Touch initiated on-demand adhesion on rough surfaces†
Zhekun
Shi
 ac,
Di
Tan
d,
Kangjian
Xiao
a,
Xiaolong
Zhang
a,
Bo
Zhu
ab,
Zhen
Lin
a,
Quan
Liu
c,
Daobing
Chen
ab,
Qinghua
Zhang
ac,
Di
Tan
d,
Kangjian
Xiao
a,
Xiaolong
Zhang
a,
Bo
Zhu
ab,
Zhen
Lin
a,
Quan
Liu
c,
Daobing
Chen
ab,
Qinghua
Zhang
 c and
Longjian
Xue
c and
Longjian
Xue
 *ab
*ab
aSchool of Power and Mechanical Engineering, the Institute of Technological Science, Wuhan University, South Donghu Road 8, 430072, Wuhan, China. E-mail: xuelongjian@whu.edu.cn
bHubei Key Laboratory of Electronic Manufacturing and Packaging Integration (Wuhan University), Wuhan University, Wuhan, Hubei 430072, China
cInstitute of Zhejiang University-Quzhou, Zhejiang Provincial Innovation Center of Advanced Chemicals Technology, Quzhou 324000, China
dInstitute of Textiles and Clothing, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong, China
First published on 20th June 2024
Abstract
Reversible adhesion with on-demand attachment and detachment is used by many animals for their locomotion. However, achieving robust and switchable adhesion on rough surfaces in artificial adhesives remains a significant challenge. Here, we present a snail mucus-inspired touch-initiate adhesive (TIA), showing robust adhesions on various surfaces. TIA is a polymeric hydrogel photo-cured with the presence of supersaturated sodium acetate (NaAc) in the precursor solution. TIA is soft and flexible at room temperature, allowing it to form conformal contact with objects with various surfaces. The contact with the target surface immediately initiates the crystallization of TIA, increasing the elastic modulus of TIA by an order of magnitude. The increased modulus and the interlocking with the target surfaces thus results in an adhesion strength up to 465.56 ± 84.05 kPa. TIA can be easily detached from the surface by heating to a temperature above 58 °C, showing an adhesion strength of 12.71 ± 2.73 kPa. The detached TIA, even cooled down to and kept at room temperature, is readily used for the subsequent adhesion. The study here not only provides a highly adhesive material for on-demand attachment to various surfaces, but also proposes a new design strategy to compose smart materials.
New conceptsIn this work, the touch-triggered on-demand adhesive has been demonstrated to achieve reversible adhesion on rough surfaces via responsive phase transition of the hydrogel. Here, we design a touch initiated adhesive (TIA) which is composed of polymeric hydrogel and saturated sodium acetate (NaAc) solution. At room temperature, TIA maintains an amorphous and soft state, facilitating the conformal adaption to rough surfaces. The contact with the target surface initiates the crystallization of NaAc, facilitated by a reduction in the energy barrier attributed to the transition from homogeneous to heterogeneous nucleation. The crystallized TIA increases the modulus by an order of magnitude and interlocks with the target surfaces, achieving an adhesion up to 465.56 ± 84.05 kPa. Upon heating, TIA returns to the soft state, allowing an easy detachment. The separated TIA remains amorphous after cooling to room temperature and can be utilized for the next touch-initiated adhesion. The work here establishes a novel strategy for developing smart adhesives. |
1. Introduction
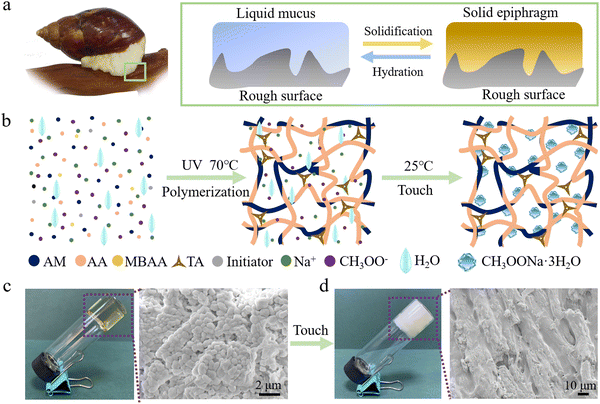
Learning from nature, reversible adhesions have been realized by constructing various micro- & nano-structures by using a limited range of materials.1–7 For example, gecko-inspired micro- & nano-pillar arrays,8–13 octopus-inspired suckers,14–16 fish-inspired adhesive disks,17,18 tree-frog-inspired polygonal patterns,19–23 and insect-inspired micropillar arrays24,25 achieved impressive adhesion performances. Meanwhile, dopamine chemistry, which is inspired by mussels, has been employed to modify surfaces with catecholamine functional groups, greatly enhancing the adhesion strength and environmental adaption.26–29 However, the adhesion on rough surfaces remains highly challenging.Snails can adhere to rough surfaces, such as rocks or tree barks, thanks to the mucus they secrete. When the mucus is in a flowing state, it can form adaptive contacts with the rough surface. Once it solidifies, the mucus transforms into a hardened epiphragm that interlocks with the rough surface, resulting in a strong adhesion (Fig. 1a).30 Inspired by snails, modulus-tunable materials have been developed to achieve strong and reversible adhesions on various surfaces. The modulus switching of materials is realized by the application of stimuli, like light, electricity, and heat.31–35 For instance, Sitti et al.31 demonstrated strong adhesion on both smooth and rough surfaces by utilizing the solid–liquid phase transition of gallium. In this approach, gallium contacts the target surface in its liquid state (at high temperature) and detaches in its solid state (at low temperature), achieving a high switching ratio of up to 178 on rough surfaces. Tan et al.33 achieved an adhesion strength of up to 278 kPa on surfaces with roughness ranging from 0.24 to 8.22 μm, and a rapid (within 10 s) and reversible adhesion (with a switching ratio of 29) by utilizing the transition between the rubbery and glassy states of polyurethane. Wang et al.34 employed the crystallinity of an ionic gel, which adheres to the target surface during crystal melting and detaches upon cooling, achieving reversible adhesion with a strength up to 9.82 MPa. In these cases, the adhesives need to be maintained in a liquid or soft state with external energy supply during the contact formation. Once the energy supply is removed, the material hardens that effective contact and strong adhesion cannot be realized. Therefore, to realize energy saving, on-demand reversible adhesion on rough surfaces remains difficult.
