In vitro analysis of β-carotene supplementation: insights from Raman spectroscopy on brain, lung, and breast cancer cells†
Karolina
Jarczewska
 ,
Monika
Kopeć
,
Monika
Kopeć
 and
Jakub Maciej
Surmacki
and
Jakub Maciej
Surmacki
 *
*
Lodz University of Technology, Faculty of Chemistry, Institute of Applied Radiation Chemistry, Laboratory of Laser Molecular Spectroscopy, Wroblewskiego 15, 93-590 Lodz, Poland. E-mail: jakub.surmacki@p.lodz.pl; Tel: +48426313188
First published on 4th July 2025
Abstract
β-carotene, a carotenoid abundant in fruits and vegetables such as carrots and tomatoes, plays a significant role in human health and its potential in cancer prevention is widely discussed. However, its precise impact on tumour cell metabolism remains unclear. Utilizing Raman spectroscopy, a technique adept at analysing complex biological systems, we investigated the effects of β-carotene on lung (A549), breast (MCF7), and brain (CRL-1718) cancer cells. We have found that the supplementation with 1 and 10 μM β-carotene altered lipid metabolism differently across cancers: increased lipid production and cytochrome c redox changes in lung cancer, and inhibited growth with reduced lipid and protein levels in breast and brain cancers. Those changes are observed based on a detailed analysis of recorded Raman spectra. We have chosen Raman bands at 1003, 1254, 1310, 1444, 1654, 2848–2964 cm−1 as biomarkers. These findings suggest that β-carotene influences cancer cell metabolism in unique ways, supporting its potential role in cancer treatment strategies.
Introduction
According to the World Health Organisation's 2024 statistics, lung cancer is the most deadly cancer in men and women, and breast cancer is the second most deadly cancer in women.1 Brain tumours are dangerous mainly because of the pressure on other parts of the brain, which can affect a person's functioning. Although brain tumours are not common, they are among the top ten most deadly cancers.1 Carcinogenesis affects mostly cellular energy metabolism, to proliferate faster, cancer cells redirect metabolism to glycolysis regardless of the presence of oxygen.2,3Lung cancer has been identified as the leading cause of cancer deaths among both sexes in the United States (population 333.3 million in 2022) and among men worldwide.4,5 The most common symptoms of lung cancer are prolonged cough, shortness of breath, wheezing, hoarseness, chest and arm pain, constant fatigue, and swelling of the face and neck. A key factor in the development of lung cancer is smoking. Lung cancer usually arises from exposure to carcinogens in tobacco smoke, not only traditional cigarettes, but also more popular electric cigarettes nowadays. Other factors include polluted air, exposure to asbestos or radon and genetic background.6 It has been reported that c-myc, mutated K-ras and overexpressed EGFR, cyclin D1 and BCL2 are oncogenes that contribute to the pathogenesis of lung cancer.7,8 There are two main types of lung cancer: small cell carcinoma and non-small cell carcinoma (adenocarcinoma, squamous cell carcinoma and large cell carcinoma).9,10 Diagnosis of lung cancer includes X-ray, CT scan, positron emission tomography, MRI, or chest ultrasound.11–13 Once the tumour is detected and localised, the main investigation is a biopsy. Usually, the tumour is surgically removed, and chemotherapy or radiotherapy is implemented. New treatments such as targeted molecular treatment or immunotherapy are currently being developed.10
Breast cancer is one of the most common cancers in women. It has been reported that malignant transformation is connected to the dysregulation of fatty acid metabolism in breast cancer.14 The change in metabolism occurs also in mitochondria.15 Breast cancer arises mainly from breast cells and, if malignant, can metastasise to lymph nodes or other organs such as the lungs.16 Most breast cancer cases originate in the ducts and far fewer in the lobules. It is a dangerous disease because it usually does not produce any symptoms for a long time. When it is detected, it has often already metastasised. Obesity, physical inactivity, alcohol abuse and ionising radiation are common factors in the development of breast cancer.17,18 Some breast cancers have a genetic basis - if there is a family history, subsequent generations are at higher risk. There are many programmes to improve breast cancer diagnosis, such as free mammography for women over sixty, but this still does not improve the diagnosis enough.19
The brain is the centre of the human nervous system. It is the most complex organ in the entire body. Brain cells are neurons that are capable of processing and conducting information in the form of an electrical signal.20,21 Neurons are present in the brain and throughout the body, where they are connected by synapses.22 The role of the brain is to control all processes in the body.
It is well known that diet is a key factor affecting health. Various diseases result from a poor diet, such as diabetes,23–27 obesity,28–31 cardiovascular disease32 or osteoporosis.33–36 It has been reported that some cancers are diet-dependent.37–39 Instead, a proper and well-balanced diet may be important in cancer prevention. During cancer treatment, such as chemotherapy, diet plays a key role in recovery. The amount of nutrients required to recover from cancer is much higher than in a standard diet, so special nutrition should be implemented. A diet rich in fruit and vegetables has been proven to be the key to preventing disease or recovering faster.
β-carotene is the most common carotenoid, present in various diets, which can be converted into vitamin A (retinol). Sources of β-carotene include plants such as carrots, spinach, lettuce, broccoli or pumpkin, fruits such as melons or oranges and egg yolks.40–42Fig. 1 shows the structural formula of β-carotene and its typical sources.
Its functions in the human body are to prevent from reactive oxygen species (ROS), participate in the formation of melanin, collagen and facilitate the binding of minerals, thus improving bone structure. β-carotene stimulates the immune system and prevents inflammation.43 Its ability to prevent from ROS is valuable in cancer prevention. Furthermore, being a precursor to vitamin A, which is responsible for cell differentiation, also indicates that β-carotene may inhibit cancer development.44–46
β-carotene in the human body is cleaved at the 15,15′ double bond by β-carotene-15,15′-oxygenase and generates two molecules of retinaldehyde. It can be oxidised to retinoic acid by the aldehyde dehydrogenase family of enzymes. Further oxidation of retinoic acid by the cytochrome P450 (CYP) 26 family converts retinoic acid to 4-oxo-retinoic acid, which is more polar. Other alcohol dehydrogenases and retinol dehydrogenases can reduce retinaldehyde to retinol. This can be esterified to retinyl esters. Apo-carotenes can be generated from β-carotene when cleavage occurs at the 9′,10′ double bond.42,47,48 In many research or industrial applications, there is a need to quantify β-carotene or its metabolites. To date, the most popular methods have been high-performance liquid chromatography (HPLC), gas chromatography (GS) and mass spectrometry (MS).49 However, these methods require extraction of the sample and thus its destruction. When there is a need to analyse β-carotene in cellular metabolism, information on the location of metabolites is crucial. To address this, we implemented Raman spectroscopy (RS) and imaging. These techniques have proven to be a valuable tool for the analysis of carotenoids and retinoids.50–54
A phenomenon of the Raman effect is based on the interaction between light and a molecule. When the energy of an incident photon is different than the energy of the molecule, the light is scattered inelastically. Around 1 in 1010 of the light is scattered inelastically at a different wavelength than the incident light.55,56 RS analyses the scattered light and enables the analysis of solid and liquid samples. In biological applications, RS has proven to be a valuable tool because it is non-destructive, label-free and requires minimal sample preparation.57,58
It is well known that β-carotene plays a significant role in human health. Although its potential in cancer prevention is widely discussed, its precise impact on tumour cell metabolism remains unclear. To answer the question of how to improve cancer prevention in human globally, we proposed a study on the effects of β-carotene on brain, breast and lung cancer cells analysed by Raman spectroscopy and imaging.
