we successfully obtained 4-aminobenzonitrile 2a. After a systematic evaluation of reaction parameters, including solvent, nickel catalyst, ligands, and base, the optimal conditions had been found to be as follows: Ni(+)/Ni(−) electrodes at 60 °C with TBAB as the electrolyte, NiCl2·DME as the catalyst, bpy (2,2′-bipyridine) as the ligand, DBU (1,8-diazabicyclo [5.4.0]undec-7-ene) as the base, TEMPO (2,2,6,6-tetramethylpiperidinyl-1-oxide) as the additive, and DMA as the solvent. The reaction was carried out under a Ar atmosphere with a constant current of 3 mA and a stirring speed of 800 rpm for 10 hours, successfully affording the desired product 2a in a 70% isolated yield (Table 1, entry 1). Firstly, control experiments were conducted, demonstrating that the reaction did not proceed in the absence of the electrolysis, ligands, and base (entries 2–4), proving that they were essential for promoting C–N cross-coupling. Since the nickel electrode generates a substantial amount of divalent nickel during anodic oxidation, the reaction can still proceed even when no additional nickel source is added (entry 5). The reaction yield slightly decreased when no additional H2O was added (entry 6). When TEMPO was absent from the reaction, the yield of monoaryl amines slightly decreased, while the conversion of diaryl amines increased (entry 7). Subsequently, we attempted to use other polar solvents, such as N,N-dimethylformamide (DMF) and acetonitrile (MeCN), which resulted in significantly reduced yields (entries 8 and 9). A base was necessary for the reaction, and the mild, soluble DBU was significantly more effective than the other bases, while the presence of Et3N promoted the conversion of diarylamine (entries 10–12). Reducing the reaction temperature had a slight impact on the yield (entry 13), whereas raising the temperature did not markedly improve the conversion rate of 1a (entry 14). The use of other bipyridine ligands also resulted in lower yields (entries 15 and 16). Switching to other electrodes, the yield of monoaryl amines decreased. The use of Al as the anode led to an increased conversion to diaryl amines 3r (entries 17 and 18).| Entry | Variations from the ‘standard’ conditions | Yield 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (%) a (%) |
Yield 3r![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (%) a (%) |
|---|---|---|---|
| Standard conditions A: aryl halides (1.0 equiv., 0.3 mmol), TMSN3 (2.0 equiv., 0.6 mmol), Ni(+)||Ni(−), NiCl2·DME (15.0 mol%, 0.045 mmol), bpy (2,2′-dipyridyl, 15.0%, 0.045 mmol), TBAB (tetrabutylammonium bromide, 1.0 equiv., 0.3 mmol), DBU (1,8-diazabicyclo[5.4.0]undec-7-ene, 2.0 equiv., 0.6 mmol), TEMPO (2,2,6,6-tetramethylpiperidinyl-1-oxide, 30 mol%, 0.09 mmol), H2O (10.0 μL), DMA (N,N-dimethylacetamide, 3.0 mL), 800 rpm, 60 °C, constant current = 3 mA in Ar for 10 h (3.7 F mol−1).a Isolated yields are shown and in an undivided cell.b No reaction. | |||