Here, drawing inspiration from the adhesive capabilities of snails (Fig. 1a), we successfully developed a touch-initiated adhesive (TIA) that exhibits remarkable on-demand adhesion on a diverse range of rough surfaces, boasting adhesion strength up to 465.56 ± 84.05 kPa. Super-saturated salts in TIA play a critical role for liquid–solid phase change and modulus regulation. In the amorphous state, which can be simply realized at room temperature without the need to remain at a high temperature, TIA can form conformal contact with rough surfaces. Once TIA is touched by the target surface, it turns into the crystallized state, forming a robust interlocking with the counterpart surface. Upon heating, the melting of the crystals softens TIA, enabling an easy detachment. The subsequent cooling to room temperature does not re-crystallize the TIA, but prepares TIA for the next round of conformal contact. That is, the facile melting and crystallization processes offer TIA with reversible adhesion that is conveniently triggered by a simple touch. The work here builds up a new strategy for the development of advanced adhesive materials and has significant implications for various applications requiring reversible and controllable adhesion.
2. Results and discussion
2.1. Synthesis of TIA
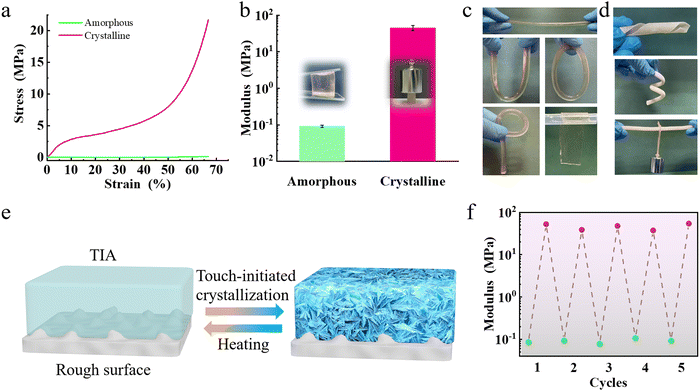
TIA was synthesized by a straightforward one-pot synthesis method. Sodium acetate (NaAc) was added to the hydrogel precursor, followed by the crosslinking of the hydrogel under UV light irradiation, resulting in the formation of TIA (Fig. 1b and c). The formulation of the hydrogel typically included acrylamide (AM, 12.5 wt%), acrylic acid (AA, 12.5 wt%) monomers, cross-linker (1.0 wt%), initiator (1.0 wt%), tannic acid (TA, 10 wt%) and NaAc (with a mass ratio to water of 1.2) (Fig. S1, ESI†). Hydrogels with different monomers and NaAc were prepared and named as “MaAb–Nc”, where M, A, and N represent AM, AA, and NaAc, respectively, and the subscripts indicate the corresponding mass ratio. For instance, “M12.5A12.5–N1.2” indicates a sample prepared with 12.5 wt% AM and 12.5 wt% AA as monomers, and a mass ratio of NaAc to water of 1.2. Once the hydrogel undergoes natural cooling to ambient temperature (∼25 °C), TIA achieves its fully operational state, poised for immediate use. At this stage, TIA was soft and transparent (Fig. 1c and Fig. S2, ESI†). After drying, minute grains which were precipitates of NaAc crystals with size ranging from 30 to 50 nm were dispersed in a random fashion (Fig. S2, ESI†). Upon touch stimulation, TIA transformed into an opaque and rigid state. Inside TIA, distinct fiber-like crystals with diameters ranging from 5 to 20 μm can be observed, showing an oriented arrangement (Fig. 1d).2.2. Reversible adhesion of TIA
