Challenges and opportunities on sustainable electrochemical transformations: application towards the synthesis of pharmaceuticals and precursors of drug-like molecules
Adrija
Ghosh
,
Vishal Kumar
Parida
and
Debasis
Banerjee
 *
*
Department of Chemistry, Laboratory of Catalysis and Organic Synthesis, Indian Institute of Technology Roorkee, Roorkee-247667, Uttarakhand, India. E-mail: debasis.banerjee@cy.iitr.ac.in
First published on 25th March 2024
Abstract
The pivotal role of electrochemistry has significantly improved the step economy of organic transformations, thereby surpassing conventional pathways. Sustainable electrochemical transformations enormously influenced the chemical synthesis and attracted significant attention towards pharmaceutical industries. More specifically, the electrochemical organic transformation streamlines the synthesis of a variety of drugs and precursors of drug-like molecules. This review summarised the electrochemical synthesis of more than 50 such drugs, therapeutic chemicals, pharmaceuticals, and precursors of drug-like molecules, which were directly or indirectly synthesised over the period of 2017–2023.
1. Introduction
Electrochemical organic transformation is an emerging research area and has gained significant attention towards sustainable development. It is noteworthy to mention that this transformation has paved a new roadmap for modern catalytic technologies over the traditional synthetic pathways via its greater resource economy and elimination of possible by-products.1 Owing to their inherent advantages, which comply with the principles of green chemistry, employing electrochemistry towards the direct synthesis of functionalised chemical space has proven to be a highly significant development. This is particularly crucial for the fact that the number of steps and class of reagents used in traditional chemical synthesis have a great impact on the product yield, as well as on the environment.2–4 Looking at the potency of electrochemistry in bridging the gap between complex molecule synthesis and sustainability, it is evident that the future scope of such emerging technologies shall be ruled by its applications in various aspects of synthetic organic chemistry including medicine to materials.5Organic electrochemistry has attracted significant interest in comparison to conventional synthetic transformations.6 The pursuit of electrochemistry is much greener and cost-effective.7 This review shall merge all of the recent developments made in the area of electrochemical pathways to drug analogues and value-added products, and pharmaceuticals.6–10 Interestingly, electrochemistry has significantly shortened the synthetic routes to various pharmaceuticals and drug-like molecules, as highlighted in Fig. 1.
Herein, we summarise all these recent advancements, including the challenges and opportunities on sustainable electrochemical transformations towards the synthesis of pharmaceuticals and precursors of drug-like molecules.
A previous report by Bisai in 2022 focused on the total synthesis of naturally occurring alkaloids and terpenoids via electrochemical C–H functionalization.11 Cantillo very briefly summarised some of the recent syntheses of active pharmaceutical ingredients. However, no attention was paid towards the importance of the electrocatalytic protocol or its scope.12 Chain reported on the total synthesis of alkaloids and terpenoids, including fatty acids, polyketides and carbohydrates.13 Ackermann presented the electro-organic synthesis of speciality chemicals mainly focused on fluorinated, organophosphorous, azido, and deuterated compounds solely based on the C–H bond functionalisation.7d The total synthesis of natural products employing electrochemical methodologies was mostly documented in earlier reviews. Moreover, the major focus was directed towards chemical catalysis, and precise attention often was paid to the synthesis of a class of compounds. However, it is necessary to pay attention towards more sustainable approaches, where potential emphasis on electrochemical transformations for such applications is in demand. It is noteworthy to mention that since the past couple of years, we are actively involved in base-metal catalysed sustainable strategies for organic transformations14,15 and have focused our attention towards emerging electrocatalysis approaches for organic transformations.
The present review focuses on the potential applications of electrochemical strategies. More specifically, the direct (one-step) or indirect (2–3 steps) synthesis of drugs, natural products, hormone analogues, neurotransmitters, metabolites, synthesis of cytotoxins, NSAIDs, etc. is the primary focus. This review shall cover the sustainable electrochemical syntheses of more than 50 such complex molecules, which were directly and indirectly synthesised over the period of 2017–2023. Herein, we plan to briefly classify electrochemical methodologies based on the type of reaction and applications in drug synthesis. Furthermore, the syntheses of value-added products that have not been highlighted yet in any of the above-mentioned previous reviews are summarised. The pivotal role of electrochemistry in all of these classified areas has significantly improved the step economy of organic transformations, thereby surpassing conventional pathways.
Moreover, to the best of our knowledge, there is no comprehensive review reported that highlights sustainable electrochemical transformations for the synthesis of therapeutic chemicals, pharmaceuticals, and precursors of drug-like molecules, including their applications in medicine to materials.
The key features summarised are as follows: (a) electrochemical cross-electrophile coupling (XEC) for the formation of new C–C bonds; (b) electrochemical C–H functionalisation towards applications to materials chemistry; (c) electrochemical intramolecular cyclisation/annulation; (d) electrochemical CO2 activation to biologically active organic moieties via elimination of toxic reducing agents; (e) asymmetric electro synthesis to optically active compounds; (f) electrochemical dimerisation; and (g) miscellaneous electrochemical transformations.
2. Electrochemical strategies for drug synthesis
2.1 Electrochemical CO2 activation
Carbon dioxide is the most abundant C1 source, and one of the major components of the greenhouse effect.16 Recent years have witnessed significant utilisation of CO2 to value-added chemicals17a,b and natural products.18 Researchers have exploited metal complexes and harsh conditions to overcome the barriers of CO2 activation and incorporation in organic compounds.19a–d In this regard, electrochemistry has emerged as a potent, sustainable and effective methodology for activation of inert carbon dioxide and its valorisation.20 For instance, Naproxen (3) and Ibuprofen (7) are regularly used, and the direct syntheses of these drugs have gained great synthetic importance over the years.21 Conventional methods for their synthesis depend on an excess over the stoichiometric amounts of reducing agents (zinc and MgCl2), which could be avoided via application of electrochemistry.Mellah and co-workers (2019) utilised highly oxophilic divalent samarium (Int. 1), generated electrochemically, and catalysed the carboxylation of benzyl halides to corresponding acids (Scheme 1).22a It was proposed that reduction of carbon dioxide and ligation with the metal centre generated a samarium carboxylate species Int. 2, followed by TMSCl catalysing the cleavage of the Sm(III)–O species (Int. 3) to silyl ester (Int. 5), and furnished the desired product after hydrolysis. During the process, trivalent samarium (Int. 4) is generated along with Int. 1 by cathodic reduction. This protocol avoids the use of any extra metal catalysts, and prevents the generation of by-products via dehalogenation and dimerisation.22b Naproxen (3) hormone, widely used for pain and inflammation, was obtained in a single step.
 | ||
| Scheme 1 Electrochemical CO2 activation for the synthesis of Naproxen (ref. 22a). | ||
Similar transformations using aryl triflates, tosylates, or ethers and allylic alcohols with CO2 were also resulted carboxylic acid motifs.23a–f An alternative metal-free carboxylation of air-stable benzyl tri-methylammonium bromide salts was also reported by Manthiram and co-workers.24
Ye and co-workers employed the electro-carboxylation approach using primary, secondary and tertiary sulfones under mild reaction conditions (Scheme 2a).25 High stability and the electron-withdrawing nature of sulfones favoured the carboxylation to desired acids. Mechanistic studies revealed that the carbon sulphur bond was cleaved via a two-electron reduction, resulting in the phenyl sulfinate and benzyl anion (Int. 7). Upon subsequent addition of CO2, the reaction resulted in phenyl acetic acid 6 in high yield. Thus, application of toxic organic halides could be avoided using the CO2 electro-carboxylation approach. Guirado and co-workers reported on sustainable and efficient electrosynthetic routes to ibuprofen using ionic liquids (Scheme 2b).26 The use of ionic liquids avoids flammable and hazardous organic solvents, and acts as both electrolyte as well as catalyst. Moreover, the use of a silver cathode decreases the reduction potential in the carbon-halide cleavage step, and around 80% of the ionic liquids could be recovered and reused. Buckley and co-workers also reported on a highly regioselective electrochemical approach for hydrocarboxylation of α,β-unsaturated esters to all-carbon quaternary centres (Scheme 2c).27 This protocol emphasised the potential of the metal-free electrochemical approach, and is different from previously reported strategies by Lan, Yu and Mikami.28a–c A key challenge associated with this includes the high competition between the double bond reduction and carbon dioxide reduction because of their similar reduction potentials. The protocol was employed in the synthesis of the drug precursor ethosuximide (14).
