Fullerene as a probe molecule for single-atom oxygen reduction electrocatalysts†
Ning
Li‡
a,
Kun
Guo‡
 *a,
Song
Lu
b,
Lipiao
Bao
*a,
Song
Lu
b,
Lipiao
Bao
 a,
Zhixin
Yu
a,
Zhixin
Yu
 c and
Xing
Lu
c and
Xing
Lu
 *a
*a
aState Key Laboratory of Materials Processing and Die & Mould Technology, School of Materials Science and Engineering, Huazhong University of Science and Technology, Wuhan 430074, China. E-mail: guok@hust.edu.cn; lux@hust.edu.cn
bInstitute of New Energy, School of Chemistry and Chemical Engineering, Shaoxing University, Shaoxing 312000, China
cDepartment of Energy and Petroleum Engineering, University of Stavanger, Stavanger 4036, Norway
First published on 25th September 2024
Abstract
Fullerenes interact positively with many metal-based catalysts via intense electron transfer. Yet, we here revealed that C60 serves as a probe due to its deactivation of the active sites of single-atom O2 reduction electrocatalysts. C60 adsorption to metal atoms creates steric hindrance that restricts the access of O2 to the active sites.
Fullerenes are π-conjugated molecular carbon allotropes with a set of fascinating structural and electronic properties that have triggered significant interest from the catalysis community.1–3 For instance, the abundant native pentagons render fullerenes an unparalleled feedstock to construct intrinsic defect-rich carbon catalysts.4–6 The metal-like electrical conductivity brings higher fullerene C96 with superior oxygen reduction reaction (ORR) activity to that of semiconducting C60 and C70.7 Besides, covalent 2D fullerene networks with suitable band gaps and high carrier mobility are potent photocatalysts for water splitting.8,9
Recently, fullerenes as powerful regulators of the localized electronic structures of metal-based catalysts are increasingly being reported. For example, the exceptional redox capability renders C60 molecules as a robust electron-buffering agent between Cu0/Cu+ and Cu2+/Cu+ couples.10 Compared to Cu/SiO2 with dominant Cu0 species, C60 can extract electrons from Cu0 to form C60− and Cu+. Meanwhile, C60− can also donate electrons to Cu2+, forming C60 and Cu+. As a result, crucial Cu+ species are stabilized by C60 to enhance the hydrogenation activity of C60-Cu/SiO2. In pursuit of the monovalent Cu+ species that play a pivotal role in Cu-based catalysts towards CO2 electroreduction, researchers have devised C60-adsorbed Cu metals as superior electrocatalysts to bare Cu.11,12 Likewise, intense interfacial electron transfer induced by fullerene molecules has also been evidenced to enhance the catalytic performance of metallic substrates, such as CoNi layered double hydroxides13 and MoS2.14,15 Nevertheless, positive electronic interactions are uncovered only between fullerenes and large-sized metallic aggregates. How sub-nanoscale fullerenes would affect the metal active sites at the atomic level remains an unresolved issue. It is thus of high interest to explore whether fullerenes can still optimize the electronic structure of the metal site where they are adsorbed to, behaving like a feasible axial ligand of metal single-atom catalysts (SACs).16–21 Or alternatively, the larger sizes of fullerene molecules than that of metal atoms may result in the deactivation of the metal site by surface blocking, functioning as a molecular probe of the active site of SACs.
Macrocycles bearing representative M–N4–C moieties have been widely exploited as model catalysts to investigate various carbon-hosted SACs.22–24 Utilizing metalloporphyrin (MP) as a molecular SAC, we unravel the unexpected inhibiting effect of C60 in the ORR activity of MP. Control experiments indicate that non-covalently anchoring either cobalt porphyrin (CoP) or C60 on carbon nanotubes (CoP/CNT and C60/CNT) leads to much higher ORR activity than individual CoP, C60, and CNTs. However, further addition of C60 to CoP/CNT (C60-CoP/CNT) results in a dramatic ORR activity decay, the same as that observed after adding the well-noted thiocyanate (SCN−) poison. Density functional theory (DFT) calculations reveal that the planar MP molecule is confined between the CNT matrix and C60 molecule, creating high steric hindrance that restricts O2 access to the active Co atoms.
