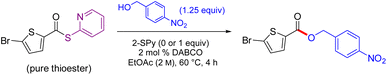Use of dipyridyldithiocarbonate (DPDTC) as an environmentally responsible reagent leading to esters and thioesters under green chemistry conditions†
Kaitlyn M.
Freiberg
 ,
Erika
Ghiglietti
,
Erika
Ghiglietti
 ,
Matthew
Scurria
and
Bruce H.
Lipshutz
,
Matthew
Scurria
and
Bruce H.
Lipshutz
 *
*
Department of Chemistry and Biochemistry, University of California, Santa Barbara, CA 93106, USA. E-mail: lipshutz@chem.ucsb.edu; kfreiberg@ucsb.edu
First published on 9th November 2023
Abstract
A new, green esterification and thioesterification method has been developed based on the intermediacy of in situ-generated 2-thiopyridine esters derived from dipyridyldithiocarbamate (DPDTC). Both are formed from initial treatment of the corresponding carboxylic acid with DPDTC under neat conditions, followed by introduction of the alcohol or thiol, together with catalytic amounts of an activating group, in concentrated EtOAc or under aqueous micellar conditions. These 1-pot processes generate high yields of esters and thioesters, avoid use of traditional coupling agents, and lead to minimal waste creation as both the EtOAc and the thiol by-product are easily recycled.
Introduction
Whether making esters or thioesters, the literature, including textbooks, will suggest a variety of traditional methods involving coupling between carboxylic acids and alcohols or thiols.1,2 Although Fischer type esterifications are the oldest and most known method, high temperatures and harsh acidic conditions are required to drive the equilibrium in the desired direction.3 As a result, several alternatives and far milder conditions that deliver the same outcomes are now frequently used. The most common reagents; acid halides,4 DCC (dicyclohexylcarbodiimide),5a,b,6 EDC (N-(3-dimethylamino-propyl)-N′-ethylcarbodiimide)/DMAP (4-dimethylamino-pyridine),7etc. are representative of a long list of environmentally egregious options (Fig. 1). Although they are usually effective leading to the targeted functionality, arriving at the designated reaction outcome is not the only consideration. The question has been broadened, and today might be rephrased as: If this process works, what is the cost to the environment?Traditional coupling agents for ester/thioester synthesis, although prominently applied to peptide synthesis, were created to effectively remove water, which could in principle, participate in an undesired equilibrium. But the evidence against their use is growing,8 given their known side-effects (see the 2023 report in C&E News; https://cen.acs.org/safety/lab-safety/Peptide-coupling-agents-cause-severe/98/web/2020/01).9a,b The reagents were designed for use in waste-generating organic solvents,10a,11 regardless of origin (i.e., whether petroleum-based or from renewable sources) and the by-products, by definition, are not recyclable. Hence, while the targeted esters and thioesters may be of great value (Fig. 2),2,12–18 their preparation using a “business as usual” mentality is no longer acceptable, as such chemistry is not sustainable.
“Rethinking” the use of water as the actual reaction medium, akin to nature's approach to esters and thioesters done in water (e.g., thioester formation in metabolism, etc.), there are opportunities to follow Nature's lead21,22 by making a reactive thioester in situ as a precursor to other functionality. There are alternatives that appear to be general; for example, the recent (2023) contribution by Wu, Zhang and co-workers from academia (among several others) utilizing elemental sulfur and feedstock chemicals looks very attractive, as it can be used to make highly functionalized thioesters and is amenable to late-stage functionalization; all very positive features.23 From the green chemistry perspective, however, there are some potential issues, including use of unrecycled CH3CN, a requirement for oxidation-prone aldehydes as educts, the involvement of radical intermediates that are generated by photochemical activation using an additive that becomes organic waste, and a 24-hour reaction time.