Materials and methods
Chemicals
The β-carotene (no. C9750) was purchased from Merck Life Science.Cell culture and preparation for microscopy
Cancer cell lines, brain astrocytoma CRL-1718 (no. CCF-STTG1), breast adenocarcinoma MCF7 (no. HTB-22) and lung A549 (CCL-185) were purchased from ATCC. We selected cancerous cell lines based on their representation of mildly aggressive tumours. CRL-1718 was cultured on RPMI-1640 medium (no. 30-2001, ATCC). MCF7 was cultured on Eagle's Minimum Essential Medium (EMEM, no. 30-2003, ATCC). A549 was cultured on Kaighn's Modification of Ham's F-12K Medium (no. 30-2004, ATCC). To each medium Fetal Bovine Serum (FBS, no. 30-2020, ATCC) was added to a final concentration of 10%. Media were renewed twice a week, and the subculturing was done once a week. Cells were cultured in T75 sterile, flat-bottom flasks (no. 90076, Techno Plastic Products). Cells were maintained in a humidified atmosphere incubator at 37 °C with 5% of CO2.Preparation for microscopy was done 24 hours before supplementation. Cells were seeded on CaF2 windows (no. CAFP25-1R Crystran Ltd, Poole, UK) in Petri dish (no. 150460, Nunc). 10 mM β-carotene solution was prepared by dissolving the proper amount of β-carotene powder in dimethyl sulfoxide (DMSO, no. 4-X, ATCC). On the day of supplementation, CRL-1718, MCF7 and A549 cell's media were changed for the solution of β-carotene in RPMI-160, EMEM and F-12K media, respectively. 1 and 10 μM concentrations were investigated. These concentrations were chosen to present two extremes - low, close to the actual conditions found in cells after dietary supplementation, and high, likely to have a therapeutic effect.
After 24 or 48 hours, media were removed, and cells were rinsed with phosphate-buffered saline (PBS, no. 10010023 Gibco) and fixed in 4% formalin (neutrally buffered). Cells in PBS were measured by Raman imaging.
Raman spectroscopy and imaging
All Raman spectra and images presented in this manuscript were obtained using a WITec Alpha 300 RSA+ confocal microscope (Ulm, Germany). This microscope was coupled to an enhanced mode CCD camera (EMCCD, Andor Newton DU970N-UVB-353). Experiments were carried out using a laser with an excitation wavelength of 532 nm and a laser power of 10 mW and a 40× objective with an aqueous solution cap (Zeiss). The integration time was 0.5 seconds for the range from 400 to 1800 cm−1 and 0.3 seconds for the range from 2700 to 3100 cm−1. Each Raman spectrum and image were pre-processed for smoothing (Savitzky–Golay method, order 4, derivative 0), cosmic ray removal (filter size 2, dynamic factor 10) and background subtraction using WITec Project Plus software. Raman images were recorded with 1 × 1 μm spatial resolution. Total amount of analysed samples was 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 control samples and 4 supplemented for each cell line. From each sample, four cells were measured, which gave the total amount of measured cells: 60. For A549 cell line, we analysed 14
3 control samples and 4 supplemented for each cell line. From each sample, four cells were measured, which gave the total amount of measured cells: 60. For A549 cell line, we analysed 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128 spectra for control cells and 24
128 spectra for control cells and 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 864 spectra for supplemented cells. For MCF7 cell line, we analysed 5191 spectra for control cells and 21
864 spectra for supplemented cells. For MCF7 cell line, we analysed 5191 spectra for control cells and 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 424 spectra for supplemented cells. For CRL-1718 cell line, we analysed 9580 spectra for control cells and 32
424 spectra for supplemented cells. For CRL-1718 cell line, we analysed 9580 spectra for control cells and 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 416 spectra for supplemented cells. The average size of one Raman image was 40 × 45 μm, which gives 1800 spectra for one cell and one spectral region. Raman maps were analysed using a Cluster Analysis method.3,59 This approach enables segmentation of spectral data into distinct groups sharing common vibrational signatures. Cluster analysis was conducted using WITec Project Plus software, with the number of clusters set to six. Each Raman map was analysed independently, preserving spatial resolution and mitigating potential misclassification arising from heterogeneity in cell morphology, size, and microenvironmental conditions. There were six clusters corresponding to cell organelles: nucleus (red), mitochondria (magenta), lipid droplets/endoplasmic reticulum (blue), cytoplasm (green), cell membrane (light grey) and the extracellular region (dark grey). Mean and standard deviation calculations were performed in Origin Pro. Statistical analysis, one-way ANOVA with Tukey's tests was performed to denote statistical differences. If the p-value was less than or equal to 0.05, we marked the difference as significant with an asterisk (*). PLS-DA, a chemometric analysis was performed using MATLAB. Prior to Partial Least Squares Discriminant Analysis (PLS-DA), spectral data were vector normalized using the “Divide by Norm” method in Origin Pro to compensate for possible variations in overall signal intensity. For the PLS-DA model, dummy variables were assigned according to the experimental groups corresponding to controls and supplemented samples (as presented on Fig. 9). The classification parameter was based on these categorical group assignments, allowing the model to discriminate between the biochemical profiles associated with each condition. The optimal number of latent variables was determined by cross-validation to avoid overfitting and ensure robustness of the model. Six discrete classes were defined for classification, with mean centring applied to enhance data variance structure. The PLS-DA model was constructed using five latent variables, optimized to capture the maximum covariance between spectral features and class membership.
416 spectra for supplemented cells. The average size of one Raman image was 40 × 45 μm, which gives 1800 spectra for one cell and one spectral region. Raman maps were analysed using a Cluster Analysis method.3,59 This approach enables segmentation of spectral data into distinct groups sharing common vibrational signatures. Cluster analysis was conducted using WITec Project Plus software, with the number of clusters set to six. Each Raman map was analysed independently, preserving spatial resolution and mitigating potential misclassification arising from heterogeneity in cell morphology, size, and microenvironmental conditions. There were six clusters corresponding to cell organelles: nucleus (red), mitochondria (magenta), lipid droplets/endoplasmic reticulum (blue), cytoplasm (green), cell membrane (light grey) and the extracellular region (dark grey). Mean and standard deviation calculations were performed in Origin Pro. Statistical analysis, one-way ANOVA with Tukey's tests was performed to denote statistical differences. If the p-value was less than or equal to 0.05, we marked the difference as significant with an asterisk (*). PLS-DA, a chemometric analysis was performed using MATLAB. Prior to Partial Least Squares Discriminant Analysis (PLS-DA), spectral data were vector normalized using the “Divide by Norm” method in Origin Pro to compensate for possible variations in overall signal intensity. For the PLS-DA model, dummy variables were assigned according to the experimental groups corresponding to controls and supplemented samples (as presented on Fig. 9). The classification parameter was based on these categorical group assignments, allowing the model to discriminate between the biochemical profiles associated with each condition. The optimal number of latent variables was determined by cross-validation to avoid overfitting and ensure robustness of the model. Six discrete classes were defined for classification, with mean centring applied to enhance data variance structure. The PLS-DA model was constructed using five latent variables, optimized to capture the maximum covariance between spectral features and class membership.
Results and discussion
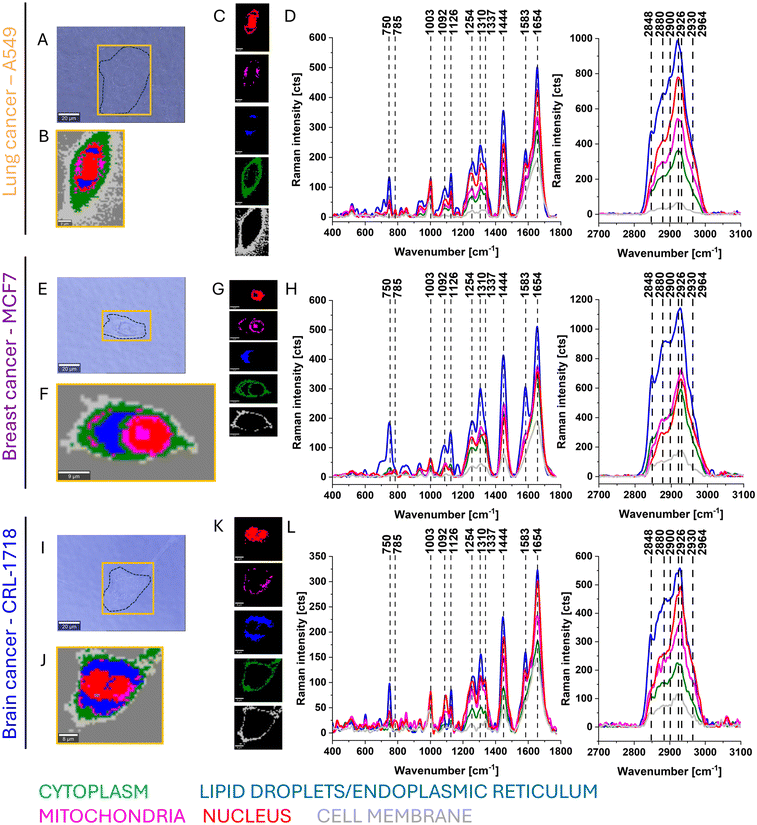
The aim of our study was to investigate how β-carotene affects lung, breast and brain cancer cells using Raman imaging. We analysed cells without supplementation (control) as reference, and cells supplemented with 1 and 10 μM β-carotene.We started our considerations with an analysis of the control cells. Fig. 2 shows typical Raman images of control cells from cell lines A549, MCF7 and CRL-1718.