CoP/CNT and C60-CoP/CNT are prepared via a straightforward π interaction-enabled adsorption strategy by mixing different precursors in CS2 solvent (Experimental section, ESI†). In particular, CoP is selected because it presents better ORR activity than the other common 3d metal MPs (Fig. S1, ESI†).25 The mass loading of CoP is optimized to be 50 wt% due to the highest ORR activity among a series of CoP/CNT (Fig. S2, ESI†). The same molar quantity of C60 as that of CoP is then added in C60-CoP/CNT. The morphology and structure of the as-prepared materials are first examined by scanning and transmission electron microscopies (SEM and TEM). The typical 1D morphology is clearly seen in the commercially-received CNTs (Fig. 1a). Despite the high tendency for the self-assembly of CoP and C60 into large molecular crystals, no obvious aggregates are discerned in either CoP/CNT (Fig. 1b) or C60-CoP/CNT (Fig. 1c), which could be attributed to the improved dispersity of both molecules on CNTs. This is further supported by their TEM images. It is found that these CNTs are composed of multi-walled tubes with diameters of dozens of nanometers (Fig. 1d). The uniform image contrasts indicate the absence of large-sized crystalline particles in CoP/CNT (Fig. 1e) and C60-CoP/CNT (Fig. 1f). Additional SEM and TEM images are presented in Fig. S3 (ESI†).
The crystallinity of the materials under study is investigated by X-ray diffraction (XRD). As shown in Fig. 1g, a major characteristic peak at ∼26°, corresponding to the (002) plane of graphitic carbon, is observed for the CNTs. Pure CoP and C60 show well-defined characteristic peaks, reflecting the fact that they are easily self-assembled into crystalline particles owing to intermolecular forces.26 After loading CoP on the CNTs, aside from the diffraction peak of the CNTs, two broad peaks that can be assigned to CoP are discerned, indicating the significantly decreased crystallinity of CoP. A similar phenomenon is also observed after further addition of C60 to form C60-CoP/CNT, in which the C60 phase presents much lower crystallinity than pure C60. The results again manifest that CNTs substantially mitigates the severe aggregation of CoP and C60 molecules.27
Raman spectroscopy is further employed to analyze the chemical structures of these materials. Typical D and G bands of graphitic carbon materials at 1340 and 1570 cm−1 are observed for CNTs, CoP/CNT, and C60-CoP/CNT (Fig. 1h). Besides, multiple weak Raman bands originating from CoP are also observed in CoP/CNT, whereas an additional band at 1460 cm−1, namely the Ag(2) mode of C60, is discerned in C60-CoP/CNT. The Raman results also confirm that only small-sized CoP and C60 aggregates are formed after their hybridization with CNTs. Elemental states and speciation information of CoP, CoP/CNT, and C60-CoP/CNT are further acquired by X-ray photoelectron spectroscopy (XPS). The full-survey XPS spectra verify the existence of C, N, and Co elements (Fig. S4, ESI†). Fig. 1i displays the high-resolution XPS spectra of the Co 2p and N 1s regions, in which the Co 2p spectra can be resolved into two spin–orbit doublets, Co 2p1/2 and Co 2p3/2, whereas the peak centered at 399.2 eV can be assigned to the pyrrolic N of CoP. Compared to CoP, the intensities of the Co 2p1/2, Co 2p3/2, and N 1s peaks for CoP/CNT become substantially lower, coinciding with the lower mass ratio of CoP in CoP/CNT. Importantly, the Co 2p and N 1s peaks are barely observable for C60-CoP/CNT, indicating that the addition of C60 may lead to the surface coverage of CoP to a certain depth that is beyond the detection limit of XPS.