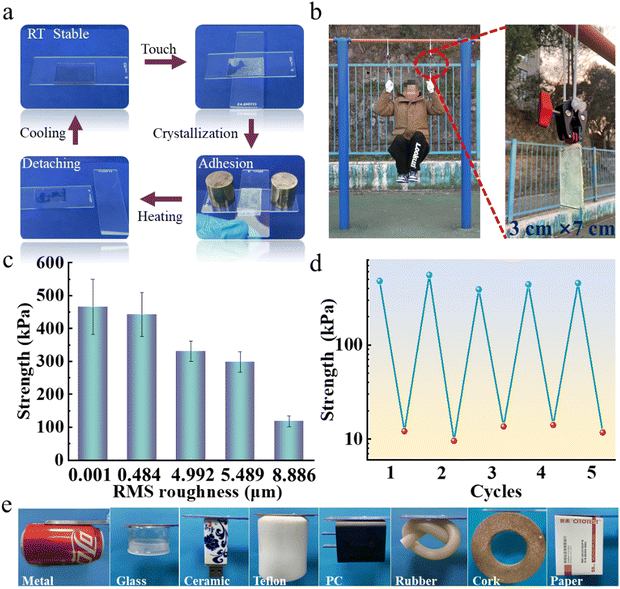
Compared with traditional phase-transition-based adhesives, which stay soft under external heating,31–34 TIA does not need continuous exposure to an external stimulus prior to usage (Fig. 2a and Video S1, ESI†). At room temperature, TIA remains transparent, rendering it primed for contact formation. The touching of TIA with the counterpart surface triggered the phase transition of TIA. Once the phase change of TIA was completed, a strong adhesion can be achieved. For demonstration, two glass plates with a roughness of 0.007 μm adhered together by TIA with an area of 3 cm × 7 cm (Fig. S3, ESI†) could support a ∼70 kg person (Fig. 2b and Video S2, ESI†). The adhesion was quite stable, allowing the performance of multiple pull-ups.TIA possesses strong adaption to surfaces with a wide range of roughnesses (Fig. S4, ESI†). An adhesion strength of 465.56 ± 84.05 kPa was demonstrated on the surface with root mean square (RMS) roughness of 1 nm (Fig. 2c and Table S1, ESI†). When the RMS roughness of the surface was ∼5000 times higher (∼4.99 μm), the adhesion strength reduced to 331.28 ± 30.73 kPa. The adhesion capabilities were confirmed on different surfaces (Fig. 2e and Fig. S5, Table S2, ESI†) which covers inorganic (iron, glass, ceramic) and organic (Teflon, PC, rubber, wood, paper) materials which include plane/curved, smooth/rough, hydrophobic/hydrophilic, and hard/soft surfaces. The adhesion capabilities were further demonstrated on 15 objects randomly selected in our office (Fig. S6 and S7, ESI†). As these objects have a surface roughness between 0.001 and 8.89 μm, the great potential of TIA was thus confirmed. On the other hand, when the RMS roughness was increased to 8.89 μm, 118.12 ± 16.15 kPa of adhesion strength remained, which remains higher than gecko adhesion.2 The decrease in adhesion strength can be attributed to the reduced effective contact area. While the crystalline speed was constant at a given temperature, the enlarged roughness would hinder the conformal contact with roughness.
The heating softened TIA and turned it back to the transparent state, greatly reducing the adhesion strength to 12.71 ± 2.73 kPa, facilitating the easy detachment (Fig. S8, ESI†). Moreover, when TIA was detached from the adhered surface and remained untouched, it remained in the transparent state at room temperature. Therefore, the heating of TIA allows the detachment and prepares it for the subsequent adhesion. Once the TIA was brought in contact with another object, strong adhesion was promptly re-established (Fig. 2d).