Fig. 2 presents Raman imaging of control, non-supplemented cells. Cluster analysis enabled the identification of five distinct cellular domains, corresponding to the nucleus (red), mitochondria (magenta), lipid droplets/endoplasmic reticulum (blue), cytoplasm (green) and cell membrane (grey). Each domain exhibited characteristic vibrational signatures. Lipid-rich regions demonstrated prominent Raman bands at 1254, 1444, 1660 cm−1 and 2848 cm−1. The nucleus and mitochondria were distinguished by well-defined bands at approximately 785, 1092 cm−1 (nucleic acids/DNA) and 750, 1126, 1310, 1583 cm−1 (mitochondrial cytochrome c), respectively. In contrast, cytoplasm and cell membrane domains displayed generally lower intensities across most Raman bands analysed. However, protein-related amide bands (Amide I and III regions, ∼1654 cm−1 and ∼1337 cm−1 respectively) are observed throughout the cytoplasm and mitochondria, reflecting the high protein content in these organelles.
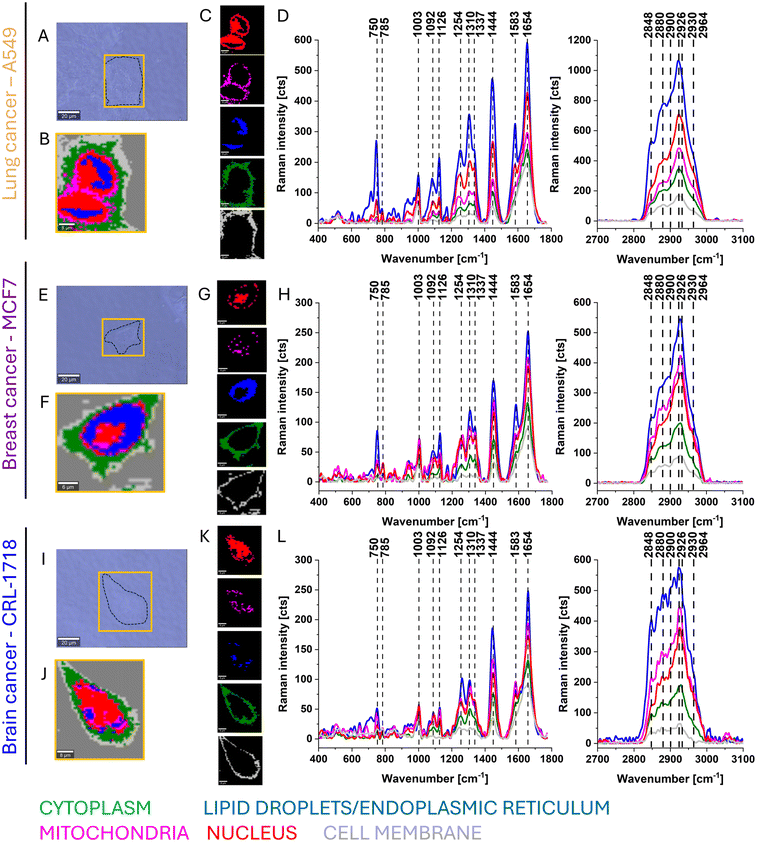
Now let us focus on the analysis of supplemented cells. Fig. 3 shows typical Raman images and spectra of β-carotene-supplemented A549, MCF7 and CRL-1718 cells.
Based on the results presented in Fig. 2 and 3, we have demonstrated that Raman spectroscopy and Raman imaging are a suitable tool for the analysis of the structure of cellular organization. Fig. 2 and 3 provide information on the distribution of chemical components in the cells. The Raman bands mode assignments are presented in Table 1.
| Raman band wavenumber [cm−1] | Raman vibrational mode assignment55,60–81 |
|---|---|
| 750 | Tryptophan (Proteins), cytochrome c |
| 785 | DNA |
| 1003 | Phenylalanine (Proteins) |
| 1092 | O–P–O backbone stretching in DNA, symmetric phosphate stretching vibrations, phosphodioxy |
| 1126 | C–N stretching in proteins, cytochrome c, disaccharides |
| 1254 | PO2− asymmetric stretching in lipids, C–N in plane stretching, formalin contamination |
| 1310 | Guanine–ring breathing mode in DNA, cytochrome c, mixed fatty acid chains |
| 1337 | Amide III and CH2 wagging vibrations in DNA, C–H deformation in protein, tryptophan, nucleic acid, cytochrome c |
| 1444 | Cholesterol, lipids and fatty acids, CH2 and CH3 deformation vibrations in lipids, mixed amide I protein |
| 1583 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bending mode of phenylalanine, cytochrome c C bending mode of phenylalanine, cytochrome c |
| 1654–1660 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching in lipids and proteins, amide I protein C stretching in lipids and proteins, amide I protein |
| 2848 | Symmetric stretching of CH2 of lipids, triglycerides, phospholipids |
| 2870 | Symmetric stretching of CH3 of lipids, CH2 asymmetric stretch of lipids and proteins |
| 2880 | Symmetric stretching of CH2 and CH3, and asymmetric stretching of CH of lipids and proteins |
| 2900 | CH stretch of lipids and proteins |
| 2926 | CH3 stretching vibrations, CH stretch of lipids and proteins |
| 2930 | Symmetric and asymmetric stretching of CH3 in proteins |
| 2964 | Asymmetric stretching of CH3 |
The prominent Raman bands are at 750, 1003, 1092, 1126, 1254, 1310, 1337, 1444, 1583, 1654, 2848, 2870, 2880, 2900, 2926, 2930 and 2964 cm−1. Moving on to the analysis of individual cell organelles, we will focus on lipid droplets. Fig. 4 shows the average Raman spectra from lipid droplets/endoplasmic reticulum. Each graph shows spectra from control cells and cells supplemented with 1 and 10 μM β-carotene after 24 and 48 hours of incubation with the compound.
Fig. 4 shows the average Raman spectra in two spectral ranges: from 400 to 1800 cm−1 and from 2700 to 3100 cm−1. It can be seen that in lung cells the highest intensity of most Raman bands is seen for 10 μM β-carotene after 48 hours. In breast cancer cells, the highest intensity can be observed for control cells. Brain cancer cells showed the highest intensity of most Raman bands for 10 μM β-carotene after 24 hours. Except for the band at 1583 cm−1, where the highest intensity was for 10 μM β-carotene for 48 hours. This indicates that β-carotene induces changes in these organelles, and these changes are different for each organ. In the lung, the highest concentration of β-carotene for the longest time results in more lipids (1254, 1444, 1654 cm−1) and proteins (1003, 1654 cm−1) in the lipid droplets/endoplasmic reticulum. In breast cancer, β-carotene supplementation reduced the intensity of most bands and thus the amount of lipids and proteins in lipid droplets/endoplasmic reticulum. In brain cancer cells, the intensities were higher after β-carotene supplementation and therefore the amount of lipids and proteins in this organelle was higher. It has been shown that, regardless of the mechanism of action, excess carotenoids are stored in adipose tissue.41,44–46 Additionally, we prepared analogous analysis for nucleus (Fig. S1, ESI†) and mitochondria (Fig. S2, ESI†).