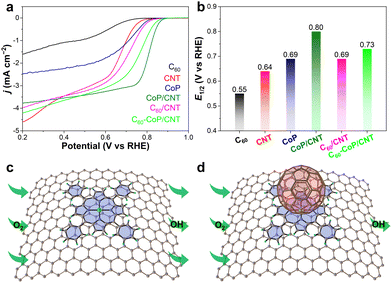
The ORR performance of the catalysts under study is then evaluated using the standard rotating disk electrode technique in 0.1 M KOH electrolyte (pH = 13). Fig. 2a shows the polarization curves of the ORR on six different catalysts at 1600 rpm. Their corresponding half-wave potentials (E1/2) are measured and compared in Fig. 2b. Either C60 or CNT alone presents rather poor ORR activity with low E1/2 values of 0.55 and 0.64 V, respectively. Despite the presence of Co SACs, CoP still displays limited ORR activity (0.69 V) due probably to severe molecular aggregation and low electrical conductivity. After non-covalently dispersing CoP molecules on conductive CNTs, a significant increase in E1/2 is observed for CoP/CNT (0.80 V), affirming the positive role of the CNT support. This is also supported by loading C60 onto the CNTs to gain C60/CNT, which gives a higher E1/2 of 0.69 V than that of individual C60 and CNTs, in good line with the results of a previous study.28 Rotating ring-disk electrode analysis reveals a highly-selective four-electron O2-to-OH− ORR pathway on CoP/CNT (Fig. S5, ESI†). More intriguingly, the further addition of C60 to CoP/CNT leads to an unexpected decrease of E1/2 to 0.73 V, indicating an eminent ORR activity decay of Co SACs caused by C60 molecules. Due to the π–π stacking interactions, it is no wonder that CoP molecules are planarly adsorbed on the CNT surface, allowing O2 reactants to be readily adsorbed to and reduced on the exposed Co sites, as illustrated in Fig. 2c. Once C60 molecules are introduced, they could be adsorbed to either CNTs or CoP owing to the analogous π–π stacking interactions. Given the deteriorated ORR activity of C60-CoP/CNT and the aforesaid material characterization results, it is rationalized that C60 shall be situated on top of CoP, potentially blocking the access of O2 to the ORR-active Co sites (Fig. 2d). On this account, C60 is not an expected facilitator, but rather can serve as a probe molecule to identify the active sites of SACs due to its site deactivation role.
Control experiments are carried out to verify the deactivation role of C60. We first tune the molar ratio between CoP and C60 (0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 1
4, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) of C60-CoP/CNT. Polarization curves (Fig. 3a) and E1/2 values (Fig. 3b) show that the ORR activity of C60-CoP/CNT decreases from 0.80 to 0.73 V with the increase of the C60 content. Such positive correlation implies a growing inhibiting effect with more C60 molecules. Moreover, SCN− is known as a probe that strongly binds to transition metals, causing chemical, physical, and electronic effects that dramatically disrupt the catalytic functions of such metals.29–32 It is thus no surprise that the addition of SCN− leads to a dramatic activity loss of CoP/CNT (Fig. 3c and d). However, the same E1/2 reduction level by adding C60 as that by adding SCN− strongly hints that C60 behaves just like SCN− as an inhibitor to the Co SACs, indicating that C60 can serve as a similarly efficient probe to examine the catalytically active sites as SCN−.
1) of C60-CoP/CNT. Polarization curves (Fig. 3a) and E1/2 values (Fig. 3b) show that the ORR activity of C60-CoP/CNT decreases from 0.80 to 0.73 V with the increase of the C60 content. Such positive correlation implies a growing inhibiting effect with more C60 molecules. Moreover, SCN− is known as a probe that strongly binds to transition metals, causing chemical, physical, and electronic effects that dramatically disrupt the catalytic functions of such metals.29–32 It is thus no surprise that the addition of SCN− leads to a dramatic activity loss of CoP/CNT (Fig. 3c and d). However, the same E1/2 reduction level by adding C60 as that by adding SCN− strongly hints that C60 behaves just like SCN− as an inhibitor to the Co SACs, indicating that C60 can serve as a similarly efficient probe to examine the catalytically active sites as SCN−.
To rule out the potential influence of the exclusiveness of Co metal and the CNT support in the deactivation role of C60, we have made further efforts by utilizing alternative MP molecules and a carbon support. Specifically, NiP/CNT and C60-NiP/CNT are synthesized following the same synthetic recipe as that of the CoP counterparts. As displayed in Fig. 3e and f, substantial ORR activity loss is also observed for NiP/CNT after C60 adsorption with the E1/2 values reduced from 0.73 to 0.69 V, again manifesting the role of C60 as an inhibitor. Analogously, a conductive carbon black, ketjenblack (KB), is adopted to replace CNTs as the support of CoP molecules following the same synthetic methods. It is discerned that CoP/KB and C60/KB exhibit almost the same ORR activities as those of CoP/CNT and C60/CNT (Fig. 3g and h). After the addition of C60, CoP/KB also experiences dramatic ORR activity decay with an evidently decreased E1/2 of 0.76 V. Therefore, it is confidently concluded that the probing role of fullerene will be universally applicable to various metal SACs and irrelevant to the carbon support.
