Functional deep eutectic solvent for lignocellulose valorization via lignin stabilization and cellulose functionalization†
Zhen
Zhang
,
Pingli
Lv
 ,
Hairui
Ji
,
Hairui
Ji
 *,
Xingxiang
Ji
,
Zhongjian
Tian
and
Jiachuan
Chen
*,
Xingxiang
Ji
,
Zhongjian
Tian
and
Jiachuan
Chen
Key Laboratory of Pulp and Paper Science & Technology of Ministry of Education, State Key Laboratory of Biobased Material and Green Papermaking, Faculty of Light Industry, Qilu University of Technology (Shandong Academy of Sciences), Jinan 250353, China. E-mail: jihairui@yeah.net
First published on 2nd February 2024
Abstract
This study shows a functional deep eutectic solvent (DES) employing choline chloride (ChCl) and glyoxylic acid (GA) for lignocellulose fractionation. This DES exhibited a higher pretreatment efficiency (92.15%) than that composed of ChCl and lactic acid (10.36%). Molecular dynamic (MD) simulations indicated that the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES showed a higher total binding energy (ΔE) with lignin than the ChCl
GA DES showed a higher total binding energy (ΔE) with lignin than the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES, resulting in a higher dissolution of lignin. GA prevented lignin condensation during pretreatment. The stabilized lignin containing 69.39% β-O-4 linkages can be used for phenolic monomer production with a bio-oil yield of 59.60% and lignin-based sunscreen preparation with a SPF of 58.46. The cellulose residues with a mild pretreatment severity exhibited a high enzymatic saccharification yield of 91.99%. Meanwhile, an esterification reaction occurred between cellulose and GA during pretreatment. The inhibition of enzymatic hydrolysis and microbial growth enables the use of functional cellulose to produce antibacterial paper. Furthermore, 96.80% of the DES could be easily recycled and reused for biorefinery purposes.
LA DES, resulting in a higher dissolution of lignin. GA prevented lignin condensation during pretreatment. The stabilized lignin containing 69.39% β-O-4 linkages can be used for phenolic monomer production with a bio-oil yield of 59.60% and lignin-based sunscreen preparation with a SPF of 58.46. The cellulose residues with a mild pretreatment severity exhibited a high enzymatic saccharification yield of 91.99%. Meanwhile, an esterification reaction occurred between cellulose and GA during pretreatment. The inhibition of enzymatic hydrolysis and microbial growth enables the use of functional cellulose to produce antibacterial paper. Furthermore, 96.80% of the DES could be easily recycled and reused for biorefinery purposes.
1. Introduction
Lignocellulosic biomass, a promising feedstock to replace petroleum resources for biofuel, chemical, and bioproduct production, has attracted much attention in recent years due to its sustainability, renewability, and carbon neutrality.1,2 In the plant cell wall, hemicellulose and lignin encircle cellulose to form a rigid structure that is recalcitrant against degradation. The complex matrix hinders their further fractionation and value-added utilization for humans.3,4 Thus, a pretreatment step is necessary to break down the recalcitrance towards efficient biomass conversion.5,6So far, various pretreatment techniques have been developed to enhance the biomass fractionation, such as physical methods (e.g., ball milling and microwave irradiation), chemical methods (e.g., using ionic liquid and organic acid), biological methods (e.g., using white-rot fungus), and physicochemical methods (e.g., ammonia or alkali-steam explosion). Although these strategies promote enzymatic saccharification of glucan, their industrial applications face many challenges, such as cost and energy consumption.7,8 DES, a typical eutectic mixture consisting of a hydrogen bond acceptor (HBA) and a hydrogen bond donor (HBD), has been identified as a promising media for biomass pretreatment due to its high lignin solubility, non-flammability, good bio-compatibility, and thermostability.5,9,10 In particular, the ChCl-based DES exhibits an excellent ability to selectively dissolve lignin and hemicellulose from lignocellulosic biomass.11,12 This is due to strong hydrogen bonds formed between hydroxyl groups (–OH) in the lignin-polysaccharide complex and the chloride ion (Cl−) from ChCl.1 Conversely, acidic protons in the HBD can promote the cleavage of ester bonds in lignin and hemicellulose, leading to their isolation from biomass.4 Currently, widely used ChCl-based DESs include ChCl as a HBA and polyol and organic acids, such as glycerol, 1,4-butanediol (BDO), lactic acid (LA), oxalic acid (OA), acetic acid (AA), and p-toluenesulfonic acid (p-TsOH), as HBDs.13–15 Although a high lignin dissolution (65%–90%) was obtained with the ChCl-based DES, an undesired condensation of lignin usually occurs by the formation of a C–C linkage during pretreatment, which dramatically affects the further upgrading of lignin.16,17 For example, Wang et al. used a DES (ChCl and p-TsOH) for miscanthus pretreatment at 80 °C for 20 min, resulting in a low β-O-4 linkage content of only 30.90% in the obtained lignin.18 Several researchers have developed diol-based DES systems containing glycol and ChCl to pretreat biomass and obtain functionalized lignin.4,19,20 However, the delignification yield and lignin stabilization efficiency need to be improved. Recently, Bertella et al. used a cosolvent containing glyoxylic acid (GA), dioxane, and HCl to extract lignin. GA addition efficiently prevented lignin condensation and simultaneously introduced a carboxyl group on the lignin backbone.21 This gave us inspiration to use GA-containing carboxyl groups as HBDs to prepare a DES with ChCl as a HBA, achieving lignin extraction, stabilization, and functionalization.
In this study, a functional DES employing ChCl and GA was developed to pretreat lignocellulose for lignin stabilization and valorization toward multiple purposes. The effect of different pretreatment conditions on changes in main components during pretreatment was investigated. The lignin dissolution mechanism of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES was explored by a molecular dynamics (MD) simulation. After that, the value-added utilization of stabilized lignin was carried out, such as preparing phenolic monomers via hydrogenative depolymerization and producing lignin-based sunscreen. Furthermore, the enzymatic hydrolysis and antibacterial testing of obtained fibers after pretreatment were performed. By blending with softwood pulp, the obtained fibers were used for papermaking. In addition, the ChCl
GA DES was explored by a molecular dynamics (MD) simulation. After that, the value-added utilization of stabilized lignin was carried out, such as preparing phenolic monomers via hydrogenative depolymerization and producing lignin-based sunscreen. Furthermore, the enzymatic hydrolysis and antibacterial testing of obtained fibers after pretreatment were performed. By blending with softwood pulp, the obtained fibers were used for papermaking. In addition, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES was recycled for further reuse. Therefore, this green refining strategy provides a valuable reference for achieving a sustainable bioeconomy and highlights the potential for efficient pretreatment and conversion of lignocellulosic biomass using the ChCl
GA DES was recycled for further reuse. Therefore, this green refining strategy provides a valuable reference for achieving a sustainable bioeconomy and highlights the potential for efficient pretreatment and conversion of lignocellulosic biomass using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES.
GA DES.