Numerous studies have explored carotenoids and their impact on human health.41,44–46,82 It has been showed that carotenoids exert a variety of biological effects, which may be driven by different mechanisms.82,83Fig. 5 shows the mechanisms of action of β-carotene. These mechanisms include direct antioxidant actions, redox-sensitive cell signalling, vitamin A signalling pathways, and others.41,44–46,82
 | ||
| Fig. 5 The mechanism of β-carotene activity inside human cell.82 Abbreviations: APAF-1 - apoptotic protease activating factor-1; Bak - Bcl-2 homologues antagonist/killer; BAX - Bcl-2 associated X; Bcl-2 - B-cell lymphoma 2; Bcl-XL - B-cell lymphoma-extra-large; c-fos, c-jun, c-myc – genes; COX-2 - cyclooxygenase 2; elk-1 - ETS transcription factor ELK1; ERK - extracellular signal-reduced kinase; IKK - inhibitory-κB kinase; Iκ-B -Ikappa-B kinase; IL-6; IL-8 - pro-inflammatory cytokines; MEK - mitogen-activated protein kinase/extracellular signal-reduced kinase; MOMP - mitochondrial outer membrane permeabilization; NF-κB - nuclear factor kappa-light-chain-enhancer of activated B cells; p38 MAPK - p38 mitogen-activated protein kinase; p53 - tumour protein; PI3K - phosphoinositide 3-kinase; RAF - member of the RAF kinase family; RAS - a guanosine-nucleotide-binding protein. | ||
Carotenoids possess anticancer properties and can prevent the development of various types of cancer, such as bladder, breast, leukaemia, lung, and prostate cancers. The underlying mechanisms of this anticancer activity may vary depending on the specific carotenoid being analysed. These mechanisms include induction of apoptosis and the inhibition of cell proliferation. It should be mentioned that β-carotene is a precursor of vitamin A (retinoid acid) and plays a key role in the differentiation and proliferation of lung epithelial cells. The deficiency or excess of vitamin A can lead to squamous metaplasia of the bronchial epithelium, confirming the critical role of β-carotene in the proper growth and function of lung cells. The metabolic mechanism driving this process is the RAR/RXR-dependent repression of AP1 activity, where AP1 is a transcriptional regulator that responds to external signals, and its activation promotes cell proliferation through the expression of cyclin D1. Squamous metaplasia, a precancerous lesion of the airway epithelium, is induced by the expression of keratins and other proteins.84 β-carotene in lung cells may undergo oxidation and cleavage to form oxidative metabolites. These metabolites can decrease retinoic acid levels and impair retinoid signalling by down-regulating RAR expression and up-regulating AP1 (activator protein 1). When present in excess, β-carotene may have procarcinogenic effects.85 In reported study86 of breast cancer cells, β-carotene has been shown to be an inhibitor of cell proliferation, where stopped the cell cycle progression of MDA-MB-231 cells at the G1 phase. Authors suggested that the antiproliferative effects of β-carotene involve the c-Jun N-terminal kinase (JNK) pathway. What is interesting, β-carotene did not affected healthy, control cells viability in this study.86 β-carotene supplementation reduced the viability of both MCF7 and MDA-MB-231 cells, and decreased the number of cells in the G2/M and G0/G1 phases, respectively.87 It has also been demonstrated that oxidation products of β-carotene (e.g. retinol and retinoic acid) exert effects on breast cancer cells derived from the MCF7 cell line, inhibiting their proliferation.88
In breast cancer cells, carotenoids have been shown to downregulate the PI3K/Akt/mTOR signalling pathway and inhibit the RAS/RAF/MEK/ERK1/2 signalling cascade, thereby inhibiting cell proliferation. Carotenoids also block the phosphorylation of IKK protein, preventing the degradation of Iκ-B. Furthermore, carotenoids inhibit the activity of prosurvival proteins (such as Bcl-2 and Bcl-xL) and promote the expression of pro-apoptotic proteins (Bax, Bak, p53), thereby enhancing caspase activity and inducing cell death.82 It has been suggested that β-carotene regulates the expression of oxidative stress-sensitive genes through the inhibition of Akt and ERK1/2 signalling. β-carotene suppresses the expression of Bcl-2 and NF-κB, thereby activating the caspase 3 family and triggering apoptosis. The influence of β-carotene on brain cancer is thought to be related to its antioxidant properties. Reactive oxygen species (ROS) activate transcription factors such as nuclear factor erythroid 2-related factor 2 (Nrf-2), which promotes the survival of cancer cells and cause NF-κB activation. NF-κB is involved in processes such as cell proliferation, invasion, and metastasis. By deactivating ROS, β-carotene helps protect brain cells from oncogenesis.89 The absorption of β-carotene occurs in the human intestinal mucosa.42 A portion of dietary β-carotene is transported with lipoproteins via the bloodstream to tissues, where it is metabolised or stored.90 It is cleaved into β-apo-carotenals and all-trans-retinal, which is subsequently converted to retinol. Central cleavage to retinal is catalysed by β-carotene-15,15′-oxygenase enzymes (CMOI). β-Apocarotenals are produced after nonspecific cleavage at several double bonds, catalysed by CMOII enzymes.47 Retinal may be converted into retinol, retinoic acid, and other polar metabolites, which are then transported to the bloodstream from intestinal mucosal cells.42 Retinoic acid binds to retinoic acid receptors (RARs) and retinoid X receptors (RXRs), which are nuclear receptors involved in steroid hormone signalling.91 Upon activating, these receptors modulate the expression of genes encoding proteins and enzymes. When retinoids bind to RARs, they stimulate the expression of genes involved in cell differentiation and apoptosis, and inhibit the metastasis of breast cancer.92
For a better understanding of the mechanisms of β-carotene action inside the cell, we prepared difference spectra for lipid droplets/endoplasmic reticulum, nucleus and mitochondria. Fig. 6–8 present difference spectra for lipid droplets/endoplasmic reticulum (Fig. 6), nucleus (Fig. 7), and mitochondria (Fig. 8) from lung, breast, and brain cells.
The Fig. 6 shows the boundary conditions (the control cells without supplementation and the higher β-carotene concentration with longer incubation time). It can be seen that in the lung cancer cells (A549) most of the bands have lower or the same intensity as the control cells, except for the band at 1444 cm−1, which has a higher intensity. The intensities of all bands in lung cancer cells between 2700 and 3100 cm−1 are higher than in control cells. In breast cancer (MCF7), all intensities in both ranges are lower than in control cells, apart from protein band at 1003 cm−1, which shows no significant changes. In the range from 400 to 1800 cm−1 in brain cancer cells (CRL-1718), most bands have lower or the same intensity as in control cells. The exception is the band at 1310 cm−1, which has a higher intensity compared to control cells. In brain cancer, the band at 2848 cm−1 has the same intensity, the other bands have a lower intensity than control cells. This could mean that in lipid droplets of lung cells β-carotene increases the number of proteins and lipids (bands at 1444 and in the range of 2700 to 3100 cm−1), in breast cells it decreases the number of cellular components, and in brain cells it also decreases the number of cellular components or makes them the same as in unsupplemented cells. Importantly, in lung and brain cancer, β-carotene supplementation resulted in lower band intensities at 750, 1126 and 1583 cm−1. In breast cancer cells, the changes in band intensities at 750 and 1126 cm−1 were not evident, but the band intensity at 1583 cm−1 was lower than in control cells. This may indicate that β-carotene alters the redox state of cytochrome c in lipid droplets. Analogous analysis was conducted for cell nucleus (Fig. 7) and mitochondria (Fig. 8). Raman spectra of A549 cells after β-carotene supplementation showed higher intensities in the mitochondria, as in the lipid droplets/endoplasmic reticulum. However, in the nucleus, in 400–1800 cm−1 the intensity was slightly lower, and in 2700–3100 cm−1 there was almost no difference. After β-carotene supplementation, the difference in the Raman spectra of MCF7 cells showed lower intensities for all bands in the 400–1800 and 2700–3100 cm−1 spectral ranges in the nucleus and mitochondria, as in the lipid droplets/endoplasmic reticulum. CRL-1718 cells after β-carotene supplementation had higher intensities of all analysed bands in both spectral ranges for the nucleus and mitochondria. This was the opposite result to that obtained in lipid droplets. In the ESI,† we present normalized difference spectra corresponding to lipid droplets (Fig. S6, ESI†), nucleus (Fig. S7, ESI†), and mitochondria (Fig. S8, ESI†).
Focusing on the differences between each organ with and without supplementation, we present a partial least squares-discriminant analysis (PLS-DA). Fig. 9 shows plots of PLS-DA results for all recorded Raman spectra of brain, breast, and lung cancer cells before and after β-carotene supplementation.