Insights of the deactivation mechanism of C60 on CoP/CNT are further provided by DFT calculations. The preferable adsorption site of C60 on carbon-supported CoP is examined by calculating the binding energy (BE) of two distinct hybrids that have C60 and CoP on the same side (Fig. 4a) and opposite sides (Fig. 4b) of a graphene substrate, respectively. The results indicate that the hybrid with C60 adsorbed on top of CoP is more stable than the other by a ΔBE value of 0.435 eV. Even when the solvent effect is included in the computation, a more stable model is obtained for C60 adsorbed to CoP (a ΔBE value of 0.280 eV). Furthermore, O2 adsorption to the Co site has to overcome the considerable interactions between C60 and CoP, as reflected by the elongated distance between Co and C60 after O2 binding (Fig. S6, ESI†). Combined with the above experimental results, it is interpreted that the molecular SACs have probably been spatially blocked by the C60 molecules that are favorably adsorbed on top of the ORR-active Co atoms, creating substantial steric hindrance to restrict the access of O2. Note that the inhibiting role of C60 to SACs does not contradict with the previously reported positive effects brought to metal particle catalysts by C60 adsorption.10–12 In fact, it rules out the possibility of the surface metal atom, to which the C60 molecule is specifically adsorbed, to serve as the potential active sites.
 | ||
| Fig. 4 Geometric configurations of C60 adsorbed on the same side (a) and opposite sides (b) of CoP adsorbed on a graphene matrix. Views of the atomic models from two distinct angles are illustrated on the left and right, respectively. Same color code as in Fig. 2. | ||
In summary, utilizing metalloporphyrin molecules that are uniformly dispersed on CNTs via non-covalent π–π interactions as model SACs, we disclose the probing role of C60 molecules in identifying the metal active sites of SACs towards the ORR. While the dispersion of CoP and C60 on CNTs leads to much superior ORR activities to bare CoP and C60, respectively, the addition of C60 to CoP/CNT results in reduced ORR activity. A set of control experiments point out that the inhibiting effect of CoP gets severe with increasing C60 molar ratios. C60 leads to the same ORR activity decay as the recognized SCN− poison. The probing role of C60 is also applicable to other metal SACs and carbon supports. Structural analysis and DFT calculations consistently indicate that the surface-stabilized CoP molecules are tightly confined between the C60 molecules and the basal plane of the CNTs. High steric hindrance is thus created to restrain the access of O2 to the central metal atoms of MP SACs.
This work was supported by the National Natural Science Foundation of China (22301088, 21925104, 92261204) and the Hubei Provincial Natural Science Foundation of China (2022CFB623, 2021CFA020). The authors also acknowledge the support from the Analytical and Testing Center in Huazhong University of Science and Technology.
Data availability
The data supporting the findings of this study are available within the article and its ESI.†Conflicts of interest
There are no conflicts to declare.References
- A. R. Puente Santiago, O. Fernandez-Delgado, A. Gomez, M. A. Ahsan and L. Echegoyen, Angew. Chem., Int. Ed., 2021, 60, 122–141 CrossRef CAS PubMed.
- K. Guo, N. Li, L. Bao and X. Lu, Green Energy Environ., 2024, 9, 7–27 CrossRef CAS.
- N. K. Gupta, A. Pashigreva, E. A. Pidko, E. J. Hensen and L. Mleczko, et al. , Angew. Chem., Int. Ed., 2016, 55, 1728–1732 CrossRef CAS PubMed.
- C. Zhang, W. Shen, K. Guo, M. Xiong and J. Zhang, et al. , J. Am. Chem. Soc., 2023, 145, 11589–11598 CrossRef CAS PubMed.
- N. Li, K. Guo, M. Li, X. Shao and Z. Du, et al. , J. Am. Chem. Soc., 2023, 145, 24580 CAS.
- N. Li, M. Li, K. Guo, Z. Guo and R. Wang, et al. , Adv. Energy Mater., 2024, 14, 2401008 CrossRef CAS.
- M. F. Sanad, H. M. Franklin, B. A. Ali, A. R. Puente Santiago and A. N. Nair, et al. , Angew. Chem., Int. Ed., 2022, 61, e202116727 CrossRef CAS PubMed.
