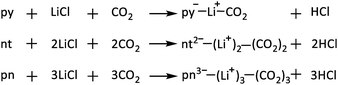Hydroxy-substituted aromatic N-heterocycles as high-affinity CO2 adsorbents: a DFT study†
Puthiyavalappil K.
Arathi
ab and
Cherumuttathu H.
Suresh
 *abc
*abc
aChemical Sciences and Technology Division, CSIR-National Institute for Interdisciplinary Science and Technology, Thiruvananthapuram, 695019, India. E-mail: sureshch@gmail.com
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad, 201002, India
cSrinivasa Ramanujan Institute for Basic Sciences, Kerala State Council for Science, Technology and Environment, Kottayam, Kerala 686501, India
First published on 23rd June 2025
Abstract
Hydroxy-substituted aromatic N-heterocycles, including hydroxy pyridine (py), dihydroxy naphthyridines (nt), and trihydroxy pyridonaphthyridines (pn), have been investigated for their potential as CO2 adsorbents using density functional theory (DFT) calculations. Building on the pioneering work of Luo et al., who demonstrated exceptional CO2 capture capacities in pyridine-containing anion-functionalized ionic liquids, this study extends the exploration to a broader range of N-heterocycles. These N-heterocycles exhibit exceptional CO2 capture capabilities, driven by cooperative interactions between nitrogen and oxygen centres with CO2. The adsorption capacity increases with the number of nitrogen centres and hydroxy groups, with py, nt, and pn systems binding one, two, and three CO2 molecules, respectively. Notably, anionic N-heterocycles exhibit dramatically improved CO2 adsorption compared to their neutral counterparts, forming covalent bonds with CO2. The presence of counter cations, such as lithium or tetramethylphosphonium ions, further stabilizes CO2 adsorption, resulting in shorter interaction distances and higher exergonic free energy values. Solvent effects modeled using monoethanolamine (MEA) indicate a modest reduction in interaction energies for neutral and anionic systems, while ion-paired systems exhibit enhanced CO2 affinity in solution. Additionally, molecular electrostatic potential (MESP) analysis highlights the key adsorption sites and charge delocalization mechanisms that facilitate CO2 capture. The study also finds that enol–keto transformations, which could lead to CO2 conversion into carboxylates, are energetically unfavorable due to the loss of aromatic stability. These findings underscore the potential of hydroxy-substituted N-heterocycles, particularly in their anionic and cation-stabilized forms, as promising candidates for efficient CO2 capture. The insights gained from this study provide valuable guidelines for the design of next-generation CO2 sequestration materials and highlight new directions for experimental validation and real-world applications.
Introduction
The rising levels of atmospheric CO2 have driven global efforts to develop efficient capture and conversion strategies.1–7 Traditional methods, such as sequestration and storage face significant challenges related to cost, efficiency, and environmental impact.8–10 In contrast, adsorption-based techniques employing nitrogen-rich molecules and materials present a promising alternative due to their operational simplicity and lower energy demands.11–15Ionic liquids (ILs) have gained attention as versatile media for both CO2 capture and conversion. The evolution of ILs, from conventional to amine-functionalized and multi-sited ILs, has led to remarkable improvements in CO2 absorption efficiency.16–27 Among these advancements, the work by Luo et al. stands out as a significant contribution to the field. In their seminal study, Luo et al. demonstrated that pyridine-containing anion-functionalized ionic liquids exhibit exceptional CO2 capture capacities, achieving up to 1.60 mol CO2 per mol IL through multiple-site cooperative interactions.28 Their work revealed that the presence of nitrogen and oxygen atoms in the anion, along with π-electron delocalization in the pyridine ring, significantly enhances CO2 binding. This cooperative interaction between the electronegative oxygen and nitrogen atoms in the anion and CO2 was shown to be the key driver of the high CO2 capture capacity.28
Building on the insights from Luo et al.,28 this study aims to further explore the potential of hydroxy-substituted aromatic N-heterocycles as high-affinity CO2 adsorbents. While Luo et al. focused on the role of pyridine-containing ILs, we extend this approach to investigate a broader range of N-heterocycles, including hydroxy pyridines (py), dihydroxy naphthyridines (nt), and trihydroxy pyridonaphthyridines (pn), to understand how the strategic incorporation of nitrogen and hydroxy groups within these molecular structures can enhance CO2 capture through cooperative binding mechanisms.
Recent studies have highlighted the significance of multiple-site cooperative interactions in enhancing CO2 capture.28–38 Inspired by Luo et al.'s findings,28 we hypothesize that the presence of multiple nitrogen and hydroxy groups in these N-heterocycles will lead to enhanced CO2 capture through similar cooperative interactions. Specifically, we aim to elucidate the electronic and structural factors governing CO2 interaction and conversion in these systems, providing valuable insights for the design of efficient CO2 capture materials.
This study will assess the CO2 capture capacity of neutral and anionic forms of these molecules, identify the preferred binding sites for CO2, investigate the thermodynamics of CO2 capture and conversion processes, explore the influence of molecular structure, charge and counter cation on CO2 interaction and conversion efficiency. By unravelling the intricate relationship between molecular structure, charge, and CO2 capture capabilities, this study aims to contribute to the development of innovative solutions for mitigating the environmental impact of greenhouse gas emissions.
In this study, we focus on the adsorption of CO2 onto functionalized organic molecules, rather than its absorption into bulk liquid or solid phases. Adsorption refers to surface-level binding at active sites, whereas absorption involves uptake into the bulk. The molecules studied here are potential precursors for integration into extended frameworks such as MOFs or ZIFs, or for surface immobilization on porous substrates to enhance CO2 capture.8 For clarity, the term adsorption is used throughout this work to describe CO2 binding to molecular surface.
Effective CO2 capture requires optimizing adsorption energy and viscosity. A moderate adsorption energy (e.g., −5 to −10 kcal mol−1) is often ideal-it indicates sufficiently strong binding for efficient capture but also reversible desorption, enabling regeneration.39 Viscosity also influences capture performance, as lower viscosity improves mass transfer. Nitrogen-functionalized molecules, as explored in this study, offer an advantageous balance of moderate binding strength and favourable rheological properties, making them promising candidates for efficient CO2 adsorption.40,41
Methodology and molecular design strategies
All geometry optimizations were performed using density functional theory (DFT) at the M06-2X/6-311++G(d,p) level, as implemented in the Gaussian 16 suite of programs.42 The inclusion of diffuse functions in the basis set was essential for accurately describing the electronic structure of anionic complexes. All optimized geometries were confirmed to have only real vibrational frequencies, ensuring they represent true minima on the potential energy surface, while transition states exhibited a single imaginary frequency, validating their nature as first-order saddle points. The M06-2X functional is well-regarded for its accuracy in modelling intermolecular non-covalent interactions, crucial in understanding CO2-adsorbent complexes.43 To identify the most stable equilibrium structures, various initial guess geometries were considered, placing CO2 near electron-rich sites of the adsorbent. The lowest energy structures from these optimizations were used for further analysis. The adsorption energy (ΔEad) of CO2 was calculated using the supermolecule approach as follows (eqn (1)):| ΔEad = Ecomplex − (Eadsorbent + ECO2) + EBSSE | (1) |
In addition to adsorption energies (ΔEad), Gibbs free energies of adsorption (ΔGad) were computed to offer a more complete thermodynamic perspective. These calculations were performed in the gas phase at 293 K and 1 atm, consistent with ambient CO2 capture conditions.
To further elucidate electronic features relevant to CO2 binding, topological analysis of the molecular electrostatic potential (MESP), V(r), was carried out. The MESP as defined in eqn (2) was computed at the M06-2X/6-311++G(d,p) level for all optimized geometries using Gaussian 16. This analysis identifies electron-rich regions such as lone pairs and π-systems – critical for understanding interaction sites in the hydroxy-substituted N-heterocycles, their anionic counterparts, and the corresponding CO2 complexes.
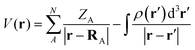 | (2) |
In eqn (2), ρ(r′) is the electron density, ZA is the charge on nucleus A located at RA, and r′ is a dummy integration variable.48,49 The MESP value at a (3, +3) critical point observed in electron rich regions such as lone pairs and π-bonds in topology analysis is called Vmin. The Vmin analysis is useful to understand the subtle variations and delocalization in electron density distribution due to the interactive behaviour of molecules.49–52
The molecular design strategy focuses on how introducing nitrogen and oxygen atoms affects the CO2 capture capacity of N-heterocycle. To explore this, we studied a series of hydroxy-substituted molecules – pyridine (py), naphthyridine (nt) and pyridonaphthyridine (pn) – featuring both two- and three-membered rings with systematically increasing numbers of carbon, nitrogen, and oxygen atoms. This enables evaluation of enhanced cooperative interactions due to the increased number of potential CO2 binding sites.
The choice of these molecular systems is grounded in prior research: hydroxy pyridines (py) have been examined for CO2 adsorption, notably by Luo et al. in the context of ionic liquids.28 While naphthyridines (nt) have been extensively studied for their pharmacological properties,53–57 their potential for CO2 capture remains largely unexplored. Similarly, some pyridonaphthyridine derivatives have shown therapeutic promise,58,59 but their abilities for CO2 adsorption have not been thoroughly investigated.