2. Experimental section
2.1 Materials
Boehmeria nivea stalks used in this study were kindly provided by Shandong Century Sunshine Paper Group Co., Ltd (Shandong, China). Chemical reagents of analytical grade, such as choline chloride (ChCl), lactic acid (LA), glyoxylic acid (GA), and dimethyl sulfoxide (DMSO), were purchased from Macklin Biochemical Technology Co., Ltd (Shanghai, China).2.2 DES synthesis
ChCl and GA were mixed at different molar ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1
2, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 1
3, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 and 1
4 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) and subsequently heated for 30 min at 80 °C and 200 rpm to obtain a DES. Additionally, a DES composed of ChCl and LA with a molar ratio of 1
5) and subsequently heated for 30 min at 80 °C and 200 rpm to obtain a DES. Additionally, a DES composed of ChCl and LA with a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 was used for comparison.
4 was used for comparison.
2.3 Boehmeria nivea stalk pretreatment
The Boehmeria nivea stalks were crushed into particles with a diameter of 0.45–0.9 mm using a grinder (FZ102, Tianjin Teste Instrument Co., Ltd, China). Subsequently, 3 g of those particles and 40 g of DES were added in a pressure vessel (P170003, Tchongqing Xinweier Glass Co., Ltd, China), and the pretreatment was carried out at different temperatures (110 °C, 120 °C, 130 °C, and 140 °C) and times (30 min, 60 min, 90 min, and 120 min), as illustrated in Table S1.† At the end of each reaction, the pressure vessel was cooled to room temperature before opening. After adding 50 mL of ethanol (EtOH) into the vessel and stirring for 30 min; solid and liquid phases were separated employing filter paper (15 cm, Shenzhen Xinkehua Experimental Instrument Co., Ltd, China). The solids were washed with ethanol (50 mL) and deionized water (DI) to a neutral pH, and subsequently subjected to ultrasonication for 10 s to form a pulp. EtOH was removed via rotary evaporation (XA-R-1010, Xi'an Yuhui Experimental Instrument Co., Ltd) at 40 °C and 100 rpm. The stabilized lignin was obtained from the liquid phase after precipitation with DI and filtration. The DES was recycled using rotary evaporation at 100 rpm for 60 °C for the next pretreatment of Boehmeria nivea stalks.The component content in the pretreated solids was analyzed following the protocol described by the National Renewable Energy Laboratory (NREL).22 This process involved adding 0.3 g of dry solids and 3 mL of 72% sulfuric acid to a pressure vessel and allowing it to soak for 2 h. After adding 84 mL of ultrapure water, the hydrolysis reaction was conducted at 121 °C for 1 h using a steam sterilizer. Monosaccharide concentrations in the liquid phase were measured using an HPLC system (Ultimate 3000, Thermo Scientific) equipped with a separating column (Aminex HPX-87H, Bio-Rad, CA, United States) and a refractive index detector (RID-20A, Shimadzu, Japan). The pretreatment efficiency was calculated using the following equation:23
 | (1) |
2.4 MD simulation of the lignin dissolution mechanism
MD simulations were performed using GROMACS 2018.4 software.24–26 Veratrylglycerol-β-guaiacyl ether (VG) was used as a model compound of lignin for computer simulation.27–29 The simulation system of the pure DES contained 510 Lac/Gac and 2040 choline chloride molecules. Before program running, the steepest descent method was used to minimize the simulation system. The simulation was equilibrated for 5 ns in the normal volume and temperature (NVT) ensemble and 40 ns in the normal pressure and temperature (NPT) ensemble (P = 1 bar). An additional 5 ns NPT ensemble simulation was carried out to collect representative snapshots. After adding a VG molecule into the DES simulation system, the DES-VG system simulation was run under same conditions as described above.2.5 Cellulose residue characterization and utilization
The crystallinity index (CrI) of the cellulose residue was analyzed on an X-ray diffractometer (Ultima IV, Japan) with a diffraction angle range from 5° to 40° and a scanning speed of 2° min−1. CrI was calculated using the following equation: | (2) |
Fiber quality analysis (FQA), including fiber length and width, was performed on a L&W Fiber tester (ABB AB/Lorentzen & Wettre, Sweden) according to the TAPPI T-pm-91 standard.
Handsheets were prepared by blending softwood pulp and Boehmeria nivea pulp of various amounts (0%, 5%, 10%, 15%, and 20%, wt%) at a basis weight of 100 g m−2 in accordance with the SCAN-C 26:76 standard. The ring crush strength of the prepared handsheets was measured on a ring compressive strength tester (17–60, Testing Machines Inc., America) based on the TAPPI T822 om-11 standard, the tear strength was tested on a tearing tester (009, ABB AB/Lorentzen & Wettre, Sweden) according to the TAPPI T414 om-88 standard, and the tensile strength was tested using an Auto Tensile Tester (066, ABB AB/Lorentzen & Wettre, Sweden) in accordance with the TAPPI T494 om-81 standard. The morphology was scanned using an SEM system (TM4000 Plus, Japan Hitachi Nake high-tech enterprise).
The enzymatic hydrolysis of the fibers was conducted on a shaking bed incubator (THZ-100, Shanghai Bluepard Instruments Co., Ltd, China) at a temperature of 50° C and a speed of 180 rpm. Specifically, dry pretreated substrate (1 g) and cellulase (Cellic® CTec 2, Novozymes, China) with a cellulase loading of 15 FPU g−1 glucan were added into a sodium citrate buffer of 50 mL (pH 4.8). At different time intervals (3 h, 6 h, 12 h, 20 h, and 30 h), 400 μL of the supernatant was taken out and centrifuged at 8000 rpm for 5 min. The glucose content was measured via a biosensor analyzer (SBA-40E, Biological Institute of Shandong Academy Sciences, China). The yield of enzymatic hydrolysis was calculated using the following equation:
 | (3) |
To assess the antimicrobial capabilities of the prepared antibacterial paper using pretreated fibers, 100 μL of Staphylococcus aureus cultured in 50 mL of liquid medium was diluted with sterile water and poured onto a plate with solid medium containing 2% (w/v) agar. The plate containing antibacterial paper and Staphylococcus aureus was incubated at 37 °C for 24 h.
The 13C NMR measurement of cellulose was carried out using a Bruker Avance NEO spectrometer (400WB, Bruker, Germany) at a spinning speed of 10 kHz (4.2 μs 90° pulses), a cross-polarization pulse of 2 ms, and a recycling delay of 3 s.
2.6 Lignin characterization and utilization
To explore the chemical structure characteristics of lignin samples, 2D-HSQC NMR measurement was carried out on an Avance NEO 600M spectrometer (Bruker, Germany). Before testing, 40 mg of lignin was dissolved in 0.5 mL of DMSO-d6 solution.17The lignin depolymerization was carried out in a high-pressure reactor containing 0.15 g of lignin and 20 mL of methanol at 230 °C for 15 h with a Ru/C catalysis loading of 0.1 g. It is worth noting that the initial hydrogen pressure and the working hydrogen pressure were 1 MPa and 8 MPa, respectively. After cooling the reactor to 25 °C, the liquid phase containing depolymerization products was separated from the reacted mixture through filtration. Then, n-hexane was added to extract the depolymerization products. The resulting liquid phase was analyzed by a GC-MS system (QP2020, Shimadzu) with a HP-5 column. The n-hexane was removed using rotary evaporation (XA-R-1010, Xi'an Yuhui Experimental Instrument Co., Ltd). The yield of bio-oil was calculated based on the following equation:
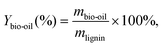 | (4) |
The UV absorbance of the lignin sample was measured on a UV-vis spectrophotometer (Shimadzu, Japan) with a wavelength range from 290 nm to 400 nm. DMSO-lignin solution was added in a dialysis bag (31 mm, Beijing Tianan United Technology Co., Ltd, China) for 48 h to generate lignin nanoparticles (LNPs). The particle size and morphology of the LNPs were further analyzed using a zeta potential tester (ZEN3690, Malvern Instruments Ltd, UK) and an atomic force microscope (AFM) (Multimode8, Bruker, Germany), respectively.