Although other “green” methodologies are available and have been developed en route to esters and thioesters,24–31 including a solvent–reagent selection guide to identify the most sustainable combination, improvements can be anticipated.10a,b Hence, in this report are described green and efficient approaches to esterification and thioesterification that make use of an in situ-formed and used (albeit stable and isolable) 2-pyridylthioester intermediate. These can be generated in the absence of any reaction medium (i.e., done neat) using DiPyridylDiThioCarbamate (DPDTC; Scheme 1), derived from solid and easily handled triphosgene (Caution: triphosgene is toxic).32 It should be noted that there is no odor associated with DPDTC, or its byproduct, 2-mercaptopyridine. Subsequent reactions involving the addition of an alcohol or thiol can be run neat as well (see ESI, Table S1†), or in the green and recyclable solvent ethyl acetate at elevated concentration (2 M; see ESI, Table S6†). Use of a third approach involving aqueous micellar catalysis is also demonstrated. The 2-mercaptopyridine by-product can be easily recovered as well, overall decreasing the waste generated.
 | ||
| Scheme 1 Use of DPDTC to form thioester intermediates in a 1-pot sequence en route to esters and thioesters. | ||
Results and discussion
Ester formation
Although expecting esterification to be straightforward, optimization was challenging. When starting with isolated, pure thioester, initial screening of additives in various amounts (i.e., triethylamine, N-methylmorpholine, DBU, DMAP, etc.), conversions to esters with phenols and benzyl alcohols consistently led to good yields. However, the yields significantly dropped when attempting to form the esters in one pot, and upon further inspection it was determined that the by-product using DPDTC, 2-mercaptopyridine, had a dramatic effect on the reaction outcome (Table 1). Addition of 2-mercaptopyridine (1 equiv.) to the reaction containing pure thioester decreased the yield by half. The acidic hydrogen on sulfur in 2-mercaptopyridine was likely involved in protonating the activating agent present (in catalytic amounts) in the reaction mixture.34 When the 2-mercaptopyridine byproduct was intentionally removed (i.e., either by using the purified thioester, or removed with 1 M NaOH after formation of thioester), the yield increased significantly. Re-optimization of the loading of the activating agent led to the inclusion of 10 mol% 1,4-diazabicyclo[2.2.2]octane (DABCO) rather than the initially used 2 mol%. This increased loading of DABCO afforded higher yields resulting from complete consumption of the 2-thiopyridyl thioester. Use of DABCO, akin to DMAP (which was avoided due to the price and toxicity), performed the most consistently and with greater success involving other types of alcohols (see ESI, Section 4.2, Tables S27 and S28†).| Entry | 2-SPy (equiv.) | Alcohol (equiv.) | Yield (%) |
|---|---|---|---|
| All reactions were run using 2 mol% DABCO at 60 °C for 4 h in 2 M EtOAc.a By crude NMR. NMR yield with internal standard 1,3,5-trimethoxybenzene.b Isolated yield.c Formed from the carboxylic acid and DPDTC, followed by addition of alcohol, no additional 2-mercaptopyridine was added. 2-Mercaptopyridine was added to thioester and alcohol in entries 2 and 3. | |||
| 1 | n/a | 1.05 | 95a |
| 2 | 1 | 1.05 | 42a |
| 3 | 1 | 1.25 | 46a |
| 4 | n/a | 1.05 | 38b,c |
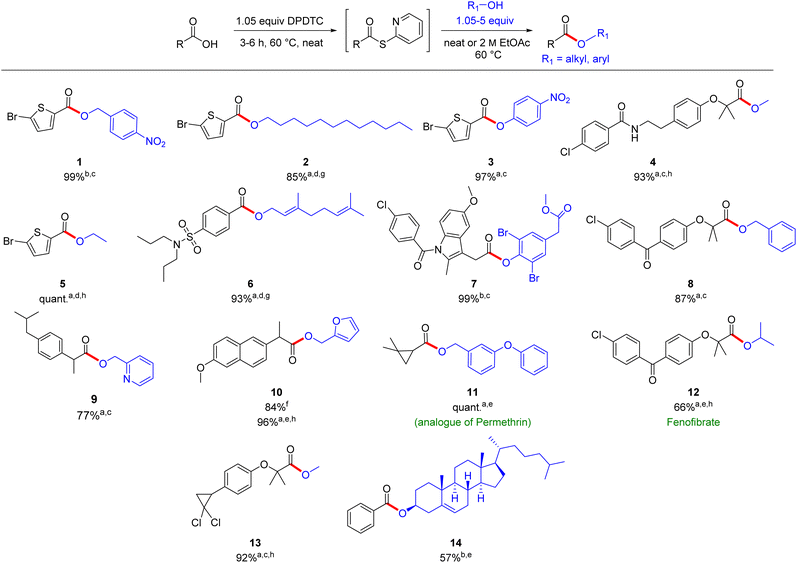
Substrates containing secondary alcohols required somewhat more forcing conditions, including catalytic DMAP (10 mol%) and longer reaction times. Thus, while the ester could be obtained, the yields were modest (see products 12 and 14; Scheme 2). Moreover, neat reaction conditions and increased levels of DABCO (1 equiv.) were needed with aliphatic alcohols, due to their higher pKa values (see products 2, 5 and 6).