| 1 | None | 70 | 10 |
| 2 | Without electrolysis | N.R.b | N.R. |
| 3 | Without bpy | Trace | Trace |
| 4 | Without DBU | Trace | Trace |
| 5 | without NiCl2·DME | 45 | Trace |
| 6 | Without H2O | 58 | 13 |
| 7 | Without TEMPO | 50 | 24 |
| 8 | DMF instead of DMA | 52 | Trace |
| 9 | MeCN instead of DMA | 30 | Trace |
| 10 | Et3N instead of DBU | 27 | 27 |
| 11 | MTBD instead of DBU | 53 | 13 |
| 12 | TMG instead of DBU | 62 | 15 |
| 13 | 25 °C instead of 60 °C | 43 | Trace |
| 14 | 80 °C instead of 60 °C | 66 | 11 |
| 15 | dtbbpy instead of bpy | 53 | 15 |
| 16 | Di(4-Me)-bpy instead of bpy | 50 | 12 |
| 17 | (+)Ni||C(−) as electrodes | 63 | 12 |
| 18 | (+)Al||C(−) as electrodes | 23 | 23 |
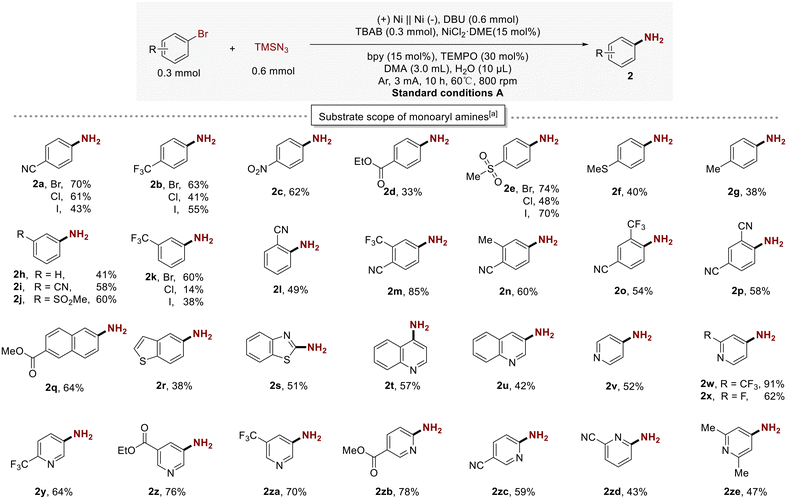
Next, the substrate scope of this protocol was investigated under the optimal conditions. As summarized in Scheme 2, the amination products of aryl halides were obtained mostly in moderate to excellent yields. Aryl bromides with para-substituted electron-withdrawing groups (2a, –CN; 2b, –CF3; 2c, NO2; 2d, –CO2Et; 2e, –SO2Me) or electron-donating groups (2f, –SMe; 2g, –Me) were efficiently converted into their corresponding monoaryl amine products. Substrates with electron-withdrawing groups afforded higher yields compared to those with electron-donating groups. However, when 4-ester-substituted haloarenes were introduced into the reaction, the yield significantly decreased (2d). In this reaction, the primary by-product of 2d is the diarylamine compound. Additionally, when disubstituted aryl bromides (1m–1p) were employed, the reactions proceeded smoothly, affording the corresponding products (2m, 85%; 2n, 60%; 2o, 54%; 2p, 58%).
Aryl bromides bearing ortho-substituents (1l, 1o, 1p) also underwent this transformation successfully to deliver the desired amination products (2l, 2o, 2p), thereby demonstrating the robustness of this protocol toward the steric hindrance imposed by the aryl halides. Naphthalene bromide was also a suitable substrate, yielding the desired products (2q, 64%). Subsequently, we began exploring whether heteroaryl halides could be effectively aminated in the reaction system. Amino derivatives of benzothiophenes (2r), benzothiazole (2s), quinolines (2t; 2u), and pyridines (2v–2ze) could be obtained in moderate to good yields. Afterwards, we started exploring whether the substrate scope of this transformation was limited to brominated aromatic hydrocarbons. The application of the coupling reaction to other aryl halides, including aryl iodides and chlorides, was also feasible. Under the same reaction conditions, aryl iodides and chlorides demonstrated excellent conversion, yielding the corresponding phenylamine compounds (2a; 2b; 2e; 2k) and showcasing a broader range of substrate adaptability.
