Electrochemical bromination of enamides with sodium bromide†
Shinan
Luan
,
Thomas
Castanheiro
 * and
Thomas
Poisson
* and
Thomas
Poisson
 *
*
INSA Rouen Normandie, Univ. Rouen Normandie, CNRS, Normandie Univ., COBRA UMR 6014, INC3M FR 3038, F-76000 Rouen, France. E-mail: thomas.castanheiro-matias@univ-rouen.fr; thomas.poisson@insa-rouen.fr
First published on 1st February 2024
Abstract
The electrochemical bromination of enamide derivatives was developed using inexpensive and non-toxic sodium bromide (NaBr). This transformation enabled the direct stereoselective formation of a C(sp2)−Br bond and was applied to a wide variety of enamides without the need for external hazardous oxidants, reductants or metal catalysts. The protocol showed a general efficiency and tolerance, allowing access to brominated enamides with yields ranging from 56% to 85%. The reaction conditions were applied to the chlorination reaction using sodium chloride (NaCl). The synthetic utility of the products was illustrated through Suzuki and Sonogashira cross-coupling reactions, offering a novel reaction procedure to access complex enamide derivatives.
Introduction
Organohalide compounds play a crucial role in society and are among the most explored chemicals. Their importance not only comes from their prevalence in Nature,1 with more than 4500 natural products discovered containing at least one halogen atom, but is also a result of their high versatility as synthetic intermediates and building blocks in a plethora of chemical reactions.2 Furthermore, as a result of the advent of transition-metal-catalyzed cross-coupling reactions, the relevance of aryl and vinyl halides has increased significantly over the past forty years.3 Moreover, carbon–halogen (C–X) bonds are present in the structural backbone of many chemicals used in materials sciences, as well as in pharmaceuticals and agrochemicals.4–6Organohalides are conventionally synthesized through well-established procedures, including electrophilic functionalization, the Sandmeyer reaction and ortho-lithiation methods, for instance.7 Aware of the limitations of such strategies, including regioselectivity and functional group tolerance, synthetic chemists have developed more efficient methods, such as transition-metal-catalyzed direct C–H halogenation, transition-metal-free oxidative halogenations, visible-light-induced halogenations and enzymatic halogenations, for instance.8
In light of the contemporary concern to discover more sustainable, cost-effective and safe protocols, there is a high demand for the development of procedures that avoid the use of expensive catalysts, hazardous chemical oxidants, and stoichiometric reagents to minimize hazards and synthetic waste. For that purpose, electrochemical transformations provide significant benefits. Organic electrosynthesis offers mild reaction conditions, while providing high chemoselectivity, and minimal waste generation since it relies on the use of electrons as the main reagent.9 Therefore, synthetic organic electrochemistry has emerged as one of the most important and environmentally benign tools in organic synthesis, particularly when the electricity is produced from renewable sources. Hence, as a part of our ongoing research program dedicated to the functionalization of organic molecules through the addition of electrogenerated radicals,10 we sought to develop a versatile methodology to access synthetically useful Br-containing building blocks, avoiding the use of harmful brominating agents. With the resurgence of electrochemistry, significant efforts have been invested in the development of sustainable bromination procedures.
Various C(sp2)– and C(sp3)–Br bonds formed on heteroaryl, aryl, alkene, alkyne, alkyl and carboxylic acid derivatives were successfully developed through an electrochemical process (Scheme 1A).11–15
In addition, the enamide scaffold is ubiquitous in a wide range of pharmaceuticals and natural products and is widely recognized as a linchpin in organic synthesis.16 Quite surprisingly, the synthesis of halogenated enamides has been underexplored and has relied on elaborate reaction systems. Indeed, to our knowledge, access to halogenated enamides has focused on transition-metal-catalyzed or transition-metal-free amido-halogenation of alkynes,17,18 radical addition onto alkynes,19 hydroamidation of halogenated alkynes,20 or haloolefination of formamides.21 Hence, in light of the conspicuous absence of a practical, sustainable and direct approach toward halogenated enamides, we surmised that the electrochemical generation of bromide radicals through anodic oxidation would offer smooth and sustainable access to these interesting scaffolds in a single operation from enamides (Scheme 1B).