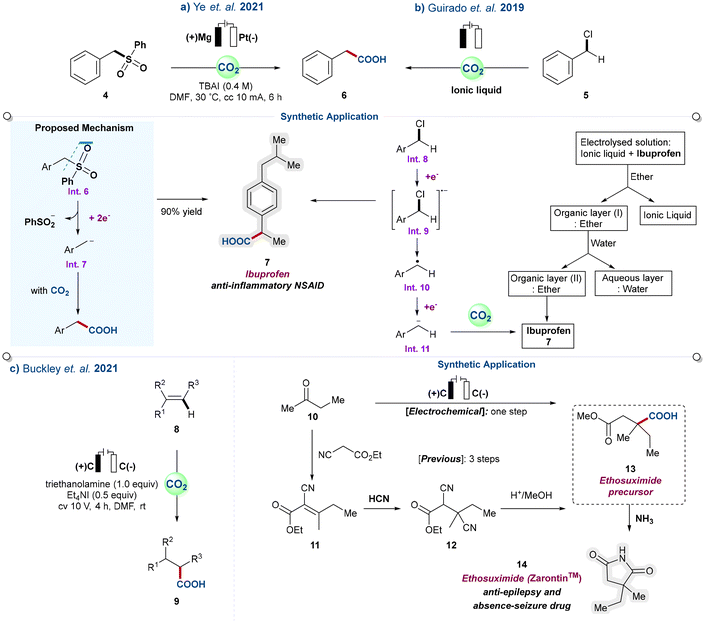 | ||
| Scheme 2 (a) Desulfonylative electrocarboxylation of sulfones for the synthesis of Ibuprofen (ref. 25). (b) Electrocarboxylation of benzyl halides in ionic liquids (ref. 26). (c) Selective electrosynthetic hydrocarboxylation of α-β unsaturated esters (ref. 27). | ||
Di-acids are ubiquitous in polymers, and much attention has been paid to the dicarboxylation of alkenes, alkynes, allenes, heteroarenes, and dienes.29,30 In 2020, Lin and co-workers reported an electro-reductive carbo-functionalisation of alkenes with alkyl halides. They focused on chemo- and regioselective C(sp3)–C(sp3) bond construction under a transition metal-free approach. The reaction proceeded via a radical polar crossover pathway, where alkyl bromide was successfully reduced at the cathode, leading to the generation of the key radical intermediate (Int. 12), followed by the desired product (17, Scheme 3). The process demonstrated the synthesis of biologically active molecules, such as the glucokinase activator (20, used to maintain the potassium channel in cells), pregabalin (23, used for neuropathic pain and anxiety disorders), and histone deacetylase inhibitors (25, used for psychiatry and neurology as mood stabilizers, and also for anti-epileptics). These molecules are being investigated as a possible alternative for treatments of cancers, and parasitic and inflammatory diseases.31
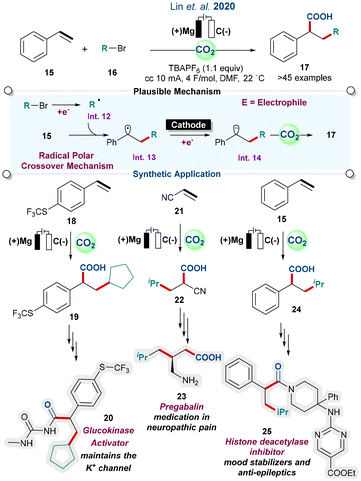 | ||
| Scheme 3 Electroreductive carbo-functionalisation of alkenes: synthesis of biologically active molecules (ref. 31). | ||
Mita and co-workers reported an electrochemical protocol for dearomative mono- and di-carboxylation of heteroaromatics using carbon dioxide as the potent dicarboxylating agent (Scheme 4).32,33a As proposed, the carbon dioxide radical anion was initially produced, which mainly yielded the trans-oriented di-carboxylic acids from protected indoles, and was supported by computational studies including control experiments. The catalytic protocol was directly applied to biologically active dicarboxylic acid derivatives; inhibitors of PDE-IV, TXA2 33, and antagonist of NMDA and AMPA receptors 34.33b,c
 | ||
| Scheme 4 Electrochemical di-carboxylation of heterocycles (ref. 33a). | ||
2.2 Electrochemical C–H activation
C–H activation has an opulent history in organic synthesis and has received considerable attention from chemists over the years. N-hydroxy phthalimide (NHPI), a HAT mediator, first introduced in 1980s by Masui and co-workers,34a,b has been used for trapping organic radicals in the presence of O2 leading to the formation of oxygenated products. Contrary to this, electrochemical NHPI oxidation allows generation of PINO (phthalimido-N-oxyl) in the absence of O-atom sources. Stahl and co-workers first reported the use of N-hydroxyphthalimide as an electrochemical mediator, employing I2 as the radical trap via the HAT process. The reaction of in-situ generated PINO with the weak benzylic C-H bond is thermoneutral and produces a radical which apparently reacts with iodine to give the desired product (Scheme 5a).35 Synthetic application of the in situ nucleophilic substitution of iodides was also an advantage of this protocol. Electrochemical iodination protocol can be utilized for sequential iodination/alkylation apart from in situ functionalization. This has been exploited for the synthesis of three pharmaceutically significant moieties namely, Permethrin (used in treatment of scabies), Sergliflozin (anti-diabetic drug), and HIV-1 integrase inhibitor (antiretroviral drug). Biochemical approaches are popular due to their catalyst-controlled selectivity,36 and often utilised for the direct abstraction of the hydrogen atom for C–H oxidation reactions.37 Due to the challenging tunability of these catalysts or reagents, which require tedious synthetic procedures, the focus has recently been shifted towards more sustainable electrochemical hydrogen atom transfer-based mediators.38N-Alkyl ammonium ylides served as a new class of reactive radicals, which have been selectively tuned by Baran and co-workers and utilised for the oxidation of inactivated C–H bonds.39,40 The developed strategy was based on a structure-agnostic approach, wherein a desired catalytic cycle was modelled prior to mediator selection (Scheme 5b). The key catalytic steps are: (a) abstraction of hydrogen by a radical mediator; (b) deprotonation to regenerate a radical precursor; and (c) generation of an oxidised product, along with the regeneration of a radical mediator. The remarkable ability for the generation of tertiary alcohol products was observed in quinuclidine-mediated oxidations.40 Such chemo-selective electro-oxidative approach was employed in the shortest route to the key intermediate 48 for the synthesis of buspirone (49, anxiolytic drug), and tiospirone 50, respectively. | ||
| Scheme 5 (a) NHPI-Mediated electrochemical iodination of methylarenes for the synthesis of Permethrin, Sergliflozin, and HIV-1 Integrase Inhibitor (ref. 35). (b) N-Ammonium ylide-mediated electrochemical C–H oxidation for the syntheses of Buspirone and Tiospirone (ref. 40). | ||
Benzyl esters are omnipresent in natural products and drugs, which have been conventionally synthesised via esterification, transesterification, and often require high reaction temperatures. Considering a sustainable and cost-efficient approach, He and co-workers designed an electrochemical protocol, where the direct acyloxylation of benzyl C(sp3)–H bond with amide was achieved.41 This strategy features metal- and oxidant-free C(sp3)–H functionalisation with hydrogen gas evolution, and exhibited good functional group tolerance. Mechanistic studies suggested that the in situ oxidation of the benzylic C–H bond led to the formation of benzylic carbocation, which transformed into an imine via nucleophilic attack of DMF. Hydrolysis of the imine intermediate furnished the benzyl-acyloxylated product. The protocol was employed for the synthesis of food additives (54, imparts vanilla essence), and a fragment of the natural product Schisantherin A 55 serves as an antagonist of adiponectin receptor 2 (Scheme 6).
 | ||
| Scheme 6 Electrochemical approach for the synthesis of Schisantherin A (ref. 41). | ||
2,5-Diaryl substituted oxazoles are omnipresent in various pharmaceutically active compounds, yet their synthesis is associated with sacrificial oxidants, expensive transition metals and generates stoichiometric waste. In 2023, Wang and co-workers reported a metal-free electro-oxidative strategy of 2,5-diaryloxazoles that follows sequential C(sp3)–H and N–H bond functionalisation, starting from aryl ketones and naturally occurring α-amino acids.42 The one-pot syntheses of O-methylhalfordinol (59, an anti-proliferative natural product) and texaline 60, and balsoxine 61 and texamine 62, known as antimycobacterial agents, were synthesised in good to excellent yield (Scheme 7).
 | ||
| Scheme 7 Electrochemical approach to 2,5-diaryloxazole: synthesis of O-methylhalfordinol, texaline, balsoxine and texamine (ref. 42). | ||
Directing group-mediated electrochemical C–H halogenation for the synthesis of haloarenes is a long-standing goal.43 In 2017, Kakiuchi and co-workers reported the Pd-catalysed ortho-chlorination using HCl under electro-oxidative conditions. Benzamides bearing the 5,7-dichloro-8-quinolinyl group, a bidentate-directing group, proved efficient for selective ortho-chlorination.44 Vismodegib, an FDA-approved drug used for the treatment of basal cell carcinoma, was obtained using this electro-oxidative protocol. Reduction of the nitro group in 67 in the presence of iron in acetic acid furnished 68, which was coupled with acid 66, and resulted in vismodegib 69 in 70% yield (Scheme 8a).
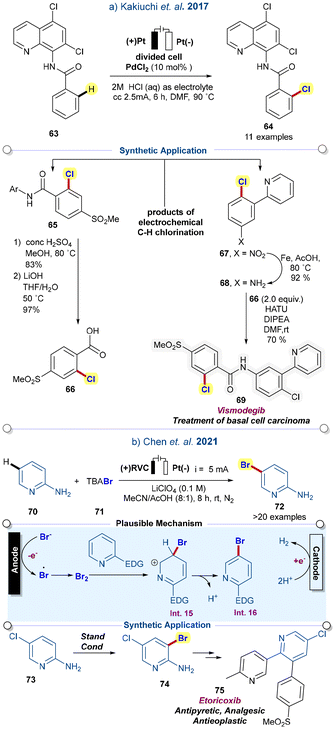 | ||
| Scheme 8 (a) Pd-catalyzed C–H chlorination of benzamide derivatives for the synthesis of Vismodegib (ref. 44), (b) electrochemical meta-bromination of pyridines for the synthesis of Etoricoxib (ref. 45). | ||
Recently, Chen and co-workers reported on the catalyst and oxidant-free electrochemical bromination of pyridine. It was observed that the pyridines (having electron-donating groups) control the regio-selective bromination. As proposed, under mild conditions, the bromine radical coupled to produce bromine gas, which further participated in aromatic electrophilic substitution to achieve Int. 15, followed by deprotonation, resulting in the desired product. This protocol was further extended to the precursor of etoricoxib 75, an NSAID drug (Scheme 8b).45
The electrochemical C-3 formylation/acylation of indoles via the one-pot Mannich reaction was reported by Zheng and co-workers (Scheme 9a).46 It was observed that the amine used for the Mannich reaction activates the aldehyde, which is then removed via electrochemical oxidative C–N bond cleavage. Using this strategy, 78 was obtained, followed by the synthesis of GSK-3β inhibitors (79, against bipolar disorders) in a single step. GSK-3β inhibitors are widely used in hepatic glycolysis regulation, cell signalling pathway and phosphorylation of various proteins, including treatments for cancer and Alzheimer's diseases.
 | ||
| Scheme 9 (a) Electrochemical C3-formylation: synthesis of GSK-3β Inhibitors (ref. 46), (b) direct synthesis of paracetamol: electrochemical Ritter type C–H amination of phenols (ref. 48). | ||
Paracetamol, or N-acetyl-para-aminophenol (APAP), is one of the highest consumed analgesic drugs. Synthesis of APAP mostly depends on the N-acetylation of p-aminophenol. However, these processes are associated with selectivity issues, as well as the use of hazardous reagents and harsh reaction conditions.47 In contrast, electrochemistry has enabled a greener synthesis route to paracetamol directly from phenols, employing the concept of the Ritter reaction (Scheme 9b).48 The site-selective Ritter type amidation using acetonitrile, as the nitrogen source, with phenol 80 resulted in paracetamol 81 in high yield.