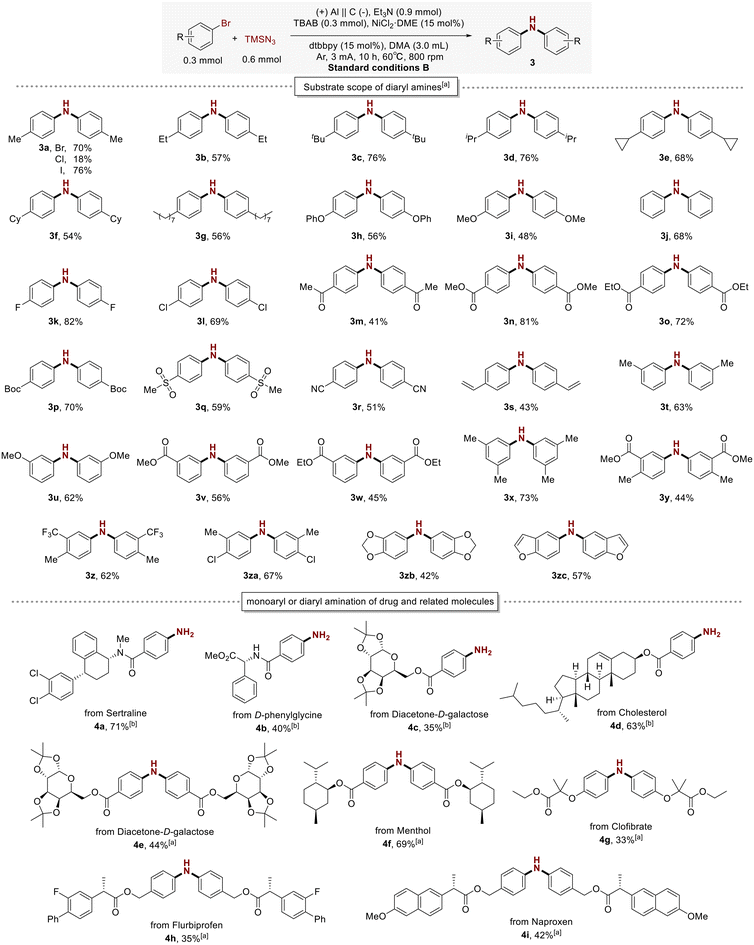
Considering the significant role of diaryl amines in antioxidants and optoelectronic materials, we subsequently investigated the direct synthesis of diaryl amines using aryl halides and TMSN3. After optimizing the reaction conditions, it was ultimately determined to use Al(+)/C(−) electrodes at 60 °C with TBAB as the electrolyte, NiCl2·DME as the catalyst, dtbbpy (4,4′-di-tert-butyl-2,2′-dipyridyl) as the ligand, Et3N (triethylamine) as the base, and DMA as the solvent. The reaction was carried out under an argon atmosphere with a constant current of 3 mA and a stirring speed of 800 rpm for 10 hours.
Subsequently, we investigated the substrate compatibility of this diaryl amination process under the optimized conditions. As shown in the top part of Scheme 3, aryl bromides substituted with para-position electron-donating groups such as alkyls (3a–3g; 3j) and ethers (3h; 3i), or electron-withdrawing groups such as halides (3k; 3l), ketone (3m), ester (3n–3p), sulfonyl (3q), cyano (3r), and alkenyl (3s) were effectively converted into diaryl amine compounds. Notably, aryl bromides with meta-substituents were also viable substrates and could provide the desired products (3t–3w) with excellent yields. Multi-substituted aryl bromides also reacted successfully under this method; for example, 5-bromo-m-xylene afforded the corresponding diarylamine compound in high yield (3x, 73%). 3,4-Disubstituted aryl bromides proceeded smoothly to deliver the desired products (3y, 44%; 3z, 62%; 3za, 67%). However, ortho-substituted aryl halides proved challenging to convert into corresponding products in this transformation, likely due to the higher steric requirements for the second oxidative addition. We then began exploring whether heteroaryl halides could be effectively used in this reaction system for diaryl amination. Examples included the synthesis of amino derivatives of 1,3-benzodioxole (3zb) and benzofuran (3zc).
The direct conversion of halogenated arenes to diaryl amines is also applicable to other aryl halides, including aryl iodides and aryl chlorides. Aryl iodides can slightly improve the yield of diarylamine 3a. However, the yield of the product tends to be lower for aryl chlorides, likely due to the stronger influence of the C–Cl bond on the reaction kinetics, which is a common trend in nickel catalysis.