Results and discussion
At the outset of our investigations, enamide 1 was chosen as the model substrate and ammonium bromide was used as the bromine source to initiate the reaction (Table 1, entry 1). Pleasingly, 2 was obtained with a 37% NMR yield by using a carbon graphite anode and a platinum cathode, under a constant current of 5 mA with a total charge of 3 F mol−1 in acetonitrile (CH3CN) under an air atmosphere in an undivided cell. Variation of the solvent of the reaction showed that DMA allowed a slight increase in the yield, reaching 43%. Thus, we pursued the optimization using DMA as the solvent. The use of NaBr in place of NH4Br as the bromine source resulted in a better yield (55%), while the use of nBu4NBr as both the electrolyte and the bromine source was deleterious for the outcome of the reaction. We then tried to replace the platinum cathode with a glassy carbon (entry 5), a stainless-steel (entry 6) or a carbon graphite (entry 7) cathode.| Entry | Br reagent | Solvent | Electrodes | Yieldsa (%) |
|---|---|---|---|---|
| a 1H NMR yield using dibromomethane as an internal standard. b Isolated yield. c Reaction without electrolyte. C: carbon graphite. GC: glassy carbon. SST: stainless steel. | ||||
| 1 | NH4Br | CH3CN | C(+)IPt(−) | 37 |
| 2 | NH4Br | DMA | C(+)IPt(−) | 43 |
| 3 | nBu4NBr | DMA | C(+)IPt(−) | 55 |
| 4 | NaBr | DMA | C(+)IPt(−) | 0 |
| 5 | NaBr | DMA | C(+)IGC(−) | 70 |
| 6 | NaBr | DMA | C(+)ISST(−) | 81b |
| 7 | NaBr | DMA | C(+)IC(−) | 83b |
| 8 | NaBr | DMA | SST(+)IC(−) | 0 |
| 9c | NaBr | DMA | C(+)IC(−) | 50 |
Pleasingly, 2 was isolated in 81% and 83% yields, respectively, using a stainless-steel or a carbon graphite cathode (entries 6 & 7). Moreover, the use of a carbon graphite anode was crucial for the formation of product 2 (entry 8). Then, to increase the atom economy and the sustainability of the process, the electrolyte was removed from the reaction conditions (entry 9). Unfortunately, its absence was deleterious for the reaction outcome, since a moderate 50% NMR yield was obtained. Although NaBr could be used as an electrolyte, increasing the amount to 8.0 equiv. did not result in a decent yield. To showcase the added value of our electrochemical bromination protocol, we compared its efficiency with classical electrophilic bromination conditions (Table 2). The reaction of 1 with NBS (N-bromosuccinimide) under classical conditions gave the product 2 in a modest yield of 36% along with a significant amount of side products (Table 2, entry 2).22 Likewise, the use of pyridinium tribromide did not improve the reaction efficiency and 2 was isolated in a modest 23% NMR yield (Table 2, entry 3).23 Overall, the electrochemical bromination outcompetes the classical brominating reagents. These results clearly highlight the added value of our electrochemical bromination protocol. In addition, it offers a sustainable approach as it avoids the use of an electrophilic brominating agent and uses the eco-friendly and harmless sodium bromide.