2.3 Electrochemical cross coupling
Cross-coupling reactions largely contributed to chemical synthesis, and are extensively utilised for complex molecular scaffolds.49 However, the generation of halide waste, the use of harsh reaction conditions, and the involvement of multi-step processes forced researchers to find more mild and sustainable synthetic routes. Often, the cross-electrophilic coupling reactions were aided by metal catalysts or metal nano-particles, and used in over-stoichiometric amounts.50 In this direction, Sevov and co-workers reported an interesting cross-coupling reaction using electrocatalysis (Scheme 10).51 Primary and secondary alkyl bromides were coupled with aryl, heteroaryl or vinyl bromides, and resulted C(sp2)–C(sp3) bonds in the presence of a redox shuttle, which attenuates the over-reduction and degeneration of the catalyst. Such redox shuttle serves as a co-catalyst to shuttle electrons from the cathode to the anode. The protocol was utilized in the synthesis of pridopidine (86), which is used for Huntington's disease and amyotrophic lateral sclerosis.51 | ||
| Scheme 10 Cross electrophile coupling for the synthesis of pridopidine (ref. 51). | ||
Electrochemical sequential C–C and C–X bond formation using decarboxylative cross-coupling has attracted potential interest in recent times.52,53 Sheng and co-workers reported an electrochemical intramolecular decarboxylative coupling of isatin and benzoyl hydrazines to 2-(1,3,4-oxadiazol-2-yl) aniline derivatives (Scheme 11).54a,b The isatin moiety behaved as an amino-attached C1 source. Late-stage functionalisation of the desired products resulted in the ATP-binding cassette transporter G2 inhibitor 90 and N-heterocycles having antibacterial activity 93.
 | ||
| Scheme 11 Electrosynthesis of 2-(1,3,4-oxadiazol-2-yl) aniline derivatives (ref. 54a). | ||
Quinazolinones have exhibited a broad range of biological activities, and were identified as essential scaffolds in drugs,55 and in natural products.56 Luotonin A and its derivatives, having a quinazolinone core, serve against cancer cell lines.57 Primitive synthetic protocols for the synthesis of the quinazolinone core involve condensation,58a,b carbonylative annulation,59a,b and transition metal catalyzed annulation.60a,b Such processes are limited by their harsh reaction conditions, toxic solvents, and poor product selectivities. In this direction, Ke and co-workers reported on the one-pot electrochemical tandem reaction towards the synthesis of quinazolinones from o-aminobenzonitrile and benzaldehyde.61 Iodine serves as a catalyst in water, which complies with the concept of green chemistry. Synthesis of the Gefitinib analogue 101 was obtained, which inhibits the growth of human colon cancer cells HCT-116, human Lung Carcinoma Cells A549, and was used for the treatment of human gastric cancer cells SGC-7901 (Scheme 12).
 | ||
| Scheme 12 Electrochemical synthesis of quinazolines (ref. 61). | ||
Oxidative carbonylation provides direct access to ynones, present in various biologically active scaffolds.62a,b However, safety issues related to its handling and the hazardous CO limited its applications in bulk industrial synthesis. Moreover, oxidative carbonylation employing direct CO/O2 mixtures is an efficient alternative for easy access to carbonyl moieties.63
In 2021, Lei and co-workers demonstrated an improved and elegant Pd-catalyzed carbonylation protocol for the synthesis of ynones using alkynes and aryl hydrazines. The anti-inflammatory drug and analgesic, 104 was synthesized using this electrochemical carbonylative strategy (Scheme 13a).64 Brown and co-workers demonstrated the impact of amides as main building blocks used in almost 50% of pharmaceuticals,65 natural products and in polymers.66a,b In 2022, Patel and co-workers reported on the electrochemical amidation of benzoyl hydrazine/carbazate, employing primary and secondary amines as coupling partners. Benzoyl hydrazine serves as a potential acyl radical donor, which couples with amine, the N-radical donor, thereby resulting in the desired amide. This greener, metal-free electrosynthetic protocol enabled the two-step synthesis of the Bezafibrate drug 108, which is widely used for the treatment of hyperlipidemia (Scheme 13b).67
 | ||
| Scheme 13 (a) Palladium-catalyzed oxidative Sonogashira carbonylation for the synthesis of the FSY drug (ref. 64), (b) electrochemical amidation: synthesis of bezafibrate (ref. 67). | ||
The Horner–Wadsworth–Emmons (HWE) reaction is primarily used for alkyl chain elongation,68 having wide applications in the synthesis natural products.69a–c Cheng and co-workers demonstrated a tandem electrochemical reaction highlighting the cathodic reduction of the substrate with ammonia as the hydrogen donor in the presence of a base (Scheme 14).70 A catalytic amount of base was generated electrochemically from methanol and resulted in the phosphonoacetate anion Int. 28. Thereafter, the HWE reaction resulted in the desired products in high yield. Such metal and oxidant-free protocol was employed in the synthesis of lipid raft modulator 112 and potential anti-influenza derivative 113.
 | ||
| Scheme 14 Electrochemical tandem olefination and hydrogenation with ammonia (ref. 70). | ||
In 2023, Baran and co-workers reported on the nickel-catalysed enantioselective double decarboxylative cross-coupling of redox active esters and malonate derivatives.71a The reaction profile featured the formation of two radicals, where coupling can be controlled by the careful choice of the ligand. A comparative study of the conventional synthetic pathways of a series of various biologically active molecules and their electrochemical pathways was proposed in Scheme 15. For instance, Fluvirucinin B1 is a 14-membered macrolactam produced by Actinomadura vulgaris, and is active against a variety of fungi, as well as influenza A.71b Penicitide A exhibited moderate cytotoxicity against pathogen Alternaria brassicae and the human hepatocellular liver carcinoma (HepG2) cell line. Again, 3-amino chroman derivatives and compounds are also moderately used in the treatment of depression and anxiety.
 | ||
| Scheme 15 Enantioselective doubly decarboxylative C(sp3)–C(sp3) cross coupling (ref. 71a). | ||
2.4 Electrochemical annulation or cyclisation
Intramolecular annulation reactions are commonly used as a tool in the assembling of cyclic scaffolds with sequential formation of new C–C or C–X bonds. Such processes are mostly influenced by transition metals and the use of very strong oxidents.72a–c However, in terms of sustainability, an alternative solution is highly desirable. In 2018, Xu and co-workers proposed the ferrocene-catalysed electrochemical dehydrogenative cyclisation of 1,3-dicarbonyls via functionalisation of C(sp3)–H/C(sp2)–H bonds. 1,3-Dicarbonyls were employed for the facile oxidative cleavage of acidic hydrogen (activated by the iron catalyst), and facilitated the generation of an electrophilic carbon-centred radical. This protocol avoids the use of organic oxidants and transition metal-catalyzed aerobic oxidations used in earlier studies. Mechanistic studies confirmed the formation of carbanion via abstraction of the α-C–H proton, followed by a single-electron oxidation in the presence of the anodically generated oxidised ferrocene. Cyclisation and re-aromatisation gave access to the indole-based skeleton and extended to the synthesis of the precursor of ketorolac 128, known as an NSAID drug (Scheme 16).73a | ||
| Scheme 16 Electrochemical dehydrogenative cyclisation to ketorolac (ref. 73a). | ||
3α-Bromopyrrolindolines are useful elementary units and provide access for late-stage functionalisation of C–Br bonds with retention of the configuration.74a–c Inspired by this idea, Vincent and co-workers reported an eco-friendly electrochemical halocyclisation protocol via dearomatisation of tryptophol, tryptamine and tryptophan derivatives, which are significantly used for the total synthesis of various alkaloids. An electrochemically generated electrophilic halogen intermediate (Br+) was formed via oxidation of the bromide salt (MgBr2), which also served as the electrolyte, thereby minimizing waste generation. The bromonium ion assisted in the C2–C3 double bond de-aromatisation, leading to the final halocyclized products (Scheme 17).75a Furthermore, the catalytic protocol was employed in the synthesis of precursors of various biologically active moieties, 132, 133, 135, and 137.