Additionally, the study extends to anionic forms of hydroxy-N-heterocycles (deprotonated form) to assess the impact of electron density on CO2 binding.60–76 We analyze how electron distribution evolves with the addition of aromatic rings and heteroatoms as the molecules transition from monoanionic to dianionic and trianionic states.
By combining the molecular design strategies with DFT calculations and MESP analysis,50–52,77,78 this study aims to provide a comprehensive understanding of the structure–activity relationships governing CO2 capture and conversion in these systems.
Results and discussion
N-Heterocycles and CO2 complexes
The chemical structure of hydroxy pyridine (py), the selected set of dihydroxy naphthyridines (nt to nt12), and the set of trihydroxy pyridonaphthyridines (pn1 to pn36) are presented in Fig. 1 (see ESI† for their IUPAC names, Fig. S1). Collectively, these compounds are referred to as N-heterocycles (Nhet). In all Nhet systems, the hydroxy groups are strategically positioned adjacent to nitrogen atoms to maximize the potential for synergistic interactions with CO2. This design rationale is based on calculated adsorption energies (ΔEad) for positional isomers of hydroxypyridine: 2-hydroxypyridine showed the most favourable interaction energy (−11.3 kcal mol−1), compared to −10.3 kcal mol−1 for 3-hydroxypyridine and −10.4 kcal mol−1 for 4-hydroxypyridine. The enhanced performance of 2-hydroxypyridine is attributed to cooperative binding between the adjacent nitrogen and hydroxyl functionalities, which strengthens both the N⋯CO2 and O–H⋯OCO interactions.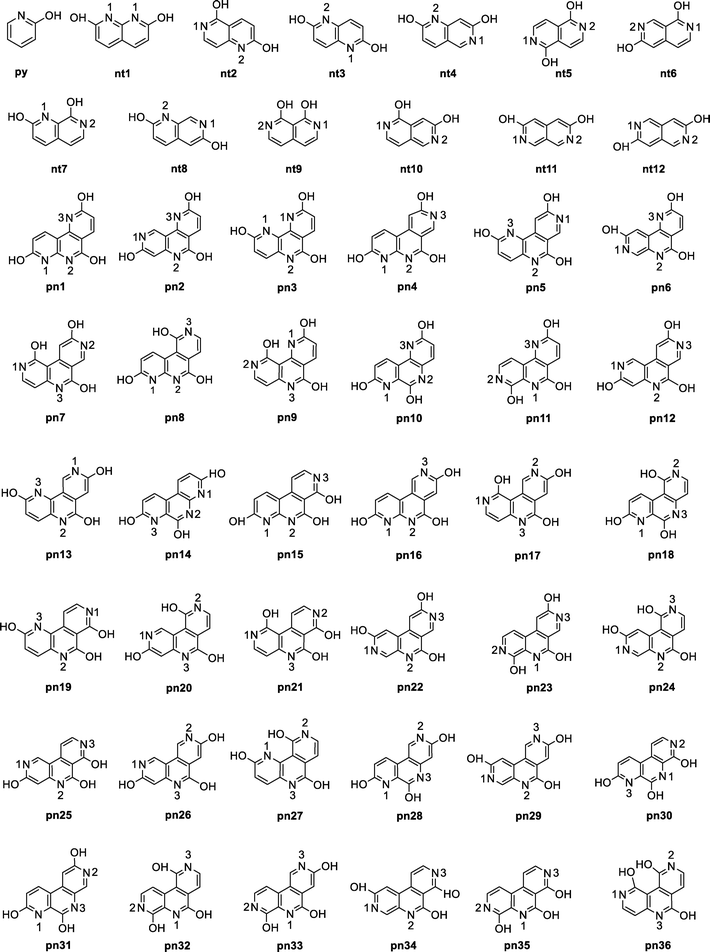 | ||
| Fig. 1 Chemical structures of hydroxy pyridine (py), 12 dihydroxy naphthyridines (nt), and 36 trihydroxy pyridonaphthyridines (pn). | ||
The minimum energy structures for each Nhet compound were identified, along with stable configurations of their CO2 complexes, denoted as Nhet⋯(CO2)n complexes, where n = 1 for py, n = 1 and 2 for nt and n = 1, 2, and 3 for pn systems. The relative energy of each structural isomers of nt and pn (ΔErel) was calculated using the equation (eqn (3)):
| ΔErel = Esample − Estandard | (3) |
| Molecules | V min | ΔErel | ΔEad1 | ΔEad2 | ΔGad1 | ΔGad2 | |
|---|---|---|---|---|---|---|---|
| N1 | N2 | ||||||
| nt1 | −53.1 | −53.1 | 0.0 | −10.5 | −10.7 | −3.7 | −2.7 |
| nt2 | −47.0 | −42.8 | 0.5 | −10.9 | −10.1 | −3.1 | −3.7 |
| nt3 | −43.3 | −43.2 | 1.5 | −10.5 | −10.0 | −3.4 | −3.5 |
| nt4 | −43.8 | −43.7 | 2.9 | −10.7 | −10.1 | −3.6 | −3.6 |
| nt5 | −44.0 | −43.9 | 3.5 | −10.8 | −10.3 | −3.9 | −3.4 |
| nt6 | −44.5 | −42.4 | 4.0 | −11.0 | −10.1 | −3.8 | −3.4 |
| nt7 | −51.4 | −47.6 | 5.1 | −10.2 | −10.1 | −3.9 | −3.6 |
| nt8 | −44.5 | −42.0 | 5.5 | −10.6 | −9.9 | −3.6 | −3.7 |
| nt9 | −45.2 | −44.9 | 6.1 | −10.8 | −10.3 | −3.4 | −3.4 |
| nt10 | −44.2 | −40.9 | 6.5 | −10.8 | −10.0 | −3.7 | −3.4 |
| nt11 | −41.2 | −41.0 | 6.5 | −10.6 | −10.2 | −3.7 | −3.6 |
| nt12 | −40.2 | −40.1 | 10.3 | −10.5 | −10.0 | −3.8 | −3.7 |
| Molecules | ΔErel | ΔEad1 | ΔEad2 | ΔEad3 | ΔGad1 | ΔGad2 | ΔGad3 |
|---|---|---|---|---|---|---|---|
| pn1 | 0.0 | −10.5 | −10.8 | −6.4 | −4.1 | −2.0 | −3.1 |
| pn2 | 2.5 | −10.8 | −10.2 | −8.1 | −3.1 | −3.8 | −3.0 |
| pn3 | 4.2 | −11.6 | −6.3 | −9.4 | −4.5 | −3.2 | −2.4 |
| pn4 | 4.7 | −10.5 | −10.8 | −9.0 | −1.7 | −3.8 | −4.7 |
| pn5 | 4.7 | −10.5 | −10.2 | −7.9 | −1.7 | −5.0 | −3.4 |
| pn6 | 5.0 | −10.6 | −10.0 | −7.8 | −3.0 | −1.9 | −2.1 |
| pn7 | 5.2 | −10.8 | −10.0 | −9.6 | −3.2 | −3.7 | −3.5 |
| pn8 | 5.2 | −10.5 | −10.7 | −9.1 | −3.3 | −3.7 | −3.6 |
| pn9 | 5.3 | −10.8 | −10.9 | −9.0 | −3.2 | −3.6 | −3.5 |
| pn10 | 5.5 | −10.2 | −9.2 | −7.8 | −2.0 | −4.9 | −3.5 |
| pn11 | 5.7 | −9.9 | −10.7 | −6.2 | −1.9 | −5.1 | −0.9 |
| pn12 | 6.2 | −10.8 | −10.1 | −9.4 | −3.5 | −3.7 | −3.6 |
| pn13 | 6.4 | −10.5 | −10.1 | −8.2 | −3.6 | −3.7 | −3.5 |
| pn14 | 6.6 | −10.5 | −10.6 | −8.6 | −3.4 | −3.7 | −3.6 |
| pn15 | 6.9 | −10.5 | −10.5 | −9.0 | −3.4 | −3.7 | −3.5 |
| pn16 | 7.4 | −10.5 | −10.7 | −9.0 | −3.7 | −3.6 | −1.8 |
| pn17 | 7.5 | −10.9 | −10.0 | −9.5 | −3.8 | −3.4 | −3.6 |
| pn18 | 7.8 | −9.7 | −10.8 | −9.2 | −3.7 | −3.7 | −3.5 |
| pn19 | 8.0 | −10.6 | −9.8 | −6.7 | −3.7 | −3.5 | −3.7 |
| pn20 | 8.0 | −10.7 | −10.1 | −9.4 | −3.7 | −3.6 | −3.6 |
| pn21 | 8.8 | −10.9 | −10.1 | −9.2 | −3.7 | −3.7 | −2.0 |
| pn22 | 8.9 | −10.6 | −9.9 | −9.4 | −3.8 | −3.7 | −3.3 |
| pn23 | 9.0 | −10.4 | −10.0 | −9.2 | −3.7 | −3.6 | −3.5 |
| pn24 | 9.2 | −10.6 | −9.8 | −9.5 | −3.7 | −3.5 | −3.4 |
| pn25 | 9.4 | −10.8 | −9.8 | −9.5 | −3.6 | −3.5 | −3.4 |
| pn26 | 9.6 | −10.8 | −10.0 | −9.4 | −3.8 | −3.5 | −3.5 |
| pn27 | 9.7 | −10.6 | −10.9 | −7.2 | −3.6 | −3.5 | −0.4 |
| pn28 | 9.7 | −9.7 | −10.3 | −9.8 | −3.5 | −3.9 | −1.7 |
| pn29 | 9.8 | −10.6 | −9.8 | −9.4 | −3.4 | −3.7 | −1.5 |
| pn30 | 10.0 | −10.3 | −10.0 | −8.9 | −3.5 | −3.5 | −3.1 |
| pn31 | 10.2 | −9.8 | −10.0 | −9.6 | −4.9 | −1.0 | −3.7 |
| pn32 | 10.3 | −9.9 | −10.6 | −9.4 | −3.9 | −2.5 | −2.1 |
| pn33 | 10.9 | −10.3 | −10.0 | −9.3 | −3.8 | −2.6 | −3.4 |
| pn34 | 12.4 | −10.5 | −9.6 | −9.4 | −4.2 | −2.1 | −3.5 |
| pn35 | 12.6 | −10.3 | −9.9 | −9.2 | −3.8 | −2.5 | −3.6 |
| pn36 | 15.5 | −10.8 | −10.1 | −9.5 | −4.0 | −2.2 | −3.8 |
The Nhet⋯CO2 complex is characterized by two key interactions: the nitrogen-to-carbon (N⋯CO2) interaction and the CO2 oxygen-to-hydroxy hydrogen (CO2⋯HO) interaction. The N⋯CO2 interaction enhances electron density at the oxygen atoms of CO2, thereby strengthening the CO2⋯HO interaction. This synergistic effect results in positive cooperativity, where an improvement in N⋯CO2 interaction leads to enhanced CO2⋯HO interaction. Consequently, the CO2 adsorption efficiency of Nhet can be attributed to the combined influence of these mutually reinforcing interactions.