2.3. Mechanism for the touch-initiated phase transition
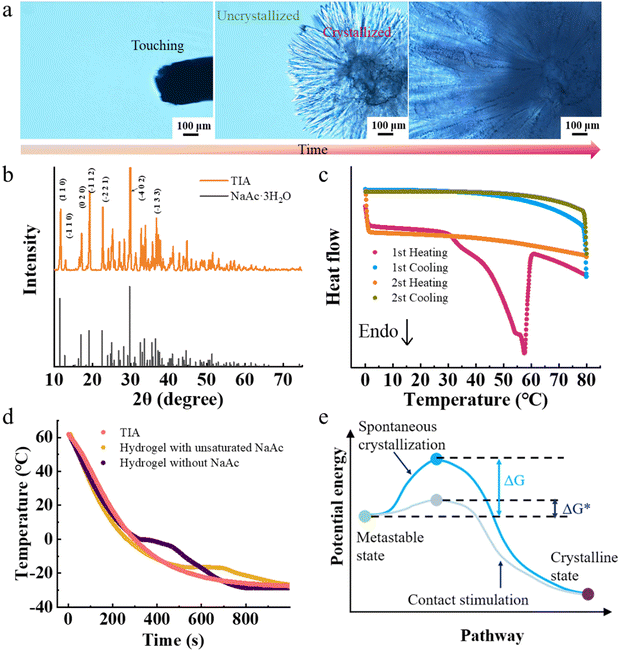
Given the pivotal role of phase transition in reversible adhesion, a comprehensive investigation was conducted to understand the nature of the phase transition of TIA. When the amorphous TIA was touched, crystallization was immediately initiated at the touched location (Fig. 3a and Video S3, ESI†). Subsequently, fiber-like crystals began to propagate outwards until the entire hydrogel was crystallized. XRD diffraction peaks unambiguously confirmed the presence of NaAc·3H2O crystals (PDF#29-1160) within the TIA (Fig. 3b). In order to validate the nature of the NaAc·3H2O crystals, differential scanning calorimetry analysis was conducted on the crystallized TIA (Fig. 3c). During the first heating cycle, a distinct endothermic peak at 58 °C was observed, providing clear evidence of crystal melting of TIA. In contrast, the hydrogel without NaAc did not have a corresponding endothermic peak (Fig. S9, ESI†). Both phenomena confirmed the existence of NaAc·3H2O crystals in TIA. On the other hand, no exothermic peak associated with the crystallization of NaAc·3H2O was detected during the subsequent cooling process. Additionally, no discernible endothermic or exothermic peaks corresponding to phase transitions were detected during the second round of heating or cooling. These phenomena indicated that TIA can maintain the amorphous state without the external heating. We can thus argue that the responsiveness of TIA primarily stems from the phase transition of NaAc·3H2O.The cooling behavior of TIA was further investigated in an extended temperature range (Fig. 3d). Conventionally, water undergoes a phase transition to ice at 0 °C under standard atmospheric conditions, leading to a characteristic plateau at 0 °C in the cooling curve until the crystallization process is complete (Fig. S10, ESI†). Similarly, hydrogels without NaAc or containing unsaturated NaAc underwent crystallization processes at 0 °C and −16 °C, mirroring the behavior of pure water and unsaturated NaAc solution, respectively. In contrast, TIA did not undergo a liquid–solid transition during cooling from 60 to −25 °C, as evidenced by the absence of a temperature plateau in the cooling curve. Consequently, TIA maintained a supercooled state at room temperature. The supercooled solution is difficult to nucleate spontaneously due to the high energy barrier associated with homogeneous nucleation (Fig. 3e).36–38 Upon being touched, TIA shifts its nucleation type from homogeneous to heterogeneous. The energy barrier for heterogeneous nucleation is significantly lower, facilitating the initiation of TIA crystallization. Meanwhile, the kinetic energy imparted by the touching disturbs the supercooled solution, fostering the crystal nucleus formation by disrupting the metastable state.39 As a result, TIA is sensitive to physical touch.
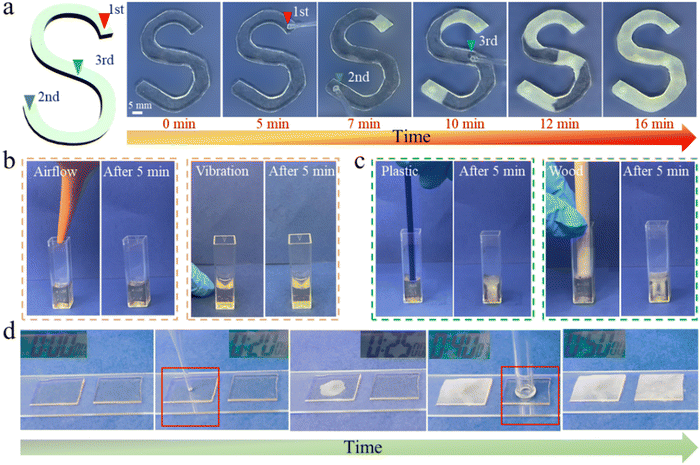
In order to investigate the touch-initiated phase transition of TIA, S-shaped TIA was simulated by a needle at different times and positions (Fig. 4a). When the top of S-shaped TIA was brought into contact with a needle, a white spot immediately showed up indicating the start of crystallization. Subsequently, the white crystalline regions expanded outward in all directions. Likewise, subsequent contacts at different locations within the uncrystallized region yielded similar phenomena, until TIA was fully crystallized. The crystallization speed was estimated to be 19 μm s−1 (Video S4, ESI†), which indicated that the crystallization process along the thickness direction of TIA (with a thickness of approximately 1 mm) would be completed in 53 s. When in contact with a rough surface, the presence of numerous contact points significantly shortens the crystallization time.