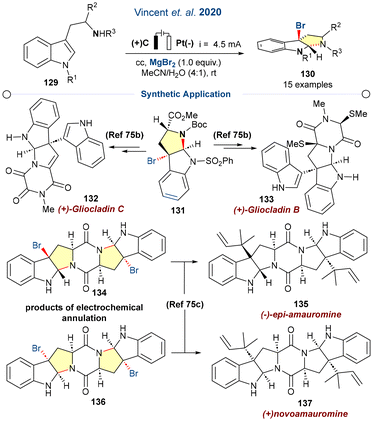 | ||
| Scheme 17 Electrochemical synthesis of 3a-bromofuranoindolines and 3a-bromopyrrolindolines (ref. 75a). | ||
In 2022, Guo and coworkers developed an electrochemical methodology for the di-functionalisation of alkynes (Scheme 18).76a Access to different indolizine aldehydes and ketones via intramolecular amino-oxidation was obtained in a single step.
 | ||
| Scheme 18 Electrochemical intramolecular 1,2-amino oxygenation (ref. 76a). | ||
Chen and co-workers developed a metal-free electrochemical protocol for de-hydrogenative C–H bond amination of arylsulfonamides.77 Iodination at the N-centre of sulfonamide in the presence of a strong base resulted in the homolytic cleavage, including the N-centred radical. The reactive radical facilitates an intramolecular cyclisation with an aryl ring. Finally, deprotonation followed by re-aromatisation furnished the carbazole scaffold. The catalytic protocol was employed in the synthesis of clausine, a naturally occurring alkaloid as presented in Scheme 19.
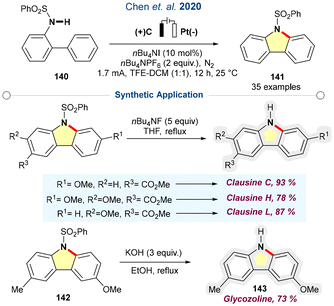 | ||
| Scheme 19 Scalable electrochemical dehydrogenative cross coupling amination-enabled alkaloid clausine syntheses (ref. 77). | ||
5-Bromoindolines are substantially found in many natural products and bioactive compounds.78a,b Classical approaches for the synthesis of these moieties required stoichiometric amounts of potent brominating regents, such as bromine gas, NBS, and BBr3.79a,b The frequent handling and storage of such brominating regents raised serious concern. In this direction, Ren and co-workers designed an electrochemical protocol for dearomative halocyclisation in the presence of NaBr, serving as a reactant as well as electrolyte in water. Considering the mutual effect of bromide electro-oxidation and reductive hydro de-bromination, two different products 145 and 146 were obtained (Scheme 20).80 The process was employed in the synthesis of the histamine derivative 148.
 | ||
| Scheme 20 Electrochemical synthesis of 3a- or 5a-bromoindolines and histamine core in water (ref. 80). | ||
2.5 Electrochemical asymmetric transformations
Chiral compounds bearing heterocycles are of significant interest in pharmaceutical industries.81a–d In this direction, the enantioselective pyridinylation that was demonstrated by Cheng and co-workers using a chiral palladium-complex is noteworthy. The chiral palladium-complex played dual roles as the chiral catalyst, as well as electron-transfer mediator, in the asymmetric allyl-pyridinylation process (Scheme 21a).82 4-Cyano-pyridine has proven to be the most adapted pyridylation reagent in photoredox-, electro- and in thermal catalyses.83a–c It was observed that the in situ generated Pd(0)-species facilitated the allyl Pd(II)-complex Int. 39 with cinnamyl acetate. This allyl complex undergoes cathodic reduction, followed by electron transfer to 4-cyano-pyridine 149, resulting in complex Int. 41. The subsequent reductive elimination furnished the pyridinylated product 151. Syntheses of the tachykinin/serotonin-reuptake inhibitor 153, used as antidepressants, and the calcium receptor modulator 155, which maintains calcium levels in blood, were obtained in high yield. | ||
| Scheme 21 (a) Palladium-catalyzed asymmetric allylic 4-pyridinylation (ref. 82). (b) Dual electrocatalysis enables enantioselective hydrocyanation (ref. 85). | ||
Chiral nitriles have wide application in pharmaceuticals, cosmetics, and in agrochemical industries.84a,b In 2020, Lin and co-workers demonstrated the synthesis of a series of chiral nitriles, merging two parallel radical cycles, which took place at the anode. It was observed that the cobalt-catalysed hydrogen atom transfer and copper-catalysed cyanation for the di-functionalisation of an alkene are the key for successful transformations (Scheme 21b).85 BOX-type ligands were used to control the product selectivity imparted by steric hindrance. Moreover, the presence of ester functionalities on the ligand coordinated with the substrate, and is the key for C–CN bond formation. Mechanistic studies revealed that cobalt-hydride species Int. 44 was generated using a hydrosilane, and reacted with alkene. As proposed, in another catalytic cycle, the CuII–CN intermediate Int. 45 is formed. Finally, the reductive elimination completed the hydrocyanation cycle, and produced reactive copper and cobalt species. Catalytic protocols were used in the synthesis of the drugs, naproxen and duocarmycin, including the precursor for palonosetron 161, which is used to prevent nausea and vomiting.
2.6 Electrochemical dimerisation
TMTD 163, or thiram (tetramethylthiuram disulphide), is an important fungicide produced by a number of plants, and is widely used in crop and seed protection.86a,b It has wide applications as an activator in the rubber industry.87 However, the conventional synthesis of such compounds is associated with over-stoichiometric waste. In this direction, electrochemical dimerisation has become increasingly attractive, owing to the facile and direct access to a plethora of biologically and pharmaceutically active compounds. In 2023, Terent'ev and co-workers utilised a combination of amines and carbon disulphide under constant current conditions in an undivided electrochemical cell for the synthesis of thiram and its analogues (Scheme 22a).88 Potassium iodide played dual roles as a redox active catalyst and an electrolyte. As proposed, mechanistic studies suggested that a hydroxide anion formed at the cathode and assisted the formation of di-thiocarbamate via nucleophilic attack of 162 on CS2. Next, iodide was oxidised to molecular iodine at the anode and simultaneously assisted the dimerisation to 163. | ||
| Scheme 22 (a) Electrochemical dimerisation for the synthesis of fungicides (ref. 88). (b) Electrochemical dimerisation of 3-substituted-2-oxindoles for the syntheses of folicanthine and chimonanthine (ref. 90). | ||
Oxindoles are omnipresent in various naturally occurring organic compounds. Similarly, dimerised 3-substituted 2-oxindoles are present in the core structure of a range of bis(cyclotryptamine) and tris(cyclotriptamine) alkaloids.89a,b Bisai and Paul demonstrated an indirect electrochemical methodology using TEMPO as the redox mediator, thereby circumventing electrode passivation, which resulted in the direct oxidation/reduction at the electrode surfaces. This protocol allows the dimerisation of 3-substituted 2-oxindoles under constant potential electrolysis in a three-electrode setup (Scheme 22b).90
The oxidation potentials of 2-oxindoles were responsible for driving either two-electron or one-electron-transfer pathways. Furthermore, this simple and environment-friendly strategy has been employed in the total synthesis of dimeric hexahydropyrrolo[2,3-b]indole alkaloids, and (±)-chimonanthine 166, which can be easily converted to another alkaloid (±)-folicanthine 167 using aqueous paraformaldehyde and sodium tri-acetoxyborohydride.
2.7 Heterogeneous electrochemical transformations
The Achmatowicz reaction converts biomass-derived furfuryl alcohols to di-hydropyranone acetals, and often generates a new chiral centre. The Achmatowicz rearrangement also allows the application of N-bromosuccinimide, meta-chloroperoxybenzoic acid, dimethyldioxiran, and mono-oxygenase for the generation of hydropyranones.91–95 However, these chemical oxidations require stoichiometric oxidants and expensive or toxic solvents. Therefore, a more sustainable and benign technology is highly desirable for such transformations. Sun and co-workers demonstrated a heterogeneous version of this rearrangement under electrochemical conditions (Scheme 23a).96 The heterogeneous Ni-electrocatalyst was synthesized via photo-induced immobilisation of 5,5′-divinyl-2,2′-bipyridine (DVBP). Incorporation of nickel was achieved in unsaturated coordination spheres, thereby establishing the interaction between furfuryl alcohol and nickel, which in turn enabled hydroxide transfer from nickel to furfuryl alcohol. This facile electrochemical protocol demonstrated the synthesis of (−)-Aspergillide A 171 and halichondrin 173. | ||
| Scheme 23 (a) Electrocatalytic synthesis of heterocycles from biomass-derived furfuryl alcohols (ref. 96). (b) Electrocatalytic synthesis of essential amino acids (ref. 98). | ||
One of the major pollutants generated via combustion of fossil fuels are nitrous oxides (NOx), which has caused hazardous environmental issues over the years. Despite the urge to develop efficient and inexpensive NOx removal techniques, many of these methods depend on harsh conditions employing high energy or the use of expensive precious metals.97a,b However, the electrochemical NOx reduction into value-added chemicals is a promising technology. In 2023, Li and co-workers reported the first method for the synthesis of essential α-amino acids from α-keto acids via an electroreductive NO fixation strategy (Scheme 23b).98 The catalytic reaction was performed in aqueous media using an atomically dispersed Fe-catalyst on a N-doped carbon matrix (defined as AD-Fe/NC). The formation of amino acids required three steps, as suggested by in situ X-ray absorption fine structure (XAFS) and synchrotron radiation infrared spectroscopy (SRIR). As proposed, the initial formation of oxime via C–N coupling is crucial. Subsequently, the hydrogenation resulted the amino acid.