MESP analysis of N-heterocycles and CO2 complexes
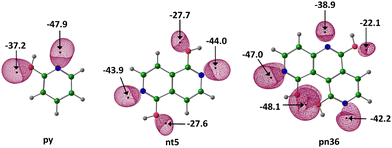
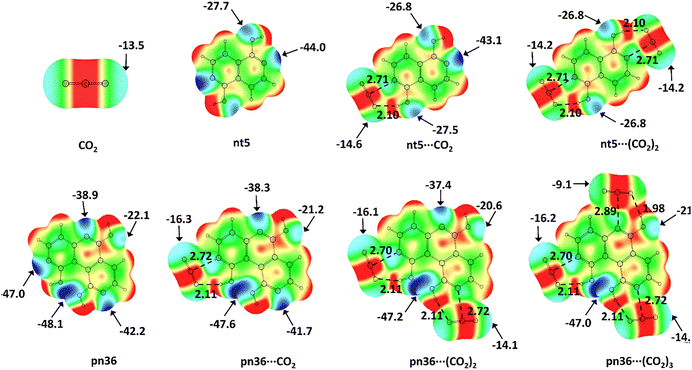
Molecular electrostatic potential (MESP) topology analysis uses the minimum electrostatic potential (Vmin) to map lone pair electron density around relevant atoms.52 As illustrated in Fig. 1, nitrogen centres in Nhet are identified based on their Vmin values and are numbered in decreasing order of magnitude, with N1 being the most electron rich, followed by N2 and N3. These nitrogen atoms serve as the primary interaction sites for CO2.The Vmin distribution of a representative set of Nhet systems is shown in Fig. 2. In py, the Vmin value for the nitrogen lone pair is −47.9 kcal mol−1. Among nt systems, nt1 exhibits the most negative Vmin (−53.1 kcal mol−1) while nt12, with a Vmin −40.1 kcal mol−1, comparatively less electron-dense. The Vmin values of N1 and N2 within the same nt molecule show minimal variation, with an average difference of approximately 10 kcal mol−1. In pn systems, Vmin varies significantly across N1, N2 and N3, ranging from −53.1 to −31.3 kcal mol−1, depending on the position of nitrogen centres (Table S2, ESI†). Based on the MESP analysis, nitrogen centres are identified as the most probable sites for CO2 interaction. The affinity of CO2 for different nitrogen centres follows a general trend, where a more negative Vmin corresponds to stronger interaction potential. MESP plotted on 0.005 a.u. isodensity surface provides a visual representation of electron reorganization during CO2 complexation with N-heterocycles (Fig. 3).79 When the carbon atom of CO2 accepts electron density from the nitrogen centre of Nhet, a counterbalancing electron density donation occurs from the oxygen of CO2 to the hydroxy hydrogen of Nhet. This process leads to delocalization of electron density around the lone pairs of nitrogen and oxygen atoms due to the N⋯CO2 and CO2⋯HO interactions. Because of the mutually compensating effect of these interactions, apart from the noncovalently bonded regions, the overall electron distribution in the Nhet⋯CO2 complex remains largely unchanged outside the noncovalently bonded regions. For instance, in pn systems, the oxygen atoms of hydroxy groups exhibit only minor changes in Vmin even with successive CO2 adsorption. Similarly, nitrogen atoms not directly involved in noncovalent bonding retain nearly the same Vmin values in Nhet⋯CO2 and Nhet⋯(CO2)2 complexes. This consistency suggests a steady interaction energy value for CO2 adsorption, allowing Nhet to effectively capture up to three CO2 molecules.
Transition state (TS) calculations
To further understand the CO2 capture process, transition state (TS) calculations were performed to evaluate the activation energies for the reaction pathways involving different CO2 binding configurations. For the py system, the transition state for the first CO2 binding (TS1) is located at −0.7 kcal mol−1, with the corresponding acid formation at −2.9 kcal mol−1. In the case of nt5, the activation energy for CO2 binding decreases as additional CO2 molecules are added, with values of −1.3 kcal mol−1 for the first binding, −5.0 kcal mol−1 for the diacid formation (TS2). The pn36 system exhibits a similar trend, with transition state energies of −2.2 kcal mol−1 for TS1 and −5.1 kcal mol−1 for TS2, with a significant drop in activation energy observed at −0.9 kcal mol−1 for the triacid formation (TS3). These calculations provide valuable insight into the energetic barriers for CO2 binding and highlight the influence of molecular structure on the activation energies and stability of the intermediates.CO2 adsorption and enol–keto transformation in N-heterocycles
The nitrogen-to-carbon interaction distance (dNC) serves as an initial indicator of interaction strength. Notably, all captured CO2 molecules bind to the nitrogen centre at distances ranging from 2.70 to 2.90 Å. The complexes are further stabilized by CO2⋯HO hydrogen bonding, with hydrogen bond distance (dOH) ranging from 1.98 to 2.12 Å (Table S3, ESI†). Representative examples of Nhet⋯(CO2)n complexes are illustrated in Fig. 4. The noncovalent interactions slightly reduce the OCO bond angle from 180°.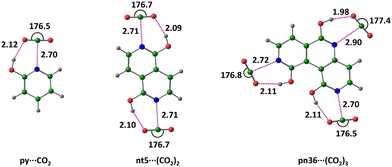 | ||
| Fig. 4 Optimized structures of Nhet⋯(CO2)n complexes at M06-2X/6-311++G(d,p) level of theory, with bond distances in Å and bond angle in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
The py⋯CO2 complex demonstrates an interaction energy (ΔEad) of −10.8 kcal mol−1, with a corresponding free energy of interaction (ΔGad) of −3.8 kcal mol−1, confirming its exergonic nature. For nt systems, ΔEad1 represents the reaction nt + CO2 → nt⋯CO2, corresponding to CO2 adsorption at the N1 site. Similarly, ΔEad2 refers to the energy released upon CO2 adsorption at the N2 site of the nt⋯CO2 complex. In pn systems, ΔEad1, ΔEad2 and ΔEad3 correspond to CO2 adsorption energies at N1, N2 and N3 atoms, respectively. The data presented in Tables 1 and 2 suggest that the combined effect of N⋯CO2 and CO2⋯HO interactions result in nearly equivalent CO2 adsorption strength across N1, N2 and N3 in all Nhet systems. The interaction energy for the first CO2 adsorption is approximately −10.5 kcal mol−1, for the second CO2 adsorption is −10.1 kcal mol−1, and for the third CO2 adsorption is −8.8 kcal mol−1. Moreover, each stage of the CO2 adsorption is exergonic, with average ΔGad1, ΔGad2 and ΔGad3 values of −3.5, −3.4 and −3.0 kcal mol−1, respectively (Tables 1 and 2). These thermodynamic data confirm that the CO2 adsorption capacity of naphthyridine is nearly double that of hydroxypyridine, while that of pyridonaphthyridine is nearly triple.