To further validate that the responsive behavior of TIA primarily originates from physical touch with a foreign object rather than non-contact forces, comparative experiments were conducted. The application of airflow onto the surface of TIA or the vibration of the supporting table was found to be insufficient to induce the crystallization of TIA (Fig. 4b and Video S5, ESI†). However, both touches with the solid plastic rod and the hollow wood tube triggered the crystallization (Fig. 4c). It is worth mentioning that, upon contact with a hollow wood tube, the untouched central area remained in the amorphous state, while the downward growth of crystallization had already been completed under the contacted area. This was reminiscent of contact with a rough surface, as it allowed sufficient time for the uncrystallized areas to form adaptive contacts. Additionally, an experiment was conducted by subjecting two TIA samples to an airflow environment (Fig. 4e and Video S6, ESI†). Initially, both TIA samples remained stable for the first 20 minutes. Crystallization commenced in one TIA sample upon touch with the needle, while the other piece remained unchanged. At 30 minutes, the touching with the second uncrystallized TIA eventually induced the crystallization. These experiments unequivocally confirm that physical touch is the crucial prerequisite for the phase transition of TIA, distinguishing it from previously reported adhesives based on phase transitions.31–34 To illustrate whether there is a critical pressure for the physical contact, we employed metal wires with a diameter of 0.9 mm and varying weights to get into contact with TIA (Fig. S11a, ESI†). The wires with weights ranging from 0.040 to 0.056 g freely falling onto TIA failed to induce crystallization such that TIA maintained a stable amorphous state (Fig. S11b, ESI†). By utilizing a 0.060 g wire, crystallization of TIA was initiated upon contact immediately (Fig. S11b, ESI†). Therefore, the critical pressure to induce the crystallization lies between 0.88 and 0.94 kPa.
2.4 Mechanism for the reversible adheison
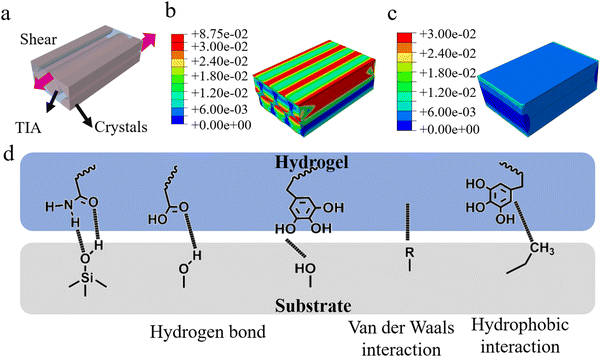
Previous researches have demonstrated that the material having low modulus is easier to gain adaptive contact with rough surfaces, while separation in a higher modulus state induces strong adhesion.40–42 Since physical contact between TIA and a rough surface at room temperature leads to the crystallization of NaAc·3H2O, the modulus of TIA at 70 °C was used to represent the amorphous state. The amorphous TIA material demonstrated a modulus of 0.091 ± 0.007 MPa and a compressive stress of 0.14 MPa at 60% strain (Fig. 5a, b and Fig. S12, ESI†). Consequently, amorphous TIA showed excellent adaptability and can be easily folded into a U-shape, O-shape, P-shape or bent shape (Fig. 5c). Crystallization renders TIA opaque and increased its modulus to 44.80 ± 12.08 MPa with a compressive stress of 18.85 MPa at 65% strain. Consequently, crystallized TIA is rigid enough, allowing it to bear load laterally. (Fig. 5d). By integrating the crystallization process of TIA and its mechanical properties in the two states, the mechanism for the touch-initiated strong adhesion of TIA was proposed (Fig. 5e). During attachment, the soft TIA can adapt to the surface asperities. The relatively low crystalline speed allows a sufficient time to adapt to the rough surface, forming conformal contacts. Meanwhile, crystallization starts from the contact points of asperities and spreads to adjacent areas, interlocking with the surface. The high modulus of TIA benefits cohesion, positively contributing to the adhesion. However, if the surface roughness is too large, and the crystallization process may be terminated before the hydrogel reaches the valleys in the roughness, impeding conformal contact between TIA and the rough surface. Consequently, decreased adhesion was observed on rougher surfaces (Fig. 2c). Moreover, TIA exhibits reversible switching between high and low moduli, thereby enabling control of adhesion by adjusting the temperature (Fig. 5f).To account for the effect of TIA structure and modulus, we established a finite element analysis model in both crystalline and amorphous states (Fig. 6a). In order to streamline the model, we assumed that the crystalline hydrogel is comprised of repetitive units of cylindrical crystals and hydrogel matrix, wherein the crystals are arranged in a specific direction throughout the hydrogel. TIA in the amorphous state was considered as a homogeneous polymer matrix. In the case of a crystalline state, the stress was primarily concentrated on the cylindrical crystals when TIA underwent shear deformation. In addition, the cylindrical crystals within the matrix can transfer stress, and thus disperse stress in the matrix (Fig. 6b). In contrast, the stress at the contacting interface of the amorphous TIA was almost identical to that inside the matrix when it was subjected to a shear deformation (Fig. 6c), and the stress is not adequately dissipated. Although the cylindrical crystal alignment is not entirely unidirectional and has an inhomogeneous size and distribution, our model still provides a feasible approach for stress dissipation in the system. Even with differences in stress alignment, the stress was primarily concentrated on the cylindrical crystals, revealing a dissipative effect resulting in higher adhesion strength.