2.8 Miscellaneous electrochemical transformations
Recently, Lennox and co-workers established an improved route towards the synthesis of difluoromethyl ketones from trifluoromethyl ketones following hydro-defluorination technology, which circumvents the need of stoichiometric metals.99 Trifluoromethyl ketones were efficiently reduced at the anode and reacted with TMSCl, followed by second reduction and subsequent dehydrofluorination, which resulted in the desired products 184 and 185 (Scheme 24). | ||
| Scheme 24 Electrochemical hydrodefluorination to difluoromethyl ketones (ref. 99). | ||
Recently, Baran and co-workers described an elegant and simple electrochemical protocol for the transformation of activated acids to their borylated analogues (Scheme 25). Borylated compounds are one of the useful intermediates used for various functional group transformations. This electrochemical transformation was employed in the synthesis of the precursor 190 of jawsamycin 191, an oligocyclopropyl containing natural product with antifungal activity.100
 | ||
| Scheme 25 Electrochemically enabled borylation for the synthesis of Jawsamycin (ref. 100). | ||
Very recently, Ma and co-workers reported an interesting electrochemical sulfonylation of organoboronic acids to functionalised sulfones under metal-free conditions (Scheme 26).101 Bench-stable sodium arylsulfinate salts 194 were used for this transformation, and overcome the associated limitations of conventional sulfonylating agents, such as sulfonyl halides and sulphur dioxide. This metal- and oxidative-free protocol enabled the synthesis of biologically active sulfone dapsone 197, which is widely used as antibacterial drug in combination with rifampicin and clofazimine for the treatment of leprosy.
 | ||
| Scheme 26 Electrochemical sulfonylation: synthesis of antibacterial drug dapsone (ref. 101). | ||
The pyrazole core is omnipresent in various anti-cancer drugs, and has attracted significant biological importance due to its anti-psychotic, anti-inflammatory, as well as anti-bacterial properties. Conventional processes for their synthesis are associated with stoichiometric oxidants and other toxic chemicals.102 However, electrochemical transformations of pyrazoles represent a greener and sustainable approach.103–106 In 2023, Waldvogel and co-workers reported an oxidative electrochemical approach for the transformations of pyrazolines to pyrazoles mediated by NaCl, acting as a supporting electrolyte, as well as mediator (Scheme 27).107 Robust and abundant boron-doped diamond electrodes, in combination with a green solvent system, water and ethyl acetate, facilitate the transformations in high yield. Analogous to previous literature, the authors have also proposed an ionic mechanism for this oxidative aromatisation via initial chlorination at the 2-position of nitrogen atom. Subsequently, a proton is elimination followed by aromatisation via removal of HCl, which results in the desired product. Synthesis of the key intermediate of fungicide pyraclostrobin 202 was obtained using this process. Again, double electrochemical conversion of ethyl glyoxalate phenyl hydrazine 203 to 204 established the synthetic potential of the protocol.108
 | ||
| Scheme 27 Electrochemical oxidative aromatisation for the synthesis of pyraclostrobin (ref. 107). | ||
3. Conclusion and outlook
Electrochemical organic transformations widely used to streamline the synthesis of complex molecules are evident from the illustrations provided in this review. Utilisation of toxic gases, such as carbon dioxide, nitrous oxides (NOx, a major air pollutant), has remained challenging owing to their activation and insertion into an organic moiety. These challenges have been successfully overcome using modern electrochemical techniques. Applications of organic electrochemistry have proven to be robust and sustainable in contrast to conventional methods via replacement of stoichiometric amounts of hazardous reagents with electrons. There are numerous advantages that rate electrochemistry as superior to conventional strategies in terms of its sustainability. For instance, electrochemical strategies minimised the waste generation, improved efficiency, can be used under mild operational conditions, and enabled direct target molecule synthesis with greater product selectivity. The ability of scaling up electrochemical reactions via utilisation of flow electrochemical cells has been the most significant advantage observed so far. This has enabled the direct commercialisation of a proposed synthetic strategy in industrial scales. Such technique considered as a major challenge in industrial synthesis using chemical reagents, as a large amount of unwanted waste is generated. Electrochemistry has immensely outshined our expectations towards achieving a greener chemical outlook, and would streamline the roadmap for further development.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors thank SERB-India (CRG/2021/002686) for financial support. MoE-STARS/STARS-2/2023-1003 and IIT Roorkee (SMILE-32) are gratefully acknowledged for instrument facilities. A. G. thanks PMRF and V. K. P. thanks UGC-India for financial support.References
-
(a) B. A. Frontana-Uribe, R. D. Little, J. G. Ibanez, A. Palma and R. Vasquez-Medrano, Green Chem., 2010, 12, 2099–2119 RSC
; (b) E. J. Horn, B. R. Rosen and P. S. Baran, ACS Cent. Sci., 2016, 2, 302–308 CrossRef CAS PubMed
; (c) Y. H. Budnikova, E. L. Dolengovski, M. V. Tarasov and T. V. Gryaznova, J. Solid State Electrochem., 2024, 28, 659–676 CrossRef CAS
.
- D. P. Rotella, ACS Chem. Neurosci., 2016, 7, 1315–1316 CrossRef CAS PubMed
.
- T. Cernak, K. D. Dykstra, S. Tyagarajan, P. Vachal and S. W. Krska, Chem. Soc. Rev., 2016, 45, 546–576 RSC
.
- D. C. Blakemore, L. Castro, I. Churcher, D. C. Rees, A. W. Thomas, D. M. Wilson and A. Wood, Nat. Chem., 2018, 10, 383–394 CrossRef CAS PubMed
.
- S. Möhle, M. Zirbes, E. Rodrigo, T. Gieshoff, A. Wiebe and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 6018–6041 CrossRef PubMed
.
-
(a) C. Zhu, N. W. J. Ang, T. H. Meyer, Y. Qiu and L. Ackermann, ACS Cent. Sci., 2021, 7, 415–431 CrossRef CAS PubMed
; (b) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 CrossRef CAS PubMed
; (c) T. H. Meyer, I. Choi, C. Tian and L. Ackermann, Chem, 2020, 6, 2484–2496 CrossRef CAS
.
-
(a) C. Schotten, T. P. Nicholls, R. A. Bourne, N. Kapur, B. N. Nguyen and C. E. Willans, Green Chem., 2020, 22, 3358–3375 RSC
; (b) L. F. T. Novaes, J. Liu, Y. Shen, L. Lu, J. M. Meinhardt and S. Lin, Chem. Soc. Rev., 2021, 50, 7941–8002 RSC
; (c) Q. Jing and K. D. Moeller, Acc. Chem. Res., 2020, 53, 135–143 CrossRef CAS PubMed
; (d) Y. Wang, S. Dana, H. Long, Y. Xu, Y. Li, N. Kaplaneris and L. Ackermann, Chem. Rev., 2023, 123, 11269–11335 CrossRef CAS PubMed
.
-
(a) F. Zhang, H. Zhang and Z. Liu, Curr. Opin. Green Sustainable Chem., 2019, 16, 77–84 CrossRef
; (b) S. Overa, B. H. Ko, Y. Zhao and F. Jiao, Acc. Chem. Res., 2022, 55, 638–648 CrossRef CAS PubMed
.
-
(a) A. M. F. Phillips and A. J. L. Pombeiro, Org. Biomol. Chem., 2020, 18, 7026–7055 RSC
; (b) D. I. Park, S. Jung, H. J. Yoon and K. Jin, Electrochim. Acta, 2021, 397, 139271 CrossRef CAS
; (c) M. Ghosh, V. S. Shinde and M. Rueping, Beilstein J. Org. Chem., 2019, 15, 2710–2746 CrossRef CAS PubMed
; (d) K. Yamamoto, M. Kuriyama and O. Onomura, Curr. Opin. Electrochem., 2019, 15, 2710–2746 Search PubMed
.
-
(a) A. G. Tamirat, X. Guan, J. Liu, J. Luo and Y. Xia, Chem. Soc. Rev., 2020, 49, 7454–7478 RSC
; (b) W. Shao, B. Lu, J. Cao, J. Zhang, H. Cao, F. Zhang and C. Zhang, Chem. – Asian J., 2023, 18, e202201093 CrossRef CAS PubMed
.
- M. Munda, S. Niyogi, K. Shaw, S. Kundu, R. Nandi and A. Bisai, Org. Biomol. Chem., 2022, 20, 727–748 RSC
.
- D. Cantillo, Chem. Commun., 2022, 58, 619–628 RSC
.
- C. E. Hatch and W. J. Chain, ChemElectroChem, 2023, 10, e202300140 CrossRef CAS PubMed
.
-
A. Ghosh, A. Bera, S. Bera and D. Banerjee, Electrochemical Oxidative C-H Functionalization: A Metal-Free Approach, in Handbook of CH-Functionalization, ed. D. Maiti, 2022, pp. 1–27. DOI:10.1002/9783527834242.chf0224
.
-
(a) M. Vellakkaran, K. Singh and D. Banerjee, ACS Catal., 2017, 7, 8152–8158 CrossRef CAS
; (b) K. Singh, M. Vellakkaran and D. Banerjee, Green Chem., 2018, 20, 2250–2256 RSC
; (c) L. M. Kabadwal, J. Das and D. Banerjee, Chem. Commun., 2018, 54, 14069–14072 RSC
; (d) J. Das, M. Vellakkaran, M. Sk and D. Banerjee, Org. Lett., 2019, 21, 7514–7518 CrossRef CAS PubMed
; (e) A. Ghosh, A. Bera and D. Banerjee, ChemCatChem, 2023, 15, e202201433 CrossRef CAS
; (f) M. Sk, A. Bera and D. Banerjee, ChemCatChem, 2023, 15, e202300412 CrossRef CAS
.
- S.-S. Yan, Q. Fu, L.-L. Liao, G.-Q. Sun, J.-H. Ye, L. Gong, Y.-Z. Bo-Xue and D.-G. Yu, Coord. Chem. Rev., 2018, 374, 439–463 CrossRef CAS
.
-
(a) A. Tortajada, F. Juliá-Hernández, M. Börjesson, T. Moragas and R. Martin, Angew. Chem., Int. Ed., 2018, 57, 15948–15982 CrossRef CAS PubMed
; (b) C. K. Ran, X. W. Chen, Y. Y. Gui, J. Liu, L. Song, K. Ren and D. G. Yu, Sci. China: Chem., 2020, 63, 1336–1351 CrossRef CAS
.
