Open Access Article
Open Access ArticleChemical characterization of Wuyi rock tea with different roasting degrees and their discrimination based on volatile profiles†
Xiaobo Liu‡
ab,
Yawen Liu‡a,
Pan Li‡a,
Jiangfan Yangc,
Fang Wangc,
Eunhye Kima,
Yuanyuan Wua,
Puming Hea,
Bo Li *a and
Youying Tu
*a and
Youying Tu *a
*a
aDepartment of Tea Science, Zhejiang University, Hangzhou 310058, China. E-mail: drlib@zju.edu.cn; youytu@zju.edu.cn
bCollege of Tea Science, Guizhou University, Guiyang 550025, China
cCollege of Tea and Food Science, Wuyi University, Wuyishan 354300, China
First published on 25th March 2021
Abstract
Wuyi rock tea is a typical and famous oolong tea in China and roasting is an important manufacturing procedure for its flavor formation. This work aimed to explore the effect of roasting on non-volatiles and volatiles of 12 Wuyi rock tea samples at three roasting levels (low, moderate and sufficient), made from four tea cultivars (Shuixian, Qizhong, Dahongpao, Rougui). Results show that different roasting had not caused significant difference on contents of soluble solids, total polyphenols, flavonoids, soluble sugar, thearubigins and theabrownins, while it slightly regulated caffeine, proteins and theaflavins, and remarkably reduced catechins and free amino acids. The ratio of polyphenol content/amino acid content, a negative-correlated indicator of fresh and brisk taste, significantly increased with the increase of roasting degree. High-level roasting not only decreased the fresh and brisk taste of the tea infusion, but also reduced the amount of bioactive ingredients including catechins and theanine. A total of 315 volatiles were detected and analyzed with OPLS-DA and HCA methods, in which 99 volatiles were found with variable importance in the projection (VIP) values greater than 1.00. Tea samples at different roasting degrees were successfully separated by this model of roasting-level discrimination. ‘Naphthalene, 1,2,3,4-tetrahydro-1,6,8-trimethyl-’, ‘1,1,5-trimethyl-1,2-dihydronaphthalene’, ‘p-Xylene’, ‘alpha.-methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol’, ‘hydrazinecarboxylic acid, phenylmethyl ester’, and ‘3-buten-2-one, 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-’ might be key characteristic markers for the roasting process of Wuyi rock tea.
1 Introduction
Tea, made from the buds and leaves of Camellia sinensis L, is a widely consumed beverage worldwide. Numerous studies have reported the health benefits of tea, in which polyphenols, caffeine, L-theanine and black tea pigments are the major active components.1 Based on the manufacturing process, tea is classified into six major types including green, yellow, white, black, oolong and dark tea.2 Oolong tea is a typical semi-fermented tea with elegant aroma and characteristic flavor. Wuyi rock tea is one of the most famous oolong teas produced in the Wuyi Mountain and nearby regions, in northern Fujian Province, China. It is well known for the rich flavor and long-lasting fragrance, which is called ‘rock charm and floral fragrance’. The characteristics of Wuyi rock tea are decided by manufacturing procedures and the quality of fresh tea leaves. The latter varies from the tea cultivars and growth environment. Among the more than 200 tea cultivars, the varieties ‘Shuixian’, ‘Rougui’, ‘Dahongpao’ and ‘Qizhong’ are commonly planted and used for the Wuyi rock tea production.3,4The primary manufacturing processes of raw oolong tea involve withering, shaking and setting, fixation, rolling and drying.5 For Wuyi rock tea, a roasting procedure is added into drying operations and plays an important role in the development of organoleptic quality. In general, by roasting degree, popular Wuyi rock tea is divided into low, moderate, and sufficient roasting level, which are determined by heating temperature and duration. It is worth noticing that the roasting parameters for the tea made from diverse varieties need to be modified, due to their different physical and chemical properties. As the roasting degree increases, tea aroma usually changes from flower scent to fruit- and caramel-like odor, color of tea infusion from orange yellow to orange red, the grassy and astringent taste decrease, and color of infused tea leaves alters from green with red edge to black auburn.
Roasting is an important manufacturing procedure for various beverage including coffee, cocoa and oat, and has impact on their sensory properties and health-related compounds.6,7 Recently, metabolomics and proteomics analysis have been used to reveal the dynamic changes of volatile and non-volatile metabolites, and interpret the enzymatic modulation of small molecules during oolong tea manufacture.8,9 However, these researches mainly focused on the processing from withering to drying. Roasting is considered as a refining and requisite procedure for production of Wuyi rock tea, but its impacts on the flavor and functional ingredients are still not well documented. The changes in tea components may have unknown pattern upon roasting. This work aimed to investigate the effects of roasting technology on the overall non-volatile and volatile compositions of Wuyi rock tea, and construct a discriminant model for roasting degree based on chemical profiles with multivariate statistical analysis.