The strong adhesion of TIA also comes from the forces including van der Waals force and hydrogen bonds originating from the carboxyl groups in AA and amide groups in AM (Fig. 6d). In TA, one tannic acid molecule contains 10 benzene rings, each of which contains 2–3 hydroxyl groups, providing a great increase in the number of hydrogen bonds. Meanwhile, the large amount of benzene in TA molecules could provide hydrophobic interactions to the contacting surfaces, especially the hydrophobic ones. Therefore, TIA can generate multiple attractive forces with rough surfaces, resulting in a strong adhesion.
3. Conclusion
A touch-initiated adhesive was successfully synthesized via the photopolymerization of compatible monomers in molten NaAc aqueous solution at 70 °C. The amorphous TIA had a low modulus of 0.091 ± 0.007 MPa, enabling it to conformally adhere to surfaces with varying roughness and materials. Upon contact, NaAc within the TIA underwent crystallization, resulting in a sharp increase in modulus to 44.80 ± 12.08 MPa, facilitating the interlocking with the contacting surfaces. Adhesion on smooth surfaces reached a strength of 465.56 ± 84.05 kPa. Although adhesion strength decreased with increasing surface roughness, adhesion on surfaces with a roughness of 8.89 μm still reached 118.12 ± 16.15 kPa. Heating the TIA above 58 °C turned it to the soft state, allowing the easy detachment from the adhered surfaces and preparing it for the subsequent attachment. Therefore, TIA is a promising adhesive for achieving on-demand adhesion and detachment. The adjustments to the monomers or crystals are expected to broaden the applications to fields like bioelectronics, electronic processing, and machine handling.4. Experimental section
4.1. Materials
Acrylamide (AM), acrylic acid (AA), and photo-initiator 1173 were obtained from Aladdin Co., Ltd (Shanghai, China). N,N′-methylenebisacrylamide (MBAA) was purchased from Macklin Biochemical Co., Ltd (Shanghai, China). PDMS elastomer kits (Sylgard 184) were purchased from Dow Corning (MI, USA). Anhydrous sodium acetate (NaAc) was purchased from Sinopharm Chemical Reagent Co., Ltd, China.Tannic acid (TA) and 2-hydroxy-2-methylpropiophenone were purchased from Aladdin, China. Deionized water (DI water) with a resistivity of 18.2 MΩ cm was used throughout all experiments. Glass slides were purchased from Citotest Labware Manufacturing Co., Ltd.
4.2. Preparation of the hydrogel
TIA was prepared by the following procedure: taking M12.5A12.5–N1.2 as the example. Firstly, 1.5 g AM, 1.5 g AA, 1.2 g TA, 30 mg MBAA (cross-linker, 1.0 wt% with respect to monomers), and 30 mg 2-hydroxy-2-methylpropiophenone (initiator, 1.0 wt% with respect to monomers) were dissolved in 7.8 mL DI water. Then, 9.36 g NaAc (the mass ratio to water is 1.2) was added to the above solution under magnetic stirring at 70 °C in a dark environment to obtain a homogenous solution. Then, the solution was transferred to a homemade mold and placed under UV irradiation (155 mW cm−2) at room temperature for 10 min. The UV lamp (XM 102, Shanghai AVENTK Electronic Technology Co., Ltd, China) with a wavelength of 365 nm was employed to initiate polymerization. The homemade mold consists of a U-shaped PDMS spacer sandwiched between two glass slides to form a chamber (10 mm × 10 mm). The thickness of the PDMS spacer was 1 mm. PDMS was prepared by mixing prepolymer and cross-linker at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and curing at 90 °C for 1 h.
1 and curing at 90 °C for 1 h.