After freeze-drying, LNPs were added to commercial sunscreen to prepare lignin-based sunscreens. Different amounts (1%, 2%, 3%, and 4%, wt%) of LNPs and commercial sunscreen were blended and stirred in the dark for 24 h. Subsequently, the UV transmittance (T) of the prepared sunscreen was tested within a wavelength region of 290 nm–400 nm and a slit width of 5 nm. Prior to testing, the prepared sunscreen was coated on a 3 M medical tape on a clean quartz glass slide with a coating weight of 2 mg cm−2. The sun protection factor (SPF) was calculated according to the following equation based on previous literature:6,30
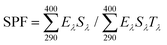 | (5) |
3. Results and discussion
3.1 Boehmeria nivea stalk pretreatment
The effect of different pretreatment parameters, such as pretreatment temperature (T), molar ratio (M), and pretreatment time (t), on the changes in the main components of the pretreated solid during pretreatment was investigated. Fig. 1a shows the effect of temperature on the cell wall compositions of the pretreated substrates. Accordingly, the content of the main components can be seen in Table S2.† Hemicellulose of 47.68% and lignin of 19.46% were removed from raw materials at 110 °C. Increasing the temperature from 110 °C to 140 °C caused obvious delignification and hemicellulose removal. Only 4.13% of hemicellulose and 4.32% of lignin were found in the pretreated solid at 130 °C compared with initial contents in the raw material (hemicellulose of 22.46% and lignin of 21.53%). Under this pretreatment temperature, an obvious fiber dissociation occurred after 10 s of ultrasonic treatment (Fig. 1b). However, an increased pretreatment temperature caused a high energy consumption and serious cellulose degradation. Therefore, a relatively mild pretreatment temperature of 130 °C was determined with a high removal of lignin (79.93%) and hemicellulose (81.61%). After that, the effect of the molar ratio of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA on the changes in the main components of the cellulose residues was further explored at 130 °C (Fig. 1c). As the molar ratio increased from 1
GA on the changes in the main components of the cellulose residues was further explored at 130 °C (Fig. 1c). As the molar ratio increased from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 to 1
2 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, the retention rate of hemicellulose and lignin decreased from 4.13% and 4.32% to 2.54% and 0.96%, respectively (Table S2†). The viscosity of the DES presented in Table S3† further supported the above observation that an increase in the GA content in the DES led to a decrease in viscosity, resulting in improved flowability, which promoted the pretreatment efficiency.31 The fibers were released from the pretreated matrix (Fig. 1d). It should be noted that the content of cellulose partially reduced from 34.50% to 31.97% (Table S2†) as the molar ratio increased. Furthermore, the presence of excessive GA in the DES solution further affected the saccharification yield in enzymatic hydrolysis. The pretreatment efficiency under molar ratios of 1
5, the retention rate of hemicellulose and lignin decreased from 4.13% and 4.32% to 2.54% and 0.96%, respectively (Table S2†). The viscosity of the DES presented in Table S3† further supported the above observation that an increase in the GA content in the DES led to a decrease in viscosity, resulting in improved flowability, which promoted the pretreatment efficiency.31 The fibers were released from the pretreated matrix (Fig. 1d). It should be noted that the content of cellulose partially reduced from 34.50% to 31.97% (Table S2†) as the molar ratio increased. Furthermore, the presence of excessive GA in the DES solution further affected the saccharification yield in enzymatic hydrolysis. The pretreatment efficiency under molar ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 reached 92.15%. Therefore, an appropriate molar ratio of 1
4 reached 92.15%. Therefore, an appropriate molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 with high-pretreatment efficiency (>90%) was chosen. According to the results in Fig. 1e and Table S2,† and the morphology of the stabilized lignin (Fig. 1f), the optimum pretreatment time was determined to be 30 min. Only 3.33% of hemicellulose and 2.15% of lignin remained in the cellulose residue. Although prolonging the pretreatment time resulted in an increase of the pretreatment efficiency, cellulose would be degraded accompanied by the destruction of the lignin structure. Higher temperature enhanced the removal of lignin and hemicellulose from the amorphous region. The CrI of the cellulose residue also increased (Fig. S1b†). Increasing the molar ratio and extending the pretreatment time showed a decrease of CrI. For example, an obvious CrI decrease from 75.45% to 72.40% was found as the pretreatment time which further prolonged from 30 min to 120 min. The main reason was that the swelling of the crystalline region of cellulose was promoted after DES penetration.32 In addition, the CrI decreased from 74.65% (1
4 with high-pretreatment efficiency (>90%) was chosen. According to the results in Fig. 1e and Table S2,† and the morphology of the stabilized lignin (Fig. 1f), the optimum pretreatment time was determined to be 30 min. Only 3.33% of hemicellulose and 2.15% of lignin remained in the cellulose residue. Although prolonging the pretreatment time resulted in an increase of the pretreatment efficiency, cellulose would be degraded accompanied by the destruction of the lignin structure. Higher temperature enhanced the removal of lignin and hemicellulose from the amorphous region. The CrI of the cellulose residue also increased (Fig. S1b†). Increasing the molar ratio and extending the pretreatment time showed a decrease of CrI. For example, an obvious CrI decrease from 75.45% to 72.40% was found as the pretreatment time which further prolonged from 30 min to 120 min. The main reason was that the swelling of the crystalline region of cellulose was promoted after DES penetration.32 In addition, the CrI decreased from 74.65% (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) to 72.48% (1
3) to 72.48% (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) because the acid hydrolysis of cellulose in the crystalline region occurred due to the presence of GA. Therefore, the optimal pretreatment conditions were determined as a pretreatment temperature of 130 °C, a molar ratio (ChCl
5) because the acid hydrolysis of cellulose in the crystalline region occurred due to the presence of GA. Therefore, the optimal pretreatment conditions were determined as a pretreatment temperature of 130 °C, a molar ratio (ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA) of 1
GA) of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, and a pretreatment time of 30 min.
4, and a pretreatment time of 30 min.