2 Materials and methods
2.1 Tea sample preparation
Twelve Wuyi rock tea samples were produced by Mr Liu in Wuyishan Institute of Tea Science. All the tea shoots, consisting of a bud and three or four leaves, were collected from tea garden located in the core region of Wuyi Mountain (latitude: 27.71 °N, longitude: 118.00 °E) in April, where is in the subtropical monsoon climate zone with light soil and yellow-red soil. The raw tea samples were made from four tea cultivars (Camellia sinensis var. sinensis) including ‘Shuixian’ (SX), ‘Qizhong’ (QZ), ‘Dahongpao’ (DHP), and ‘Rougui’ (RG), according to the conventional manufacturing processes.10 Briefly, the harvested tea shoots were withered under sunlight (28–32 °C) for 30 min, following wilted in an air-conditioned room (20 °C) for another 1 h. Then the samples underwent alternating steps of shaking and setting for 8 h, which was a special process for oolong tea processing. The semifermented tea samples were fixed at 260 °C for 8 min, rolled at 25 °C for 12 min, and dried at 120 °C for 20 min. Afterwards, the roasting process was operated in a special room with tea samples put in roasting baskets, with burny charcoal below in tunnel to heat. Every sample was roasted separately at low (L), moderate (M) and sufficient (S) levels according to diverse temperature and duration, details were shown in Table S1.† The quantity of charcoal and a digimite were to make sure the planned temperature. Sensory analysis were performed by six professional tea assessors according to the tea sensory evaluation procedure of the Chinese National Standards.11 All tea samples used in this work were assessed to meet the corresponding roasting degree.2.2 Chemicals and reagents
The catechins including (−)-gallocatechin (GC), (−)-epigallocatechin (EGC), (+)-catechin (C), (−)-epicatechin (EC), (−)-epigallocatechin gallate (EGCG), (−)-gallocatechin gallate (GCG), (−)-epicatechin gallate (ECG), (−)-catechin gallate (CG), caffeine, L-theanine and eighteen amino acid standards (kit) were purchased from Sigma (St. Louis, MO, USA). Four theaflavin monomers including theaflavin (TF1, 95%), theaflavin-3-gallate (TF2A, 92%), theaflavin-3′-gallate (TF2B, 90%) and theaflavin-3, 3′-digallate (TF3, 90%) were prepared according to the method developed in our lab, and their purities were determined by high-performance liquid chromatography-ultraviolet-mass spectrum (HPLC-UV-MS), with the standard samples from sigma company.12 Methanol and acetonitrile of HPLC grade were purchased from Tianjin Shield Company (Tianjin, China). Other reagents of analytical reagent grade were purchased from National Medicine Chemical Reagent Co., Ltd. (Shanghai, China).2.3 Analysis of major non-volatile constituents in tea samples
The tea samples were milled into powder and passed through a 20-mesh sieve. Three grams of tea powder was extracted with 450 ml of boiling distilled water for 45 min. Then the decoction was filtered using a qualitative filter paper, and cooled to room temperature for chemical composition analysis except for polyphenols, thearubigins (TRs) and theabrownins (TBs). The contents of total proteins, free amino acids, flavonoids and sugars were determined by spectrophotometric methods as follows: proteins by Coomassie Brilliant Blue method, free amino acids by ninhydrin, flavonoids by aluminum chloride colorimetric assay, and sugars by anthrone–sulfuric acid method. The contents of moisture and soluble solids were determined by the constant weight methods.5,13 The content of total polyphenols was determined by folin–ciocalteu assay after 0.2 g of tea powder was infused twice with 5 ml of 70% methanol solution, which dissolves polyphenols better than water, at 70 °C for 10 min.14 For determin of thearubigins (TRs) and theabrownins (TBs), a total of 3.0 g sample was extracted with 125 ml boiling pure water for 10 min, and the filtrate was further separated by EtOA, 95% ethanol, distilled water and NaHCO3 solution was used to separate TRs and TBs into different layers, which were measured at 380 nm. It's a system approach by spectrophotometry similar to other researches.15 The compositions of gallic acid, caffeine, eight catechins and four theaflavin monomers were analysed by a high-performance liquid chromatography-ultraviolet (HPLC-UV) method developed in our lab.12The nineteen amino acids were determined by a HPLC-fluorescence method via pre-column derivatization as described by R. M. Kazan (2019),16 with some modifications. For derivatization of amino acids, 10 μl of tea sample solution or standard solution was mixed with 500 μl of borate buffer (pH 10), 90 μl of o-phtalaldehyde (OPA, 1 mg ml−1), 10 μl of 3-mercaptopropionic acid (3-MPA), 100 μl of acetonitrile and 300 μl of ultra-distilled water. The mixture was vortexed quickly and incubated at room temperature for 30 minutes before sample injection. HPLC analysis was performed using an LC-20A system (Shimadzu, Kyoto, Japan) equipped with a Shimadzu SPD ultraviolet detector (SPD-20A) and a fluorescence detector (RF-20A). Chromatographic separation was carried out on an Agilent Zorbax Eclipse-AAA column (3 μm, 150 mm × 4.6 mm i.d.) at the flow rate of 1.5 ml min−1. The mobile phase A was 40 mM NaH2PO4 solution (pH 7.8), and B was methanol/acetonitrile/water (45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, v/v/v/v). Gradient elution was performed as follows: 0–1.9 min, 5% B; 1.9–18.1 min, 5–57% B; 18.1–18.6 min, 57–100% B; 18.6–22.3 min, 100% B; 22.3–23.2 min, 100–5% B; 23.2–30.0 min, 5% B. The sample injection volume was 10 μL, and the excitation wavelength was set at 338 nm.
10, v/v/v/v). Gradient elution was performed as follows: 0–1.9 min, 5% B; 1.9–18.1 min, 5–57% B; 18.1–18.6 min, 57–100% B; 18.6–22.3 min, 100% B; 22.3–23.2 min, 100–5% B; 23.2–30.0 min, 5% B. The sample injection volume was 10 μL, and the excitation wavelength was set at 338 nm.
2.4 Volatile analysis by HS-SPME/GC-MS
The extraction of volatile compounds was carried out by a headspace solid-phase microextraction (HS-SPME) method using a 50/30 μm DVB/CAR/PDMS fiber (Supelco, Bellafonte, PA, USA). For each sample, two grams of tea were transferred into a 20 ml glass septum flask, and infused with 6 ml boiling water. Subsequently sample was kept at 60 °C in a water bath for 5 min to equilibrate, and then the SPME fiber was exposed to the headspace of flask at 60 °C for 60 min. The fiber was preconditioned in the injection port of the GC at 250 °C for 5 min before each extraction.GC-MS analysis was performed on a Shimadzu single quadrupole GCMS-QP2010SE gas chromatograph-mass spectrometer (Shimadzu, Shiga, Japan). An SH-Rxi-5Sil MS capillary column (30 m, 0.25 mm, 0.25 μm; Shimadzu, Shiga, Japan) was equipped, and purified helium (>99.999%) was used as carrier gas with a flow rate of 1 ml min−1. The oven temperature was programmed as follows: held at 50 °C for 5 min, increased to 210 °C at a rate of 3 °C min−1 and maintain for 5 min, then increased to 230 °C at a rate of 15 °C min−1 and held for 5 min. The mass spectrometer was operated in the full scan and EI (electron ionization) mode. The injector and ion source temperatures were 250 and 230 °C respectively, and the mass spectrometry scan range was 20–500 amu. The volatile compounds were identified basing on the NIST 17 mass spectral library. The relative proportions of volatiles were obtained by peak area normalization and the results were expressed as the ratio of single constituent area to total area.