The CO2⋯HO hydrogen bond interaction in Nhet⋯CO2 can lead to a proton transfer, potentially converting the adsorbed CO2 into a carboxylate functionality. In other words, the CO2⋯HO–C interaction can transition to a CO2H⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C interaction, corresponding to an enol–keto transformation of the Nhet system. The schematic representation of CO2 complex formation and enol–keto transformation is illustrated in Fig. 5 for a representative pn5 system. This transformation simultaneously alters the N⋯CO2 noncovalent interaction into a covalent N–CO2H bond. However, the enol–keto transformation is expected to be energetically unfavourable, as it significantly disrupts the π-electron distribution within the ring, eliminating the alternating single and double bond pattern necessary for aromatic stabilization. In Fig. 5, the regions with reduced aromatic character are highlighted in red. When one hydroxy group is converted to the keto form, the nt moiety is denoted as nt′, while the notation nt′′ represents the conversion of both hydroxy groups to the keto form. Similarly, py′ refers to the keto form of hydroxypyridine, while pn′, pn′′, and pn′′′ represent the keto forms of pyridonaphthyridines.
C interaction, corresponding to an enol–keto transformation of the Nhet system. The schematic representation of CO2 complex formation and enol–keto transformation is illustrated in Fig. 5 for a representative pn5 system. This transformation simultaneously alters the N⋯CO2 noncovalent interaction into a covalent N–CO2H bond. However, the enol–keto transformation is expected to be energetically unfavourable, as it significantly disrupts the π-electron distribution within the ring, eliminating the alternating single and double bond pattern necessary for aromatic stabilization. In Fig. 5, the regions with reduced aromatic character are highlighted in red. When one hydroxy group is converted to the keto form, the nt moiety is denoted as nt′, while the notation nt′′ represents the conversion of both hydroxy groups to the keto form. Similarly, py′ refers to the keto form of hydroxypyridine, while pn′, pn′′, and pn′′′ represent the keto forms of pyridonaphthyridines.
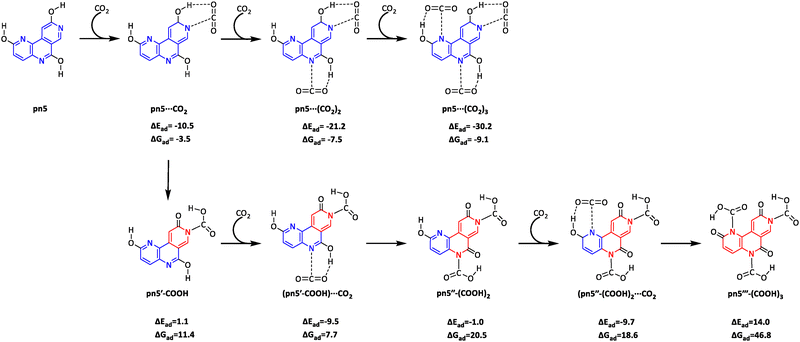 | ||
| Fig. 5 Schematic illustration of CO2 complex formation with pn5 and subsequent enol–keto transformations. A ring that cannot be represented by alternate single and double bonds is shown in red. | ||
Although transformation of py⋯CO2 to py′-COOH is exothermic by −2.9 kcal mol−1, it is endergonic by 7.8 kcal mol−1, indicating that py′-COOH formation is highly unlikely due to the loss of aromaticity in the ring structure. This trend is consistently observed across most nt and pn systems (Fig. S2, ESI†). In some cases, the formation of carboxylated products is even endothermic. As shown in Fig. 6, the CO2 adsorption is energetically more favourable than its conversion to carboxylates across all systems due to the partial loss of aromatic character in the keto form. However, certain nt′-COOH and pn′-COOH (the keto forms) exhibit relatively favourable energy values compared to the others. For instance, in nt systems, the formation of nt2′-COOH, nt5′-COOH, nt6′-COOH, nt9′-COOH, and nt10′-COOH is exothermic, while the rest display endothermic behavior. Similarly, among monocarboxylated pn derivatives, pn2′–COOH, pn7′–COOH, pn9′–COOH, pn12′–COOH, pn17′–COOH, pn19′–COOH, pn20′–COOH, pn21′–COOH, pn25′–COOH, pn26′–COOH and pn36′–COOH show better energy data than the rest of the monocarboxylic acids. Compared to pn′–COOH, the relative energy of pn′′–(COOH)2 is more negative only in two cases: pn21′′–(COOH)2 and pn36′′–(COOH)2. In contrast, all tricarboxylated pn′′′–(COOH)3 systems have significantly higher energy compared to the corresponding pn⋯(CO2)3 complexes. Thus, the energy data presented in Fig. 6 strongly support the efficient capture of CO2 by Nhet, while its conversion to carboxylates remains energetically demanding due to the loss of aromatic stabilization in the core structure.
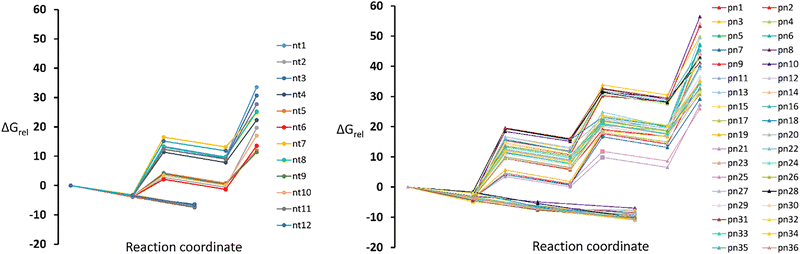
To provide a more thermodynamically meaningful comparison of the different CO2 adsorption processes, we calculated the relative Gibbs free energies (ΔGrel) for all systems studied. ΔGrel values offer insight into the spontaneity and favourability of each reaction under standard conditions (293 K, 1 atm), incorporating both enthalpic and entropic contributions. These values complement ΔErel and allow for a more accurate assessment of the adsorption–desorption equilibrium, which is particularly important for evaluating practical CO2 capture applications (Fig. S2, ESI†). By analyzing ΔGrel, we identify systems where CO2 binding remains favourable not only in terms of interaction strength but also in terms of thermodynamic feasibility, thereby guiding the selection of optimal candidates for efficient and reversible CO2 capture.
Interaction of anionic N-heterocycles with CO2
Luo et al. explored the application of pyridine-containing anion-functionalized ionic liquids (ILs) for CO2 capture.28 The ILs were synthesized via the neutralization of hydroxypyridine using an ethanol solution of trihexyl(tetradecyl)phosphonium hydroxide. Their study proposed a plausible mechanism for CO2 interaction, identifying the 2-pyridonate ion as the key active species. This ion engages in a two-site interaction with CO2, where both the oxygen and nitrogen atoms within the anion participate in coordinating and binding CO2. Building on this mechanism, this study delves into the interaction behavior of the 2-pyridonate ion (py−) with CO2. Additionally, as a representative case, the naphthyridonate system (nt5−) is examined to explore the CO2 adsorption properties of both its monoanionic (nt5−) and dianionic (nt52−) forms. Similarly, for the pyridonaphthyridine system, a representative case, pn36, is selected to investigate the complexation behaviour of its monoanionic (pn36−), dianionic (pn362−) and trianionic (pn363−) forms with CO2.Among the various hydroxy-substituted N-heterocycles examined, nt5 and pn36 emerged as the most promising candidates for CO2 capture based on their computed interaction energies. The nt5 system, representing the naphthyridine class, demonstrated the highest CO2 adsorption energy among the twelve derivatives studied, suggesting a strong and favourable interaction with CO2. Similarly, pn36, a trihydroxy-substituted pyridonaphthyridine, exhibited superior CO2 binding performance compared to its analogues. These results highlight the importance of both the number and position of hydroxy groups relative to nitrogen centres in facilitating cooperative binding effects. Consequently, nt5 and pn36 were selected for detailed analysis in their anionic forms to further explore the impact of charge and substitution patterns on CO2 adsorption efficiency.
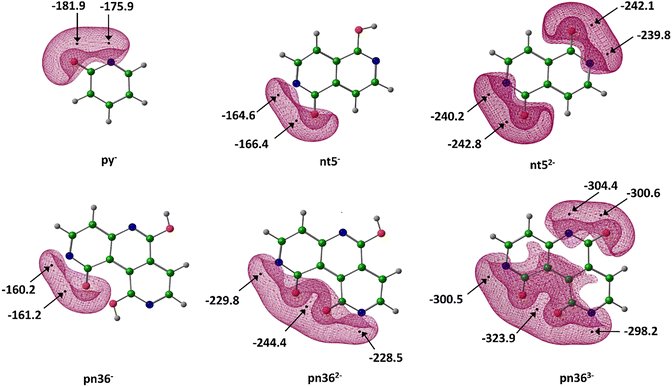
Fig. 7 illustrates the MESP characteristics of these anions. The MESP minimum (Vmin) in the vicinity of oxygen atom is consistently more negative than that near than the nitrogen centre. Relative to their neutral counterparts, the monoanions exhibit a significant increase in the magnitude of Vmin, specifically by 134.0.0, 122.5 and 113.1 kcal mol−1, respectively for py−, nt−, and pn−, respectively. Similarly, the dianions exhibit even more negative Vmin values than monoanions, with increases of 76.4 and 83.2 kcal mol−1 for nt2−and pn2−, respectively. The Vmin of pn3− is 79.5 kcal mol−1 more negative than that of pn2−. This data suggests that the electron-donating interactive behavior of these anionic systems with CO2 follows the order: trianion > dianion > monoanion » neutral.