A pretreatment comparison was carried out between ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA and ChCl
GA and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA with the same pretreatment conditions as for T130M1:4t30. Fig. 1g and h show the changes in the three main components and morphology of the pulp (top) and stabilized lignin (bottom). Only 28.66% of lignin and 45.73% of hemicellulose were removed from the Boehmeria nivea stalks for ChCl
LA with the same pretreatment conditions as for T130M1:4t30. Fig. 1g and h show the changes in the three main components and morphology of the pulp (top) and stabilized lignin (bottom). Only 28.66% of lignin and 45.73% of hemicellulose were removed from the Boehmeria nivea stalks for ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA. After pretreatment with ChCl
LA. After pretreatment with ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA, the CrI of the cellulose residue increased from 65.96% to 75.45% as hemicellulose and lignin were dissolved out from the raw materials. Compared with the ChCl
GA, the CrI of the cellulose residue increased from 65.96% to 75.45% as hemicellulose and lignin were dissolved out from the raw materials. Compared with the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment, the ChCl
GA DES pretreatment, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES pretreatment led to a lower CrI under same pretreatment conditions (Fig. S1a†). The pretreatment efficiency of the ChCl
LA DES pretreatment led to a lower CrI under same pretreatment conditions (Fig. S1a†). The pretreatment efficiency of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES reached 92.15%, which was 8.89 times higher than that of the ChCl
GA DES reached 92.15%, which was 8.89 times higher than that of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES (10.36%) under same conditions of T130M1:4t30, demonstrating a strong ability for lignin and hemicellulose extraction. Moreover, the obtained lignin with ChCl
LA DES (10.36%) under same conditions of T130M1:4t30, demonstrating a strong ability for lignin and hemicellulose extraction. Moreover, the obtained lignin with ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES exhibited a darker brown color than that of ChCl
LA DES exhibited a darker brown color than that of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES. Furthermore, the collected cellulose residue cannot be effectively dispersed in water to form a pulp after ultrasonic treatment (Fig. 1h).
GA DES. Furthermore, the collected cellulose residue cannot be effectively dispersed in water to form a pulp after ultrasonic treatment (Fig. 1h).
3.2 MD simulation of the lignin dissolution mechanism
To understand the interactions between lignin and DES molecules, the lignin dissolution mechanism of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES, and the reason for differences in lignin removal between ChCl
GA DES, and the reason for differences in lignin removal between ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA and ChCl
GA and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DESs, MD simulations were performed using VG as a model compound of lignin. The densities of the two DESs were first calculated to ensure the accuracy of the parameters employed in the MD simulations.13 The calculated densities of ChCl
LA DESs, MD simulations were performed using VG as a model compound of lignin. The densities of the two DESs were first calculated to ensure the accuracy of the parameters employed in the MD simulations.13 The calculated densities of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA and ChCl
GA and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DESs were 1.25 g cm−3 and 1.39 g cm−3, as shown in Fig. S2,† respectively, which was consistent with the measured densities of 1.23 g cm−3 and 1.36 g cm−3 with a permitted error of below 2.00%. The force field parameters used in the simulation are very reliable. Fig. 2 shows the volumetric density map (isosurface) of cosolvent molecules around the VG molecule at different isovalues. Based on the results from Fig. 2a and the movie in the ESI,† the distance between ChCl
LA DESs were 1.25 g cm−3 and 1.39 g cm−3, as shown in Fig. S2,† respectively, which was consistent with the measured densities of 1.23 g cm−3 and 1.36 g cm−3 with a permitted error of below 2.00%. The force field parameters used in the simulation are very reliable. Fig. 2 shows the volumetric density map (isosurface) of cosolvent molecules around the VG molecule at different isovalues. Based on the results from Fig. 2a and the movie in the ESI,† the distance between ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA and VG is closer than that between ChCl
GA and VG is closer than that between ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA and VG. Subsequently, the local organization and bonding of the DES molecules in the immediate vicinity of VG were investigated by calculating radial distribution functions (RDFs) between the DES and VG molecules, or more precisely, by calculating the center of mass (the molecule) RDFs, as shown in Fig. 2b. For the simulation system of Lac/ChCl-VG, an obvious sharp peak was found at 5.36 Å with a strength value of 1.97 for the centroids of the ChCl
LA and VG. Subsequently, the local organization and bonding of the DES molecules in the immediate vicinity of VG were investigated by calculating radial distribution functions (RDFs) between the DES and VG molecules, or more precisely, by calculating the center of mass (the molecule) RDFs, as shown in Fig. 2b. For the simulation system of Lac/ChCl-VG, an obvious sharp peak was found at 5.36 Å with a strength value of 1.97 for the centroids of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA and VG benzene ring, while a strong peak with a strength value of 2.23 appearing at 4.50 Å for the centroids of the ChCl
LA and VG benzene ring, while a strong peak with a strength value of 2.23 appearing at 4.50 Å for the centroids of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA and VG benzene ring in the simulation system of Gac/ChCl-VG. The two strong peaks approaching around 5.00 Å were attributed to the H–π interaction between the VG benzene ring and DES molecules, thus resulting in high strength of van der Waals interaction.12 The results were consistent with those in Fig. 2a. Furthermore, interaction energies (ΔE), mainly van der Waals (LJ-SR) and electrostatic (Coul-SR) interaction energies, between the DES and VG molecules were calculated, as shown in Fig. 2c. The electrostatic interaction energies of ChCl/LA-VG and ChCl/GA-VG were calculated to be 161.19 kJ mol−1 and 158.35 kJ mol−1, respectively. No significant difference in electrostatic interaction energy was found between the two simulation systems. The electrostatic interactions were cumulated according to the cutoff method with a distance of 3.0 nm. The calculated van der Waals interaction energies of Lac/ChCl-VG and Gac/ChCl-VG were 215.63 kJ mol−1 and 234.55 kJ mol−1, respectively, indicating that van der Waals interaction plays an important role in the interaction between the DES and VG molecules. The ChCl
GA and VG benzene ring in the simulation system of Gac/ChCl-VG. The two strong peaks approaching around 5.00 Å were attributed to the H–π interaction between the VG benzene ring and DES molecules, thus resulting in high strength of van der Waals interaction.12 The results were consistent with those in Fig. 2a. Furthermore, interaction energies (ΔE), mainly van der Waals (LJ-SR) and electrostatic (Coul-SR) interaction energies, between the DES and VG molecules were calculated, as shown in Fig. 2c. The electrostatic interaction energies of ChCl/LA-VG and ChCl/GA-VG were calculated to be 161.19 kJ mol−1 and 158.35 kJ mol−1, respectively. No significant difference in electrostatic interaction energy was found between the two simulation systems. The electrostatic interactions were cumulated according to the cutoff method with a distance of 3.0 nm. The calculated van der Waals interaction energies of Lac/ChCl-VG and Gac/ChCl-VG were 215.63 kJ mol−1 and 234.55 kJ mol−1, respectively, indicating that van der Waals interaction plays an important role in the interaction between the DES and VG molecules. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES system with a high total ΔE of 392.90 kJ mol−1 caused a higher dissolution of lignin than the ChCl
GA DES system with a high total ΔE of 392.90 kJ mol−1 caused a higher dissolution of lignin than the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES system (376.82 kJ mol−1), which explains why using the ChCl
LA DES system (376.82 kJ mol−1), which explains why using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES lead to a higher lignin removal during pretreatment than using the ChCl
GA DES lead to a higher lignin removal during pretreatment than using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES.
LA DES.