2.5 Statistical analysis
All tea samples were analysed in triplicate. The data were presented as means ± standard deviations (SD) of three determinations. One-way analysis of variance (ANOVA) and Pearson's correlation analysis was performed by employing the SPSS statistics 26.0 software (SPSS Inc., Chicago, IL, USA). A probability value of <0.05 was considered significant, and <0.01 was extremely significant. Orthogonal partial least squares discriminant analysis (OPLS-DA) and hierarchical cluster analysis (HCA) were performed using SIMCA 14.1 (Umetrics, Umea, Sweden).3 Results and discussion
3.1 Comparison of non-volatile constituents in Wuyi rock tea with different roasting degrees
Major non-volatile constituents of 12 Wuyi rock tea samples, made from four tea cultivars, were determined to explore the effect of roasting on tea quality. As shown in Table 1, the contents of moisture, total free amino acids and theaflavins decreased significantly for all the tea cultivar groups, and the protein content reduced for ‘QZ’ and ‘RG’ as the increase of roasting level (P < 0.05). Compared with the L-roasted samples, moisture contents decreased by 2.4–5.8% for most tea samples at the M- and S-roasting levels. Contents of total free amino acids and theaflavins in the S-roasted tea were 72–85% and 71–80% amount of that in L-roasted tea, respectively. For contents of soluble solids, total polyphenols, flavonoids, soluble sugar, as can be seen most of their values are with same superscripted letters within the same cultivar groups, indicating that roasting procedure had no remarkable influence on them, except that the maximum content values of polyphenols for ‘QZ’, flavonoids and soluble sugars for ‘DHP’ were observed in the M-roasted samples (P < 0.05), the difference of these ingredients were mainly between different varieties. For thearubigins and theabrownins, all the samples showed no significant difference (P > 0.05). Caffeine contents reached the maximum in all the M-roasted tea samples, and their values were 3–16% higher than the L- and S-roasted samples. The difference was statistically significant for three tea cultivar groups including ‘SX’, ‘DHP’ and ‘RG’ (P < 0.05). A relative increase in caffeine content was also found in the drying of green tea, but the reason was unclear.17 Caffeine has thermal stability below 200 °C, and decomposes completely at around 285 °C.18 Previous studies showed that heating increased the caffeine content of the regular robusta and civet coffee, due to damage of the cell membrane and the vacuoles that enhance the leaching yield.19 This may explain that the highest levels of caffeine and several constituents occurred in the M-roasted tea sample. Caffeine's sublimation is 120 °C, it may be the reason for its decreasing in sufficient roasting samples.| Tea samples | SX-L | SX-M | SX-S | QZ-L | QZ-M | QZ-S | DHP-L | DHP-M | DHP-S | RG-L | RG-M | RG-S | P |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Data are expressed as mean ± SD (n = 3). Values in a row followed by different letters are significantly different (P < 0.05). Data marked with different superscripted letters are significantly different at P < 0.05 level by the S–N–K test using SPSS 19.0 software. Capital letters (A, B, C…) represent a statistically significant difference within each row. | |||||||||||||
| Moisture content (%) | 3.8 ± 0.1C | 1.4 ± 0.0D | 1.2 ± 0.1FE | 7.1 ± 0.1A | 1.3 ± 0.0E | 1.2 ± 0.0E | 1.0 ± 0.0FG | 1.1 ± 0.0F | 0.9 ± 0.0GH | 5.9 ± 0.1B | 1.0 ± 0.1FGH | 0.9 ± 0.0H | P < 0.001 |
| Soluble solids (%) | 43 ± 1B | 43 ± 1B | 42 ± 1B | 46 ± 1A | 44 ± 2BA | 44 ± 1BA | 36 ± 1C | 37 ± 1C | 35 ± 1C | 35 ± 0C | 37 ± 1C | 35 ± 1C | |
| Proteins (%) | 4.8 ± 0.1B | 4.6 ± 0.1B | 4.7 ± 0.1B | 5.0 ± 0.1A | 4.8 ± 0.1B | 4.7 ± 0.1B | 4.0 ± 0.1DE | 4.1 ± 0.1DC | 4.0 ± 0.0DE | 4.2 ± 0.1C | 4.0 ± 0.1DE | 3.9 ± 0.1E | |
| Free amino acids (%) | 2.1 ± 0.1A | 1.7 ± 0.0C | 1.5 ± 0.1E | 1.9 ± 0.0B | 1.4 ± 0.0FE | 1.3 ± 0.0G | 1.7 ± 0.0DC | 1.6 ± 0.1D | 1.4 ± 0.1F | 1.6 ± 0.0DC | 1.3 ± 0.0G | 1.2 ± 0.0G | |