The complexation of py− with CO2 is more stable when CO2 binds via the nitrogen centre rather than the oxygen centre, with a stability difference of 0.9 kcal mol−1. In both cases, CO2 forms a covalent bond, as indicated by N–CO2 and O–CO2 bond distances within the covalent range. The nitrogen-bound covalent complex (py–CO2−)N and the oxygen-bound covalent complex (py–CO2−)O (Fig. 8) were further analysed for interactions with a second CO2 molecule. In the scenario where one CO2 binds at nitrogen and the second at oxygen, the total interaction energy (ΔEad) is −26.9 kcal mol−1. This complex is denoted as (py⋯(CO2−)2)N&O wherein the dotted line indicates that CO2 molecules are connected noncovalently to N and O. In the case of (py–CO2−)O, the second CO2 interacts noncovalently with the oxygen of the already covalently bound CO2. This complex (py–CO2−)O⋯CO2. A similar structure, (py–CO2−)N⋯CO2 is also identified. Among the complexes the two-CO2 complexes, the most stable configuration is (py–CO2−)N⋯CO2 with an ΔEad of −31.4 kcal mol−1.
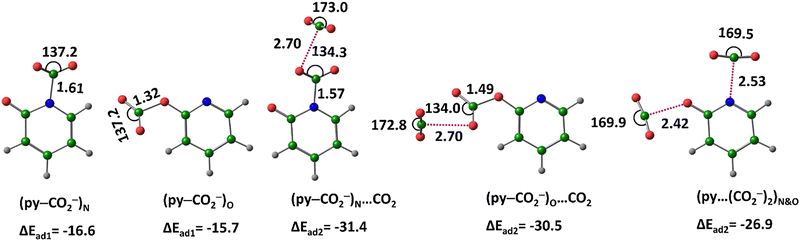 | ||
| Fig. 8 Optimized structures of the CO2–adsorbed by py−, at M06-2X/6-311++G(d,p) level of theory. Distances in Å and angles in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
For the nt5− anion, the nitrogen-bound (covalent) complex (nt5–CO2−)N exhibits an ΔEad of −13.6 kcal mol−1, which is 1.5 kcal mol−1 more stable than the oxygen-bound (noncovalent) complex (nt5−⋯CO2)O. Notably, an oxygen-bound covalent complex is not observed. When two CO2 molecules are adsorbed, one binds covalently to nitrogen while the other interacts noncovalently with oxygen, forming the complex (CO2⋯nt5–CO2−)N&O, with an ΔEad of −29.4 kcal mol−1. Another identified structure, (nt5–CO2−)N⋯CO2 describes the interaction of the second CO2 with the oxygen of the initially adsorbed CO2 molecule (Fig. 9).
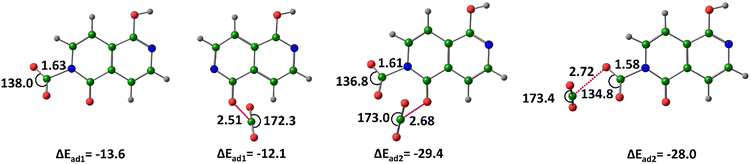 | ||
| Fig. 9 Optimized structures of the CO2–adsorbed by nt5−, at M06-2X/6-311++G(d,p) level of theory. Distances in Å and angles in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
For the nt52− dianion interacting with two CO2 molecules, among the various optimized structures (Fig. S3, ESI†), the most stable configuration is (nt5–(CO2−)2)N&N. Here, both CO2 molecules form covalent bonds with nitrogen, acquiring carboxylate character and delocalization of the excess negative charge. The ΔEad of this complex is −59.3 kcal mol−1, meaning the interaction energy per CO2 molecule adsorbed is −29.7 kcal mol−1-more than twice that of the nt5− anion and nearly three times that of neutral systems. Further CO2 adsorption occurs via an oxygen connection (noncovalent), forming (CO2⋯nt5–(CO2−)2)N&N&O, which stabilizes the system by an additional 17.7 kcal mol−1 (Fig. 10).
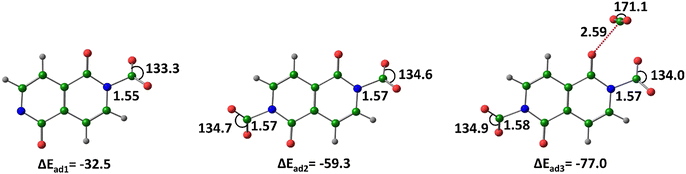 | ||
| Fig. 10 Optimized structures of the CO2-adsorbed by nt52−, at M06-2X/6-311++G(d,p) level of theory. Distances in Å and angles in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
For the pn36− anion, the oxygen-bound (noncovalent) complex (pn36⋯CO2−)O is slightly more stable than the nitrogen-bound (covalent) complex (pn36–CO2−)N (Fig. S4, ESI†). However, the most stable configuration with two CO2 molecules originates from (pn36–CO2−)N. The complex (pn36–CO2−)N⋯CO2, where a second CO2 interacts with the oxygen of the first adsorbed CO2, has an energy of −29.4 kcal mol−1-more than twice the interaction energy of (pn36–CO2−)N, indicating strong positive cooperativity.
For the pn362− dianion, the nitrogen-bound (covalent) CO2 complex (pn36−–CO2−)N is more stable than the oxygen-bound (covalent) complex (pn36−–CO2−)O by 8.8 kcal mol−1 (Fig. S5, ESI†). The most stable configuration for two pn362− dianion with two CO2 molecules is (pn36–(CO2−)2)N&N, with an ΔEad of −50.0 kcal mol−1, meaning that the ΔEad per CO2 molecule adsorbed is −25.0 kcal mol−1-approximately 12.0 kcal mol−1 higher than that of the monoanion. When a third CO2 molecule is adsorbed through an oxygen connection (noncovalent), forming (CO2⋯pn36–(CO2−)2)N&N&O, the interaction energy further improves by 17.6 kcal mol−1 (Fig. 11).
 | ||
| Fig. 11 Optimized structures of the CO2-adsorbed by pn36− and pn362−, at M06-2X/6-311++G(d,p) level of theory. Distances in Å and angles in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
For the pn363− trianion, the nitrogen-bound (covalent) CO2 complex (pn362−–CO2−)N, is the most stable among all configurations, with ΔEad = −46.1 kcal mol−1 (Fig. 12). Similarly, the nitrogen-bound (covalent) complex with two CO2 molecules, (pn36−–(CO2−)2)N&N, shows the highest stability with ΔEad = −85.8 kcal mol−1. Furthermore, when three CO2 molecules are adsorbed in a nitrogen-connected (covalent) manner, forming (pn36–(CO2−)3)N&N&N, the ΔEad reaches −115.1 kcal mol−1, indicating that interaction energy per CO2 molecule adsorbed is −38.4 kcal mol−1.
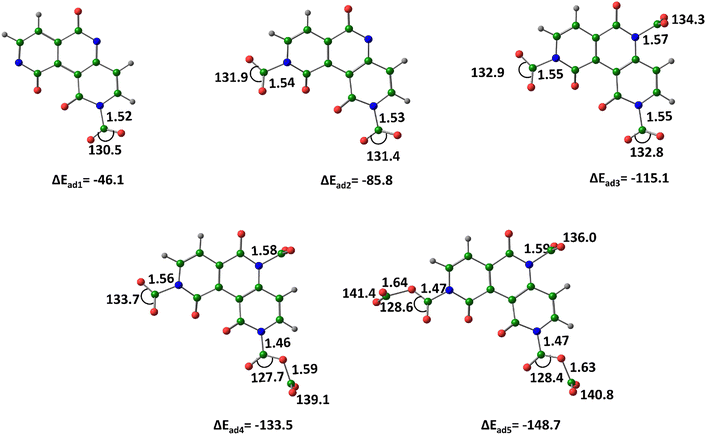 | ||
| Fig. 12 Optimized structures of the CO2–adsorbed by pn363−, at M06-2X/6-311++G(d,p) level of theory. Distances in Å and angles in degrees. (Color key: green-C, grey-H, blue-N, red-O). | ||
Across all anionic, dianionic, and trianionic species, the CO2 adsorption preferentially occurs at nitrogen rather than oxygen, except in one case (Tables S4 and S5, ESI†). Unlike neutral systems, where N to CO2 interactions are noncovalent, the anionic systems exhibit covalent N–CO2 bonding. For dianions, the interaction energy nearly doubles with the adsorption of two CO2 molecules, while for trianions, it almost triples with three CO2 molecules. In all cases, carboxylate formation facilitates charge delocalization, centralizing the anionic charge within carboxylate groups, which in turn promotes further CO2 interactions. As a result, multiple CO2 molecules are effectively adsorbed, demonstrating significantly enhanced CO2-binding affinity for anionic species compared to their neutral counterparts. With increasing anionic charge, a substantial increase in ΔEad is observed, with the trianionic system exhibiting the highest interaction energy. Notably, exergonic CO2 adsorption involving up to five CO2 molecules is reported here (Fig. 13 and Table 3). The exergonic nature of these systems is further confirmed by the calculated free energies (Table S6, ESI†).