Next, the applicability of this method in the amination and diarylamination of various bromine-substituted drug molecules was investigated. As shown in Scheme 3 (bottom), a series of drug molecules containing Csp2–Br bonds, such as sertraline (4a), D-phenylglycine (4b), diacetone-D-galactose (4c), and cholesterol (4d), were efficiently converted to the desired aryl amines; diacetone-D-galactose (4e), menthol (4f), clofibrate (4g), flurbiprofen (4h), and naproxen (4i) were efficiently converted to the desired diaryl amines.
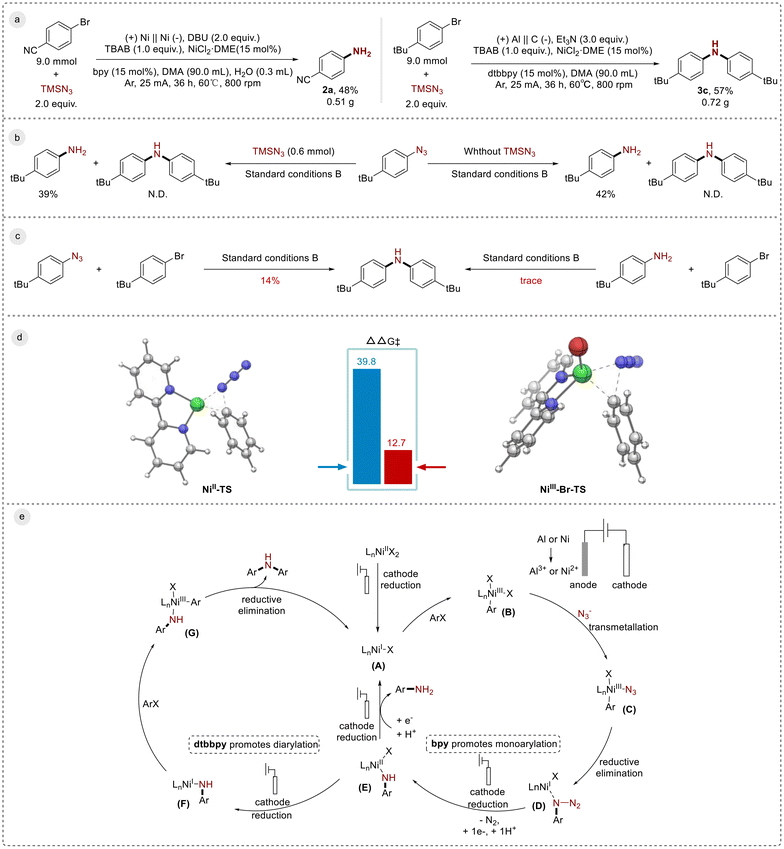
In order to further investigate this reaction, we conducted gram-scale experiments and a series of mechanistic experiments (Scheme 4). To verify the practicality and scalability of the electrochemical nickel dual-catalysis amination, we conducted gram-scale experiments. We utilized 9.0 mmol of 4-bromobenzonitrile and 4-tert-butylbromobenzene for monoaryl amination and diaryl amination reactions, respectively. As shown in Scheme 4a, by simply increasing the amount of each reagent and maintaining a constant current of 25 mA for 36 hours, we were able to obtain 2a (0.51 g) with a yield of 48% and 3c (0.72 g) with a yield of 57%, respectively. To confirm that phenyl azide was an intermediate, it was tested under standard reaction conditions both with and without TMSN3, resulting in the formation of monoaryl amines in all cases. However, the formation of diarylamine was not observed (Scheme 4b). Experiments involving phenyl azide and aniline with aryl bromides as reactants showed that the azide participated in the formation of diarylamine, whereas aniline, in contrast, did not participate in the formation of diarylamine (Scheme 4c). This suggested the existence of a mechanism that did not involve naked aniline. To further validate the intermediates in the reaction, we used DFT (density functional theory) calculations (Scheme 4d). The calculations indicated that the reduction elimination energy barriers for NiII and NiIII complexes were 39.8 and 12.7 kcal mol−1, respectively. These results confirmed that in this amination scheme, the NiIII complex was more prone to undergo reduction elimination.