- G. Wang, J. Chen, Y. Ding, P. Cai, L. Yi, Y. Li, C. Tu, Y. Hou, Z. Wen and L. Dai, Chem. Soc. Rev., 2021, 50, 4993–5061 RSC
.
-
(a) K. Nemoto, S. Onozawa, N. Egusa, N. Morohashi and T. Hattori, Tetrahedron Lett., 2009, 50, 4512–4514 CrossRef CAS
; (b) K. Nemoto, S. Onozawa, M. Konno, N. Morohashi and T. Hattori, Bull. Chem. Soc. Jpn., 2012, 85, 369–371 CrossRef CAS
; (c) K. Inamoto, N. Asano, Y. Nakamura, M. Yonemoto and Y. Kondo, Org. Lett., 2012, 14, 2622–2625 CrossRef CAS PubMed
; (d) I. I. F. Boogaerts, G. C. Fortman, M. R. L. Furst, C. S. J. Cazin and S. P. Nolan, Angew. Chem., Int. Ed., 2010, 49, 8674–8677 CrossRef CAS PubMed
.
-
(a) I. Reche, I. Gallardo and G. Guirado, RSC Adv., 2014, 4, 65176–65183 RSC
; (b) J. Albo, M. Alvarez-Guerra, P. Castaño and A. Irabien, Green Chem., 2015, 17, 2304–2324 RSC
.
- H. Maag, Prodrugs, 2008, 703–729 Search PubMed
.
-
(a) S. Bazzi, E. Schulz and M. Mellah, Org. Lett., 2019, 21, 10033–10037 CrossRef CAS PubMed
; (b) T. León, A. Correa and R. Martin, J. Am. Chem. Soc., 2013, 135, 1221–1224 CrossRef PubMed
.
-
(a) K. Nogi, T. Fujihara, J. Terao and Y. Tsuji, J. Org. Chem., 2015, 80, 11618–11623 CrossRef CAS PubMed
; (b) F. Rebih, M. Andreini, A. Moncomble, A. Harrison-Marchand, J. Maddaluno and M. Durandetti, Chem. – Eur. J., 2016, 22, 3758–3763 CrossRef CAS PubMed
; (c) A. Correa, T. Leon and R. Martin, J. Am. Chem. Soc., 2014, 136, 1062–1069 CrossRef CAS PubMed
; (d) T. Mita, Y. Higuchi and Y. Sato, Chem. – Eur. J., 2015, 21, 16391–16394 CrossRef CAS PubMed
; (e) M. van Gemmeren, M. Börjesson, A. Tortajada, S. Sun, K. Okura and R. Martin, Angew. Chem., 2017, 129, 6658–6662 CrossRef
; (f) Y. G. Chen, B. Shuai, C. Ma, X. J. Zhang, P. Fang and T. S. Mei, Org. Lett., 2017, 19, 2969–2972 CrossRef CAS PubMed
.
- D. T. Yang, M. Zhu, Z. J. Schiffer, K. Williams, X. Song, X. Liu and K. Manthiram, ACS Catal., 2019, 9, 4699–4705 CrossRef CAS
.
- J.-S. Zhong, Z.-X. Yang, C.-L. Ding, Y.-F. Huang, Y. Zhao, H. Yan and K.-Y. Ye, J. Org. Chem., 2021, 86, 16162–16170 CrossRef CAS PubMed
.
- S. Mena, J. Sanchez and G. Guirado, RSC Adv., 2019, 9, 15115–15123 RSC
.
- A. M. Sheta, A. Alkayal, M. A. Mashaly, S. B. Said, S. S. Elmorsy, A. V. Malkov and B. R. Buckley, Angew. Chem., Int. Ed., 2021, 60, 21832–21837 CrossRef CAS PubMed
.
-
(a) H. Huang, J.-H. Ye, L. Zhu, C.-K. Ran, M. Miao, W. Wang, H. Chen, W.-J. Zhou, Y. Lan, B. Yu and D.-G. Yu, CCS Chem., 2020, 2, 1746–1756 Search PubMed
; (b) H. Wang, Y.-F. Du, M.-Y. Lin, K. Zhang and J.-X. Lu, Chin. J. Chem., 2008, 26, 1745–1748 CrossRef CAS
; (c) S. Kawashima, K. Aikawa and K. Mikami, Eur. J. Org. Chem., 2016, 3166–3170 CrossRef CAS
.
-
(a) C. Li, G. Yuan and H. Jiang, Chin. J. Chem., 2010, 28, 1685–1689 CrossRef CAS
; (b) S. Dérien, J.-C. Clinet, E. Duñach and J. Périchon, Tetrahedron, 1992, 48, 5235–5248 CrossRef
; (c) T. Fujihara, Y. Horimoto, T. Mizoe, F. B. Sayyed, Y. Tani, J. Terao, S. Sakaki and Y. Tsuji, Org. Lett., 2014, 16, 4960–4963 CrossRef CAS PubMed
.
-
(a) T. Ju, Y.-Q. Zhou, K.-G. Cao, Q. Fu, J.-H. Ye, G.-Q. Sun, X.-F. Liu, L. Chen, L.-L. Liao and D.-G. Yu, Nat. Catal., 2021, 4, 304–311 CrossRef CAS
; (b) A. Tortajada, R. Ninokata and R. Martin, J. Am. Chem. Soc., 2018, 140, 2050–2053 CrossRef CAS PubMed
; (c) M. Shigeno, K. Sasaki, K. Nozawa-Kumada and Y. Kondo, Org. Lett., 2019, 21, 4515–4519 CrossRef CAS PubMed
.
- W. Zhang and S. Lin, J. Am. Chem. Soc., 2020, 142, 20661–20670 CrossRef CAS PubMed
.
-
(a) K. Nemoto, S. Onozawa, N. Egusa, N. Morohashi and T. Hattori, Tetrahedron Lett., 2009, 50, 4512–4514 CrossRef CAS
; (b) K. Nemoto, S. Tanaka, M. Konno, S. Onozawa, M. Chiba, Y. Tanaka, Y. Sasaki, R. Okubo and T. Hattori, Tetrahedron, 2016, 72, 734–745 CrossRef CAS
; (c) M. Shigeno, I. Tohara, K. Nozawa-Kumada and Y. Kondo, Eur. J. Org. Chem., 2020, 2020, 1987–1991 CrossRef CAS
.
-
(a) Y. You, W. Kanna, H. Takano, H. Hayashi, S. Maeda and T. Mita, J. Am. Chem. Soc., 2022, 144, 3685–3695 CrossRef CAS PubMed
; (b) A. Monge, I. Aldana, T. Alvarez, M. Font, E. Santigo, J. A. Latre, M. J. Bermejillo, M. J. Lopez-Unzu and E. Fernandez-Alvarez, J. Med. Chem., 1991, 34, 3023–3029 CrossRef CAS PubMed
; (c) T. Ladduwahetty and A. M. MacLeod, Pyridazino-Indole Derivatives, US Patent5693640, 1997 Search PubMed
.
-
(a) M. Masui, S. Hara, T. Ueshima, T. Kawaguchi and S. Ozaki, Chem. Pharm. Bull., 1983, 31, 4209–4211 CrossRef CAS
; (b) M. Masui, T. Kawaguchi and S. Ozaki, J. Chem. Soc., Chem. Commun., 1985, 1484–1485 RSC
.
- M. Rafiee, F. Wang, D. P. Hruszkewycz and S. S. Stahl, J. Am. Chem. Soc., 2018, 140, 22–25 CrossRef CAS PubMed
.
- J. C. Lewis, P. S. Coelho and F. H. Arnold, Chem. Soc. Rev., 2011, 40, 2003–2021 RSC
.
-
(a) T. Newhouse and P. S. Baran, Angew. Chem., Int. Ed., 2011, 50, 3362–3374 CrossRef CAS PubMed
; (b) H. Sterckx, B. Morel and B. U. W. Maes, Angew. Chem., Int. Ed., 2019, 58, 7946–7970 CrossRef CAS PubMed
; (c) L. M. Stateman, K. M. Nakafuku and D. A. Nagib, Synthesis, 2018, 50, 1569–1586 CrossRef CAS PubMed
; (d) A. Gunay and K. H. Theopold, Chem. Rev., 2010, 110, 1060–1081 CrossRef CAS PubMed
; (e) L. Zou, R. S. Paton, A. Eschenmoser, T. R. Newhouse, P. S. Baran and K. N. Houk, J. Org. Chem., 2013, 78, 4037–4048 CrossRef CAS PubMed
.
- Y. Kawamata, M. Yan, Z. Liu, D. H. Bao, J. Chen, J. T. Starr and P. S. Baran, J. Am. Chem. Soc., 2017, 139, 7448–7451 CrossRef CAS PubMed
.
- J. M. Howell, K. Feng, J. R. Clark, L. J. Trzepkowski and M. C. White, J. Am. Chem. Soc., 2015, 137, 14590–14593 CrossRef CAS PubMed
.
- M. Saito, Y. Kawamata, M. Meanwell, R. Navratil, D. Chiodi, E. Carlson, P. Hu, L. Chen, S. Udyavara, C. Kingston, M. Tanwar, S. Tyagi, B. P. McKillican, M. G. Gichinga, M. A. Schmidt, M. D. Eastgate, M. Lamberto, C. He, T. Tang, C. A. Malapit, M. S. Sigman, S. D. Minteer, M. Neurock and P. S. Baran, J. Am. Chem. Soc., 2021, 143, 7859–7867 CrossRef CAS PubMed
.