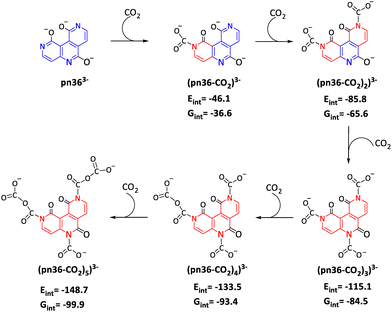 | ||
| Fig. 13 Schematic illustration of CO2 complex formation with pn36 trianion. A ring that cannot be represented by alternate single and double bonds is shown in red. | ||
| Molecule | ΔEad1 | ΔEad2 | ΔEad3 | ΔEad4 | ΔEad5 |
|---|---|---|---|---|---|
| py− | −16.6 | ||||
| nt5− | −13.6 | ||||
| nt52− | −32.5 | −59.3 | |||
| pn36− | −13.3 | ||||
| pn362− | −27.5 | −50.0 | |||
| pn363− | −46.1 | −85.8 | −115.1 | −133.5 | −148.7 |
To assess the practical relevance of CO2 capture, we computed the CO2 uptake in terms of weight percent (wt%) using the molar mass ratio of adsorbed CO2 to the to the total mass of the anion–CO2 complex (Table S9, ESI†). The calculated values reveal substantial uptake across all anionic systems studied. For instance, the anion of 2-hydroxypyridine captures up to 48% CO2 by mass with two CO2 molecules, while the dianion of dihydroxy naphthyridine reaches 45% with three. The trianion of trihydroxy pyridonaphthyridine exhibits the highest uptake, achieving nearly 49 wt% upon binding five CO2 molecules. These results underscore the strong potential of these N-heterocyclic systems as high-capacity CO2 adsorbents.
Influence of counter cations on CO2 adsorption
To evaluate the impact of counter cations on CO2 adsorption, the py, nt, and pn systems were studied in the presence of lithium chloride (LiCl), leading to the formation of Li+-incorporated complexes and HCl (Scheme 1). In these reactions, CO2 forms nitrogen-bound (covalent) complexes (Fig. 14), which are further stabilized by bidentate Li⋯O interactions involving both the oxygen of the adsorbent system and the oxygen of the adsorbed CO2. The energy of reactions (Erea1) given in Scheme 1 was calculated using the equation (eqn (4)):| Erea1 = ΣEproducts − ΣEreactants | (4) |
The Erea1 serves as a measure of the adsorption efficiency in the presence of Li+. These reactions are exothermic, with Erea1 values of −12.4, −29.3, and −37.5 kcal mol−1, indicating that, compared to neutral systems, the presence of Li+ significantly enhances covalent CO2 binding.
To further investigate the effect of a larger countercation on CO2 adsorption, tetramethylphosphonium hydroxide ((CH3)4P+(OH)−) was selected as a model for the bulky trihexyl(tetradecyl)phosphonium hydroxide used by Luo et al.28Table 4 presents the reaction energies for (CH3)4P+(OH)− with py, nt, and pn molecules both in the absence and presence of CO2. The reaction energy (Erea3) in the presence of CO2 (entries 7–12 in Table 4) is significantly more favourable than in its absence (Erea2; entries 1–6). In both cases, the Gibbs free energy of reaction (Grea2 and Grea3) confirms exergonic behaviour. The enhancement of Erea3 compared to Erea2 (by 40–80%) highlights the strong positive influence of the large counter cation on CO2 adsorption. The structural analysis of cation-incorporated complexes indicates covalent CO2 binding at the nitrogen centre (Fig. 15). The acquired anionic character of CO2 in these complexes leads to stabilizing electrostatic interactions with the surrounding cation. Notably, Erea3 improves in the order: complexes with three CO2 > complexes with two CO2 > complexes with one CO2, suggesting that CO2 adsorption efficiency follows the trend pn > nt > py.
| No. | Reaction | E rea2 (1–6) | G rea2 (1–6) |
|---|---|---|---|
| E rea3 (7–12) | G rea3 (7–12) | ||
| 1 | py + (CH3)4P+(OH)− → py−⋯+P(CH3)4 + H2O | −24.9 | −22.8 |
| 2 | nt5 + (CH3)4P+(OH)− → nt5−⋯+P(CH3)4 + H2O | −24.5 | −23.8 |
| 3 | pn36 + (CH3)4P+(OH)− → pn36−⋯+P(CH3)4 + H2O | −24.3 | −23.5 |
| 4 | nt5 + 2(CH3)4P+(OH)− → nt52−⋯+(P(CH3)4)2 + 2H2O | −42.8 | −39.2 |
| 5 | pn36 + 2(CH3)4P+(OH)− → pn362−⋯+(P(CH3)4)2 + 2H2O | −56.2 | −52.7 |
| 6 | pn36 + 3(CH3)4P+(OH)− → pn363−⋯+(P(CH3)4)3 + 3H2O | −72.7 | −69.9 |
| 7 | py + (CH3)4P+(OH)− + CO2 → py–(CO2)−⋯+P(CH3)4 + H2O | −37.1 | −24.6 |
| 8 | nt5 + (CH3)4P+(OH)− + CO2 → nt5–(CO2)−⋯+P(CH3)4 + H2O | −41.6 | −29.6 |
| 9 | pn36 + (CH3)4P+(OH)− + CO2 → pn36–(CO2)−⋯+P(CH3)4 + H2O | −41.6 | −29.3 |
| 10 | nt5 + 2(CH3)4P+(OH)− + 2CO2 → (nt5–(CO2)2)2−⋯+(P(CH3)4)2 + 2H2O | −80.7 | −56.7 |
| 11 | pn36 + 2(CH3)4P+(OH)− + 2CO2 → (pn36–(CO2)2)2−⋯+(P(CH3)4)2 + 2H2O | −78.9 | −55.0 |
| 12 | pn36 + 3(CH3)4P+(OH)− + 3CO2 → (pn36–(CO2)3)3−⋯+(P(CH3)4)3 + 3H2O | −116.9 | −79.8 |
 | ||
| Fig. 15 CO2 complexes of anionic N-heterocycles in the presence of trimethyl phosphonium cation. Distances in Å. (Color key: green-C, grey-H, blue-N, red-O, magenta-P). | ||
Solvation effect and Gibbs free energy changes
For neutral and anionic Nhet species such as hydroxypyridine (py), dihydroxy naphthyridines (nt), and trihydroxy pyridonaphthyridines (pn), solvation generally led to a modest reduction in the CO2 adsorption energies (ΔEad). This reflects the stabilizing influence of the polar solvent on individual charged and polar components (Table S10, ESI†). For example, the strength of CO2 adsorption in py weakened with ΔEad shifting from −10.8 kcal mol−1 in the gas phase to −7.6 kcal mol−1 in MEA. Similar reductions were observed across nt and pn systems (e.g., ΔEad for nt5 changed from −10.3 to −7.0 kcal mol−1). As a result, the Gibbs free energy of adsorption (ΔGad) in the solvent phase hovered around thermoneutrality, ranging from −1.6 to +3.2 kcal mol−1 for all Nhet⋯(CO2)n complexes (Table S13, ESI†). For anionic species, ΔGad values were more favourable, falling between −8.7 and 0.1 kcal mol−1, while dianionic systems exhibited ΔGad values in the range of −23.4 to −1.8 kcal mol−1 (Tables S14 and S15, ESI†). Notably, trianions showed the most exergonic adsorption, with ΔGad values between −26.3 and −7.8 kcal mol−1 (Table S16, ESI†).In comparison to the gas phase, a substantial reduction (71–81%) of the reaction energy (Erea1) is observed in the solvent phase for the reactions described in Scheme 1, which involve the formation of CO2 complexes of Nhet in the presence of Li+. A similar trend is noted for the formation of ion pairs between Nhet and the bulky cation (CH3)4P+(OH)− with a marked reduction in exothermicity (Erea2) in solution—by 52% for py, and more modest reductions for nt (16%) and pn (6%) systems (entries 1–6, Table 4). These observations are consistent with the strong solvation of the small, highly charged Li+ ion, which stabilizes the free cation more than the associated complex, thereby lowering the net driving force for complexation. In contrast, the bulky cation (CH3)4P+(OH)− is less strongly solvated due to its diffuse charge distribution, resulting in a smaller energetic penalty upon complex formation. The nt and pn systems may further resist tight ion pairing due to steric or electronic factors, contributing to the relatively lower impact of solvation.