- T. Wang, L. Zhang, J. Wu, M. Chen and S. Yang, et al. , Angew. Chem., Int. Ed., 2023, 62, e202311352 CrossRef CAS PubMed.
- B. Peng, J. Am. Chem. Soc., 2022, 144, 19921–19931 CrossRef CAS PubMed.
- J. Zheng, L. Huang, C. H. Cui, Z. C. Chen and X. F. Liu, et al. , Science, 2022, 376, 288–292 CrossRef CAS PubMed.
- B. Zhao, F. Chen, C. Cheng, L. Li and C. Liu, et al. , Adv. Energy Mater., 2023, 13, 2204346 CrossRef CAS.
- Y. Li, H. Zhang, T. Chen, Y. Sun and F. Rosei, et al. , Adv. Funct. Mater., 2024, 34, 2312970 CrossRef CAS.
- Y. Feng, X. Wang, J. Huang, P. Dong and J. Ji, et al. , Chem. Eng. J., 2020, 390, 124525 CrossRef CAS.
- Y. H. Choi, J. Lee, A. Parija, J. Cho and S. V. Verkhoturov, et al. , ACS Catal., 2016, 6, 6246–6254 CrossRef CAS.
- A. R. Puente Santiago, T. He, O. Eraso, M. A. Ahsan and A. N. Nair, et al. , J. Am. Chem. Soc., 2020, 142, 17923–17927 CrossRef CAS PubMed.
- A. Zitolo, V. Goellner, V. Armel, M. T. Sougrati and T. Mineva, et al. , Nat. Mater., 2015, 14, 937–942 CrossRef CAS PubMed.
- F. Luo, A. Roy, L. Silvioli, D. A. Cullen and A. Zitolo, et al. , Nat. Mater., 2020, 19, 1215–1223 CrossRef CAS PubMed.
- K. M. Zhao, S. Liu, Y. Y. Li, X. Wei and G. Ye, et al. , Adv. Energy Mater., 2022, 12, 2103588 CrossRef CAS.
- X. Yang, D. Xia, Y. Kang, H. Du and F. Kang, et al. , Adv. Sci., 2020, 7, 2000176 CrossRef CAS PubMed.
- P. Hutchison, P. S. Rice, R. E. Warburton, S. Raugei and S. Hammes-Schiffer, J. Am. Chem. Soc., 2022, 144, 16524–16534 CrossRef CAS PubMed.
- T. Zhang, J. Jin, J. Chen, Y. Fang and X. Han, et al. , Nat. Commun., 2022, 13, 6875 CrossRef CAS PubMed.
- M. L. Pegis, C. F. Wise, D. J. Martin and J. M. Mayer, Chem. Rev., 2018, 118, 2340–2391 CrossRef CAS PubMed.
- J. Wu and Y. X. Yu, J. Colloid Interface Sci., 2022, 623, 432–444 CrossRef CAS PubMed.
- J. Wu and Y. X. Yu, Int. J. Hydrogen Energy, 2023, 48, 5961–5975 CrossRef CAS.
- C.-X. Zhao, B.-Q. Li, J.-N. Liu, J.-Q. Huang and Q. Zhang, Chin. Chem. Lett., 2019, 30, 911–914 CrossRef CAS.
- E. M. Perez and N. Martin, Chem. Soc. Rev., 2015, 44, 6425–6433 RSC.
- A. J. Néel, M. J. Hilton, M. S. Sigman and F. D. Toste, Nature, 2017, 543, 637–646 CrossRef PubMed.
- R. Gao, Q. Dai, F. Du, D. Yan and L. Dai, J. Am. Chem. Soc., 2019, 141, 11658–11666 CrossRef CAS PubMed.
- S. Gupta, C. Fierro and E. Yeager, J. Electroanal. Chem. Interfacial Electrochem., 1991, 306, 239–250 CrossRef CAS.
- C. H. Bartholomew, Appl. Catal., A, 2001, 212, 17–60 CrossRef CAS.
- M. S. Thorum, J. M. Hankett and A. A. Gewirth, J. Phys. Chem. Lett., 2011, 2, 295–298 CrossRef CAS.
- Q. Wang, Z. Y. Zhou, Y. J. Lai, Y. You and J. G. Liu, et al. , J. Am. Chem. Soc., 2014, 136, 10882–10885 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Experimental section, characterization and electrochemical data. See DOI: https://doi.org/10.1039/d4cc03901g |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2024 |