For liquid alcohols, reactions were run using the alcohol as solvent (1.05–5 equiv.; e.g., methanol) resulting in greatly increased reaction rates due mainly to improved stirring, and increased yields (such as with products 4, 5, 12, and 13). Both aromatic and heteroaromatic acids appear to be amenable, as are aliphatic cases. Alcohols, likewise, of various types (e.g., phenolic and aliphatic) readily participate, including hindered examples (e.g., products 3, 7, and 14). When the alcohol is non-trivial, only a slight excess is needed for high levels of conversion, and thus, yields. The esters shown in Scheme 2 were all formed in a 1-pot operation (i.e., initial conversion of the acid to the thioester, followed by esterification), although the sequence could be interrupted at the 2-pyridyl thioester intermediate stage, as these can be isolated and stored for later use. This includes their transformation into several other derivatives (e.g., amides, aldehydes, and alcohols).32,35 Also worthy of note is that, in addition, treatment of 2-pyridyl thioester intermediates under aqueous micellar conditions is also an option leading to the desired product (see compound 10). Targets such as 4 (bezafibrate), 6 (probenecid), 7 (indomethacin), 10 (naproxen), 8 and 12 (fenofibric acid), and 13 (ciprofibrate) could all be made from the associated drug-related carboxylic acids, each being realized in moderate-to-good yield. Also, analogues of insecticides permethrin (11) and pharmaceutical 12 (fenofibrate) were readily prepared in moderate-to-good yields.
Thioester formation
2-Pyridylthioester-to-thioester conversions shown in Scheme 3 did not typically require additional carbonyl activation, although aliphatic thiols are best used together with small amounts of DBU (see, e.g., products 17, 27, and 28). Thioesters derived from drug-like carboxylic acids successfully afforded the desired thioesters, typically in good isolated yields. Some examples include 21 (probenecid), 22 (ibuprofen), 23 (bezafibrate), and 28 (ciprofibrate). Scheme 4 highlights examples of thioesters made in water containing the surfactant TPGS-750-M (Fig. 3), including a derivative of gemfibrozil (31).33 These highlight the compatibility of DPDTC with water as the reaction medium. The success of DPDTC in water is likely due to both the increased stability of DPDTC-derived thioesters and the ability of the surfactant to reduce hydrolysis, although the same surfactant was also used in previous trials that were unsuccessful with traditional reagents.10 While these reactions tend to require longer reaction times than running the reaction neat, the overall efficiencies can still be quite good as compared to other methods using alternative coupling reagents in organic solvents.10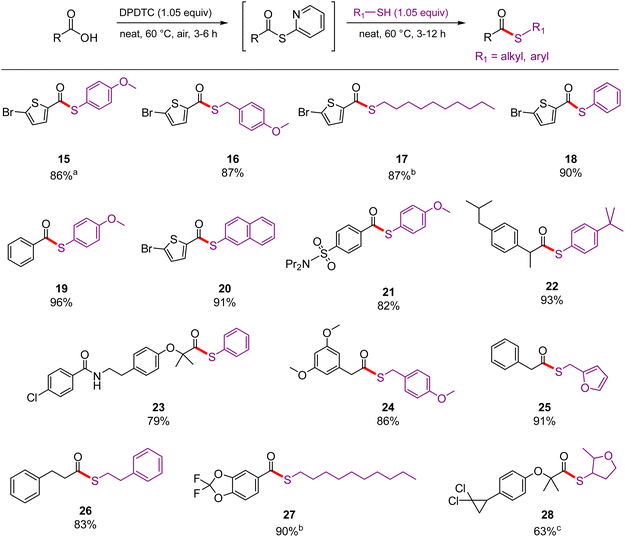 | ||
Scheme 3 Representative examples of thioester-to-thioester interconversion. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 equiv. thiol. b 1.00 equiv. thiol. b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (2 M), DBU (0.2 mol%); c EtOAc (2 M), DBU (0.2 mol%); c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (2 M), DBU (2.5 mol%). EtOAc (2 M), DBU (2.5 mol%). | ||
 | ||