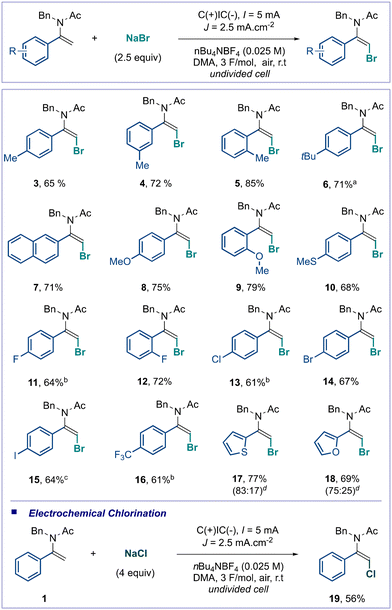
Having delineated the optimal reaction conditions (see Table 1, entry 7), the scope of this electrochemical bromination of α-aryl enamides was investigated (Scheme 2). The reaction efficiency was not affected by the substitution pattern. α-Aryl enamides substituted by a methyl group at the para-, meta- or ortho-position afforded the desired products (3–5) in good yields as a single stereoisomer, showing no impact of the steric hindrance. The reaction with enamides bearing electron-donating groups on the aryl residue such as tertbutyl, methoxy and thiomethyl ether, as well as the naphthyl derivative, proceeded well, producing the corresponding products (6–10) in high yields. Pleasingly, no further oxidation of the sulfur atom was observed under our electrochemical conditions. It should be noted that once again the steric hindrance did not impact the reaction outcome as a larger methoxy group was installed at the ortho-position, allowing for electrochemical bromination with 79% yield (see product 9). Halogen atoms were well tolerated under our electrochemical conditions as the aryl moiety substitution with fluorine (11 and 12), chlorine (13), bromine (14) and iodine (15) gave the corresponding products with good yields, enabling opportunities for further transformations through metal-catalyzed cross-coupling, for instance. The reaction with the trifluoromethyl group at the para-position of the aryl moiety efficiently afforded the desired brominated enamides with a good yield of 61% at the cost of an increased charge from 3 F mol−1 to 4.5 F mol−1. Due to the high interest in heteroaromatic compounds in medicinal chemistry and crop science, heteroaryl enamides were also reacted under our optimal reaction conditions and were proved to be efficient substrates, yielding two valuable heteroaromatic molecules (17 and 18). In these cases, the bromoenamides were obtained as a separable E/Z mixture of stereoisomers, the E being the major one. To showcase the versatility of our transformation, sodium bromide was replaced by sodium chloride. To our delight, the electrochemical chlorination occurred under similar reaction conditions with a slightly lower efficiency, giving access to the unknown chlorinated enamide 19, with a good yield of 56%. Importantly, the reaction was highly stereoselective, as all substrates were exclusively obtained as (E)-isomers, except the heteroaromatic derivatives 17 and 18. Unfortunately, some enamide substrates remained reluctant or poorly reactive under our reaction conditions. Aliphatic enamides showed no reactivity, highlighting the importance of the formation of a stabilized benzylic radical in our transformation, and enamides derived from cyclic olefins were poorly reactive, leading to trace formation of the desired products.24
Then, to highlight the synthetic utility of the obtained products, we envisioned taking advantage of a halogen atom to develop original C–C bond formation on the enamide scaffold (Scheme 3). To the best our knowledge, no report dealing with the functionalization of halogenated enamides has been reported to date. Thus, we took advantage of the bromide atom to develop a protocol for the Suzuki cross-coupling reaction, a pivotal transformation in medicinal chemistry and API manufacturing.25 Pleasingly, with our developed reaction conditions, excellent yields were obtained using aryl boronic acids bearing electron-donating or -withdrawing substituents. Methyl, trifluoromethyl, ethyl ester, cyano and aldehyde substituents were very well tolerated, affording the desired products (20–24) with yields ranging from 91% to 99%. In addition, an excellent yield of 99% was obtained by reacting 2 with an heteroaryl boronic acid, i.e. benzofuran-2-ylboronic acid. Then, the reaction with β-styrylboronic acid afforded the corresponding (E,E)-dienes in a very good yield of 96%, with a complete stereoretention of the starting olefins. Finally, we showcased the possible use of alkyl boronic acid, through the reaction with cyclopropyl boronic acids, which afforded the product 27 in a fairly decent yield of 59%.
Finally, we developed a procedure for the Sonogashira cross-coupling reaction to access enyne derivatives. The use of standard reaction conditions (Pd(PPh3)2Cl2, CuI in Et3N) allowed the reaction of 2 with ethynyl-4-methylbenzene, 1-ethynylcyclohexene and ethynyltrimethylsilane. The corresponding enynes 28, 29 and 30 were obtained with excellent yields ranging from 93% to more than 99%. Finally, we applied our protocol to the reaction of 2 with mestranol, a complex bioactive molecule. The reaction proceeded smoothly and the product 31 was obtained in an excellent yield of 82%, showcasing the versatility of the transformation toward complex structures. Overall, these novel transformations highlighted the synthetic utility of these building blocks. Then, to gain insights into the reaction mechanism, cyclic voltammetry measurements were carried out (Scheme 4A).