Interestingly, a moderate enhancement (6–9%) in exothermic character (Erea3) is observed in the solvent phase for the formation of CO2 complexes from pre-formed Nhet⋯(CH3)4P+(OH)− ion pairs (entries 7–12, Table 4). This likely arises from solvent-induced weakening of the cation–anion interaction, which renders the anion more accessible for CO2 binding, thereby enhancing the overall adsorption energy in the solvated environment. The corresponding Gibbs free energies of reaction Grea3 in solution range from −26.8 kcal mol−1 for the monoanionic complex py–(CO2)−⋯+P(CH3)4 to −81.2 kcal mol−1 for the trianionic complex (pn–(CO2)3)3−⋯+(P(CH3)4)3.
The strongly negative ΔGad values observed in solution for many dianionic and trianionic systems along with the highly exergonic character of Grea3 suggest that these Nhet-based framework systems have the potential to bind more CO2 molecules than currently explored. These findings highlight the promising CO2 uptake capacity of such systems and underscore the critical role of solvation in modulating both the thermodynamics and the cooperative effects involved in CO2 capture. They emphasize that solvent-specific interactions must be carefully considered in the rational design of efficient and regenerable CO2 adsorbents for solution-phase applications.
Conclusions
This comprehensive DFT study highlights the exceptional CO2 capture capabilities of hydroxy-substituted aromatic N-heterocycles, revealing their potential as powerful adsorbents for mitigating greenhouse gas emissions. Through density functional theory (DFT) calculations, we have demonstrated that strategic incorporation of nitrogen and hydroxy groups within the molecular structures of pyridines, naphthyridines, and pyridonaphthyridines fosters cooperative binding mechanisms, leading to enhanced CO2 adsorption. The findings reveal that the adsorption CO2 capacity increases with the number of nitrogen and hydroxy centres.A key breakthrough in this study is the significantly enhanced CO2 adsorption observed in the anionic forms of these N-heterocycles. Compared to their neutral counterparts, anionic species exhibit significantly stronger CO2 interactions, with the formation of covalent bonds between nitrogen and CO2. As the anionic charge increases from monoanion to dianion and trianion, the CO2-binding affinity improves dramatically due to charge delocalization, resulting in more exergonic adsorption energies. This trend suggests that designing anionic or negatively charged frameworks could be an effective strategy for developing highly efficient CO2 capture materials. Importantly, solvation effects modeled using monoethanolamine (MEA) show that while polar solvents modestly reduce adsorption energies in neutral and anionic systems, they can enhance CO2 binding in ion-paired complexes by disrupting ion-pairing and increasing CO2 accessibility.
Energetic analysis further supports these findings, showing that CO2 adsorption is generally exergonic under post-combustion conditions (293 K, 1 atm). Notably, the interaction energy per CO2 molecule in trianionic species is nearly three times higher than in neutral systems, underscoring the role of charge modulation in enhancing CO2 capture efficiency. The study also establishes that enol–keto transformations, which could potentially convert the adsorbed CO2 into carboxylate groups, are energetically unfavourable. This is attributed to the loss of aromaticity in the core structure of N-heterocycles, reinforcing the importance of preserving aromatic stability for maintaining efficient CO2 adsorption.
Additionally, the influence of counter cations, such as lithium and tetramethylphosphonium, in stabilizing CO2 adsorption has been explored. The presence of these cations further strengthens CO2 binding by reducing interaction distances and increasing exergonicity, thereby improving the overall adsorption performance. These results suggest that incorporating appropriate cations into CO2 capture materials could enhance their efficiency, offering another avenue for material optimization.
Overall, this study provides valuable molecular-level insights into the structure–activity relationships governing CO2 capture in hydroxy-substituted N-heterocycles. The findings suggest that these molecules, particularly in their anionic and cation-stabilized forms, hold significant promise for developing next-generation CO2 capture technologies. Future research should focus on experimentally validating these computational predictions and exploring the practical implementation of these materials under real-world conditions. Furthermore, extending this approach to solid-state frameworks, such as functionalized porous materials or ionic liquids, could offer additional pathways for enhancing CO2 capture efficiency and selectivity.
Author contributions
CHS conceptualized the project, supervised the work, and contributed to the writing of the manuscript. PKA conducted research, curated the data, and contributed to the writing of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the ESI.†Acknowledgements
PKA is thankful to DST, Inspire, for a Senior Research Fellowship. CHS acknowledges CSIR-NIIST, Trivandrum for his deputation assignment as Director to SRIBS, Kottayam.References
- E. J. Beckman, Ind. Eng. Chem. Res., 2003, 42, 1598–1602 CrossRef.
- D. J. Darensbourg, Inorg. Chem., 2010, 49, 10765–10780 CrossRef PubMed.
- M. Mikkelsen, M. Jørgensen and F. C. Krebs, Energy Environ. Sci., 2010, 3, 43–81 RSC.
- K. S. Lackner, Science, 2003, 300, 1677–1678 CrossRef.
- N. MacDowell, N. Florin, A. Buchard, J. Hallett, A. Galindo, G. Jackson, C. S. Adjiman, C. K. Williams, N. Shah and P. Fennell, Energy Environ. Sci., 2010, 3, 1645–1669 RSC.
- P. Vadivelu and C. H. Suresh, Dalton Trans., 2015, 44, 16847–16853 RSC.
- M. J. Ajitha and C. H. Suresh, Tetrahedron Lett., 2011, 52, 5403–5406 CrossRef.
- S. Choi, J. H. Drese and C. W. Jones, ChemSusChem, 2009, 2, 796–854 CrossRef.
- E. S. Rubin, H. Mantripragada, A. Marks, P. Versteeg and J. Kitchin, Prog. Energy Combust. Sci., 2012, 38, 630–671 CrossRef CAS.
- M. E. Boot-Handford, J. C. Abanades, E. J. Anthony, M. J. Blunt, S. Brandani, N. Mac Dowell, J. R. Fernández, M.-C. Ferrari, R. Gross and J. P. Hallett, Energy Environ. Sci., 2014, 7, 130–189 RSC.
- M. Younas, M. Sohail, L. Leong, M. Bashir and S. Sumathi, Int. J. Environ. Sci. Technol., 2016, 13, 1839–1860 CrossRef.
- M. Plaza, C. Pevida, A. Arenillas, F. Rubiera and J. Pis, Fuel, 2007, 86, 2204–2212 CrossRef.
- J. Hack, N. Maeda and D. M. Meier, ACS Omega, 2022, 7, 39520–39530 CrossRef PubMed.
- M. J. Ajitha and C. H. Suresh, J. Org. Chem., 2012, 77, 1087–1094 CrossRef.
- S. Anila and C. H. Suresh, New J. Chem., 2023, 47, 3047–3054 RSC.
- M. J. Muldoon, S. N. Aki, J. L. Anderson, J. K. Dixon and J. F. Brennecke, J. Phys. Chem. B, 2007, 111, 9001–9009 CrossRef PubMed.
- B. E. Gurkan, J. C. de la Fuente, E. M. Mindrup, L. E. Ficke, B. F. Goodrich, E. A. Price, W. F. Schneider and J. F. Brennecke, J. Am. Chem. Soc., 2010, 132, 2116–2117 CrossRef.
- Z. Xue, Z. Zhang, J. Han, Y. Chen and T. Mu, Int. J. Greenhouse Gas Control, 2011, 5, 628–633 CrossRef.
- E. D. Bates, R. D. Mayton, I. Ntai and J. H. Davis, J. Am. Chem. Soc., 2002, 124, 926–927 CrossRef PubMed.
- I. Niedermaier, M. Bahlmann, C. Papp, C. Kolbeck, W. Wei, S. Krick Calderón, M. Grabau, P. S. Schulz, P. Wasserscheid and H.-P. Steinrück, J. Am. Chem. Soc., 2014, 136, 436–441 CrossRef PubMed.
- L. G. Sánchez, G. Meindersma and A. De Haan, J. Chem. Eng., 2011, 166, 1104–1115 CrossRef.
- P. Sharma, S. Do Park, K. T. Park, S. C. Nam, S. K. Jeong, Y. I. Yoon and I. H. Baek, J. Chem. Eng., 2012, 193, 267–275 CrossRef.
- J.-G. Lu, C.-T. Lu, Y. Chen, L. Gao, X. Zhao, H. Zhang and Z.-W. Xu, Appl. Energy, 2014, 115, 573–581 CrossRef.
- Y. S. Sistla and A. Khanna, J. Chem. Eng., 2015, 273, 268–276 CrossRef.
- B. Lv, G. Jing, Y. Qian and Z. Zhou, J. Chem. Eng., 2016, 289, 212–218 CrossRef.
- X. Y. Luo, X. Y. Lv, G. L. Shi, Q. Meng, H. R. Li and C. M. Wang, AIChE J., 2019, 65, 230–238 CrossRef.
- S. Anila and C. H. Suresh, Phys. Chem. Chem. Phys., 2022, 24, 22144–22153 RSC.