| Scheme 4 Representative examples of thioesters made under aqueous micellar catalysis conditions. See ref. 10a in which it is claimed that thioester 29 cannot be made under aqueous micellar conditions. | ||
In direct comparisons to literature methods, both esters and thioesters can typically be made under milder conditions and with equal or better yields, as shown in Table 2. For example, product 19 was prepared without the use of Pd, at a lower temperature, with significantly fewer additives, and in 20% higher yield using this technology.36 Likewise, 26 was formed in >20% higher yield in the absence of both elemental sulfur and TBAT, in less than half the time.23 Additionally, product 29 was made using an aqueous surfactant medium in the absence of HATU and an organic solvent (CH3CN), in comparable yield.10a
| This work | Literature | |
|---|---|---|
| 92%, 5 equiv. MeOH (5 M), 8 h |
|
79%,30 100 equiv. MeOH (0.25 M), 48 h |
| 57%, DPDTC (1.05 equiv.), DMAP (10 mol%), 2 M EtOAc |

|
34%,10b Mukaiyama's reagent (1.05 equiv.), 2,6-lutidine (2 equiv.), 0.5 M DMC |
| 96%, neat, 1 equiv. thiol, no additive |
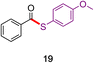
|
74%,36 1 mol% PdCl2, 2 mol% ligand, 2 equiv. thiol, PhSiH3, 1 equiv. NaOAc, NMP, 80 °C |
| 83%, neat, 1.05 equiv. thiol, no activation, 9 h total |
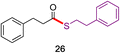
|
61%,23 S8, TBAT, 1.5 equiv. aldehyde, CH3CN, 24 h |
| 87%, DPDTC (1.05 equiv.), TEA to neutralize, TPGS-750-M/H2O |
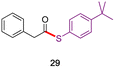
|
96%,10a HATU (1.5 equiv.), 2 equiv. DIPEA, CH3CN |
Common green chemistry metrics used to evaluate sustainability include process mass intensity (PMI), E factor, and reaction mass efficiency (RME).10b These can be readily calculated to provide a preliminary assessment as to the extent of greenness associated with any reaction. The esterification process outlined herein offers low metric values, indicative of the limited organic waste being created. For example, as illustrated in Table 3, ester 14 was prepared in almost twice the yield using DPDTC, with a greatly reduced E factor, PMI and an increased RME as compared to literature values with the use of a “green” esterification method (i.e., using Muk) (see ESI Section 7 of ref. 32 for PMI of products 1, 2, 19).10b,36 Generally, isolation of the products does not require workup; a simple silica plug or column is sufficient. Additionally, although not considered by these green chemistry metrics, the 2-mercaptopyridine by-product of the reaction can be recycled and reused to make DPDTC.32
| Cholesteryl benzoate (14) | DPDTC | Mukaiyama's reagent10b |
|---|---|---|
| Yield | 57% | 34% |
| E factor for reaction | 3.34 | 20.2 |
| RME | 35.8 | 21.2 |
| PMI for reaction | 4.34 | 21.2 |
These processes could be smoothly scaled to the gram level to give ester 1 in 95% yield and thioester 20 in 91% isolated yield, while the EtOAc could be recovered (on an academic scale) to the extent of 62% (see ESI, Sections 5 and 6 of ref. 32).
A representative 1-pot sequence consisting of 4-steps is shown in Scheme 5. Thus, after generating the initial C–N bond via an SNAr reaction (neat),37 the ester is formed with the newly formed primary alcohol to afford the crude derivative of indomethacin. The nitro group present in this resulting ester was then reduced using carbonyl iron powder (CIP)38 affording the final product 32 in 49% overall yield.