The CV measurements of NaBr (blue curve) showed two successive oxidative waves (Scheme 4A), while no oxidation of enamide (grey curve) was observed in the potential range studied (up to +1.5 V). Moreover, the analysis of a mixture of NaBr and enamide demonstrated a similar profile without any increase of oxidative potential, meaning that there is no bromine mediation in the plausible enamide oxidation (Scheme 4). With all these data to hand, we suggested a plausible reaction mechanism, starting with the anodic oxidation of bromine from sodium bromide (Scheme 4B). The resulting electrophilic bromine radical could then add onto the enamide, leading to a stabilized nucleophilic carbon centered radical A. A subsequent anodic oxidation of this open-shell intermediate generated the carbocation B, which then readily underwent E1 elimination to furnish the desired and less congested brominated enamide. A final cathodic reduction of H+, resulting from the elimination, would release H2, equilibrating the redox balance.
Conclusions
In conclusion, we depicted herein the unprecedent electrochemical bromination of enamide derivatives using non-toxic and inexpensive sodium bromine as a precursor of Br˙. The reaction conditions proved to be tolerant to various electron-donating or -withdrawing chemical substituents and provided the resulting products with excellent (E)-stereoselectivity in good to excellent yields (56–85%). The standard reaction conditions proved to be efficient for the electrochemical chlorination of enamide upon the simple replacement of NaBr by NaCl. The synthetic utility of the products was highlighted through Suzuki and Sonogashira cross-coupling reactions. Pleasingly, excellent yields were obtained, and the tri-substituted enamides were stereoselectively obtained from the brominated (E)-enamide under simple and practical reaction conditions. The efficiency of these novel reaction manifolds on enamide demonstrated their versatility toward the expansion of the chemical space. A mechanistic analysis of the reaction supported a plausible reaction pathway involving a Br˙ radical, generated from the anodic oxidation of bromide. We hope that the sustainable synthesis of these novel building blocks will offer new opportunities to expand the chemical space, particularly in medicinal chemistry.Author contributions
S. L., T. C. and T. P. conceived and designed the experiments. S. L. performed the experiments. S. L., T.C. and T.P. analyzed the data. T. C. wrote the manuscript with the input from all authors.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was partially supported by Normandie Université (NU), Région Normandie, the Centre National de la Recherche Scientifique (CNRS), Université de Rouen Normandie (URN), INSA Rouen Normandie, Labex SynOrg (ANR-11-LABX-0029), the Graduate School for Research XL-Chem (ANR-18-EURE-0020 XLCHEM) and Innovation Chimie Carnot (I2C). This work is part of the EFLUX program supported by the European Union through the operational program FEDER/FSE 2014-202. T. P. thanks the Région Normandie for support (RIN TREMPLIN EFLUX). S. L. thanks the Chinese Scholarship Council (CSC) for a doctoral fellowship.References
- (a) G. W. Gribble, Acc. Chem. Res., 1998, 31, 141–152 CrossRef CAS; (b) G. W. Gribble, J. Chem. Educ., 2004, 81, 1441–1449 CrossRef CAS.
- (a) S. Caron, in Practical Synthetic Organic Chemistry: Reactions, Principles, and Techniques, Wiley, Hoboken, New Jersey, 2011 CrossRef; (b) R. C. Larock, Comprehensive Organic Transformations, 2nd ed, Wiley-VCH, New York, 1999 Search PubMed; (c) H. House, in Modern Synthetic Reactions, 2nd ed, W. A. Benjamin, New York, 1972, pp. 459–478 Search PubMed; (d) N. De Kimpe and R. Verhé, The Chemistry of α-Haloketones, α-Haloaldehydes, and α-Haloimines, John Wiley & Sons, New York, 1988 CrossRef; (e) R. Lin, A. P. Amrute and J. Pérez-Ramírez, Chem. Rev., 2017, 117, 4182–4247 CrossRef CAS PubMed.
- (a) S. Bräse and A. De Meijere, in Metal-Catalyzed Cross-Coupling Reactions, Wiley-VCH, New York, 2004 Search PubMed; (b) N. Kambe, T. Iwasaki and J. Terao, Chem. Soc. Rev., 2011, 40, 4937–4947 RSC.