Fig. 9 shows the PLS-DA results on control cells and cells supplemented with 10 μM β-carotene. In panel (C) we can see Raman spectra for each latent variable: LV1 (58.15%), LV2 (9.84%), LV3 (4.50%), LV4 (5.11%) and LV5 (4.51%). The following Raman bands can be distinguished on the spectra: 750, 1003, 1092, 1126, 1254, 1310, 1337, 1444, 1583 and 1654 cm−1. LV1 represents most of the analysed data (58.15%) and includes the following bands at: 1003, 1254, 1310, 1337, 1444 and 1654 cm−1. It can be assumed that most of the analysed data corresponds to lipids (1254, 1444, 1654 cm−1) and proteins (1003, 1337, 1654 cm−1). LV2–LV5 have different spectral patterns, but the main Raman bands at 750, 1003, 1126, 1254, 1310, 1337, 1444, 1583 and 1654 cm−1 can be distinguished. Although we also see bands from lipids and proteins in these spectra, bands from cytochrome c (750, 1126, 1310, 1583 cm−1) are very prominent. This may indicate that LV2–LV5 are a representation of the redox state of cytochrome c. The PLS-DA score plot shows a good separation between the three control classes (orange, violet and blue squares with black edges). We can also see the difference between control (squares) and supplemented (rhombuses) classes. The most isolated are the MCF7 controls located in the upper left corner of panel A. In addition, the A549 cell control class is also separated from the other classes. Therefore, the Raman data allowed us to distinguish non-supplemented cells from β-carotene-supplemented cells. The supplemented cells did not form three separate groups, they were mixed. This may indicate that β-carotene supplementation produced similar results in each organ on the cellular level. However, at the cell organelle level, the β-carotene supplementation caused different results in each analysed organ.
To illustrate statistically significant differences in individual Raman bands of cytochrome c, lipids, and proteins, which may be Raman biomarkers of cellular responses to β-carotene supplementation, we used a one-way ANOVA test. Fig. 10 shows the differences in the Raman bands of cytochrome c (750, 1126 cm−1) and lipids (2848 cm−1) in lipid droplets/endoplasmic reticulum. These differences occurred after β-carotene supplementation for 24 and 48 hours.
A one-way ANOVA with Tukey's test was performed to verify differences between control and supplemented cells, as well as between β-carotene doses and incubation time. We analysed lipid droplets/endoplasmic reticulum on Raman bands at 750, 1126 (cytochrome c) and 2848 (lipids) cm−1. In all bands, significant differences were found between control and supplemented cells and between β-carotene concentrations (doses), except for the band at 1126 cm−1 in lung cancer cells and 2848 cm−1 in breast cancer cells, where differences only occurred between control and supplemented cells. In all cases analysed, there were differences at both incubation times, except for the band at 750 cm−1 in breast cancer cells, where differences only occurred after 24 hours of incubation. This suggests that β-carotene supplementation modifies cellular metabolism in lipid droplet organelles, alters the redox state of cytochrome c and the amount of lipids significantly depending on supplementation or lack of there, β-carotene dose, and incubation time with β-carotene.
The results presented so far have shown changes in lung (A549), breast (MCF7) and brain (CRL-1718) cancer cells after β-carotene supplementation. Furthermore, we have demonstrated that Raman imaging is an excellent tool for label-free monitoring of tumour metabolism at the cellular level. In lung cancer cells, β-carotene increased the intensities of bands corresponding to lipids (1444, 2848 cm−1) and lipids/proteins (bands at 2880–2964 cm−1). Furthermore, the intensity of bands at 750, 1126, 1583 cm−1 was lower after supplementation than in control cells, indicating that β-carotene altered the redox state of cytochrome c in lipid droplets in lung cancer cells. In breast cancer cells, β-carotene supplementation resulted in a lower intensity of all Raman bands, which may indicate that β-carotene inhibits cellular metabolism in lipid droplets and therefore the amount of most cellular components is lower than in control cells. In lipid droplets of brain cancer cells, the addition of β-carotene decreased the intensity of most bands, except for an increase in the intensity of the band at 1310 cm−1, which corresponds to mixed fat chains (lipids), and maintained the intensity of the band at 2848 cm−1. Thus, β-carotene can inhibit the growth of brain cancer cells, but to a lesser extent than in breast cancer cells. These results generally show that β-carotene alters the metabolism of a single cell, especially in lipid droplets/endoplasmic reticulum, and that this alteration is different in lung, breast, and brain cancer cells.
We found that β-carotene supplementation increased the amount of lipids in lipid droplets of human lung cancer cells, which may be related to a redirection of cellular metabolism to lipid overproduction. This is an unconventional finding, as β-carotene has been labelled effective in cancer prevention, the only exception being the smoking group, where β-carotene increased lung cancer development.93,94 Our results agree with Wright et al. who found the molecular origin of this effect. They suggested that β-carotene supplementation results in aberrant cell growth.95 Some studies have shown that even if β-carotene induces cancer development in smokers, it has a beneficial effect on non-smokers.84 If our results can be understood as an increase in cancer development, this would be consistent with Jeon et al. who found that β-carotene supplementation did not express a preventive effect on cancer incidence and cancer mortality.96 Our findings agree with Kordiak et al. who claimed that β-carotene supplementation increased the risk of lung cancer.97 They analysed clinical trials on lung cancer risk. Ruano-Ravina et al. analysed the effects of carotenoids and vitamins on the development of lung cancer and found that β-carotene alone did not prevent this cancer.98 In contrast to our findings, Yu et al. has showed that higher diet β-carotene and vitamin A intake reduce the risk of lung cancer.99 Another example of the opposite of our results is the research of Shareck et al.100 They analysed clinical trials and found that β-carotene has preventive influence on lung cancer. However, the anticancer properties of β-carotene were due to its antioxidant function.
Our findings on the effects of β-carotene on breast and brain cancer cells are similar. We found that β-carotene acts on these cancers by reducing the amount of most cellular components, such as lipids (Raman bands at 1254, 1444, 2848 cm−1) or proteins (Raman bands at 1003, 1310, 2880–2964 cm−1) in lipid droplets. Our results agree with Gloria et al.87 who found that β-carotene supplementation induces cell cycle arrest and apoptosis in human breast cancer. They also studied MCF7 cells and proved by MTT assays that β-carotene supplementation induces apoptosis. This could be biological evidence for our findings. Another biological proof was provided by Sowmya-Shree et al.101 who claimed that β-carotene increased apoptosis in MCF7 cells, which was associated to increased capsase-3 activity. In opposition to our research results stands Nagel et al. who claimed that β-carotene has weak protective effect.102 They analysed data from clinical trials. We analysed the influence of all-trans-retinal (ALTR) on brain cancer cells and found that ALTR as a metabolite of β-carotene inhibits brain cancer development.54 Our results have proven that β-carotene supplementation changes human metabolism on the cellular level. It agrees with Kim et al. who analysed neuroblastoma and proved that β-carotene inhibits cell invasion and metastasis by decreasing the level of hypoxia-inducible factor-1α.103 Holick et al.104 stands in opposition to our results. They claim that carotenoid consumption is not likely associated with the risk of adult glioma. Lin et al. also claimed that β-carotene intake does not benefit the incidence or mortality of brain cancer.105 However, β-carotene has a confirmed influence on brain diseases like Alzheimer's.106 Thus, it is proven that β-carotene affects brain cells. In summary, the mechanism of action varies on the organ.
Conclusions
Here, we employed Raman spectroscopy to analyse the potential impact of β-carotene on lung, breast, and brain cancer cells in the context of cancer prevention. Our results revealed organ-specific effects: β-carotene enhanced lipid production in lung cancer cells, as evidenced by changes in the intensity of lipid-associated Raman bands (1444, 2848 cm−1). Conversely, it inhibited cell growth and reduced lipid and protein levels in breast and brain cancer cells, demonstrated by decreased intensities in Raman bands associated with lipids and proteins (1003, 1254, 1310, 1444, 1654, 2848–2964 cm−1).In summary, the results of this study highlight the organ-specific effects of β-carotene on cancer cell metabolism, suggesting its potential role in cancer prevention and treatment.
In the context of the current state of knowledge on the effects of β-carotene on human lung, breast, and brain cancer, we have demonstrated by Raman imaging that β-carotene modifies the metabolism in cancer cells. This study involved recording Raman spectra and images in human lung (A549), breast (MCF7) and brain (CRL-1718) cancer cells in vitro with and without β-carotene at concentrations of 1 and 10 μM after 24 and 48 hours of incubation. Therefore, we demonstrated that Raman imaging is an effective technique for qualitative and quantitative analysis of individual cellular organelles. We found that β-carotene modifies the functional activity of cells, causing changes in lipids, proteins, and redox state of cytochrome c. We focused on lipid droplets and demonstrated that in this cell organelle, β-carotene supplementation improves the amount of lipids in proteins in lung cancer, reduces the amount of lipids and proteins in breast cancer, and mainly reduces the amount of lipids and proteins in brain cancer. An important finding is that β-carotene altered the redox state of cytochrome c (bands at 750, 1126 and 1583 cm−1) in lipid droplets. These findings provide promising support for understanding the effects of β-carotene on three common human cancers - lung, breast, and brain.