| Total polyphenols (%) | 9.7 ± 0.6C | 9.8 ± 0.5C | 9.8 ± 0.7C | 10.6 ± 0.6BC | 12.1 ± 0.5A | 10.7 ± 0.8BC | 9.9 ± 0.2C | 9.7 ± 0.3C | 10.2 ± 0.5BC | 10.5 ± 0.5BC | 10.8 ± 0.4BC | 11.3 ± 0.2BA | |
| Flavonoids (%) | 0.47 ± 0.01E | 0.47 ± 0.01E | 0.47 ± 0.01E | 0.52 ± 0.02DC | 0.51 ± 0.00DC | 0.48 ± 0.01ED | 0.53 ± 0.01BC | 0.56 ± 0.02A | 0.51 ± 0.02DC | 0.53 ± 0.01BC | 0.55 ± 0.01BA | 0.53 ± 0.01BAC | |
| Soluble sugars (%) | 15 ± 1EF | 13 ± 0F | 14 ± 1F | 17 ± 2ED | 15 ± 1EF | 16 ± 0E | 19 ± 1BC | 22 ± 2A | 20 ± 1B | 18 ± 1CD | 19 ± 1BC | 18 ± 1CD | |
| Caffeine (mg g−1) | 19 ± 0B | 23 ± 0A | 20 ± 0B | 22 ± 0A | 23 ± 0A | 23 ± 0A | 18 ± 0C | 20 ± 1B | 18 ± 0C | 18 ± 0C | 20 ± 0B | 18 ± 0C | |
| Total theaflavins (mg g−1) | 0.4 ± 0.0A | 0.3 ± 0.0B | 0.3 ± 0.0BC | 0.2 ± 0.0EF | 0.1 ± 0.0FG | 0.1 ± 0.0G | 0.2 ± 0.0DE | 0.2 ± 0.0D | 0.2 ± 0.0FG | 0.3 ± 0.0C | 0.3 ± 0.0C | 0.2 ± 0.0DE | |
| TF1 | 0.19 ± 0.01A | 0.14 ± 0.01B | 0.14 ± 0.01B | 0.05 ± 0.00F | 0.05 ± 0.03F | 0.04 ± 0.02F | 0.08 ± 0.00DE | 0.08 ± 0.00DE | 0.06 ± 0.00EF | 0.11 ± 0.00C | 0.10 ± 0.01CD | 0.09 ± 0.01CD | |
| TF2A | 0.08 ± 0.01A | 0.08 ± 0.00A | 0.07 ± 0.00B | 0.04 ± 0.00E | 0.03 ± 0.00F | 0.02 ± 0.00G | 0.05 ± 0.00D | 0.06 ± 0.00C | 0.04 ± 0.00E | 0.06 ± 0.00C | 0.05 ± 0.00D | 0.04 ± 0.00E | |
| TF2B | 0.04 ± 0.00A | 0.04 ± 0.00A | 0.04 ± 0.00A | 0.02 ± 0.00C | 0.01 ± 0.00D | 0.01 ± 0.00D | 0.02 ± 0.00C | 0.02 ± 0.00C | 0.01 ± 0.00D | 0.03 ± 0.00B | 0.02 ± 0.00C | 0.02 ± 0.00C | |
| TF3 | 0.09 ± 0.01B | 0.07 ± 0.00BCD | 0.07 ± 0.01BCD | 0.06 ± 0.01CDE | 0.05 ± 0.01DE | 0.05 ± 0.00DE | 0.05 ± 0.00DE | 0.07 ± 0.00BCD | 0.04 ± 0.00E | 0.08 ± 0.01BC | 0.11 ± 0.02A | 0.06 ± 0.01CDE | |
| Thearubigins (%) | 1.9 ± 0.2A | 1.9 ± 0.7A | 1.8 ± 0.2A | 1.8 ± 0.6A | 2.1 ± 0.3A | 2.2 ± 0.3A | 2.1 ± 0.7A | 2.2 ± 0.6A | 2.3 ± 0.3A | 2.1 ± 0.4A | 2.1 ± 0.5A | 2.2 ± 0.4A | P = 0.781 |
| Theabrownins (%) | 2.6 ± 0.2A | 3.5 ± 0.3A | 3.0 ± 0.2A | 2.7 ± 0.4A | 2.7 ± 0.7A | 2.9 ± 0.3A | 3.1 ± 0.2A | 3.3 ± 0.6A | 3.3 ± 0.7A | 2.6 ± 0.2A | 2.6 ± 0.4A | 3.0 ± 0.6A | P = 0.074 |
Although the folin–ciocalteu assay showed that roasting procedure had no significant influence on total polyphenol contents for the most tea cultivar groups used in this work, HPLC analysis revealed that contents of total and individual catechins tended to reach the maximum value at the M-roasting level, or decrease with the rise of roasting degree (Table 2). The total catechin contents of the S-roasted samples were 13–17% lower than the highest values in each cultivar groups. It's known that catechins undergo epimerization and degradation during thermal processing. The epistructured catechins (EGCG, EGC, ECG, EC) transform to be non-epistructured catechins (GCG GC, CG, C), and total catechin content decreases in dried green tea leaves.17 In this work, no association between the content alterations of epi- and non-epi catechins was observed, indicating that epimerization was not the main cause of catechin changes during roasting. This conversion may have been almost finished in the previous procedures including fixation and drying, which are similar with that in the green tea processing. Different trends in catechin content might be related to the physicochemical properties of fresh tea leaves. For SX-M sample, it had significant increase for most of non-epistructured catechins from SX-L, which was unique, it might because of the degradation of some unstable polymers of polyphenols since epistructured catechins didn't change much.
| Tea samples | SX-L | SX-M | SX-S | QZ-L | QZ-M | QZ-S | DHP-L | DHP-M | DHP-S | RG-L | RG-M | RG-S | P |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Data are expressed as mean ± SD (n = 3). Values in a row followed by different letters are significantly different (P < 0.05). Data marked with different superscripted letters are significantly different at P < 0.05 level by the S–N–K test using SPSS 19.0 software. Capital letters (A, B, C…) represent a statistically significant difference within each row. | |||||||||||||