- (a) K. Müller, C. Faeh and F. Diederich, Science, 2007, 317, 1881–1886 CrossRef PubMed; (b) S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Chem. Soc. Rev., 2008, 37, 320–330 RSC; (c) P. Jeschke, in Modern Crop Protection Compounds, Wiley-VCH, Weinheim, 2013 Search PubMed; (d) P. Jeschke, Pest Manage. Sci., 2017, 73, 1053–1066 CrossRef CAS PubMed; (e) R. Wilcken, M. O. Zimmermann, A. Lange, A. C. Joerger and F. M. Boeckler, J. Med. Chem., 2013, 56, 1363–1388 CrossRef CAS PubMed; (f) Y. Lu, T. Shi, Y. Wang, H. Yang, X. Yan, X. Luo, H. Jiang and W. Zhu, J. Med. Chem., 2009, 52, 2854–2862 CrossRef CAS PubMed; (g) M. J. Dagani, H. J. Barda, T. J. Benya and D. C. Sanders, Bromine Compounds In Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH Verlag GmbH & Co, KGaA, 2000 Search PubMed; (h) B. R. Smith, C. M. Eastman and J. T. Njardarson, J. Med. Chem., 2014, 57, 9764–9773 CrossRef CAS PubMed.
- https://www.essentialchemicalindustry.org/chemicals/bromine.html .
- (a) D. Ioffe and A. Kampf, in Kirk-Othmer Encyclopedia of Chemical Technology, 5th ed, John Wiley &Sons, Inc., New York, 2004, Vol. 4, pp 340–345 Search PubMed; (b) D. L. Turner, J. Food Sci., 1972, 37, 791–792 CrossRef CAS; (c) D. A. Laskowski, Rev. Environ. Contam. Toxicol., 2002, 174, 49–170 CAS; (d) J. Green, J. Fire Sci., 1996, 14, 426–442 CrossRef CAS; (e) G. W. Gribble, Chem. Soc. Rev., 1999, 28, 335–346 RSC; (f) S. J. Decanio, Contemp. Econ. Policy, 2005, 23, 376–393 CrossRef; (g) J. Kaspersma, C. Doumen, S. Munro and A.-M. Prins, Polym. Degrad. Stab., 2002, 77, 325–331 CrossRef CAS.
- (a) H. Gilman, in Organic Chemistry: An Advanced Treatise, Wiley, New York, 1938, Vol. 1, pp. 36–43 Search PubMed; (b) P. B. D. de la Mare, in Electrophilic Halogenation, Cambridge University Press, New York, 1976 Search PubMed; (c) I. Saikia, A. J. Borah and P. Phukan, Chem. Rev., 2016, 116, 6837–7042 CrossRef CAS PubMed; (d) Y. Xiang, P.-Y. Caron, B. M. Lillie and R. Vaidyanathan, Org. Process Res. Dev., 2008, 12, 116–119 CrossRef CAS; (e) Z. Wang, in Comprehensive Organic Name Reactions and Reagents, Wiley, 2010 CrossRef; (f) V. Snieckus, Chem. Rev., 1990, 90, 879–933 CrossRef CAS; (g) H. Hodgson, Chem. Rev., 1947, 47, 251–277 CrossRef PubMed.
- Selected reviews: (a) A. Paik, S. Paul, S. Bhowmik, R. Das, T. Naveen and S. Rana, Asian J. Org. Chem., 2022, 11, e202200060 CrossRef CAS; (b) R. Das and M. Kapu, Asian J. Org. Chem., 2018, 7, 1524–1541 CrossRef CAS; (c) F. H. Vaillancourt, E. Yeh, D. A. Vosburg, S. Garneau-Tsodikova and C. T. Walsh, Chem. Rev., 2006, 106, 3364–3378 CrossRef CAS PubMed; (d) A. Podogoršek, M. Zupan and J. Iskra, Angew. Chem., Int. Ed., 2009, 48, 8424–8450 CrossRef PubMed; (e) X.-W. Liang, C. Zheng and S.-L. You, Chem. – Eur. J., 2016, 22, 11918–11933 CrossRef CAS PubMed; (f) S. E. Denmark, W. E. Kuester and M. T. Burk, Angew. Chem., Int. Ed., 2012, 51, 10938–10953 CrossRef CAS PubMed; (g) C. Crove, S. Molyneux, S. V. Sharma, Y. Zhang, D. S. Gkotsi, H. Connaris and R. J. M. Goss, Chem. Soc. Rev., 2021, 50, 9443–9481 RSC; (h) H. Tu, S. Zhu, F.-L. Qing and L. Chu, Tetrahedron Lett., 2018, 59, 173–179 CrossRef CAS; (i) S. France, A. Weatherwax and T. Lectka, Eur. J. Org. Chem., 2005, 475–479 CrossRef CAS; (j) A. M. Arnold, A. Ulmer and T. Gulder, Chem. – Eur. J., 2016, 22, 8728–8739 CrossRef CAS PubMed.