Author contributions
Conceptualization: K. J., J. S.; investigation: K. J.; methodology: J. S.; writing – original draft: K. J., J. S.; manuscript editing: J. S., K. J.; M. K. manuscript revision: J. S., M. K., K. J. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the ESI.†Acknowledgements
This work was supported by the FU²N – Young Scientists' Skills Improvement Fund, Grant: “Application of Raman spectroscopy in the analysis of carotenoid-supplemented cancer cells-determining the impact.” Number 503/3-34-4-1 and the National Science Centre of Poland (OPUS, UMO 2021/43/B/ST4/01547). This paper has been completed while the first author was the Doctoral Candidate in the Interdisciplinary Doctoral School at the Lodz University of Technology, Poland. The research was supported by the Statutory Funding.References
- R. L. Siegel, A. N. Giaquinto and A. Jemal, Cancer statistics, 2024, CA. Cancer J. Clin., 2024, 74, 12–49 CrossRef PubMed.
- S. K. N. Marie and S. M. O. Shinjo, Metabolism and Brain Cancer, Clinics, 2011, 66, 33–43 CrossRef PubMed.
- M. Kopeć, K. Beton, K. Jarczewska and H. Abramczyk, Hyperglycemia and cancer in human lung carcinoma by means of Raman spectroscopy and imaging, Sci. Rep., 2022, 12, 18561 CrossRef PubMed.
- L. A. Torre, R. L. Siegel and A. Jemal, Lung Cancer Statistics, Advances in Experimental Medicine and Biology, 2016, vol. 893, pp. 1–19 Search PubMed.
- M. B. Schabath and M. L. Cote, Cancer Progress and Priorities: Lung Cancer, Cancer Epidemiol., Biomarkers Prev., 2019, 28, 1563–1579 CrossRef PubMed.
- K. C. Thandra, A. Barsouk, K. Saginala, J. S. Aluru and A. Barsouk, Epidemiology of lung cancer, Contemp. Oncol., 2021, 25, 45–52 CAS.
- J. D. Minna, J. A. Roth and A. F. Gazdar, Focus on lung cancer, Cancer Cell, 2002, 1, 49–52 CrossRef CAS PubMed.
- K. M. Fong, Y. Sekido and J. D. Minna, The Molecular Basis of Lung Carcinogenesis. in The Molecular Basis of Human Cancer ed. W. B. Coleman and G. J. Tsongalis, Humana Press, Totowa, NJ, 2002, pp. 379–405 DOI:10.1007/978-1-59259-125-1_17.
- W. D. Travis, et al., International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma, J. Thorac. Oncol., 2011, 6, 244–285 CrossRef PubMed.
- L. G. Collins, C. Haines, R. Perkel and R. E. Enck, Lung Cancer: Diagnosis and Management, Am. Fam. Physician, 2007, 75, 56–63 Search PubMed.
- W. D. Travis, Pathology of Lung Cancer, Clin. Chest Med., 2011, 32, 669–692 CrossRef PubMed.
- M.-H. Tao, Epidemiology of lung cancer. in Lung Cancer and Imaging, IOP Publishing, 2019 DOI:10.1088/978-0-7503-2540-0ch4.
- S. J. Adams, et al., Lung cancer screening, Lancet, 2023, 401, 390–408 CrossRef PubMed.
- M. E. Monaco, Fatty acid metabolism in breast cancer subtypes, Oncotarget, 2017, 8, 29487–29500 CrossRef PubMed.
- M. Cazzaniga and B. Bonanni, Breast Cancer Metabolism and Mitochondrial Activity: The Possibility of Chemoprevention with Metformin, BioMed Res. Int., 2015, 2015, 972193 Search PubMed.
- B. A. Gusterson and T. Stein, Human breast development, Semin. Cell Dev. Biol., 2012, 23, 567–573 CrossRef PubMed.
- Y.-S. Sun, et al., Risk Factors and Preventions of Breast Cancer, Int. J. Biol. Sci., 2017, 13, 1387–1397 CrossRef CAS PubMed.
- Z. Momenimovahed and H. Salehiniya, Epidemiological characteristics of and risk factors for breast cancer in the world, Breast Cancer Targets Ther., 2019, 11, 151–164 CrossRef PubMed.
- N. Karssemeijer, et al., Breast Cancer Screening Results 5 Years after Introduction of Digital Mammography in a Population-based Screening Program, Radiology, 2009, 253, 353–358 CrossRef PubMed.
- M. S. Gazzaniga, Organization of the Human Brain, Science, 1989, 245, 947–952 CrossRef CAS PubMed.
- R. Free, J. Clark, S. Amara and D. R. Sibley, Neurotransmission in the Central Nervous System, in Goodman & Gilman's: The Pharmacological Basis of Therapeutics, 13e, ed. L. L. Brunton, R. Hilal-Dandan and B. C. Knollmann, McGraw-Hill Education, 2017, https://accessmedicine.mhmedical.com/content.aspx?bookid=2189§ionid=170349771 Search PubMed.
- P. Brodal, The Central Nervous System, Oxford University Press, 2010 Search PubMed.
- J. Salas-Salvadó, M. Á. Martinez-González, M. Bulló and E. Ros, The role of diet in the prevention of type 2 diabetes, Nutr., Metab. Cardiovasc. Dis., 2011, 21, B32–B48 CrossRef PubMed.
- C. Liang, K. DeCourcy and M. R. Prater, High–saturated-fat diet induces gestational diabetes and placental vasculopathy in C57BL/6 mice, Metabolism, 2010, 59, 943–950 CrossRef CAS PubMed.
- A. M. Hodge, M. N. Karim, J. R. Hébert, N. Shivappa and B. de Courten, Association between Diet Quality Indices and Incidence of Type 2 Diabetes in the Melbourne Collaborative Cohort Study, Nutrients, 2021, 13, 4162 CrossRef CAS PubMed.
- V. Lazar, L.-M. Ditu, G. G. Pircalabioru, A. Picu, L. Petcu, N. Cucu and M. C. Chifiriuc, Gut Microbiota, Host Organism, and Diet Trialogue in Diabetes and Obesity, Front. Nutr., 2019, 6, 21 CrossRef PubMed.
- M. Verboven, D. Deluyker, D. Hansen, B. O. Eijnde and V. Bito, P1463The origin of diabetes: high-sugar diet to induce diabetic cardiomyopathy in rats, Eur. Heart J., 2017, 38, ehx502.P1463 CrossRef.
- K. Lim, B. Barzel, S. L. Burke, J. A. Armitage and G. A. Head, Origin of Aberrant Blood Pressure and Sympathetic Regulation in Diet-Induced Obesity, Hypertension, 2016, 68, 491–500 CrossRef CAS PubMed.
- N. Hariri and L. Thibault, High-fat diet-induced obesity in animal models, Nutr. Res. Rev., 2010, 23, 270–299 CrossRef CAS PubMed.
- L. D. Ruiz, M. L. Zuelch, S. M. Dimitratos and R. E. Scherr, Adolescent Obesity: Diet Quality, Psychosocial Health, and Cardiometabolic Risk Factors, Nutrients, 2020, 12, 43 CrossRef CAS PubMed.
- P. Huypens, et al., Epigenetic germline inheritance of diet-induced obesity and insulin resistance, Nat. Genet., 2016, 48, 497–499 CrossRef CAS PubMed.
- A. Mente, et al., Diet, cardiovascular disease, and mortality in 80 countries, Eur. Heart J., 2023, 44, 2560–2579 CrossRef CAS PubMed.
- C. Cooper, et al., Review: developmental origins of osteoporotic fracture, Osteoporos. Int., 2006, 17, 337–347 CrossRef PubMed.
- B. Teucher and S. Fairweather-Tait, Dietary sodium as a risk factor for osteoporosis: where is the evidence?, Proc. Nutr. Soc., 2003, 62, 859–866 CrossRef CAS PubMed.
- M. Martiniakova, et al., The Role of Macronutrients, Micronutrients and Flavonoid Polyphenols in the Prevention and Treatment of Osteoporosis, Nutrients, 2022, 14, 523 CrossRef CAS PubMed.
- D. A. Nelson, Evolutionary Origins of the Differences in Osteoporosis Risk in US Populations, J. Clin. Densitom., 2019, 22, 301–304 CrossRef PubMed.
