Utilization of DNA and 2D metal oxide interaction for an optical biosensor†
Abstract
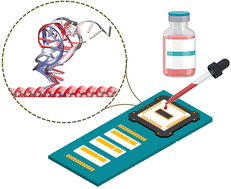
The efficient monitoring and early detection of viruses may provide essential information about diseases. In this work, we have highlighted the interaction between DNA and a two-dimensional (2D) metal oxide for developing biosensors for further detection of viral infections. Spectroscopic measurements have been used to probe the efficient interactions between single-stranded DNA (ssDNA) and the 2D metal oxide and make them ideal candidates for detecting viral infections. We have also used fully atomistic molecular dynamics (MD) simulation to give a microscopic understanding of the experimentally observed ssDNA-metal oxide interaction. The adsorption of ssDNA on the inorganic surface was found to be driven by favourable enthalpy change, and 5′-guanine was identified as the interacting nucleotide base. Additionally, the in silico assessment of the conformational changes of the ssDNA chain during the adsorption process was also performed in a quantitative manner. Finally, we comment on the practical implications of these developments for sensing that could help design advanced systems for preventing virus-related pandemics.
- This article is part of the themed collection: Emerging concepts in nucleic acids: structures, functions and applications


 Please wait while we load your content...
Please wait while we load your content...