- (a) Organic Electrochemistry, 5th Ed, ed. O. Hammerich and B. Speiser, CRC Press, Taylor and Francis, New-York, 2016 Search PubMed; (b) C. Kingston, M. D. Palkowitz, Y. Takahira, J. C. Vantourout, B. K. Peters, Y. Kawamata and P. S. Baran, Acc. Chem. Res., 2020, 53, 72–83 CrossRef CAS PubMed; (c) D. Pollok and S. R. Waldvogel, Chem. Sci., 2020, 11, 12386–12400 RSC; (d) K. Yamamoto, M. Kuriyama and O. Onomura, Acc. Chem. Res., 2020, 53, 105–120 CrossRef CAS PubMed; (e) S. Cembellin and B. Batanero, Chem. Rec., 2021, 21, 2453–2471 CrossRef CAS PubMed; (f) C. Zhu, N. W. J. Ang, T. H. Meyer, Y. Qiu and L. Ackermann, ACS Cent. Sci., 2021, 7, 415–431 CrossRef CAS PubMed; (g) Y. Yuan and A. Lei, Acc. Chem. Res., 2019, 52, 3309–3324 CrossRef CAS PubMed; (h) K. Mitsudo, Y. Kurimoto, K. Yoshioka and S. Suga, Chem. Rev., 2018, 118, 5985–5999 CrossRef CAS PubMed; (i) S. R. Waldvogel, S. Lips, M. Selt, B. Riehl and C. J. Kampf, Chem. Rev., 2018, 118, 6706–6765 CrossRef CAS PubMed; (j) M. Yan, Y. Kawamata and P. S. Baran, Chem. Rev., 2017, 117, 13230–13319 CrossRef CAS PubMed; (k) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 CrossRef CAS PubMed; (l) D. Pollok and S. R. Waldvogel, Chem. Sci., 2020, 11, 12386–12400 RSC.
- (a) S. Luan, T. Castanheiro and T. Poisson, Org. Lett., 2023, 25, 1678–1682 CrossRef CAS PubMed; (b) M. Aelterman, M. Sayes, P. Jubault and T. Poisson, Chem. – Eur. J., 2021, 27, 8277–8282 CrossRef CAS PubMed; (c) T. Biremond, P. Jubault and T. Poisson, ACS Org. Inorg. Au, 2022, 2, 148–152 CrossRef CAS PubMed; (d) T. Cantin, A. B. Charette, T. Poisson and P. Jubault, Synthesis, 2023, 55, 2943–2950 CrossRef CAS; (e) M. Aelterman, P. Jubault and T. Poisson, Eur. J. Org. Chem., 2023, e202300063 CrossRef CAS; (f) M. Aelterman, T. Biremond, P. Jubault and T. Poisson, Chem. – Eur. J., 2022, 28, e2022002194 CrossRef PubMed; (g) S. Luan, T. Castanheiro and T. Poisson, Adv. Synth. Catal., 2022, 364, 2741–2747 CrossRef CAS.