| GC | 8.5 ± 0.6BCD | 14.9 ± 0.3A | 10.4 ± 1.5B | 5.3 ± 0.6EF | 5.5 ± 1.1EF | 4.5 ± 0.1F | 6.8 ± 0.2DE | 7.6 ± 0.9CDE | 5.6 ± 0.7EF | 8.4 ± 0.3BCD | 9.2 ± 0.2BC | 7.6 ± 1.0CDE | P < 0.001 |
| EGC | 18.2 ± 0.6BC | 22.2 ± 0.2A | 16.9 ± 0.4CD | 13.9 ± 0.2EFG | 12.5 ± 0.1GH | 10.8 ± 0.1H | 15.2 ± 0.1DEF | 15.8 ± 2.8DE | 13.0 ± 0.1FG | 19.1 ± 0.2B | 16.6 ± 0.7CD | 14.1 ± 0.2EFG | |
| C | 1.1 ± 0.1CD | 1.9 ± 0.0A | 1.0 ± 0.0CD | 0.8 ± 0.1CD | 0.8 ± 0.1CD | 0.7 ± 0.0D | 1.1 ± 0.0C | 1.5 ± 0.5B | 1.1 ± 0.1CD | 0.9 ± 0.0CD | 0.9 ± 0.0CD | 0.9 ± 0.0CD | |
| EC | 3.9 ± 0.0A | 3.5 ± 0.1C | 3.5 ± 0.0CD | 3.1 ± 0.0E | 2.9 ± 0.1F | 2.6 ± 0.0G | 3.7 ± 0.0B | 3.5 ± 0.1CD | 3.2 ± 0.0E | 3.5 ± 0.0C | 3.4 ± 0.1D | 3.0 ± 0.1E | |
| EGCG | 29.1 ± 0.6CD | 30.2 ± 0.3C | 28.5 ± 0.6DE | 37.5 ± 0.3A | 37.7 ± 0.6A | 35.2 ± 0.7B | 27.6 ± 0.1DEF | 27.6 ± 1.3DEF | 26.0 ± 0.1F | 29.3 ± 0.3CD | 30.4 ± 0.9C | 27.0 ± 0.7EF | |
| GCG | 7.6 ± 0.7ABC | 7.8 ± 0.3AB | 6.0 ± 0.3C | 8.4 ± 0.9A | 6.9 ± 0.7ABC | 6.0 ± 0.2C | 6.5 ± 0.3BC | 7.3 ± 0.4ABC | 5.9 ± 0.5C | 7.2 ± 0.3ABC | 7.0 ± 0.6ABC | 6.7 ± 0.3BC | |
| ECG | 10.0 ± 0.2E | 9.8 ± 0.1E | 9.3 ± 0.3F | 12.5 ± 0.1B | 8.3 ± 0.9G | 10.9 ± 0.3D | 12.8 ± 0.1A | 13.1 ± 0.7AB | 11.8 ± 0.1C | 9.7 ± 0.6EF | 9.4 ± 0.5F | 9.0 ± 0.3FG | |
| CG | 0.9 ± 0.1BC | 1.0 ± 0.0B | 0.6 ± 0.0CD | 0.9 ± 0.1BC | 0.7 ± 0.1CD | 0.6 ± 0.0D | 1.0 ± 0.1B | 1.2 ± 0.2A | 0.9 ± 0.1BC | 0.7 ± 0.0CD | 0.7 ± 0.1CD | 0.7 ± 0.0CD | |
| Total | 78.0 ± 2.8BC | 89.6 ± 1.2A | 74.7 ± 3.1BCDE | 81.3 ± 2.4B | 73.9 ± 7.6BCDE | 69.8 ± 1.3CDE | 73.7 ± 0.9BCDE | 76.2 ± 6.5BCD | 66.2 ± 1.7E | 77.6 ± 1.7BC | 76.0 ± 2.9BCD | 67.4 ± 2.4DE | |
HPLC analysis showed contents of total and most individual free amino acids significantly dropped with the increase of roasting levels, which was consistent with the results determined by ninhydrin assay. Compared with caffeine and catechins, amino acids were more sensitive to heat, and their total contents decreased by around 50% at the M- and S-roasting degrees (Table 3). Thermal processing is the usual and traditional procedure in food industries, and has a remarkably drastic effects on proteins and amino acids.20 During fixation in green tea manufacturing, total amino acids decreased slightly at 100 to 120 °C, and declined significantly at 140 to 160 °C.21.Amino acids usually react with carbonyl compounds (such as reducing sugars) through heating, and they continue with a large set of chain reactions, namely the Maillard reaction. These reactions may contribute a lot to the changes in aroma, color and flavor of tea.22 The infusion color of Wuyi rock tea usually turns from yellowish to reddish with the increase of roasting level. Our data showed the contents of three black tea pigments including theaflavins, thearubigins and theabrownins changed slightly for each cultivar groups (Table 2), indicating that products of Maillard reaction might account for the color alteration of Wuyi rock tea during roasting. L-theanine, the most abundant amino acid in tea, was reported to react with D-glucose in a thermal model, and produce methylpyrazine, 2,5-dimethylpyrazine as well as other volatiles.23 The transformation of amino acids through heating and their derivatives' influence on tea qualities need to be further explored.
| Tea samples | SX-L | SX-M | SX-S | QZ-L | QZ-M | QZ-S | DHP-L | DHP-M | DHP-S | RG-L | RG-M | RG-S | P |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a ND: not detected. Data are expressed as mean ± SD (n = 3). Values in a row followed by different letters are significantly different (P < 0.05). Data marked with different superscripted letters are significantly different at P < 0.05 level by the S–N–K test using SPSS 19.0 software. Capital letters (A, B, C…) represent a statistically significant difference within each row. | |||||||||||||
| ASP | 1.02 ± 0.08A | 0.78 ± 0.01B | 0.78 ± 0.03B | 0.95 ± 0.09A | 0.54 ± 0.03C | 0.54 ± 0.10C | 0.44 ± 0.00CDE | 0.41 ± 0.01DE | 0.36 ± 0.01EF | 0.49 ± 0.01CD | 0.35 ± 0.02EF | 0.29 ± 0.01F | P < 0.001 |