Conclusions
In summary, a safe, effective, and green procedure leading to functionalized esters and thioesters has been developed that avoids traditional coupling reagents that can be dangerous and are waste-generating. The sequence relies on in situ generation and use of an isolable and bench stable thioester intermediate, formed using the dithiocarbonate, DPDTC. Reactions are typically amenable to use under neat conditions, require no formal (and waste-generating) workup, giving product formation within a few hours. Overall, therefore, this work represents a much-needed and timely advance leading to formation of important esters and thioesters for the chemical industry.Author contributions
K. M. F. drafted the manuscript, optimized, and carried out studies on the scope of ester formation. E. G. optimized and carried out studies on the scope of thioester formation. M. S. worked on substrate scope and the sequence described herein. B. H. L. edited and oversaw the work.Conflicts of interest
There are no conflicts to declare.Acknowledgements
Financial support from the NSF (CHE-2152566), PHT International, and Anthem Biosciences is warmly acknowledged with thanks.Notes and references
- J. Otera and J. Nishikido, Esterification: Methods, Reactions, and Applications, Wiley-VCH, New York, 2009 Search PubMed.
- M. Kouichi, Y. Rina and O. Yohei, Recent Advances in the Synthesis of Carboxylic Acid Esters, in Carboxylic Acid – Key Role in Life Sciences, ed. G. I. Badea and G. L. Radu, IntechOpen, 2018 Search PubMed.
- E. Fischer and A. Speier, Ber. Dtsch. Chem. Ges., 1895, 28, 3252–3258 CrossRef CAS.
- T. Chatterji, M. Kizil, K. Keerthi, G. Chowdhury, T. Pospísil and K. S. Gates, J. Am. Chem. Soc., 2003, 125, 4996–4997 CrossRef CAS PubMed.
- (a) G. Höfle, W. Steglich and H. Vorbrüggen, Angew. Chem., Int. Ed. Engl., 1978, 17, 569–583 CrossRef; (b) B. Neises and W. Steglich, Angew. Chem., Int. Ed. Engl., 1978, 17, 522–524 CrossRef.
- E. L. Myers and R. T. Raines, Angew. Chem., Int. Ed., 2009, 48, 2359–2363 CrossRef CAS PubMed.
- A. M. Royer, M. Salomone-Stagni, T. B. Rauchfuss and W. Meyer-Klaucke, J. Am. Chem. Soc., 2010, 132, 16997–17003 CrossRef CAS PubMed.
- S. Y. Han and Y. A. Kim, Tetrahedron, 2004, 60, 2447–2467 CrossRef CAS.
- (a) L. K. Boerner, in C&E News, Researchers can develop sensitivities to the compounds, to the point of anaphylaxis, https://cen.acs.org/safety/lab-safety/Peptide-coupling-agents-cause-severe/98/web/2020/01, accessed 01 August 2023. (b) K. J. McKnelly, W. Sokol and J. S. Nowick, J. Org. Chem., 2020, 85, 1764–1768 CrossRef CAS PubMed.
- (a) A. Jordan and H. F. Sneddon, Green Chem., 2019, 21, 1900–1906 RSC; (b) A. Jordan, K. D. Whymark, J. Sydenham and H. F. Sneddon, Green Chem., 2021, 23, 6405–6413 RSC.
- C. M. Alder, J. D. Hayler, R. K. Henderson, A. M. Redman, L. Shukla, L. E. Shuster and H. F. Sneddon, Green Chem., 2016, 18, 3879–3890 RSC.
- P. Ertl, E. Altmann and J. M. McKenna, J. Med. Chem., 2020, 63, 8408–8418 CrossRef CAS PubMed.
- M. Tsakos, E. S. Schaffert, L. L. Clement, N. L. Villadsen and T. B. Poulsen, Nat. Prod. Rep., 2015, 32, 605–632 RSC.
- A. D. Godwin, in Applied Polymer Science: 21st Century, ed. C. D. Craver and C. E. Carraher, Pergamon, Oxford, 2000, pp. 157–175, DOI:10.1016/B978-008043417-9/50011-8.
- R. J. Ouellette and J. D. Rawn, Organic Chemistry, Elsevier, Boston, 1st edn, 2014, pp. 699–745, DOI:10.1016/B978-0-12-800780-8.00021-8.
- P. Ertl, E. Altmann and J. M. McKenna, J. Med. Chem., 2020, 63, 8408–8418, DOI:10.1021/acs.jmedchem.0c00754.
- G. D. Yadav and M. S. M. Mujeebur Rahuman, Synthesis of Fragrance and Flavour Grade Esters: Activities of Different Ion Exchange Resins and Kinetic Studies, Clean Technol. Environ. Policy, 2003, 5, 128–135, DOI:10.1007/s10098-003-0196-9.