- S.-C. Chuang, et al., Diet and the risk of head and neck cancer: a pooled analysis in the INHANCE consortium, Cancer, Causes Control, Pap. Symp., 2012, 23, 69–88 CrossRef PubMed.
- C. Sapienza and J.-P. Issa, Diet, Nutrition, and Cancer Epigenetics, Annu. Rev. Nutr., 2016, 36, 665–681 CrossRef CAS PubMed.
- M. C. Stern, J. Barnoya, J. P. Elder and K. Gallegos-Carrillo, Diet, physical activity, obesity and related cancer risk: strategies to reduce cancer burden in the Americas, Rev. Invest. Salud Publica, 2020, 61, 448–455 Search PubMed.
- H. Marona, A. Gunia and E. Pękala, Retinoidy – rola w farmakoterapii w aspekcie komórkowego mechanizmu działania, 2010 Search PubMed.
- S. Agarwal and A. V. Rao, Carotenoids and chronic diseases, Drug Metabol. Drug Interact., 2000, 17, 189–210 CrossRef CAS PubMed.
- X. D. Wang, Review: absorption and metabolism of beta-carotene, J. Am. Coll. Nutr., 1994, 13, 314–325 CrossRef CAS PubMed.
- D. A. Hughes, Effects of carotenoids on human immune function, Proc. Nutr. Soc., 1999, 58, 713–718 CrossRef CAS PubMed.
- H. Stähelin, K. Gey, M. Eichholzer and E. Lüdin, β-Carotene and cancer prevention: the Basel Study, Am. J. Clin. Nutr., 1991, 53, 265S–269S CrossRef PubMed.
- N. Druesne-Pecollo, et al., Beta-carotene supplementation and cancer risk: a systematic review and metaanalysis of randomized controlled trials, Int. J. Cancer, 2010, 127, 172–184 CrossRef CAS PubMed.
- G. van Poppel and R. Goldbohm, Epidemiologic evidence for beta-carotene and cancer prevention, Am. J. Clin. Nutr., 1995, 62, 1393S–1402S CrossRef CAS PubMed.
- V. Shete and L. Quadro, Mammalian Metabolism of β-Carotene: Gaps in Knowledge, Nutrients, 2013, 5, 4849–4868 CrossRef CAS PubMed.
- R. S. Parker, Absorption, metabolism, and transport of carotenoids, FASEB J., 1996, 10, 542–551 CrossRef CAS PubMed.
- H. Stutz, N. Bresgen and P. M. Eckl, Analytical tools for the analysis of β-carotene and its degradation products, Free Radic. Res., 2015, 49, 650–680 CrossRef CAS PubMed.
- V. E. de Oliveira, H. V. Castro, H. G. M. Edwards and L. F. C. de Oliveira, Carotenes and carotenoids in natural biological samples: a Raman spectroscopic analysis, J. Raman Spectrosc., 2010, 41, 642–650 CrossRef CAS.
- R. Withnall, B. Z. Chowdhry, J. Silver, H. G. M. Edwards and L. F. C. de Oliveira, Raman spectra of carotenoids in natural products, Spectrochim. Acta, Part A, 2003, 59, 2207–2212 CrossRef PubMed.
- M. Baranska, M. Roman, J. Cz. Dobrowolski, H. Schulz and R. Baranski, Recent Advances in Raman Analysis of Plants: Alkaloids, Carotenoids, and Polyacetylenes, Curr. Anal. Chem., 2013, 9, 108–127 CrossRef CAS.
- C. P. Marshall and A. Olcott Marshall, The potential of Raman spectroscopy for the analysis of diagenetically transformed carotenoids, Philos. Trans. R. Soc., A, 2010, 368, 3137–3144 CrossRef CAS PubMed.
- K. Jarczewska, M. Kopeć, H. Abramczyk and J. M. Surmacki, Monitoring alterations of all-trans-retinal in human brain cancer cells by label-free confocal Raman imaging: regulation of the redox status of cytochrome c, RSC Adv., 2024, 14, 20982–20991 RSC.
- Z. Movasaghi, S. Rehman and I. U. Rehman, Raman Spectroscopy of Biological Tissues, Appl. Spectrosc. Rev., 2007, 42, 493–541 CrossRef CAS.
- J. Conroy, A. G. Ryder, M. N. Leger, K. Hennessey and M. G. Madden, Qualitative and quantitative analysis of chlorinated solvents using Raman spectroscopy and machine learning, Opto-Ireland 2005: Optical Sensing and Spectroscopy, SPIE, 2005, vol. 5826, pp. 131–142 Search PubMed.
- K. A. Antonio and Z. D. Schultz, Advances in Biomedical Raman Microscopy, Anal. Chem., 2014, 86, 30–46 CrossRef CAS PubMed.
- E. E. Lawson, B. W. Barry, A. C. Williams and H. G. M. Edwards, Biomedical Applications of Raman Spectroscopy, J. Raman Spectrosc., 1997, 28, 111–117 CrossRef CAS.
- B. Brozek-Pluska, J. Musial, R. Kordek and H. Abramczyk, Analysis of Human Colon by Raman Spectroscopy and Imaging-Elucidation of Biochemical Changes in Carcinogenesis, Int. J. Mol. Sci., 2019, 20, 3398 CrossRef CAS PubMed.
- K. Jarczewska, M. Kopeć and J. M. Surmacki, Monitoring cellular human breast adenocarcinoma cells’ response to xanthophylls by label-free Raman spectroscopy and imaging, Spectrochim. Acta, Part A, 2025, 339, 126263 CrossRef CAS PubMed.
- K. Jarczewska, M. Kopeć and J. M. Surmacki, Raman signatures of astrocytoma metabolism alterations induced by crocin, fucoxanthin and lutein, Spectrochim. Acta, Part A, 2025, 342, 126465 CrossRef CAS PubMed.
- J. Binoy, et al., NIR-FT Raman and FT-IR spectral studies and ab initio calculations of the anti-cancer drug combretastatin-A4, J. Raman Spectrosc., 2004, 35, 939–946 CrossRef CAS.
- J. W. Chan, et al., Micro-Raman spectroscopy detects individual neoplastic and normal hematopoietic cells, Biophys. J., 2006, 90, 648–656 CrossRef CAS PubMed.
- W.-T. Cheng, M.-T. Liu, H.-N. Liu and S.-Y. Lin, Micro-Raman spectroscopy used to identify and grade human skin pilomatrixoma, Microsc. Res. Tech., 2005, 68, 75–79 CrossRef CAS PubMed.
- R. K. Dukor, Vibrational Spectroscopy in the Detection of Cancer, in Handbook of Vibrational Spectroscopy, John Wiley & Sons, Ltd, 2006 DOI:10.1002/0470027320.s8107.
- A. J. Ruiz-Chica, M. A. Medina, F. Sánchez-Jiménez and F. J. Ramírez, Characterization by Raman spectroscopy of conformational changes on guanine–cytosine and adenine–thymine oligonucleotides induced by aminooxy analogues of spermidine, J. Raman Spectrosc., 2004, 35, 93–100 CrossRef CAS.
- G. Shetty, C. Kendall, N. Shepherd, N. Stone and H. Barr, Raman spectroscopy: elucidation of biochemical changes in carcinogenesis of oesophagus, Br. J. Cancer, 2006, 94, 1460–1464 CrossRef CAS PubMed.
- D. Naumann, Infrared and NIR Raman spectroscopy in medical microbiology, Proceedings, 1998, 3257, 245–257 CAS.
- Z. Huang, et al., Effect of formalin fixation on the near-infrared Raman spectroscopy of normal and cancerous human bronchial tissues, Int. J. Oncol., 2003, 23, 649–655 Search PubMed.
- L. Silveira Jr., et al., Correlation between near-infrared Raman spectroscopy and the histopathological analysis of atherosclerosis in human coronary arteries, Lasers Surg. Med., 2002, 30, 290–297 CrossRef PubMed.
- D. P. Lau, et al., Raman spectroscopy for optical diagnosis in normal and cancerous tissue of the nasopharynx-preliminary findings, Lasers Surg. Med., 2003, 32, 210–214 CrossRef PubMed.
- H. Abramczyk, B. Brozek-Pluska and M. Kopeć, Double face of cytochrome c in cancers by Raman imaging, Sci. Rep., 2022, 12, 2120 CrossRef CAS PubMed.
- C. J. Frank, R. L. McCreery and D. C. Redd, Raman spectroscopy of normal and diseased human breast tissues, Anal. Chem., 1995, 67, 777–783 CrossRef CAS PubMed.