4.3. Equipment/characterization
The morphology of the samples was characterized by field emission scanning electron microscopy (SEM, MIRA3, LMH, TESCAN). The sample underwent brittle fracturing in liquid nitrogen. After freeze-drying for 24 hours, the fractured cross-section was selected and subjected to gold sputtering for 60 s. Surface roughness was characterized by a 3D Optical Surface Profiler (NewView 9000, Zygo, USA). The samples were positioned horizontally on the profilometer moving stage, with the Z-axis accuracy set to 0.1 nm. A field of view measuring 350 × 350 μm was selected for imaging. The RMS roughness values were extracted from the images. Compression test and lap shear adhesion was performed on a universal testing machine (UTM 2503, Shenzhen Suns Technology STOCK Co., Ltd, China). In the compression test, cylindrical samples with a diameter of 15 mm and a height of 15 mm were conducted at a speed of 2 mm min−1. For the lap shear adhesion test, the substrates were washed with DI water 3 times and then dried. The TIA with dimensions of 25 mm (length) × 15 mm (width) × 1 mm (thickness) was laid flat on one substrate. The other same substrate was used to touch the TIA at RT with a pressure of 12 kPa to induce its crystallization. After 1 hour, a lap shear adhesion test was conducted at a tensile speed of 2 mm min−1. At least three samples were tested under identical specimen conditions. The UV light-emitting diode (LED) lamp (XM 102, Shanghai AVENTK Electronic Technology Co., Ltd, China) with a wavelength of 365 nm was employed to initiate polymerization.4.4. Ethical approval
All human body-related experiments with TIA were performed under ethical approval from the Ethics Committee of School of Medicine, Wuhan University (WHU-LFMD-IRB2023006). Informed written consent was obtained from all participants prior to data collection for the research.Author contributions
Zhekun Shi: methodology, data curation, formal analysis and writing original draft; Di Tan, Kangjian Xiao and Xiaolong Zhang, methodology, formal analysis; Bo Zhu, Zhen Lin and Quan Liu, investigation; Daobing Chen and Qinghua Zhang, writing – review & editing; Longjian Xue, conceptualization, resources, writing – review & editing and funding acquisition.Conflicts of interest
The authors declare that they have no conflict of interest.Acknowledgements
This work was supported by the National Key R&D Program of China (2022YFB4701000), National Natural Science Foundation of China (52373119), Open Fund of Hubei Key Laboratory of Electronic Manufacturing and Packaging Integration (Wuhan University) (EMPI2023020), Research Funds of the Institute of Zhejiang University-Quzhou (IZQ2023RCZX017, IZQ2023KJ2003), and Zhejiang Provincial Natural Science Foundation of China (LQ24E030016). The authors acknowledge the nanofabrication assistance from the Center for Nanoscience and Nanotechnology at Wuhan University.References
- Y. Chen, J. Meng, Z. Gu, X. Wan, L. Jiang and S. Wang, Adv. Funct. Mater., 2020, 30, 1905287 CrossRef CAS.
- K. Autumn, Y. A. Liang, S. T. Hsieh, W. Zesch, W. P. Chan, T. W. Kenny, R. Fearing and R. J. Full, Nature, 2000, 405, 681–685 CrossRef CAS PubMed.
- E. Arzt, S. Gorb and R. Spolenak, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 10603–10606 CrossRef CAS PubMed.
- T. Priemel, G. Palia, F. Förste, F. Jehle, S. Sviben, I. Mantouvalou, P. Zaslansky, L. Bertinetti and J. Harrington Matthew, Science, 2021, 374, 206–211 CrossRef CAS PubMed.
- E. Arzt, H. Quan, R. M. McMeeking and R. Hensel, Prog. Mater. Sci., 2021, 120, 100823 CrossRef.
- C. Lee, S.-E. Choi, J. W. Kim and S.-Y. Lee, Small, 2020, 16, 1904282 CrossRef CAS PubMed.
- T. Eisner and D. J. Aneshansley, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 6568–6573 CrossRef CAS PubMed.
- L. Qu, L. Dai, M. Stone, Z. Xia and L. Wang Zhong, Science, 2008, 322, 238–242 CrossRef CAS PubMed.
- Y. Wang and R. Hensel, Adv. Funct. Mater., 2021, 31, 2101787 CrossRef CAS.
- Q. Liu, D. Tan, F. Meng, B. Yang, Z. Shi, X. Wang, Q. Li, C. Nie, S. Liu and L. Xue, Small, 2021, 17, 2005493 CrossRef CAS PubMed.
- X. Wang, D. Tan, S. Hu, Q. Li, B. Yang, Z. Shi, R. Das, X. Xu, Z.-S. Wu and L. Xue, ACS Appl. Mater. Interfaces, 2019, 11, 46337–46343 CrossRef CAS PubMed.
- A. del
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Campo, C. Greiner, I. Álvarez and E. Arzt, Adv. Mater., 2007, 19, 1973–1977 CrossRef.
Campo, C. Greiner, I. Álvarez and E. Arzt, Adv. Mater., 2007, 19, 1973–1977 CrossRef. - Z. Wang, ACS Nano, 2018, 12, 1273–1284 CrossRef CAS PubMed.
- H. Lee, D.-S. Um, Y. Lee, S. Lim, H.-J. Kim and H. Ko, Adv. Mater., 2016, 28, 7457–7465 CrossRef CAS PubMed.
- S. Baik, D. W. Kim, Y. Park, T.-J. Lee, S. Ho Bhang and C. Pang, Nature, 2017, 546, 396–400 CrossRef CAS PubMed.
- S. T. Frey, A. B. M. T. Haque, R. Tutika, E. V. Krotz, C. Lee, C. B. Haverkamp, E. J. Markvicka and M. D. Bartlett, Sci. Adv., 2022, 8, eabq1905 CrossRef PubMed.
- Y. Wang, X. Yang, Y. Chen, D. K. Wainwright, C. P. Kenaley, Z. Gong, Z. Liu, H. Liu, J. Guan, T. Wang, J. C. Weaver, R. J. Wood and L. Wen, Sci. Robot., 2017, 2, eaan8072 CrossRef PubMed.
- S. Su, S. Wang, L. Li, Z. Xie, F. Hao, J. Xu, S. Wang, J. Guan and L. Wen, Matter, 2020, 2, 1207–1221 CrossRef.