- (a) T. Yuan, A. Yao, Y. Zheng, M. Gao, Z. Zhou, J. Qiao, J. Hu, B. Ye, J. Zhao, H. Wen and A. Lei, iScience, 2019, 12, 293–303 CrossRef PubMed; (b) W. Xie, S. Ning, N. Liu, Y. Bai, S. Wang, S. Wang, L. Shi, X. Che and J. Xiang, Synlett, 2019, 30, 1313–1316 CrossRef CAS; (c) P. Zhang, J. Chen, W. Gao, Y. Xiao, C. Liu, S. Xu, X. Yan and D. Qin, Molecules, 2019, 24, 696 CrossRef CAS PubMed; (d) J. Wu, H. Abou-Hamdan, R. Guillot, C. Kouklovsky and G. Vincent, Chem. Commun., 2020, 56, 1713–1716 RSC; (e) L. Sun, X. Zhang, Z. Li, J. Ma, Z. Zeng and H. Jiang, Eur. J. Org. Chem., 2018, 4949–4952 CrossRef CAS; (f) X. Liu, Z. Wu, C. Feng, W. Liu, M. Li and Z. Shen, Eur. J. Org. Chem., 2022, e202200262 CrossRef CAS; (g) Z. Zhou, Y. Yuan, Y. Cao, J. Qiao, A. Yao, J. Zhao, W. Zuo, W. Chen and A. Lei, Chin. J. Chem., 2019, 37, 611–615 CrossRef CAS; (h) Z. Tan, Y. Liu, R. Helmy, N. R. Rivera, D. Hesk, S. Tyagarajan, L. Yang and J. Su, Tetrahedron Lett., 2017, 58, 3014–3018 CrossRef CAS.
- (a) K. Kulangiappar, G. Karthik and M. Anbu Kulandainathan, Synth. Commun., 2013, 39, 2304–2309 CrossRef; (b) T. Raju, K. Kulangiappar, M. Anbu Kulandainathan, U. Uma, R. Malini and A. Muthukumaran, Tetrahedron Lett., 2006, 47, 4581–4584 CrossRef CAS; (c) T. Raju, K. Kulangiappar, M. Anbu Kulandainathan and A. Muthukumaran, Tetrahedron Lett., 2005, 46, 7047–7050 CrossRef CAS; (d) X. Yang, Q.-L. Yang, X.-Y. Wang, H.-H. Xu, T.-S. Mei, Y. Huang and P. Fang, J. Org. Chem., 2020, 85, 3497–3507 CrossRef CAS PubMed; (e) Q. Liu, B. Sun, Z. Liu, Y. Kao, B.-W. Dong, S.-D. Jiang, F. Li, G. Liu, Y. Yang and F. Mo, Chem. Sci., 2018, 9, 8731–8737 RSC.
- (a) K. Kulangiappar, M. Ramprakash, D. Vasudevan and T. Raju, Synth. Commun., 2015, 46, 145–153 CrossRef; (b) C. Wan, R.-J. Song and J.-H. Li, Org. Lett., 2019, 21, 2800–2803 CrossRef CAS PubMed; (c) T.-T. Zhang, M.-J. Luo, Y. Li, R.-J. Song and J.-H. Li, Org. Lett., 2020, 22, 7250–7254 CrossRef CAS PubMed; (d) M.-X. Bi, P. Qian, Y.-K. Wang, Z.-G. Zha and Z.-Y. Wang, Chin. Chem. Lett., 2017, 28, 1159–1162 CrossRef CAS.
- R. Jagatheesan, K. Joseph Santhana Raj, S. Lawrence and C. Christopher, RSC Adv., 2016, 6, 35602–35608 RSC.
- Q.-L. Yag, X.-Y. Wang, T.-L. Wang, X. Yang, D. Liu, X. Tong, X.-Y. Wu and T.-S. Mei, Org. Lett., 2019, 21, 2645–2649 CrossRef PubMed.
- For selected reviews, see: (a) K. Gopalaiah and H. B. Kagan, Chem. Rev., 2011, 111, 4599–4657 CrossRef CAS PubMed; (b) R. Matsubara and S. Kobayashi, Acc. Chem. Res., 2008, 41, 292–301 CrossRef CAS PubMed; (c) T. Courant, G. Dagousset and G. Masson, Synthesis, 2015, 47, 1799–1826 CrossRef CAS; (d) G. Bernadat and G. Masson, Synlett, 2014, 25, 2842–2867 CrossRef CAS; (e) D. R. Carbery, Org. Biomol. Chem., 2008, 6, 3455–3460 RSC; (f) N. Gigant, L. Chausset-Boissarie and I. Gillaizeau, Chem. – Eur. J., 2014, 20, 7548–7564 CrossRef CAS PubMed.