| GLU | 0.53 ± 0.04B | 0.23 ± 0.00DE | 0.23 ± 0.04DE | 0.61 ± 0.08A | 0.15 ± 0.01E | 0.13 ± 0.02E | 0.27 ± 0.01D | 0.21 ± 0.00DE | 0.18 ± 0.01DE | 0.44 ± 0.07C | 0.14 ± 0.01E | 0.17 ± 0.05DE | |
| ASN | 0.27 ± 0.02AB | 0.17 ± 0.01BC | 0.11 ± 0.04C | 0.38 ± 0.02A | 0.08 ± 0.01C | 0.07 ± 0.02C | 0.10 ± 0.00C | 0.09 ± 0.00C | 0.06 ± 0.00C | 0.08 ± 0.00C | 0.05 ± 0.00C | 0.05 ± 0.00C | |
| SER | 0.30 ± 0.03B | 0.21 ± 0.01BC | 0.18 ± 0.02C | 0.44 ± 0.12A | 0.19 ± 0.01BC | 0.18 ± 0.04C | 0.26 ± 0.01BC | 0.23 ± 0.01BC | 0.21 ± 0.02BC | 0.29 ± 0.01B | 0.19 ± 0.01BC | 0.17 ± 0.00C | |
| GLN | 0.15 ± 0.01A | 0.08 ± 0.00B | 0.06 ± 0.02B | 0.17 ± 0.02A | 0.08 ± 0.00B | 0.00 ± 0.00E | 0.07 ± 0.00B | 0.06 ± 0.00B | 0.05 ± 0.00B | 0.08 ± 0.01BC | 0.03 ± 0.00CD | 0.02 ± 0.02D | |
| HIS | ND | ND | ND | 0.06 ± 0.00A | ND | ND | 0.03 ± 0.02B | 0.02 ± 0.02BC | ND | 0.03 ± 0.00B | ND | ND | P = 0.0395 |
| GLY | 0.11 ± 0.01C | 0.10 ± 0.00C | 0.10 ± 0.00C | 0.19 ± 0.04A | 0.11 ± 0.00C | 0.11 ± 0.01C | 0.16 ± 0.00B | 0.15 ± 0.00B | 0.15 ± 0.01B | 0.15 ± 0.01B | 0.13 ± 0.00BC | 0.14 ± 0.01B | P < 0.001 |
| THR | 0.22 ± 0.02A | 0.16 ± 0.00B | 0.15 ± 0.01B | 0.21 ± 0.03A | 0.10 ± 0.01DE | 0.09 ± 0.03E | 0.12 ± 0.01CD | 0.10 ± 0.00ED | 0.09 ± 0.00E | 0.14 ± 0.01BC | 0.08 ± 0.00E | 0.07 ± 0.01E | |
| ARG | 0.25 ± 0.03B | 0.17 ± 0.02CD | 0.14 ± 0.03D | 0.49 ± 0.10A | 0.26 ± 0.03B | 0.23 ± 0.04BC | 0.12 ± 0.00D | 0.13 ± 0.00D | 0.09 ± 0.00D | 0.14 ± 0.01D | 0.09 ± 0.01D | 0.09 ± 0.01D | |
| ALA | 0.51 ± 0.01D | 0.52 ± 0.01D | 0.57 ± 0.02CD | 0.85 ± 0.07A | 0.67 ± 0.00B | 0.70 ± 0.04B | 0.37 ± 0.02F | 0.41 ± 0.01EF | 0.44 ± 0.01E | 0.62 ± 0.02C | 0.54 ± 0.01D | 0.55 ± 0.01D | |
| Theanine | 5.02 ± 0.39B | 2.35 ± 0.03CD | 1.96 ± 0.22E | 5.49 ± 0.30A | 1.38 ± 0.10F | 1.08 ± 0.24FG | 2.61 ± 0.00C | 1.89 ± 0.04E | 1.26 ± 0.01F | 2.24 ± 0.07DE | 0.81 ± 0.05G | 0.51 ± 0.00HI | |
| TYR | 0.51 ± 0.04A | 0.39 ± 0.00C | 0.43 ± 0.03B | 0.33 ± 0.05D | 0.16 ± 0.01G | 0.16 ± 0.03G | 0.26 ± 0.00E | 0.24 ± 0.01EF | 0.21 ± 0.01FG | 0.16 ± 0.01G | 0.10 ± 0.01H | 0.09 ± 0.00H | |
| VAL | 0.31 ± 0.03A | 0.23 ± 0.00B | 0.22 ± 0.00B | 0.29 ± 0.07A | 0.15 ± 0.01CD | 0.16 ± 0.03CD | 0.20 ± 0.00BC | 0.17 ± 0.00CD | 0.15 ± 0.00CD | 0.17 ± 0.01CD | 0.12 ± 0.01D | 0.13 ± 0.03D | |
| MET | 0.10 ± 0.00E | 0.11 ± 0.00D | 0.12 ± 0.00C | 0.14 ± 0.00B | 0.15 ± 0.00A | 0.16 ± 0.01A | ND | ND | ND | ND | ND | ND | |
| TRP | 0.42 ± 0.03A | 0.28 ± 0.01B | 0.29 ± 0.02B | 0.41 ± 0.07A | 0.17 ± 0.01C | 0.18 ± 0.00C | 0.19 ± 0.00C | 0.16 ± 0.00C | 0.14 ± 0.00CD | 0.19 ± 0.00C | 0.11 ± 0.01DE | 0.08 ± 0.00E | |
| PHE | 0.21 ± 0.02A | 0.15 ± 0.00BC | 0.15 ± 0.01BC | 0.16 ± 0.03BC | 0.06 ± 0.00D | 0.06 ± 0.01D | 0.20 ± 0.00A | 0.17 ± 0.00B | 0.14 ± 0.00C | 0.14 ± 0.01C | 0.08 ± 0.01D | 0.07 ± 0.01D | |
| ILE | 0.17 ± 0.01A | 0.12 ± 0.00BC | 0.10 ± 0.01CD | 0.14 ± 0.05B | 0.05 ± 0.00E | 0.05 ± 0.01E | 0.07 ± 0.00DE | 0.06 ± 0.00E | 0.05 ± 0.00E | 0.07 ± 0.01E | 0.04 ± 0.00E | 0.04 ± 0.01E | |
| LEU | 0.11 ± 0.01B | 0.13 ± 0.00A | 0.14 ± 0.01A | 0.10 ± 0.00B | 0.11 ± 0.01B | 0.11 ± 0.02B | 0.07 ± 0.00C | 0.06 ± 0.00CD | 0.05 ± 0.00CD | 0.06 ± 0.00DE | 0.04 ± 0.00E | 0.04 ± 0.01E | |
| LYS | 0.62 ± 0.05A | 0.43 ± 0.01B | 0.35 ± 0.06C | 0.45 ± 0.13B | 0.19 ± 0.01D | 0.18 ± 0.03DE | 0.14 ± 0.00DE | 0.12 ± 0.00DE | 0.10 ± 0.01DE | 0.11 ± 0.01DE | 0.07 ± 0.00E | 0.06 ± 0.01E | |
| Total | 10.8 ± 0.8B | 6.6 ± 0.1C | 6.1 ± 0.2C | 11.8 ± 1.5A | 4.6 ± 0.2DE | 4.2 ± 0.7E | 5.7 ± 0.0CD | 4.7 ± 0.1DE | 3.7 ± 0.1EF | 5.6 ± 0.3CD | 3.0 ± 0.2FG | 2.6 ± 0.2G | |
Catechins, caffeine and theanine are the major components contributing to the health benefits of tea. The contents of the former two ingredients changed less than 20% in this work, while the theanine content decreased by 52–80% after moderate and sufficient roasting. It was reported that different drying methods influenced the antioxidant activity of green tea.24 Our results indicated that sufficient roasting procedure might have negative effects on the bioactivity of Wuyi rock tea, especially that associated with theanine.