- L. D. Lavis, Ester Bonds in Prodrugs, ACS Chem. Biol., 2008, 3, 203–206, DOI:10.1021/cb800065s.
- J. T. Suh, J. W. Skiles, B. E. Williams, R. D. Youssefyeh, H. Jones, B. Loev, E. S. Neiss, A. Schwab and W. S. Mann, J. Med. Chem., 1985, 28, 57–66 CrossRef CAS PubMed.
- K. Yamada, M. Satoh, S. Shimura, T. Sasaki, T. Takishima and K. Shirato, Respiration, 1994, 61, 42–47 CrossRef CAS PubMed.
- S. Hanessian, V. Mascitti and O. Rogel, J. Org. Chem., 2002, 67, 3346–3354 CrossRef CAS PubMed.
- N. Scardovi, P. P. Garner and J. D. Protasiewicz, Org. Lett., 2003, 5, 1633–1635 CrossRef CAS PubMed.
- H. Tang, M. Zhang, Y. Zhang, P. Luo, D. Ravelli and J. Wu, J. Am. Chem. Soc., 2023, 145, 5846–5854 CrossRef CAS PubMed.
- Z. Khan, F. Javed, Z. Shamair, A. Hafeez, T. Fazal, A. Aslam, W. B. Zimmerman and F. Rehman, J. Ind. Eng. Chem., 2021, 103, 80–101, DOI:10.1016/j.jiec.2021.07.018.
- V. Singhania, M. Cortes-Clerget, J. Dussart-Gautheret, B. Akkachairin, J. Yu, N. Akporji, F. Gallou and B. H. Lipshutz, Chem. Sci., 2022, 13, 1440–1445 RSC.
- V. V. Rekha, M. V. Ramani, A. Ratnamala, V. Rupakalpana, G. V. Subbaraju, C. Satyanarayana and C. S. Rao, Org. Process Res. Dev., 2009, 13, 769–773 CrossRef CAS.
- P. A. Turhanen, J. Leppänen and J. J. Vepsäläinen, ACS Omega, 2019, 4, 8974–8984 CrossRef CAS PubMed.
- L. An, C. Si, G. Wang, C. S. Choi, Y. H. Yu, J. H. Bae, S. M. Lee and Y. S. Kim, BioResources, 2020, 15, 89–104 CAS.
- M. Minakawa, H. Baek, Y. M. A. Yamada, J. W. Han and Y. Uozumi, Org. Lett., 2013, 15, 5798–5801 CrossRef CAS PubMed.
- Y. Zheng, Y. Zhao, S. Tao, X. Li, X. Cheng, G. Jiang and X. Wan, Eur. J. Org. Chem., 2021, 2713–2718 CrossRef CAS.
- B. Basu, S. Paul and A. K. Nanda, Green Chem., 2010, 12, 767–771 RSC.
- K. M. Freiberg, R. D. Kavthe, R. M. Thomas, D. M. Fialho, P. Dee, M. Scurria and B. H. Lipshutz, Chem. Sci., 2023, 14, 3462–3469 RSC . There is no odor whatsoever associated with the use of DPDTC or the byproduct, 2-mercaptopyridine.
- S. Ghorai, A. R. Abela, R. Moser, T. Nishikata, C. Duplais, A. Krasovskiy, R. D. Gaston, R. C. Gadwood and B. H. Lipshutz, J. Org. Chem., 2011, 76, 4379–4391 CrossRef PubMed.
- V. K. Aggarwal, I. Emme and S. Y. Fulford, J. Org. Chem., 2003, 68, 692–700, DOI:10.1021/jo026671s.
- K. S. Iyer, C. Nelson and B. H. Lipshutz, Green Chem., 2023, 25, 2663–2671 RSC.
- H. Wang, Y. Li, Y. Sun and Y. Li, J. Org. Chem., 2023, 88, 8835–8842 CrossRef CAS PubMed.
- N. A. Isley, R. T. H. Linstadt, S. M. Kelly, F. Gallou and B. H. Lipshutz, Org. Lett., 2015, 17, 4734–4737 CrossRef CAS PubMed.
- N. R. Lee, A. A. Bikovtseva, M. Cortes-Clerget, F. Gallou and B. H. Lipshutz, Org. Lett., 2017, 19, 6518–6521 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3gc03093h |
| This journal is © The Royal Society of Chemistry 2023 |