- X.-W. Wang, Y. Deng, R.-X. Li, J.-F. Lv, M.-Q.-H. Fu, Z. Guan, Y.-N. Zhao and Y.-H. He, ACS Sustainable Chem. Eng., 2023, 11, 1624–1630 CrossRef CAS
.
- T. Li, L. Pan, Y. Zhang, J. Su, K. Li, K. Li, H. Chen, Q. Sun and Z. Wang, Chin. Chem. Lett., 2023, 108897 Search PubMed
.
- D. A. Petrone, J. Ye and M. Lautens, Chem. Rev., 2016, 116, 8003–8104 CrossRef CAS PubMed
.
- M. Konishi, K. Tsuchida, K. Sano, T. Kochi and F. Kakiuchi, J. Org. Chem., 2017, 82, 8716–8724 CrossRef CAS PubMed
.
- Y. Wu, S. Xu, H. Wang, D. Shao, Q. Qi, Y. Lu, L. Ma, J. Zhou, W. Hu, W. Gao and J. Chen, J. Org. Chem., 2021, 86, 16144–16150 CrossRef CAS PubMed
.
- L. Yang, Z. Liu, Y. Li, N. Lei, Y. Shen and K. Zheng, Org. Lett., 2019, 21, 7702–7707 CrossRef CAS PubMed
.
- J. Park, M. A. Kelly, J. X. Kang, S. S. Seemakurti, J. L. Ramirez, M. C. Hatzell, C. Sievers and A. S. Bommarius, Green Chem., 2021, 23, 7488–7498 RSC
.
- I. M. Taily, D. Saha and P. Banerjee, Org. Lett., 2022, 24, 2310–2314 CrossRef CAS PubMed
.
-
(a) D. A. Everson and D. J. Weix, J. Org. Chem., 2014, 79, 4793–4798 CrossRef CAS PubMed
; (b) D. G. Yu, B. J. Li and Z. J. Shi, Acc. Chem. Res., 2010, 43, 1486–1495 CrossRef CAS PubMed
.
-
(a) D. J. Weix, Acc. Chem. Res., 2015, 48, 1767–1775 CrossRef CAS PubMed
; (b) J. Gu, C. Qiu, W. Lu, Q. Qian, K. Lin and H. Gong, Synthesis, 2017, 49, 1867–1873 CrossRef CAS
; (c) P. Zhang, C. C. Le and D. W. C. MacMillan, J. Am. Chem. Soc., 2016, 138, 8084–8087 CrossRef CAS PubMed
; (d) M. O. Konev, L. E. Hanna and E. R. Jarvo, Angew. Chem., Int. Ed., 2016, 55, 6730–6733 CrossRef CAS PubMed
; (e) A. H. Cherney and S. E. Reisman, J. Am. Chem. Soc., 2014, 136, 14365–14368 CrossRef CAS PubMed
.
- B. L. Truesdell, T. B. Hamby and C. S. Sevov, J. Am. Chem. Soc., 2020, 142, 5884–5893 CrossRef CAS PubMed
.
-
(a) R. Shang and L. Liu, Sci. China: Chem., 2011, 54, 1670–1687 CrossRef CAS
; (b) N. Rodríguez and L. J. Goossen, Chem. Soc. Rev., 2011, 40, 5030–5048 RSC
.
- Y. Wei, P. Hu, M. Zhang and W. Su, Chem. Rev., 2017, 117, 8864–8907 CrossRef CAS PubMed
.
-
(a) P. Qian, Z. Zhou, L. Wang, Z. Wang, Z. Wang, Z. Zhang and L. Sheng, J. Org. Chem., 2020, 85, 13029–13036 CrossRef CAS PubMed
; (b) N. Venkatagiri, T. Krishna, P. Thirupathi, K. Bhavani and C. K. Reddy, Russ. J. Gen. Chem., 2018, 88, 1488–1494 CrossRef CAS
.
- G. Bonola, P. D. Re, M. J. Magistretti, E. Massarani and I. Setnikar, J. Med. Chem., 1968, 11, 1136–1139 CrossRef CAS PubMed
.
- A. Cagir, S. H. Jones, R. Gao, B. M. Eisenhauer and S. M. Hecht, J. Am. Chem. Soc., 2003, 125, 13628–13629 CrossRef CAS PubMed
.
- K. M. Khan, S. M. Saad, N. N. Shaikh, S. Hussain, M. I. Fakhri, S. Perveen, M. Taha and M. I. Choudhary, Bioorg. Med. Chem., 2014, 22, 3449–3454 CrossRef CAS PubMed
.
-
(a) R. J. Abdel-Jalil, W. Voelter and M. Saeed, Tetrahedron Lett., 2004, 45, 3475–3476 CrossRef CAS
; (b) D. Zhan, T. Li, H. Wei, W. Weng, K. Ghandi and Q. Zeng, RSC Adv., 2013, 3, 9325–9329 RSC
.
-
(a) S. You, B. Huang, T. Yan and M. Cai, J. Organomet. Chem., 2018, 875, 35–45 CrossRef CAS
; (b) H. Li, L. He, H. Neumann, M. Beller and X. F. Wu, Green Chem., 2014, 16, 1336–1343 RSC
.
-
(a) L. X. Wang, J. F. Xiang and Y. L. Tang, Eur. J. Org. Chem., 2014, 2014, 2682–2685 CrossRef CAS
; (b) N. Yan, C. You and M. Cai, J. Organomet. Chem., 2019, 897, 161–169 CrossRef CAS
.
- L. Yang, H. Hou, L. Li, J. Wang, S. Zhou, M. Wu and F. Ke, Org. Biomol. Chem., 2021, 19, 998–1003 RSC
.
-
(a) P. N. P. Rao, Q. H. Chen and E. E. Knaus, J. Med. Chem., 2006, 49, 1668–1683 CrossRef CAS PubMed
; (b) M. Meng, G. Wang, L. Yang, K. Cheng and C. Qi, Adv. Synth. Catal., 2018, 360, 1218–1231 CrossRef CAS
.
- J. Liu and J. E. Bäckvall, Chem. – Eur. J., 2020, 26, 15513–15518 CrossRef CAS PubMed
.
- Y. Wu, L. Zeng, H. Li, Y. Cao, J. Hu, M. Xu, R. Shi, H. Yi and A. Lei, J. Am. Chem. Soc., 2021, 143, 12460–12466 CrossRef CAS PubMed
.
- D. G. Brown and J. Boström, J. Med. Chem., 2016, 59, 4443–4458 CrossRef CAS PubMed
.
-
(a) V. R. Pattabiraman and J. W. Bode, Nature, 2011, 480, 471–479 CrossRef CAS PubMed
; (b) A. B. Hughes, Amino Acids, Peptides and Proteins in Organic Chemistry, Wiley-VCH, Germany, 2011 CrossRef
.
- T. Alam, A. Rakshit, H. N. Dhara, A. Palai and B. K. Patel, Org. Lett., 2022, 24, 6619–6624 CrossRef CAS PubMed
.
- A. B. Juan and L. R. Orelli, Curr. Org. Chem., 2015, 19, 744–775 CrossRef
.
-
(a) L. Wang and T. L. Lowary, Org. Lett., 2020, 22, 9633–9637 CrossRef CAS PubMed
; (b) T. Noshita, K. Matsumoto, H. Nishikawa, H. Ouchi, Y. Hamada, A. Saito and T. Yamada, Nat. Prod. Res., 2017, 31, 163–168 CrossRef CAS PubMed
; (c) J. Boudreault, F. Lévesque and G. Bélanger, J. Org. Chem., 2016, 81, 9247–9268 CrossRef CAS PubMed
.
- X. Zhang, R. Jiang and X. Cheng, J. Org. Chem., 2021, 86, 16016–16025 CrossRef CAS PubMed
.
-
(a) Y. Gao, B. Zhang, J. He and P. S. Baran, J. Am. Chem. Soc., 2023, 145, 11518–11523 CrossRef CAS PubMed
; (b) G. Guignard, N. Llor, E. Molins, J. Bosch and M. Amat, Org. Lett., 2016, 18, 1788–1791 CrossRef CAS PubMed
.
-
(a) B. B. Snider, Chem. Rev., 1996, 96, 339–363 CrossRef CAS PubMed
; (b) V. Nair and A. Deepthi, Tetrahedron, 2009, 65, 10745–10755 CrossRef CAS
; (c) J. E. M. N. Klein, A. Perry, D. S. Pugh and R. J. K. Taylor, Org. Lett., 2010, 12, 3446–3449 CrossRef CAS PubMed
.
-
(a) Z.-J. Wu, S.-R. Li, H. Long and H.-C. Xu, Chem. Commun., 2018, 54, 4601–4604 RSC
; (b) D. R. Artis, I.-S. Cho and J. M. Muchowski, Can. J. Chem., 1992, 70, 1838–1842 CrossRef CAS
.
-
(a) M. Movassaghi and M. A. Schmidt, Angew. Chem., Int. Ed., 2007, 46, 3725–3728 CrossRef CAS PubMed
; (b) L. Furst, J. M. R. Narayanam and C. R. J. Stephenson, Angew. Chem., Int. Ed., 2011, 50, 9655–9659 CrossRef CAS PubMed
; (c) H. Hakamata, S. Sato, H. Ueda and H. Tokuyama, Org. Lett., 2017, 19, 5308–5311 CrossRef CAS PubMed
.
-
(a) J. Wu, H. Abou-Hamdan, R. Guillot, C. Kouklovsky and G. Vincent, Chem. Commun., 2020, 56, 1713–1716 RSC
; (b) H. Lei, L. Wang, Z. Xu and T. Ye, Org. Lett., 2017, 19, 5134–5137 CrossRef CAS PubMed
; (c) H. Hakamata, S. Sato, H. Ueda and H. Tokuyama, Org. Lett., 2017, 19, 5308–5311 CrossRef CAS PubMed
.