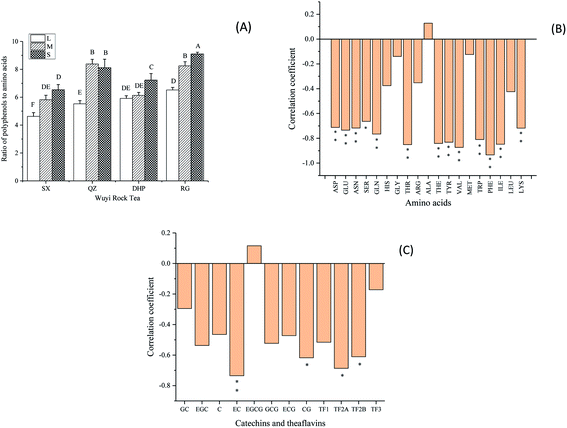
3.2 Effect of roasting on the polyphenols/amino acids ratio
Polyphenols contribute to bitterness and astringency of tea, and amino acids are closely related to tea freshness, sweetness, and briskness degree. The polyphenols/amino acids ratio (P/A) was regarded as an important indicator of tea flavor, and the lower value reflects the fresher and brisker taste of tea infusion.22 Considering obvious variation of the total and individual free amino acids in the tea samples, the ratio of total content of tea polyphenols to total content of amino acids was calculated.As shown in Fig. 1A, the P/A values significantly increased with the rise of roasting degree (P < 0.05), indicating the roasting procedure reduced the fresh and brisk taste of Wuyi rock tea. Pearson correlation coefficients between the P/A values and the contents of individual amino acids were calculated to quantify their contributions (Fig. 1B). Results showed that 13 amino acids were negatively correlated with the P/A values significantly (P < 0.05 or <0.01). The six amino acids, including His, Gly, Arg, Ala, Met and Leu, contributed little to the P/A values due to their slight changes. Amino acids can be divided into four groups according to their taste, including umami amino acids (Asp, Glu and theanine), bitter amino acids (Arg, His, Ile, Leu, Lys, Phe, Trp, Tyr and Val), sweet amino acids (Ala, Gly, Met, Pro, Ser and Thr) and sour amino acids (Gln).22 For the four L-roasted tea samples, Asp, Glu and theanine accounted for 56.8–60.7% of the total amino acid content, and decreased by 34.3–65.0% in the S-roasted tea samples. This result was consistent with the previous report that contents of umami amino acids were found to decrease faster than other amino acids during the fixation of green tea.21 Although the contents of catechins and theaflavins trended to decrease at higher roasting levels, no significant positive correlations between the P/A values and these polyphenol compounds were found (Fig. 1C). It's usually thought that the reduction of polyphenols contributes a lot to the mellow of tea infusion after heating. The lower content of catechins, especially galloylated catechins results in lower astringency perception of oolong tea (Chen, Yang, Lee, Wu, & Tzen, 2014). However, our results indicated that amino acids might play more important role in the taste change of Wuyi rock tea within a reasonable degree of roasting.
3.3 Volatile analysis
The volatiles in the Wuyi rock tea samples were analysed by HS-SPME/GC-MS. The number of compounds identified in each sample ranged from 61 to 87 (Table S2†). These compounds were classified into 11 categories according to their chemical structure, including alcohols, esters, nitrogen heterocyclic compounds, oxygen heterocyclic compounds, aromatic hydrocarbons, ketones, aldehydes, alkanes, olefins, acids, and sulphur compounds. Alcohols, esters, and nitrogen heterocyclic compounds were the major classes, and accounted for around 50% to 60% of the total aroma constituents. While acids and sulphur compounds were the minor groups.A total of 315 volatile compounds were detected in the 12 samples (Table S3†). Among then, 159 compounds were only found in one sample, and 87 in more than 3 samples. The relative contents of volatiles varied, dependent on tea cultivar and roasting degree. Some compounds were only detected in one tea cultivar group with high relative contents, indicating that they might contribute a lot to the distinct aroma named “cultivated varieties flavor”.4 For example, o-xylene and (Z)-3-ethyl-2-methyl-1,3-hexadiene were only detected in SX-M with the relative content of 9.86% and in SX-L with the relative content of 4.95%, respectively. o-Xylene possesses sweet odour, and has been found in Tieguanyin oolong tea and Kangra orthodox black tea.5,25 (Z)-3-Ethyl-2-methyl-1,3-hexadiene was considered as a major aroma compound in the flowers of cymbidium goeringii and sweet corn.26,27 cis-Verbenol was a unique volatile in QZ-L with the relative content of 2.79%, and had a fresh piney note with an ozonic character. Benzyl nitrile, trans-beta-ionon and hexadecane were only detected in RG-L, and their relative contents were more than 2%. Benzyl nitrile is a volatile compound that accumulates in tea under stresses, and has almond-like odour.28 Beta-ionone has an odour of cedar wood, and resembles odour of violets in dilute alcoholic solution.29 Hexadecane was detected in the shoots of tea variety ‘Rougui’ previously, but not in variety of ‘Tieguanyin’.30
3.4 OPLS and HCA analysis of differential volatile compounds accounting for roasting level
OPLS is a supervised multivariate projection method for analysis of correlation and discrimination. In order to maximize the separation among tea samples with different roasting levels and identify the volatiles responsible for the separation, OPLS was applied to remove noncorrelated variation in X variables (volatile compounds) to Y variables (roasting level). The OPLS score plot showed a clear separation among roasting degrees, with a Q2 value of 0.915 (Fig. 2A). HCA is a strategy of cluster analysis which seeks to build a hierarchy of clusters. In order to have a better visualization of the aroma differences among the Wuyi rock teas, HCA was performed based on data set in Table S2.† As shown in Fig. 2B, 12 samples were divided into two groups by distance 229.09 on log scale, and the second group was further divided into two group by distance 44.92 on log scale. The tea samples with three different roasting levels were clearly separated, and aromatic profiles of the low- and moderate-roasted samples are much more similar in contrast to the sufficient-roasted samples. The HCA result was consistent with that of the OPLS, indicating data processing method used in this work was reliable.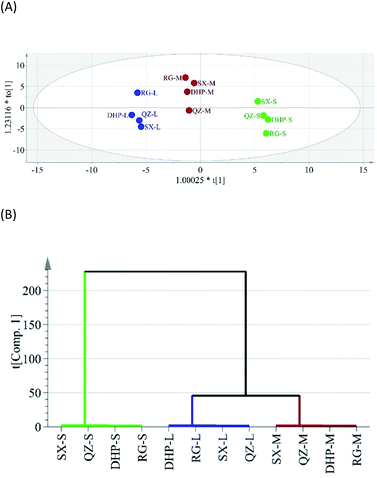 | ||