3.3 Characterization and utilization of cellulose
Fiber length and width were measured through FQA as presented in Table S4.† The fiber length and width decreased from 0.48 mm and 44.90 μm to 0.37 mm and 25.10 μm, respectively, with an increase in temperature from 110 °C to 140 °C. At 130 °C, almost all of the fibers had dissociated from the pretreated solid to form a pulp (Fig. 1b). The length and width were measured to be 0.35 mm and 25.30 μm, respectively. High temperature caused hydrolysis of the cellulose chain, resulting in a short fiber length. This conclusion is further substantiated by the SEM image presented in Fig. S3.† Thus, the fibers from the pretreatment conditions of T130M1:4t30 were used for papermaking by blending them with commercial softwood pulp.The saccharification yield of the cellulose residues after various pretreatment conditions (Table S5†) was investigated, as shown in Fig. 3a. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment resulted in a higher cellulose digestibility rate of 70.61% than that using the ChCl
GA DES pretreatment resulted in a higher cellulose digestibility rate of 70.61% than that using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES pretreatment (60.91%) under same conditions (130 °C). This was due to the removal of more hemicellulose and lignin during the pretreatment process, which led to an increased surface area for enzyme adsorption.16 The SEM images of the pretreated substrates also revealed a loose structure (Fig. 3c). After 10 s of ultrasonic treatment, the free fibers were released from the pretreated matrix (Fig. 3c) to form a pulp (Fig. 1b). Interestingly, the highest saccharification yield of 91.99% was found after 30 h of enzymatic hydrolysis for the cellulose residue with a pretreatment temperature of 110 °C. The increased pretreatment temperature caused a decrease of the enzymatic efficiency (Fig. 3a). The main reason for this was the chemical modification of cellulose by the esterification reaction between the carboxyl groups in GA and hydroxyl groups in cellulose during pretreatment (Fig. 3d). The aldehyde groups on the surface of cellulose caused a possible inhibition or deactivation effect on the individual cellobiohydrolase (CBH) and β-glucosidase (BG) activities. In particular, enzyme deactivation is usually indicated by a time-dependent loss of enzyme activity. Aldehyde groups could interact with different functional groups on the enzyme/protein surface through covalent and/or exceptionally strong noncovalent bonds, which sometimes remain even after complete protein breakdown.33,34 This finding was demonstrated in the FTIR spectrum of the pretreated substrates (Fig. 3b). A new signal was found at 1202 cm−1 originating from the stretching vibration of the CO–O–C structure in the ester bond.35 A clearer peak was observed in the substrate as the pretreatment temperature was raised. Furthermore, the solid-state 13C NMR spectra of the pretreated substrates exhibited a typical carbonyl group peak at 178 ppm in Fig. 3e, further revealing that an esterification occurred between cellulose and GA during the pretreatment process.36 In short, the ChCl
LA DES pretreatment (60.91%) under same conditions (130 °C). This was due to the removal of more hemicellulose and lignin during the pretreatment process, which led to an increased surface area for enzyme adsorption.16 The SEM images of the pretreated substrates also revealed a loose structure (Fig. 3c). After 10 s of ultrasonic treatment, the free fibers were released from the pretreated matrix (Fig. 3c) to form a pulp (Fig. 1b). Interestingly, the highest saccharification yield of 91.99% was found after 30 h of enzymatic hydrolysis for the cellulose residue with a pretreatment temperature of 110 °C. The increased pretreatment temperature caused a decrease of the enzymatic efficiency (Fig. 3a). The main reason for this was the chemical modification of cellulose by the esterification reaction between the carboxyl groups in GA and hydroxyl groups in cellulose during pretreatment (Fig. 3d). The aldehyde groups on the surface of cellulose caused a possible inhibition or deactivation effect on the individual cellobiohydrolase (CBH) and β-glucosidase (BG) activities. In particular, enzyme deactivation is usually indicated by a time-dependent loss of enzyme activity. Aldehyde groups could interact with different functional groups on the enzyme/protein surface through covalent and/or exceptionally strong noncovalent bonds, which sometimes remain even after complete protein breakdown.33,34 This finding was demonstrated in the FTIR spectrum of the pretreated substrates (Fig. 3b). A new signal was found at 1202 cm−1 originating from the stretching vibration of the CO–O–C structure in the ester bond.35 A clearer peak was observed in the substrate as the pretreatment temperature was raised. Furthermore, the solid-state 13C NMR spectra of the pretreated substrates exhibited a typical carbonyl group peak at 178 ppm in Fig. 3e, further revealing that an esterification occurred between cellulose and GA during the pretreatment process.36 In short, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment resulted in not only a high yield (91.99%) of enzymatic saccharification when converting the cellulose residue to glucose but also cellulose functionalization by introducing aldehyde groups on the cellulose surface. The inhibition on enzymatic hydrolysis gave us inspiration to use the esterified cellulose to produce antibacterial paper. Fig. 3f demonstrates the good inhibitor effect of the prepared antibacterial paper towards the growth of Staphylococcus aureus within 24 h.
GA DES pretreatment resulted in not only a high yield (91.99%) of enzymatic saccharification when converting the cellulose residue to glucose but also cellulose functionalization by introducing aldehyde groups on the cellulose surface. The inhibition on enzymatic hydrolysis gave us inspiration to use the esterified cellulose to produce antibacterial paper. Fig. 3f demonstrates the good inhibitor effect of the prepared antibacterial paper towards the growth of Staphylococcus aureus within 24 h.
The physical properties, such as ring crush strength index, tear index, and tensile index, of the handsheets produced by mixing commercial softwood pulp with Boehmeria nivea pulp are shown in Fig. 4. Although the average length of the Boehmeria nivea pulp is shorter than that of the commercial softwood pulp, some studies have shown that adding short fibers into the softwood pulp could improve the physical properties, particularly the ring crush strength index. This is because short fibers can fill up gaps between long softwood pulp fibers, leading to an increase in binding sites between the fibers. The enhancement of hydrogen bonding between hydroxyl groups on the fiber surface resulted in an improvement in the paper strength. In this study, although adding different amounts of Boehmeria nivea pulp into the softwood pulp did not significantly increase the mechanical strength of the handsheets, when the added amount of Boehmeria nivea pulp reached 10%, the mechanical strength was very close to that of the pure softwood pulp handsheets without Boehmeria nivea pulp addition. The ring crush strength index of 4.34 Nm g−1 (Fig. 4a), tear index of 8.07 mN m2 g−1 (Fig. 4b), and tensile index of 16.09 Nm g−1 (Fig. 4c) of the prepared handsheets were 83.30%, 89.37%, and 97.16% of those of the pure softwood pulp handsheets, respectively. The probable reason was that the Boehmeria nivea fibers acted as a filler in the softwood fiber frame, and the gaps between the fibers were filled, not only increasing the binding sites but also improving the interactions of intermolecular hydrogen bonds (Fig. 4d).6 However, further raising the amount of produced pulp significantly reduces the mechanical strength of the handsheets due to the decrease of softwood fibers. Thus, using Boehmeria nivea pulp to replace commercial softwood pulp at an optimum amount of 10% can reduce the cost of papermaking with a bit of bacterial inhibition function while providing a strategy for the utilization of agricultural waste, Boehmeria nivea stalks.