- (a) J. Qian, Y. Liu, J. Zhu, B. Jiang and Z. Xu, Org. Lett., 2011, 13, 4220–4223 CrossRef CAS PubMed; (b) T. Xu and G. Liu, Org. Lett., 2012, 14, 5416–5419 CrossRef CAS PubMed; (c) C. Jonasson, A. Horvath and J.-E. Backvall, J. Am. Chem. Soc., 2000, 122, 9600–9609 CrossRef CAS; (d) S. Karur, S. R. S. S. Kotti, X. Xu, J. F. Cannon, A. Headley and G. Li, J. Am. Chem. Soc., 2003, 125, 13340–13341 CrossRef CAS PubMed; (e) X. Ji, H. Huang, W. Wu and H. Jiang, J. Am. Chem. Soc., 2013, 135, 5286–5289 CrossRef CAS PubMed; (f) C. Liu and F. Yang, Eur. J. Org. Chem., 2019, 6867–6870 CrossRef CAS; (g) W. Wang, L. Liu, W. Chang and J. Li, Chem. – Eur. J., 2018, 24, 8542–8547 CrossRef CAS PubMed; (h) X. Ji, H. Huang, W. Xiong, K. Huang, W. Wu and H. Jiang, J. Org. Chem., 2014, 79, 7005–7011 CrossRef CAS PubMed.
- (a) J. Chai, W. Ding, C. Wang, S. Ito, J. Wu and N. Yoshikai, Chem. Sci., 2021, 12, 15128–15133 RSC; (b) T. Wang, Z. Tang, H. Luo, Y. Tian, M. Xu, Q. Lu and B. Li, Org. Lett., 2021, 23, 6293–6298 CrossRef CAS PubMed; (c) S. Arepally, A. Chamuah, N. Katta and D. S. Sharada, Eur. J. Org. Chem., 2019, 1542–1547 CrossRef CAS; (d) M. Li, H. Yuan, B. Zhao, F. Liang and J. Zhang, Chem. Commun., 2014, 50, 2360–2363 RSC.
- U. Wille, O. Krüger, A. Kirsch and U. Lüning, Eur. J. Org. Chem., 1999, 3185–3189 CrossRef CAS.
- (a) M. Yamagishi, K. Nishigai, T. Hata and H. Urabe, Org. Lett., 2011, 13, 4873–4875 CrossRef CAS PubMed; (b) M. Yamagishi, K. Nishigai, A. Ishii, T. Hata and H. Urabe, Angew. Chem., Int. Ed., 2012, 51, 6471–6474 CrossRef CAS PubMed.
- A. E. Pasqua, J. J. Crawford, D.-L. Long and R. Marquez, J. Org. Chem., 2012, 77, 2149 CrossRef CAS PubMed.
- R. H. Mitchell, Y.-H. Lai and R. W. Williams, J. Org. Chem., 1979, 44, 4733 CrossRef CAS.
- D. Bliman, M. Pettersson, M. Bood and M. Grotli, Tetrahedron Lett., 2014, 55, 2929 CrossRef CAS.
- See ESI† for details.
- (a) A. J. J. Lennox and G. C. Lloyd-Jones, Chem. Soc. Rev., 2014, 43, 412–443 RSC; (b) N. Miyaura and A. Suzuki, Chem. Rev., 1995, 95, 2457–2483 CrossRef CAS; (c) S. Namirembe and J. P. Morken, Chem. Soc. Rev., 2019, 48, 3464–3474 RSC; (d) J. W. B. Fyfe and A. J. B. Watson, Chem, 2017, 3, 31–55 CrossRef CAS; (e) J. Carreras, A. Caballero and P. J. Pérez, Chem. – Asian J., 2019, 14, 329–343 CrossRef CAS PubMed; (f) L. Xu, S. Zhang and P. Li, Chem. Soc. Rev., 2015, 44, 8848–8858 RSC; (g) G. Duret, R. Quinlan, P. Bisseret and N. Blanchard, Chem. Sci., 2015, 6, 5366–5382 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3gc04723g |
| This journal is © The Royal Society of Chemistry 2024 |




![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.5 F mol−1 charge was applied.
3.5 F mol−1 charge was applied. 