Based on literature reports21,23c,25 and the above experimental results, we propose a mechanism for the formation of monoaryl amines and diaryl amines (Scheme 4e). Initially, the NiII catalyst was reduced to NiI (A) via cathodic reduction. This was followed by the oxidative addition of the aryl bromide, generating the ArNiIII intermediate (B). The intermediate (B) underwent transmetalation with the azide anion to form the Ar–NiIII–N3 intermediate (C), which then proceeded through a subsequent reductive elimination step to produce the NiI intermediate (D). Successive N2 liberation followed by hydrogenation resulted in NiII. The cathodic reduction then provided monoaryl aniline and regenerated NiI (A). NiII (E) could be reduced to the NiI amine intermediate (F) via cathodic reduction and could undergo oxidative addition with the aryl bromide. Different ligands on complex F affected the electronic density of nickel during the second oxidative addition step; when the ligand was changed to dtbbpy, the electronic density on Ni increased, facilitating the second oxidative addition step to generate NiIII (G). The reductive elimination of (G) provided diarylamine and regenerated (A), starting another catalytic cycle. Throughout the entire reaction process, electrochemical reduction played a key role and served as the driving force for the entire reaction.
Conclusions
In summary, we report the chemoselective synthesis of both monoaryl amines and diaryl amines from aryl halides, utilizing trimethylsilyl azides (TMSN3) as an effective nitrogen source. This methodology not only addresses a long-standing challenge in the direct generation of monoaryl amines under electrochemical conditions but also enables sequential double C–N cross-couplings to yield diaryl amines. This strategy is simple to operate, exhibits good functional group tolerance, and is applicable to drug-related compounds, thereby underscoring its practical significance and broad applicability. A mechanism involving NiI/NiIII catalytic pathways has been proposed, and our laboratory is currently further investigating the mechanism and expanding this strategy.Author contributions
J. H. contributed to the acquisition, analysis, and interpretation of data and the preparation of the manuscript; X. L. and X. Z. contributed to the acquisition of data; Y. W. and L. X. contributed to the conception of the work, supervision, and preparation of the manuscript.Data availability
Experimental and analytical data supporting this article are available in the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
We thank for the financial support from the National Natural Science Foundation of China (22061036).References
- (a) H.-Y. Zhou, H.-T. Tang and W.-M. He, Chin. J. Catal., 2023, 46, 4–10 CrossRef CAS; (b) S. Möhle, M. Zirbes, E. Rodrigo, T. Gieshoff, A. Wiebe and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 6018–6041 CrossRef PubMed; (c) C.-K. Ran and D.-G. Yu, Nat. Rev. Chem., 2022, 6, 679–680 CrossRef PubMed; (d) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 Search PubMed; (e) Y. Jiang, K. Xu and C. Zeng, Chem. Rev., 2017, 118, 4485–4540 Search PubMed.
- (a) L. Zeng, J. Wang, D. Wang, H. Yi and A. Lei, Angew. Chem., Int. Ed., 2023, 62, e202309620 CrossRef CAS PubMed; (b) Y. Yuan, J. Yang and A. Lei, Chem. Soc. Rev., 2021, 50, 10058–10086 RSC; (c) M. Elsherbini and T. Wirth, Acc. Chem. Res., 2019, 52, 3287–3296 Search PubMed.
- (a) S. Zhu, H. Li, Y. Li, Z. Huang and L. Chu, Org. Chem. Front., 2023, 10, 548–569 RSC; (b) C. Zhu, H. Yue, J. Jia and M. Rueping, Angew. Chem., Int. Ed., 2021, 60, 17810–17831 CrossRef CAS PubMed; (c) S. Zhu, X. Zhao, H. Li and L. Chu, Chem. Soc. Rev., 2021, 50, 10836–10856 RSC; (d) R. Sun, Y. Qin and D. G. Nocera, Angew. Chem., Int. Ed., 2020, 59, 9527–9533 CrossRef CAS PubMed; (e) S. Z. Tasker, E. A. Standley and T. F. Jamison, Nature, 2014, 509, 299–309 CrossRef CAS PubMed.