- A. Haka, et al., Haka AS, Shafer-Peltier KE, Fitzmaurice M, Crowe J, Dasari RR, Feld MSIdentifying microcalcifications in benign and malignant breast lesions by probing differences in their chemical composition using Raman spectroscopy, Cancer Res., 2002, 62, 5375–5380 CAS.
- N. Stone, C. Kendall, J. Smith, P. Crow and H. Barr, Raman spectroscopy for identification of epithelial cancers, Faraday Discuss., 2004, 126, 141–157 RSC.
- S. Koljenović, T. B. Schut, A. Vincent, J. M. Kros and G. J. Puppels, Detection of meningioma in dura mater by Raman spectroscopy, Anal. Chem., 2005, 77, 7958–7965 CrossRef PubMed.
- N. J. Kline and P. J. Treado, Raman Chemical Imaging of Breast Tissue, J. Raman Spectrosc., 1997, 28, 119–124 CrossRef CAS.
- P. J. Caspers, G. W. Lucassen, E. A. Carter, H. A. Bruining and G. J. Puppels, In vivo confocal Raman microspectroscopy of the skin: noninvasive determination of molecular concentration profiles, J. Invest. Dermatol., 2001, 116, 434–442 CrossRef CAS PubMed.
- H. Abramczyk, et al., Epigenetic changes in cancer by Raman imaging, fluorescence imaging, AFM and scanning near-field optical microscopy (SNOM). Acetylation in normal and human cancer breast cells MCF10A, MCF7 and MDA-MB-231, Analyst, 2016, 141, 5646–5658 RSC.
- C. S. B. Teixeira, et al., Thyroid tissue analysis through Raman spectroscopy, Analyst, 2009, 134, 2361–2370 RSC.
- J. Rau, et al., RAMAN spectroscopy imaging improves the diagnosis of papillary thyroid carcinoma, Sci. Rep., 2016, 6, 35117 CrossRef CAS PubMed.
- J. A. Kim, J.-H. Jang and S.-Y. Lee, An Updated Comprehensive Review on Vitamin A and Carotenoids in Breast Cancer: Mechanisms, Genetics, Assessment, Current Evidence, and Future Clinical Implications, Nutrients, 2021, 13, 3162 CrossRef CAS PubMed.
- R. Sathasivam and J.-S. Ki, A Review of the Biological Activities of Microalgal Carotenoids and Their Potential Use in Healthcare and Cosmetic Industries, Mar. Drugs, 2018, 16, 1–31 CrossRef PubMed.
- R. Goralczyk, β-Carotene and Lung Cancer in Smokers: Review of Hypotheses and Status of Research, Nutr. Cancer, 2009, 61, 767–774 CrossRef CAS PubMed.
- R. M. Russell, Beta-carotene and lung cancer, Pure Appl. Chem., 2002, 74, 1461–1467 CrossRef CAS.
- A. Antunes, F. Carmo, S. Pinto, N. Andrade and F. Martel, The anti-proliferative effect of β-carotene against a triple-negative breast cancer cell line is cancer cell-specific and JNK-dependent, PharmaNutrition, 2022, 22, 100320 CrossRef CAS.
- N. F. Gloria, et al., Lycopene and Beta-carotene Induce Cell-Cycle Arrest and Apoptosis in Human Breast Cancer Cell Lines, Anticancer Res., 2014, 34, 1377–1386 CAS.
- X. Hu, K. M. White, N. E. Jacobsen, D. J. Mangelsdorf and L. M. Canfield, Inhibition of growth and cholesterol synthesis in breast cancer cells by oxidation products of β-carotene 1, J. Nutr. Biochem., 1998, 9, 567–574 CrossRef CAS.
- X. Qi, et al., Antioxidants in brain tumors: current therapeutic significance and future prospects, Mol. Cancer, 2022, 21, 204 CrossRef PubMed.
- T. Bohn, et al., β-Carotene in the human body: metabolic bioactivation pathways – from digestion to tissue distribution and excretion, Proc. Nutr. Soc., 2019, 78, 68–87 CrossRef CAS PubMed.
- X.-C. Xu, Tumor-suppressive activity of retinoic acid receptor-β in cancer, Cancer Lett., 2007, 253, 14–24 CrossRef CAS PubMed.
- R. M. Connolly, N. K. Nguyen and S. Sukumar, Molecular Pathways: Current Role and Future Directions of the Retinoic Acid Pathway in Cancer Prevention and Treatment, Clin. Cancer Res., 2013, 19, 1651–1659 CrossRef CAS PubMed.
- R. Lotan, Lung Cancer Promotion by β-Carotene and Tobacco Smoke: Relationship to Suppression of Retinoic Acid Receptor-β and Increased Activator Protein-1?, JNCI, J. Natl. Cancer Inst., 1999, 91, 7–9 CrossRef CAS PubMed.
- P. Middha, S. J. Weinstein, S. Männistö, D. Albanes and A. M. Mondul, β-Carotene Supplementation and Lung Cancer Incidence in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study: The Role of Tar and Nicotine, Nicotine Tob. Res, 2019, 21, 1045–1050 CrossRef CAS PubMed.
- M. E. Wright, et al., Effects of β-Carotene Supplementation on Molecular Markers of Lung Carcinogenesis in Male Smokers, Cancer Prev. Res. (Phila. Pa.), 2010, 3, 745–752 CrossRef CAS PubMed.
- Y.-J. Jeon, et al., Effects of Beta-Carotene Supplements on Cancer Prevention: Meta-Analysis of Randomized Controlled Trials, Nutr. Cancer, 2011, 63, 1196–1207 CrossRef CAS PubMed.
- J. Kordiak, F. Bielec, S. Jabłoński and D. Pastuszak-Lewandoska, Role of Beta-Carotene in Lung Cancer Primary Chemoprevention: A Systematic Review with Meta-Analysis and Meta-Regression, Nutrients, 2022, 14, 1361 CrossRef CAS PubMed.
- A. Ruano-Ravina, A. Figueiras, M. Freire-Garabal and J. M. Barros-Dios, Antioxidant Vitamins and Risk of Lung Cancer, Curr. Pharm. Des., 2006, 12, 599–613 CrossRef CAS PubMed.
- N. Yu, X. Su, Z. Wang, B. Dai and J. Kang, Association of Dietary Vitamin A and β-Carotene Intake with the Risk of Lung Cancer: A Meta-Analysis of 19 Publications, Nutrients, 2015, 7, 9309–9324 CrossRef CAS PubMed.
- M. Shareck, M.-C. Rousseau, A. Koushik, J. Siemiatycki and M.-E. Parent, Inverse Association between Dietary Intake of Selected Carotenoids and Vitamin C and Risk of Lung Cancer, Front. Oncol., 2017, 7, 1–12 Search PubMed.
- G. Sowmya Shree, et al., β-carotene at physiologically attainable concentration induces apoptosis and down-regulates cell survival and antioxidant markers in human breast cancer (MCF-7) cells, Mol. Cell. Biochem., 2017, 436, 1–12 CrossRef CAS PubMed.
- G. Nagel, et al., Dietary β-carotene, vitamin C and E intake and breast cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC), Breast Cancer Res. Treat., 2010, 119, 753–765 CrossRef CAS PubMed.
- Y.-S. Kim, et al., β-Carotene inhibits neuroblastoma cell invasion and metastasis in vitro and in vivo by decreasing level of hypoxia-inducible factor-1α, J. Nutr. Biochem., 2014, 25, 655–664 CrossRef CAS PubMed.
- C. N. Holick, E. L. Giovannucci, B. Rosner, M. J. Stampfer and D. S. Michaud, Prospective study of intake of fruit, vegetables, and carotenoids and the risk of adult glioma2, Am. J. Clin. Nutr., 2007, 85, 877–886 CrossRef CAS PubMed.
- J. Lin, et al., Vitamins C and E and Beta Carotene Supplementation and Cancer Risk: A Randomized Controlled Trial, JNCI, J. Natl. Cancer Inst., 2009, 101, 14–23 CrossRef CAS PubMed.
- S. Hira, et al., β-Carotene: A Natural Compound Improves Cognitive Impairment and Oxidative Stress in a Mouse Model of Streptozotocin-Induced Alzheimer's Disease, Biomolecules, 2019, 9, 441 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5cp01289a |
| This journal is © the Owner Societies 2025 |