3.4 Lignin characterization and utilization
The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES not only showed a higher lignin removal than ChCl
GA DES not only showed a higher lignin removal than ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA but also obtained a lighter colored lignin. Traditional lignin fractionation usually involves reactions of condensation and repolymerization to produce a highly stable C–C linkage. Aldehyde-assisted fractionation with GA can prevent lignin fractionation to form condensed lignin, as shown in Fig. 5a.37 The structural characteristics of the extracted lignin with ChCl
LA but also obtained a lighter colored lignin. Traditional lignin fractionation usually involves reactions of condensation and repolymerization to produce a highly stable C–C linkage. Aldehyde-assisted fractionation with GA can prevent lignin fractionation to form condensed lignin, as shown in Fig. 5a.37 The structural characteristics of the extracted lignin with ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES, labeled as GSL, were analyzed via 2D HSQC NMR as demonstrated in Fig. 5g and h. Milled wood lignin that was labeled as MWL (Fig. 5c and d) and the extracted lignin with ChCl
GA DES, labeled as GSL, were analyzed via 2D HSQC NMR as demonstrated in Fig. 5g and h. Milled wood lignin that was labeled as MWL (Fig. 5c and d) and the extracted lignin with ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES that was labeled as LAL (Fig. 5e and f) were used for comparison.
LA DES that was labeled as LAL (Fig. 5e and f) were used for comparison.
As shown in Fig. 5, the 2D HSQC NMR spectrum was divided into a side chain region and a benzene ring region at δC/δH 52.0–100.0/2.70–5.30 ppm and δC/δH 100.0–136.0/6.30–8.00 ppm, respectively. The main cross-peaks are listed in Table S6† based on a previous publication.38
In the side chain region, a clear cross-signal of methoxy groups (–OCH3) was observed at δC/δH 56.1/3.76 ppm. The typical interunit linkages, such as β-O-4 structure (A and A′ unit), phenyl-coumaran structure (β-5, B unit), and resinol (β-β, C unit), were also determined. Specifically, the cross-peaks for β-O-4 were displayed at δC/δH 72.1/4.88 ppm (Aα), δC/δH 84.1/4.35 ppm (Aβ(G)), δC/δH 87.0/4.13 ppm (Aβ(S)), δC/δH 60.0/3.72 ppm (Aγ), and δC/δH 63.5/4.25 ppm (A′γ). The phenyl-coumaran structures were found at δC/δH 85.2/4.69 ppm (Bα) and δC/δH 71.9/3.87–4.21 ppm (Bγ). The cross-signals at δC/δH 87.9/5.48 ppm (Cα), δC/δH 53.8/3.08 ppm (Cβ), and δC/δH 62.8/3.85 ppm (Cγ) were assigned to the resinol structure.14 Unlike the spectra of MWL (Fig. 5c) and LAL (Fig. 5e), the cross-peak at δC/δH 97.0/5.06–5.27 ppm was attributed to the newly formed acetal structure (Fig. 5g).39 Additionally, the α, β, and γ signals of stable lignin were observed at δC/δH 82.0/4.63 ppm, δC/δH 73.5/4.30–4.40 ppm, and δC/δH 67.0/3.82 ppm in Fig. 5g, respectively, which indicated that GA prevented further lignin condensation during the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment.21 After stabilization and functionalization, the GSL exhibited a very poor solubility in GVL, THF, and 1,4-dioxane, as shown in Fig. 5b.
GA DES pretreatment.21 After stabilization and functionalization, the GSL exhibited a very poor solubility in GVL, THF, and 1,4-dioxane, as shown in Fig. 5b.
In the benzene ring region, the typical cross-signals of lignin were found, such as syringyl (S) and guaiacyl (G) units at δC/δH 104.2/6.72 ppm (C2,6–H2,6, S unit), δC/δH 111.1/6.95 ppm (C2–H2, G unit), δC/δH 116.1/6.81 ppm (C5–H5, G unit), and δC/δH 119.5/6.82 ppm (C6–H6, G unit),40 respectively. Additionally, the peaks at δC/δH 131.1/7.57 ppm and δC/δH 130.8/7.50 ppm originated from p-benzoate units (C2,6–H2,6) and p-coumarate moieties (C2,6–H2,6, PCA unit). Table S7† illustrates the content of main inter-unit linkages (β-O-4, β-5, and β–β) and S/G ratio in the lignin samples. The GSL sample exhibited a high content of β-O-4 linkages (69.39%), which is very close to that of MWL (85.57%). The results indicated that the DES containing GA can effectively hinder the polymerization of lignin during lignin extraction.
The depolymerization of GSL was performed using Ru/C as a catalyst and methanol as a solvent at 230 °C for 15 h in hydrogen atmosphere. The original depolymerization analysis of the three lignin samples by gas chromatogram (MWL, LAL and GSL) is listed in Fig. S4.† As shown in Fig. 6, nine main aromatic monomers were found in the depolymerization products of MWL with a bio-oil yield of 81.10%. Six major lignin monomers were identified in the products of GSL with a bio-oil yield of 59.60%. Without GA stabilization during pretreatment, the bio-oil yield of 58.95% was obtained in the products of LAL. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES pretreatment showed a monomer yield of only 48.20% and low lignin removal (28.66%). The ChCl
LA DES pretreatment showed a monomer yield of only 48.20% and low lignin removal (28.66%). The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment led to not only a high lignin removal (90.01%) but also a high final monomer yield after hydrogenolysis of the extracted lignin (57.80%). The increased monomer yield can be attributed to the stabilization mechanism in Fig. 5a, which is consistent with previous publications with FA stabilization.40 The β-O-4 structure was largely preserved via preventing the formation of stable C–C bond linkages during the ChCl
GA DES pretreatment led to not only a high lignin removal (90.01%) but also a high final monomer yield after hydrogenolysis of the extracted lignin (57.80%). The increased monomer yield can be attributed to the stabilization mechanism in Fig. 5a, which is consistent with previous publications with FA stabilization.40 The β-O-4 structure was largely preserved via preventing the formation of stable C–C bond linkages during the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment process. Therefore, GSL has great potential in the preparation of biofuels with excellent combustion performance by further catalytic hydrogenation.41,42
GA DES pretreatment process. Therefore, GSL has great potential in the preparation of biofuels with excellent combustion performance by further catalytic hydrogenation.41,42
 | ||
| Fig. 6 The bio-oil and aromatic monomer yields of lignin (MWL, LAL, and GSL) after depolymerization. | ||
The stabilized lignin showed a strong UV absorption ability and could be used as an anti-ultraviolet additive for lignin-based sunscreen preparation.6 After introducing an amount of carboxyl groups, the UV absorption ability of GSL was improved (Fig. 5a). Thus, the possibility of using the obtained GSL for lignin-based sunscreen production was investigated. To improve the dispersibility and UV resistance in sunscreen, the GSL was prepared into lignin nanoparticles (LNPs). The size distributions of the produced LNPs from different pretreatment conditions are shown in Fig. S5.† The different LNPs exhibited a similar size distribution between 100 and 1000 nm, indicating that these LNPs had a larger specific surface area and could provide stronger UV shielding function. Furthermore, these uniform-size LNPs were dispersed in commercial emulsions to prepare lignin-based sunscreens (Fig. 7a).
The UV absorptions of the LNPs extracted at different temperatures were measured prior to preparing sunscreen as presented in Fig. 7b. The UV absorption of the LNPs decreased with the temperature rising. The LNPs obtained at 110 °C exhibited the highest UV absorption capacity among all LNP samples. Many publications have reported that the presence of the conjunction structure formed by double bonds and/or carbonyl groups with a phenyl group may provide lignin with a strong UV absorption capacity.43,44 However, the content of this conjunction structure in the extracted lignin was reduced with the enhancement of the pretreatment severity, which resulted in a decrease in the UV absorption capacity.14,45 Combining the highest enzymatic efficiency at 110 °C (Fig. 3a), the LNPs obtained from 110 °C were selected to prepare the lignin-based sunscreen. Besides, an AFM image of the LNPs obtained under 110 °C (Fig. S6†) exhibited their nanoscale size, further demonstrating that the collected LNPs might be a desirable anti-ultraviolet additive for sunscreen production.