- M. D. Palkowitz, M. A. Emmanuel and M. S. Oderinde, Acc. Chem. Res., 2023, 56, 2851–2865 CrossRef CAS PubMed.
- (a) S. Wang, M. Yuan, Q. Zhang and S. Huang, Curr. Opin. Green Sustainable Chem., 2022, 38, 100698–100706 CrossRef CAS; (b) C. Uyeda, Y. Tan, G. C. Fu and J. C. Peters, J. Am. Chem. Soc., 2013, 135, 9548–9552 CrossRef CAS PubMed; (c) I. P. Beletskaya and V. P. Ananikov, Chem. Rev., 2011, 111, 1596–1636 CrossRef CAS PubMed; (d) S. E. Creutz, K. J. Lotito, G. C. Fu and J. C. Peters, Science, 2012, 338, 647–651 Search PubMed; (e) J. F. Hartwig, Acc. Chem. Res., 2008, 41, 1534–1544 CrossRef CAS PubMed.
- D. Liu, H. X. Ma, P. Fang and T. S. Mei, Angew. Chem., Int. Ed., 2019, 58, 5033–5037 CrossRef CAS PubMed.
- Y. Wang, L. Deng, X. Wang, Z. Wu, Y. Wang and Y. Pan, ACS Catal., 2019, 9, 1630–1634 Search PubMed.
- Y. Bai, N. Liu, S. Wang, S. Wang, S. Ning, L. Shi, L. Cui, Z. Zhang and J. Xiang, Org. Lett., 2019, 21, 6835–6838 CrossRef CAS PubMed.
- C. Zhu, H. Yue, P. Nikolaienko and M. Rueping, CCS Chem., 2020, 2, 179–190 Search PubMed.
- Y. Mo, Z. Lu, G. Rughoobur, P. Patil, N. Gershenfeld, A. I. Akinwande, S. L. Buchwald and K. F. Jensen, Science, 2020, 368, 1352–1357 Search PubMed.
- H. J. Zhang, L. Chen, M. S. Oderinde, J. T. Edwards, Y. Kawamata and P. S. Baran, Angew. Chem., Int. Ed., 2021, 60, 20700–20705 CrossRef CAS PubMed.
- (a) L. Zhao, C. Hu, X. Cong, G. Deng, L. L. Liu, M. Luo and X. Zeng, J. Am. Chem. Soc., 2021, 143, 1618–1629 CrossRef CAS PubMed; (b) C. Chen, Z. Wang, S. Wang, L. Xu and X. Zeng, Org. Lett., 2023, 25, 4241–4246 Search PubMed.
- J. Luo, M. T. Davenport, C. Callister, S. D. Minteer, D. H. Ess and T. L. Liu, J. Am. Chem. Soc., 2023, 145, 16130–16141 Search PubMed.
- C. Li, Y. Kawamata, H. Nakamura, J. C. Vantourout, Z. Liu, Q. Hou, D. Bao, J. T. Starr, J. Chen, M. Yan and P. S. Baran, Angew. Chem., Int. Ed., 2017, 56, 13088–13093 Search PubMed.
- Y. Kawamata, J. C. Vantourout, D. P. Hickey, P. Bai, L. Chen, Q. Hou, W. Qiao, K. Barman, M. A. Edwards, A. F. Garrido-Castro, J. N. deGruyter, H. Nakamura, K. Knouse, C. Qin, K. J. Clay, D. Bao, C. Li, J. T. Starr, C. Garcia-Irizarry, N. Sach, H. S. White, M. Neurock, S. D. Minteer and P. S. Baran, J. Am. Chem. Soc., 2019, 141, 6392–6402 Search PubMed.