- X. Luo, Y. Guo, F. Ding, H. Zhao, G. Cui, H. Li and C. Wang, Angew. Chem., Int. Ed., 2014, 126, 7173–7177 CrossRef.
- C. Wang, H. Luo, D. E. Jiang, H. Li and S. Dai, Angew. Chem., Int. Ed., 2010, 122, 6114–6117 CrossRef.
- X. Y. Luo, F. Ding, W. J. Lin, Y. Q. Qi, H. R. Li and C. M. Wang, J. Phys. Chem. Lett., 2014, 5, 381–386 CrossRef.
- C. Wang, X. Luo, H. Luo, D. e Jiang, H. Li and S. Dai, Angew. Chem., Int. Ed., 2011, 21, 5020–5024 CrossRef.
- C. Wang, S. M. Mahurin, H. Luo, G. A. Baker, H. Li and S. Dai, Green Chem., 2010, 12, 870–874 RSC.
- C. Wang, H. Luo, X. Luo, H. Li and S. Dai, Green Chem., 2010, 12, 2019–2023 RSC.
- Y. Huang, G. Cui, Y. Zhao, H. Wang, Z. Li, S. Dai and J. Wang, Angew. Chem., Int. Ed., 2017, 56, 13293–13297 CrossRef.
- X. Luo and C. Wang, Curr. Opin. Green Sustainable Chem., 2017, 3, 33–38 CrossRef.
- S. Anila and C. H. Suresh, Phys. Chem. Chem. Phys., 2021, 23, 13662–13671 RSC.
- S. Anila and C. H. Suresh, Phys. Chem. Chem. Phys., 2019, 21, 23143–23153 RSC.
- S. Anila, C. H. Suresh and H. F. Schaefer III, J. Phys. Chem. A, 2022, 126, 4952–4961 CrossRef.
- J. Wang, L. Huang, R. Yang, Z. Zhang, J. Wu, Y. Gao, Q. Wang, D. O’Hare and Z. Zhong, J. Energy Environ. Sci., 2014, 7, 3478–3518 RSC.
- B. Gurkan, B. Goodrich, E. Mindrup, L. Ficke, M. Massel, S. Seo, T. Senftle, H. Wu, M. Glaser and J. Shah, J. Phys. Chem. Lett., 2010, 1, 3494–3499 CrossRef.
- T. Su, T. Lyu, S. Wang, X. Tian, Z. Chen, W. Chu, M. Xie, S. Wang and Y. Zhuo, Energy Fuels, 2025, 39, 1216–1225 CrossRef.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. GScalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision A. 03, Gaussian, Wallingford, CT, 2016 Search PubMed.
- Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed.
- S. F. Boys and F. Bernardi, Mol. Phys., 1970, 19, 553–566 CrossRef CAS.
- C. Reichardt, Org. Process Res. Dev., 2007, 11, 105–113 CrossRef CAS.
- A. J. Reynolds, T. V. Verheyen, S. B. Adeloju, E. Meuleman and P. Feron, Environ. Sci. Technol., 2012, 46, 3643–3654 CrossRef CAS PubMed.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378–6396 CrossRef CAS.
- P. Politzer and D. G. Truhlar, Chemical applications of atomic and molecular electrostatic potentials: reactivity, structure, scattering, and energetics of organic, inorganic, and biological systems, New York, 1981 Search PubMed.
- S. R. Gadre and R. N. Shirsat, Electrostatics of atoms and molecules, Hyderabad, 2000 Search PubMed.
- N. Mohan and C. H. Suresh, J. Phys. Chem. A, 2014, 118, 1697–1705 CrossRef CAS PubMed.
- P. V. Bijina and C. H. Suresh, J. Chem. Sci., 2016, 128, 1677–1686 CrossRef CAS.
- C. H. Suresh and S. Anila, Acc. Chem. Res., 2023, 56, 1884–1895 CrossRef CAS PubMed.
- G. R. Newkome, S. J. Garbis, V. K. Majestic, F. R. Fronczek and G. Chiari, J. Org. Chem., 1981, 46, 833–839 CrossRef CAS.
- A. M. Thompson, A. M. Delaney, J. M. Hamby, M. C. Schroeder, T. A. Spoon, S. M. Crean, H. H. Showalter and W. A. Denny, J. Med. Chem., 2005, 48, 4628–4653 CrossRef CAS PubMed.
- A. M. Thompson, G. W. Rewcastle, S. L. Boushelle, B. G. Hartl, A. J. Kraker, G. H. Lu, B. L. Batley, R. L. Panek, H. H. Showalter and W. A. Denny, J. Med. Chem., 2000, 43, 3134–3147 CrossRef CAS.
- T. K. Dawson, P. Dziedzic, M. J. Robertson, J. A. Cisneros, S. G. Krimmer, A. S. Newton, J. Tirado-Rives and W. L. Jorgensen, ACS Med. Chem. Lett., 2017, 8, 1287–1291 CrossRef CAS PubMed.
- H. Natsugari, Y. Ikeura, Y. Kiyota, Y. Ishichi, T. Ishimaru, O. Saga, H. Shirafuji, T. Tanaka and I. Kamo, J. Med. Chem., 1995, 38, 3106–3120 CrossRef CAS.
- T. Cailly, F. Fabis, R. Legay, H. Oulyadi and S. Rault, Tetrahedron, 2007, 63, 71–76 CrossRef CAS.
- Y. Hamada, M. Sato and I. Takeuchi, J. Pharm. Soc. Jpn., 1975, 95, 1492–1497 CrossRef CAS.
- H.-J. Fan and C.-W. Liu, Chem. Phys. Lett., 1999, 300, 351–358 CrossRef CAS.
- B. Bhargava and S. Balasubramanian, Chem. Phys. Lett., 2007, 444, 242–246 CrossRef CAS.
- B. Bhargava, M. Saharay and S. Balasubramanian, Bull. Mater. Sci., 2008, 31, 327–334 CrossRef CAS.
- K. M. Gupta and J. Jiang, J. Phys. Chem. C, 2014, 118, 3110–3118 CrossRef CAS.
- J. X. Mao, J. A. Steckel, F. Yan, N. Dhumal, H. Kim and K. Damodaran, Phys. Chem. Chem. Phys., 2016, 18, 1911–1917 RSC.
- A. R. Shaikh, H. Karkhanechi, E. Kamio, T. Yoshioka and H. Matsuyama, J. Phys. Chem. C, 2016, 120, 27734–27745 CrossRef CAS.
- X. Zhang, X. Zhang, H. Dong, Z. Zhao, S. Zhang and Y. Huang, Energy Environ. Sci., 2012, 5, 6668–6681 RSC.
- A. P. Hallenbeck and J. R. Kitchin, Eng. Chem. Res., 2013, 52, 10788–10794 CrossRef CAS.
- J. Wang, L. Huang, R. Yang, Z. Zhang, J. Wu, Y. Gao, Q. Wang, D. O'Hare and Z. Zhong, Energy Environ. Sci., 2014, 7, 3478–3518 RSC.
- A. Sanna, M. Uibu, G. Caramanna, R. Kuusik and M. Maroto-Valer, Chem. Soc. Rev., 2014, 43, 8049–8080 RSC.
- A. Goeppert, M. Czaun, J.-P. Jones, G. S. Prakash and G. A. Olah, Chem. Soc. Rev., 2014, 43, 7995–8048 RSC.
- N. Hollingsworth, S. R. Taylor, M. T. Galante, J. Jacquemin, C. Longo, K. B. Holt, N. H. De Leeuw and C. Hardacre, Angew. Chem., Int. Ed., 2015, 127, 14370–14374 CrossRef.
- D. Chen, X. Zhang and A. F. Lee, J. Mater. Chem. A, 2015, 3, 14487–14516 RSC.
- M. Marszewski, S. Cao, J. Yu and M. Jaroniec, Mater. Horiz., 2015, 2, 261–278 RSC.
- G. A. Ozin, Adv. Mater., 2015, 27, 1957–1963 CrossRef CAS PubMed.
- J. Kothandaraman, A. Goeppert, M. Czaun, G. A. Olah and G. S. Prakash, J. Am. Chem. Soc., 2016, 138, 778–781 CrossRef CAS PubMed.
- I. H. Arellano, S. H. Madani, J. Huang and P. Pendleton, J. Chem. Eng., 2016, 283, 692–702 CrossRef CAS.
- S. R. Gadre, C. H. Suresh and N. Mohan, Molecules, 2021, 26, 3289 CrossRef CAS.
- C. H. Suresh, G. S. Remya and P. K. Anjalikrishna, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1601 CAS.
- P. V. Bijina and C. H. Suresh, J. Phys. Chem. A, 2020, 124, 2231–2241 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: SCF energies, zero-point correction, thermal correction to Gibbs, free energies and BSSE (Table S1), relative interaction energies of all complexes (Table S7), Rrelative free energies of all complexes (Table S8), Ssolvation energies of anion (Table S11), Ssolvation energies of anions in presence of counter cation (Table S12) and Cartesian coordinates of all the systems (Table S17). See DOI: https://doi.org/10.1039/d5cp01075f |
| This journal is © the Owner Societies 2025 |