As exhibited in Fig. 7a, different amounts of LNPs were mixed with a commercial sunscreen with an SPF of 15. The addition of 4% LNPs resulted in a similar light-yellow color as that of the blank sample without LNP addition. The UV transmittances for produced sunscreens are shown in Fig. 7c, and their SPF values were further calculated as demonstrated in Fig. 7d according to a previously reported method.6 The produced sunscreen displayed a decrease in UV transmittance within UVA (320–400 nm) and UVB (290–320 nm) ranges with an increasing content of LNPs, indicating an excellent UV blocking performance. The SPF of the lignin-based sunscreen adding 2% LNPs reached 34.15, exceeding that of commercial SPF 30 sunscreen with a measured SPF value of 32.06. With the addition of LNPs (4%), the SPF value of the obtained sunscreen reached up to 58.46, which was 1.82 times and 1.28 times higher than those of SPF 30 and SPF 50 sunscreens, respectively. The measured results indicated that the prepared sunscreen possessed excellent UV resistant properties. The produced sunscreen did not cause any contamination on the human skin as demonstrated in Fig. 7e. Thus, the GSL extracted from Boehmeria nivea stalks can be used as an anti-ultraviolet additive for lignin-based sunscreen preparation.
3.5 DES recovery
Fig. 8 shows an approach of lignocellulose fractionation using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES and DES recovery. After filtration, the anti-solvent (ethanol) was used to wash the cellulose residue. Pulp was obtained via further ultrasonic treatment for 10 s in step 1. Subsequently, the mixture containing ethanol, lignin, and DES was further concentrated by removing ethanol using rotary evaporation at 40 °C (step 2). The addition of deionized water in step 3 led to the precipitation of GSL. After filtration and washing with DI, the protected lignin with a purity of 95.60% was obtained in step 4. DES was recovered by removing deionized water in step 5 and reused for lignocellulose fractionation in step 6. In this system, the cellulose fraction and lignin fraction were effectively separated from Boehmeria nivea stalks. The ChCl
GA DES and DES recovery. After filtration, the anti-solvent (ethanol) was used to wash the cellulose residue. Pulp was obtained via further ultrasonic treatment for 10 s in step 1. Subsequently, the mixture containing ethanol, lignin, and DES was further concentrated by removing ethanol using rotary evaporation at 40 °C (step 2). The addition of deionized water in step 3 led to the precipitation of GSL. After filtration and washing with DI, the protected lignin with a purity of 95.60% was obtained in step 4. DES was recovered by removing deionized water in step 5 and reused for lignocellulose fractionation in step 6. In this system, the cellulose fraction and lignin fraction were effectively separated from Boehmeria nivea stalks. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES used in the biomass fractionation process was easily recycled with an ideal yield of 96.80%.
GA DES used in the biomass fractionation process was easily recycled with an ideal yield of 96.80%.
 | ||
Fig. 8 The fractionation protocol of Boehmeria nivea stalks using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES and recovery of DES. GA DES and recovery of DES. | ||
4. Conclusion
Herein, a new DES composed of ChCl and GA was developed for lignocellulose valorization. Under optimal pretreatment conditions (T130M1:4t30), the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES exhibited higher removal of lignin (90.01%) and hemicellulose (85.17%) from Boehmeria nivea stalks than the ChCl
GA DES exhibited higher removal of lignin (90.01%) and hemicellulose (85.17%) from Boehmeria nivea stalks than the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES. MD simulations indicated that the ChCl
LA DES. MD simulations indicated that the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES showed a higher total binding energy (ΔE) with lignin than the ChCl
GA DES showed a higher total binding energy (ΔE) with lignin than the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES, resulting in a higher dissolution of lignin. Furthermore, the ChCl
LA DES, resulting in a higher dissolution of lignin. Furthermore, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES pretreatment caused cellulose functionalization by introducing aldehyde groups on the cellulose surface, which was not found in the pretreatment using the ChCl
GA DES pretreatment caused cellulose functionalization by introducing aldehyde groups on the cellulose surface, which was not found in the pretreatment using the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA DES. The cellulose residue can be used for antibacterial paper fabrication or industrial papermaking. Meanwhile, GA prevented lignin condensation by a stabilization approach. The stabilized lignin containing 69.39% β-O-4 linkages can be used for phenolic monomer production with a bio-oil yield of 59.60% and lignin-based sunscreen preparation with a SPF of 58.46. Moreover, the ChCl
LA DES. The cellulose residue can be used for antibacterial paper fabrication or industrial papermaking. Meanwhile, GA prevented lignin condensation by a stabilization approach. The stabilized lignin containing 69.39% β-O-4 linkages can be used for phenolic monomer production with a bio-oil yield of 59.60% and lignin-based sunscreen preparation with a SPF of 58.46. Moreover, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) GA DES was recycled with a desirable yield of 96.80%. Therefore, this study provided a functional DES for lignocellulose valorization via lignin stabilization and cellulose functionalization.
GA DES was recycled with a desirable yield of 96.80%. Therefore, this study provided a functional DES for lignocellulose valorization via lignin stabilization and cellulose functionalization.
Author contributions
Zhen Zhang: date analysis and writing-original draft; Hairui Ji: conceptualization, investigation, and writing-review & editing; Pingli Lv: software and methodology; Xingxiang Ji: supervision and writing-review & editing; Zhongjian Tian: supervision and writing-review & editing; Jiachuan Chen: supervision and writing-review & editing.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors would like to express gratitude for the support from the Program of National Natural Science Foundation of China (32271800), the Basic Research Integration Projects of Science, Education, and Industry of Qilu University of Technology (2022PY039), the Jinan Innovation Team (2021GXRC023), and the QUTJBZ Program (2022JBZ01-05).References
- M. G. Morán-Aguilar, M. Calderón-Santoyo, R. P. S. Oliveira, M. G. Aguilar-Uscanga and J. M. Domínguez, Carbohydr. Polym., 2022, 298, 120097 CrossRef PubMed.
- X.-J. Shen, J.-L. Wen, Q.-Q. Mei, X. Chen, D. Sun, T.-Q. Yuan and R.-C. Sun, Green Chem., 2019, 21, 275–283 RSC.
- M. Wu, L. Gong, C. Ma and Y.-C. He, Bioresour. Technol., 2021, 340, 125695 CrossRef CAS PubMed.
- J. Cheng, C. Huang, Y. Zhan, S. Han, J. Wang, X. Meng, C. G. Yoo, G. Fang and A. J. Ragauskas, Chem. Eng. J., 2022, 443, 136395 CrossRef CAS.
- A. Ullah, Y. Zhang, C. Liu, Q. Qiao, Q. Shao and J. Shi, Bioresour. Technol., 2023, 369, 128394 CrossRef CAS PubMed.
- Y. Lyu, X.-X. Ji, Z. Tian, H. Ji, F. Zhang, L. Dai, H. Xie and C. Si, Int. J. Biol. Macromol., 2023, 230, 123122 CrossRef CAS PubMed.