- C. Zhu, A. P. Kale, H. Yue and M. Rueping, JACS Au, 2021, 1, 1057–1065 Search PubMed.
- D. Liu, Z. R. Liu, C. Ma, K. J. Jiao, B. Sun, L. Wei, J. Lefranc, S. Herbert and T. S. Mei, Angew. Chem., Int. Ed., 2021, 60, 9444–9449 CrossRef CAS PubMed.
- J. Wang, S. Li, C. Yang, H. Gao, L. Zuo, Z. Guo, P. Yang, Y. Jiang, J. Li, L.-Z. Wu and Z. Tang, Nat. Commun., 2024, 15, 6907–6914 CrossRef PubMed.
- (a) P. Ruiz-Castillo and S. L. Buchwald, Chem. Rev., 2016, 116, 12564–12649 Search PubMed; (b) J. J. Hanthorn, R. Amorati, L. Valgimigli and D. A. Pratt, J. Org. Chem., 2012, 77, 6895–6907 Search PubMed; (c) D. M. Roundhiu, Chem. Rev., 1992, 92, 1–27 CrossRef.
- A. Vijeta, C. Casadevall and E. Reisner, Angew. Chem., Int. Ed., 2022, 61, e202203176 CrossRef CAS PubMed.
- L. Karpova, M. Daniel, R. Kancherla, K. Muralirajan, B. Maity and M. Rueping, Org. Lett., 2024, 26, 1657–1661 Search PubMed.
- (a) X. Li, J. Huang, L. Xu, J. Liu and Y. Wei, Adv. Synth. Catal., 2023, 365, 4647–4653 Search PubMed; (b) M. Liu, L. Xu and Y. Wei, Chin. Chem. Lett., 2022, 33, 1559–1562 Search PubMed; (c) D. Ding, L. Xu and Y. Wei, J. Org. Chem., 2022, 87, 4912–4917 Search PubMed.
- (a) G. Song, J. Song, Q. Li, T. Kang, J. Dong, G. Li, J. Fan, C. Wang and D. Xue, J. Am. Chem. Soc., 2024, 146, 26936–26946 Search PubMed; (b) Z. Xu, J. Dong, G. Song, F. Kong, G. Li and D. Xue, Org. Chem. Front., 2024, 11, 2313–2318 Search PubMed; (c) G. Song, J. Song, Q. Li, D. Z. Nong, J. Dong, G. Li, J. Fan, C. Wang, J. Xiao and D. Xue, Angew. Chem., Int. Ed., 2023, 63, e202314355 CrossRef PubMed; (d) R. Kancherla, K. Muralirajan, S. Dutta, K. Pal, B. Li, B. Maity, L. Cavallo and M. Rueping, Angew. Chem., Int. Ed., 2023, 63, e202314508 Search PubMed; (e) G. Song, D.-Z. Nong, Q. Li, Y. Yan, G. Li, J. Fan, W. Zhang, R. Cao, C. Wang, J. Xiao and D. Xue, ACS Catal., 2022, 12, 15590–15599 CrossRef CAS.
- (a) M. Roemer, I. Luck and N. Proschogo, Adv. Synth. Catal., 2022, 364, 2957–2971 Search PubMed; (b) Á. Georgiádes, S. B. Ötvös and F. Fülöp, Adv. Synth. Catal., 2018, 360, 1841–1849 Search PubMed; (c) R. J. Lundgren, B. D. Peters, P. G. Alsabeh and M. Stradiotto, Angew. Chem., Int. Ed., 2010, 49, 4071–4074 CrossRef CAS PubMed; (d) C. W. Cheung, D. S. Surry and S. L. Buchwald, in Patai's Chemistry of Functional Groups, 2009, vol. 15, pp. 3734–3737 Search PubMed.
- M. O. Konev, T. A. McTeague and J. W. Johannes, ACS Catal., 2018, 8, 9120–9124 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc06307d |
| This journal is © The Royal Society of Chemistry 2025 |