- D.-M. Drotlef, L. Stepien, M. Kappl, W. J. P. Barnes, H.-J. Butt and A. del Campo, Adv. Funct. Mater., 2013, 23, 1137–1146 CrossRef CAS.
- J. Iturri, L. Xue, M. Kappl, L. García-Fernández, W. J. P. Barnes, H.-J. Butt and A. del Campo, Adv. Funct. Mater., 2015, 25, 1499–1505 CrossRef CAS.
- H. Chen, L. Zhang, D. Zhang, P. Zhang and Z. Han, ACS Appl. Mater. Interfaces, 2015, 7, 13987–13995 CrossRef CAS PubMed.
- L. Zhang, H. Chen, Y. Guo, Y. Wang, Y. Jiang, D. Zhang, L. Ma, J. Luo and L. Jiang, Adv. Sci., 2020, 7, 2001125 CrossRef CAS PubMed.
- L. Xue, B. Sanz, A. Luo, K. T. Turner, X. Wang, D. Tan, R. Zhang, H. Du, M. Steinhart, C. Mijangos, M. Guttmann, M. Kappl and A. Del Campo, ACS Nano, 2017, 11, 9711–9719 CrossRef CAS PubMed.
- S. Baik, J. Lee, E. J. Jeon, B.-Y. Park, D. W. Kim, J. H. Song, H. J. Lee, S. Y. Han, S.-W. Cho and C. Pang, Sci. Adv., 2021, 7, eabf5695 CrossRef CAS PubMed.
- S. Li, H. Liu, H. Tian, C. Wang, D. Wang, Y. Wu and J. Shao, ACS Appl. Mater. Interfaces, 2021, 13, 42287–42296 CrossRef CAS PubMed.
- H. Lee, B. P. Lee and P. B. Messersmith, Nature, 2007, 448, 338–341 CrossRef CAS PubMed.
- Y. Ma, S. Ma, Y. Wu, X. Pei, S. N. Gorb, Z. Wang, W. Liu and F. Zhou, Adv. Mater., 2018, 30, 1801595 CrossRef PubMed.
- L. Gao, S. Ma, L. Bao, X. Zhao, Y. Xiang, Z. Zhang, Y. Ma, Z. Ma, Y.-M. Liang and F. Zhou, ACS Appl. Mater. Interfaces, 2022, 14, 12684–12692 CrossRef CAS PubMed.
- W. Zhang, R. Wang, Z. Sun, X. Zhu, Q. Zhao, T. Zhang, A. Cholewinski, F. Yang, B. Zhao, R. Pinnaratip, P. K. Forooshani and B. P. Lee, Chem. Soc. Rev., 2020, 49, 433–464 RSC.
- M. W. Denny, Am. Zool., 1984, 24, 23–36 CrossRef.
- Z. Ye, G. Z. Lum, S. Song, S. Rich and M. Sitti, Adv. Mater., 2016, 28, 5088–5092 CrossRef CAS PubMed.
- S. Li, H. Tian, J. Shao, H. Liu, D. Wang and W. Zhang, ACS Appl. Mater. Interfaces, 2020, 12, 39745–39755 CrossRef CAS PubMed.
- D. Tan, X. Wang, Q. Liu, K. Shi, B. Yang, S. Liu, Z.-S. Wu and L. Xue, Small, 2019, 15, 1904248 CrossRef CAS PubMed.
- S. Xi, F. Tian, G. Wei, X. He, Y. Shang, Y. Ju, W. Li, Q. Lu and Q. Wang, Adv. Mater., 2021, 33, 2103174 CrossRef CAS PubMed.
- T. Ohzono and E. Koyama, Polymer, 2022, 260, 125377 CrossRef CAS.
- L. Hu, H. Lu, X. Ma and X. Chen, J. Cryst. Grow., 2021, 575, 126340 CrossRef CAS.
- Z. Kožíšek, J. Cryst. Grow., 2019, 522, 53–60 CrossRef.
- M. Hou, Z. Jiang, W. Sun, Z. Chen, F. Chu and N.-C. Lai, Adv. Mater., 2023, 2310312 Search PubMed.
- T. B. H. Schroeder and J. Aizenberg, Nat. Commun., 2022, 13, 259 CrossRef CAS PubMed.
- C. Linghu, Y. Liu, Y. Y. Tan, J. H. M. Sing, Y. Tang, A. Zhou, X. Wang, D. Li, H. Gao and K. J. Hsia, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2221049120 CrossRef CAS PubMed.
- H. Wang, S. Chen, H. Li, X. Chen, J. Cheng, Y. Shao, C. Zhang, J. Zhang, L. Fan, H. Chang, R. Guo, X. Wang, N. Li, L. Hu, Y. Wei and J. Liu, Adv. Funct. Mater., 2021, 31, 2100274 CrossRef CAS.
- D. Wang, H. Hu, S. Li, H. Tian, W. Fan, X. Li, X. Chen, A. C. Taylor and J. Shao, Sci. Adv., 2023, 9, eadf4051 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4mh00331d |
| This journal is © The Royal Society of Chemistry 2024 |