- J. Yang, S. Wang, F. Bai, X. Lu, W. He, Z. Fang, N. Zhu and K. Guo, Chem. Eng. J., 2023, 454, 140480 CrossRef CAS.
- A. Mena-García, A. I. Ruiz-Matute, A. C. Soria and M. L. Sanz, TrAC, Trends Anal. Chem., 2019, 119, 115612 CrossRef.
- J. Xu, P. Zhou, X. Liu, L. Yuan, C. Zhang and L. Dai, ChemSusChem, 2021, 14, 2740–2748 CrossRef CAS PubMed.
- Y. Yu, D. Wang, L. Chen, H. Qi, A. Liu, M. Deng, X. Wu and K. Wang, Ind. Crops Prod., 2022, 187, 115542 CrossRef CAS.
- Y. Wang, X. Meng, K. Jeong, S. Li, G. Leem, K. H. Kim, Y. Pu, A. J. Ragauskas and C. G. Yoo, ACS Sustainable Chem. Eng., 2020, 8, 12542–12553 CrossRef CAS.
- Q. Ji, X. Yu, P. Wu, A. E.-G. A. Yagoub, L. Chen, A. T. Mustapha and C. Zhou, Ind. Crops Prod., 2021, 173, 114108 CrossRef CAS.
- H. Ji and P. Lv, Green Chem., 2020, 22, 1378–1387 RSC.
- H. Ji, Y. Song, X. Zhang and T. Tan, Bioresour. Technol., 2017, 238, 575–581 CrossRef CAS PubMed.
- P. D. Muley, J. K. Mobley, X. Tong, B. Novak, J. Stevens, D. Moldovan, J. Shi and D. Boldor, Energy Convers. Manage., 2019, 196, 1080–1088 CrossRef CAS.
- L. Yao, P. Cui, X. Chen, C. G. Yoo, Q. Liu, X. Meng, L. Xiong, A. J. Ragauskas and H. Yang, Bioresour. Technol., 2022, 350, 126885 CrossRef CAS PubMed.
- Z. Chen, W. A. Jacoby and C. Wan, Bioresour. Technol., 2019, 279, 281–286 CrossRef CAS PubMed.
- W. Wang, F. Gu, J. Y. Zhu, K. Sun, Z. Cai, S. Yao, W. Wu and Y. Jin, Ind. Crops Prod., 2020, 150, 112423 CrossRef CAS.
- Z. Chen, X. Bai, L. A., H. Zhang and C. Wan, ACS Sustainable Chem. Eng., 2020, 8, 9783–9793 CrossRef CAS.
- Y. Liu, N. Deak, Z. Wang, H. Yu, L. Hameleers, E. Jurak, P. J. Deuss and K. Barta, Nat. Commun., 2021, 12, 5424 CrossRef CAS PubMed.
- S. Bertella, M. B. Figueirêdo, G. De Angelis, M. Mourez, C. Bourmaud, E. Amstad and J. S. Luterbacher, ChemSusChem, 2022, 15, e202200270 CrossRef CAS PubMed.
- A. Sluiter, B. Hames, R. Ruiz, C. Scarlata and D. Crocker, Determination of Structural Carbohydrates and Lignin in Biomass—NREL/TP-510-42618, 2008 Search PubMed.
- P. D. Maibam and A. Goyal, Bioresour. Technol., 2022, 351, 127057 CrossRef CAS PubMed.
- H. J. C. Berendsen, D. van der Spoel and R. van Drunen, Comput. Phys. Commun., 1995, 91, 43–56 CrossRef CAS.
- B. Hess, C. Kutzner, D. van der Spoel and E. Lindahl, J. Chem. Theory Comput., 2008, 4, 435–447 CrossRef CAS PubMed.
- D. Van Der Spoel, E. Lindahl, B. Hess, G. Groenhof, A. E. Mark and H. J. C. Berendsen, J. Comput. Chem., 2005, 26, 1701–1718 CrossRef CAS PubMed.
- B. G. Janesko, Phys. Chem. Chem. Phys., 2011, 13, 11393–11401 RSC.
- W. Ji, Z. Ding, J. Liu, Q. Song, X. Xia, H. Gao, H. Wang and W. Gu, Energy Fuels, 2012, 26, 6393–6403 CrossRef CAS.
- Y. Zhu, J. Yan, C. Liu and D. Zhang, Biopolymers, 2017, 107, e23022 CrossRef PubMed.
- R. Liu, L. Dai, C. Xu, K. Wang, C. Zheng and C. Si, ChemSusChem, 2020, 13, 4266–4283 CrossRef CAS PubMed.
- Z. Zhang, J. Xu, J. Xie, S. Zhu, B. Wang, J. Li and K. Chen, Carbohydr. Polym., 2022, 290, 119472 CrossRef CAS PubMed.
- Z. Ling, Z. Guo, C. Huang, L. Yao and F. Xu, Bioresour. Technol., 2020, 305, 123025 CrossRef CAS PubMed.
- A. Mnasri, H. Dhaouadi, R. Khiari, S. Halila and E. Mauret, Carbohydr. Polym., 2022, 292, 119606 CrossRef CAS PubMed.
- R. Zhai, J. Hu and J. N. Saddler, Sustainable Energy Fuels, 2018, 2, 1048–1056 RSC.
- J. A. Ávila Ramírez, C. J. Suriano, P. Cerrutti and M. L. Foresti, Carbohydr. Polym., 2014, 114, 416–423 CrossRef PubMed.
- G. Zeng, L. Zhang, B. Qi, J. Luo and Y. Wan, Bioresour. Technol., 2023, 380, 129085 CrossRef CAS PubMed.
- M. T. Amiri, G. R. Dick, Y. M. Questell-Santiago and J. S. Luterbacher, Nat. Protoc., 2019, 14, 921–954 CrossRef PubMed.
- H. Cao, R. Liu, B. Li, Y. Wu, K. Wang, Y. Yang, A. Li, Y. Zhuang, D. Cai and P. Qin, Ind. Crops Prod., 2022, 177, 114480 CrossRef CAS.
- W. Lan, M. T. Amiri, C. M. Hunston and J. S. Luterbacher, Angew. Chem., Int. Ed., 2018, 57, 1356–1360 CrossRef CAS PubMed.
- L. Shuai, M. T. Amiri, Y. M. Questell-Santiago, F. Héroguel, Y. Li, H. Kim, R. Meilan, C. Chapple, J. Ralph and J. S. Luterbacher, Science, 2016, 354, 329–333 CrossRef CAS PubMed.
- S. Liu and H. Ji, Cellulose, 2023, 30, 1503–1515 CrossRef CAS.
- X. Liu, Z. Jiang, S. Feng, H. Zhang, J. Li and C. Hu, Fuel, 2019, 244, 247–257 CrossRef CAS.
- M. H. Tran, D.-P. Phan and E. Y. Lee, Green Chem., 2021, 23, 4633–4646 RSC.
- Y. Qian, X. Qiu and S. Zhu, Green Chem., 2015, 17, 320–324 RSC.
- H. Ji, L. Wang, Z. Pang, W. Zhu, G. Yang and C. Dong, Cellulose, 2020, 27, 7561–7573 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3gc04749k |
| This journal is © The Royal Society of Chemistry 2024 |