-
(a) Q.-L. Yang, R. C. Ma, Z. H. Li, W. W. Li, G. R. Qu and H. M. Guo, Org. Chem. Front., 2022, 9, 4990–4997 RSC
; (b) G. S. Singh and E. E. Mmatli, Eur. J. Med. Chem., 2011, 46, 5237–5257 CrossRef CAS PubMed
.
- P. Zhang, B. Li, L. Niu, L. Wang, G. Zhang, X. Jia, G. Zhang, S. Liu, L. Ma, W. Gao, D. Qin and J. Chen, Adv. Synth. Catal., 2020, 362, 2342–2347 CrossRef CAS
.
-
(a) W. Kong, Q. Wang and J. Zhu, J. Am. Chem. Soc., 2015, 137, 16028–16031 CrossRef CAS PubMed
; (b) A. E. M. Blom, J. Y. Su, L. M. Repka, S. E. Reisman and D. A. Dougherty, ACS Med. Chem. Lett., 2020, 11, 2204–2211 CrossRef CAS PubMed
.
-
(a) M. G. Kulkarni, A. P. Dhondge, A. S. Borhade, D. D. Gaikwad, S. W. Chavhan, Y. B. Shaikh, V. B. Nigdale, M. P. Desai, D. R. Birhade and M. P. Shinde, Eur. J. Org. Chem., 2009, 3875–3877 CrossRef CAS
; (b) M. Movassaghi, M. A. Schmidt and J. A. Ashenhurst, Angew. Chem., Int. Ed., 2008, 47, 1485–1487 CrossRef CAS PubMed
.
- Y.-A. Wu, R.-A. Wang, S.-Y. Jiang, T.-B. Jiang, J.-R. Song, J. Shi, W. Wu, W.-D. Pan and H. Ren, Green Chem., 2022, 6720–6726 RSC
.
-
(a) T. J. Tucker, W. C. Lumma, S. D. Lewis, S. J. Gardell, B. J. Lucas, J. T. Sisko, J. J. Lynch, E. A. Lyle, E. P. Baskin, R. F. Woltmann, S. D. Appleby, I. W. Chen, K. B. Dancheck, A. M. Naylor-Olsen, J. A. Krueger, C. M. Cooper and J. P. Vacca, J. Med. Chem., 1997, 40, 3687–3693 CrossRef CAS PubMed
; (b) R. P. Alexander, G. J. Warrellow, M. A. W. Eaton, E. C. Boyd, J. C. Head, J. R. Porter, J. A. Brown, J. T. Reuberson, B. Hutchinson, P. Turner, B. Boyce, D. Barnes, B. Mason, A. Cannell, R. J. Taylor, A. Zomaya, A. Millican, J. Leonard, R. Morphy, M. Wales, M. Perry, R. A. Allen, N. Gozzard, B. Hughes and G. Higgs, Bioorg. Med. Chem. Lett., 2002, 12, 1451–1456 CrossRef CAS PubMed
; (c) J. F. Hartwig and L. M. Stanley, Acc. Chem. Res., 2010, 43, 1461–1475 CrossRef CAS PubMed
; (d) J. C. Hethcox, S. E. Shockley and B. M. Stoltz, ACS Catal., 2016, 6, 6207–6213 CrossRef CAS PubMed
.
- W. Ding, M. Li, J. Fan and X. Cheng, Nat. Commun., 2022, 13, 1–11 Search PubMed
.
-
(a) F. Lima, M. A. Kabeshov, D. N. Tran, C. Battilocchio, J. Sedelmeier, G. Sedelmeier, B. Schenkel and S. V. Ley, Angew. Chem., 2016, 128, 14291–14295 CrossRef
; (b) D. Lehnherr, Y. H. Lam, M. C. Nicastri, J. Liu, J. A. Newman, E. L. Regalado, D. A. Dirocco and T. Rovis, J. Am. Chem. Soc., 2020, 142, 468–478 CrossRef CAS PubMed
; (c) L. Gao, G. Wang, J. Cao, H. Chen, Y. Gu, X. Liu, X. Cheng, J. Ma and S. Li, ACS Catal., 2019, 9, 10142–10151 CrossRef CAS
.
-
(a) W. A. Nugent and R. J. McKinney, J. Org. Chem., 1985, 50, 5370–5372 CrossRef CAS
; (b) X. Fang, P. Yu and B. Morandi, Science, 2016, 351, 832–836 CrossRef CAS PubMed
.
- L. Song, N. Fu, B. G. Ernst, W. H. Lee, M. O. Frederick, R. A. DiStasio and S. Lin, Nat. Chem., 2020, 12, 747–754 CrossRef CAS PubMed
.
-
(a) V. K. Sharma, J. S. Aulakh and A. K. Malik, J. Environ. Monit., 2003, 5, 717–723 RSC
; (b) V. Mancini and G. Romanazzi, Pest Manage. Sci., 2014, 70, 860–868 CrossRef CAS PubMed
.
-
J. V. Alphen, Rubber Chemicals, D. Reidel Publishing Company, 1973. pp. 128–148 Search PubMed
.
- O. M. Mulina, E. D. Bokova, M. M. Doronin and A. O. Terent'ev, ACS Agric. Sci. Technol., 2023, 3, 720–724 CrossRef CAS
.
-
(a) R. Long, J. Huang, J. Gong and Z. Yang, Nat. Prod. Rep., 2015, 32, 1584–1601 RSC
; (b) L. Verotta, T. Pilati, M. Tato, E. Elisabetsky, T. A. Amador and D. S. Nunes, J. Nat. Prod., 1998, 61, 392–396 CrossRef CAS PubMed
.
- S. Sharma, S. Shaheeda, K. Shaw, A. Bisai and A. Paul, ACS Catal., 2023, 13, 2118–2134 CrossRef CAS
.
- O. Achmatowicz, P. Bukowski, B. Szechner, Z. Zwierzchowska and A. Zamojski, Tetrahedron, 1971, 27, 1973–1996 CrossRef CAS
.
- M. P. Georgiadis and E. A. Couladouros, J. Org. Chem., 1986, 51, 2725–2727 CrossRef
.
- M. P. Georgiadis, E. A. Couladouros, M. G. Polissiou, S. E. Fililpakis, D. Mentzafos and A. Terzis, J. Org. Chem., 1982, 47, 3054–3058 CrossRef CAS
.
- B. M. Adger, C. Barrett, J. Brennan, M. A. McKervey and R. W. Murray, J. Chem. Soc. Chem. Commun., 1991, 1553–1554 RSC
.
- D. Thiel, D. Doknić and J. Deska, Nat. Commun., 2014, 5, 1–7 Search PubMed
.
- X. Liu, B. Li, G. Han, X. Liu, Z. Cao, D. Jiang and Y. Sun, Nat. Commun., 2021, 12, 1868 CrossRef CAS PubMed
.
-
(a) D. Li, Z. Xiao, T. B. Aftab and S. Xu, Environ. Eng. Sci., 2018, 35, 1151–1164 CrossRef CAS
; (b) L. Alves, L. I. V. Holz, C. Fernandes, P. Ribeirinha, D. Mendes, D. P. Fagg and A. Mendes, Renewable Sustainable Energy Rev., 2022, 155, 111916 CrossRef CAS
.
- J. Xian, S. Li, H. Su, P. Liao, S. Wang, Y. Zhang, W. Yang, J. Yang, Y. Sun, Y. Jia, Q. Liu, Q. Liu and G. Li, Angew. Chem., Int. Ed., 2023, 62, e202304007 CrossRef CAS PubMed
.
- J. R. Box, A. P. Atkins and A. J. J. Lennox, Chem. Sci., 2021, 12, 10252–10258 RSC
.
- L. M. Barton, L. Chen, D. G. Blackmond and P. S. Baran, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2109408118 CrossRef CAS PubMed
.
- W. Yao, K. Lv, Z. Xie, H. Qiu and M. Ma, J. Org. Chem., 2023, 88, 2296–2305 CrossRef CAS PubMed
.
-
(a) D. B. Grotjahn, S. Van, D. Combs, D. A. Lev, C. Schneider, M. Rideout, C. Meyer, G. Hernandez and L. Mejorado, J. Org. Chem., 2002, 67, 9200–9209 CrossRef CAS PubMed
; (b) S. T. Heller and S. R. Natarajan, Org. Lett., 2006, 8, 2675–2678 CrossRef CAS PubMed
.
-
(a) B. V. Lyalin, V. L. Sigacheva, A. S. Kudinova, S. V. Neverov, V. A. Kokorekin and V. A. Petrosyan, Molecules, 2021, 26, 4749 CrossRef CAS PubMed
; (b) B. V. Lyalin, V. A. Petrosyan and B. I. Ugrak, Russ. J. Electrochem., 2008, 44, 1320–1326 CrossRef CAS
.
- B. V. Lyalin and V. A. Petrosyan, Chem. Heterocycl. Compd., 2014, 49, 1599–1610 CrossRef CAS
.
- S. Zandi and F. Nikpour, Z. Naturforsch., B: J. Chem. Sci., 2022, 77, 35 CrossRef CAS
.
- B. V. Lyalin and V. A. Petrosyan, Russ. Chem. Bull., 2014, 63, 360–367 CrossRef CAS
.
- S. Hoffamn, M. Linden, J. Neuner, F. N. Weber and S. R. Waldvogel, Org. Biomol. Chem., 2023, 21, 4694–4701 RSC
.
- M. Linden, S. Hofmann, A. Herman, N. Ehler, R. M. Bär and S. R. Waldvogel, Angew. Chem., Int. Ed., 2023, 62, e202214820 CrossRef CAS PubMed
.
| This journal is © The Royal Society of Chemistry 2024 |