| Fig. 2 Discriminant analysis of all the Wuyi rock tea samples basing on the GC-MS data. (A) OPLS score plot. (B) Hierarchical clustering of 12 tea samples. | ||
In order to identify the volatiles responsible for the separation, variable importance in the projection (VIP) statistics was introduced to select the key compounds. Among the 315 volatiles, 99 compounds were considered important in the OPLS model with VIP values larger than 1.00, and the values ranged from 1.01 to 3.28 (Table 4). Twenty-one compounds exhibited high VIP values more than 2.00. Among them, ‘Naphthalene, 1,2,3,4-tetrahydro-1,6,8-trimethyl-’ and ‘1,1,5-Trimethyl-1,2-dihydronaphthalene’ had the highest VIP values of 3.28 and 2.96, and increased from almost zero to around 1–3% with the rise of roasting degree in all the tea cultivar groups. Naphthalene derivatives were also found in the aroma of green tea, Liubao tea, panned and unpanned oolong tea from Taiwan, and large-leaf yellow tea,31–35 and some of the ordor charastetistics of them are known. For example, 1,1,5-trimethyl-1,2-dihydronaphthalene were reported to have floral scent. Naphthalene, 1,2-dihydro-1,1,6-trimethyl- has a licorice-like or woody aroma and its threshold in water is 2.5 μg L−1.35 These compounds belong to polycyclic aromatic hydrocarbons (PAHs), which not only exist in the fresh leaves and roots of tea plants due to the absorption and accumulation from the surrounding environment, but are also produced during the tea making process.36 Our data showed that the percentage of other naphthalene derivatives including ‘Naphthalene, 1,2,3,4-tetrahydro-1,5,8-trimethyl-’, ‘Naphthalene, 1,2,3,5,6,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-, (1S-cis)- ’, ‘Naphthalene, 1,2-dihydro-1,1,6-trimethyl-’ and ‘Naphthalene, 1,2-dihydro-1,5,8-trimethyl-’ changed little or declined with the increased roasting level in several tested tea groups, indicating not all cortho-fused bicyclic hydrocarbons were positively correlated with the roasting degree of Wuyi rock tea in this work.
| Number | Compounds | VIP | Correlation |
|---|---|---|---|
| a P: relative content of compound is positively correlated with roasting degree. N: relative content of compound is negatively correlated with roasting degree. | |||
| 1 | Naphthalene, 1,2,3,4-tetrahydro-1,6,8-trimethyl- | 3.28 | P |
| 2 | 1,1,5-Trimethyl-1,2-dihydronaphthalene | 2.96 | P |
| 3 | Pentadecane | 2.56 | N |
| 4 | p-Xylene | 2.52 | P |
| 5 | Alpha.-methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol | 2.49 | P |
| 6 | Benzaldehyde, 3-benzyloxy-2-fluoro-4-methoxy- | 2.49 | N |
| 7 | Hydrazinecarboxylic acid, phenylmethyl ester | 2.32 | P |
| 8 | Ethinamate | 2.29 | N |
| 9 | Alpha.-acorenol | 2.29 | N |
| 10 | E-2-Hexenyl benzoate | 2.28 | N |
| 11 | Disulfide, di-tert-dodecyl | 2.27 | N |
| 12 | Bicyclo[3.1.1]hept-2-en-4-ol, 2,6,6-trimethyl-, acetate | 2.21 | P |
| 13 | (9-Oxabicyclo[3.3.1]non-6-en-3-yl)methanol | 2.21 | P |
| 14 | 1-Methoxyadamantane | 2.20 | N |
| 15 | Phytol, acetate | 2.19 | P |
| 16 | 3-Buten-2-one, 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)- | 2.19 | N |
| 17 | R-Limonene | 2.19 | P |
| 18 | 1,6-Octadien-3-ol, 3,7-dimethyl- | 2.17 | N |
| 19 | 3-Buten-2-one, 4-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | 2.16 | N |
| 20 | Heptadecane, 2,6,10,15-tetramethyl- | 2.13 | N |
| 21 | Hexanoic acid, 3-hexenyl ester, (Z)- | 2.08 | P |
| 22 | 5-Ethyl-5-methyl-2-phenyl-2-oxazoline | 1.97 | N |
| 23 | 1,2-Propanediol diformate | 1.95 | P |
| 24 | 2H-Pyran-3-ol, 6-ethenyltetrahydro-2,2,6-trimethyl- | 1.92 | P |
| 25 | 4-Epi-cubedol | 1.92 | N |
| 26 | 4-(2,4,4-Trimethyl-cyclohexa-1,5-dienyl)-but-3-en-2-one | 1.91 | N |
| 27 | Terpinyl formate | 1.87 | P |
| 28 | 1,6,10-Dodecatrien-3-ol, 3,7,11-trimethyl- | 1.87 | N |
| 29 | Benzenepropanoic acid, alpha.-(hydroxyimino)- | 1.87 | N |
| 30 | Formic acid, 3,7,11-trimethyl-1,6,10-dodecatrien-3-yl ester | 1.84 | P |
| 31 | Cyclopentanone, 2-cyclopentylidene- | 1.77 | N |
| 32 | Acetic acid, (1,2,3,4,5,6,7,8-octahydro-3,8,8-trimethylnaphth-2-yl)methyl ester | 1.76 | P |
| 33 | N-[3-[N-Aziridyl]propylidene]tetrahydrofurfurylamine | 1.74 | N |
| 34 | Cyclohexanol, 4-methyl-, trans- | 1.74 | P |
| 35 | Hexanoic acid, hexyl ester | 1.74 | P |
| 36 | 2-Octen-1-ol, 3,7-dimethyl-, isobutyrate, (Z)- | 1.71 | N |
| 37 | [5,5-Dimethyl-6-(3-methyl-buta-1,3-dienyl)-7-oxa-bicyclo[4.1.0]hept-1-yl]-methanol | 1.69 | N |
| 38 | D-Limonene | 1.69 | N |
| 39 | 2,5-Octadecadiynoic acid, methyl ester | 1.69 | N |
| 40 | 1-Penten-3-one, 1-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | 1.65 | P |
| 41 | 10-Undecynoic acid, methyl ester | 1.64 | P |
| 42 | Phenol, 2,4,6-tris(1,1-dimethylethyl)- | 1.59 | N |
| 43 | 1-Heptatriacotanol | 1.57 | N |
| 44 | Tetrapentacontane, 1,54-dibromo- | 1.56 | P |
| 45 | Hexanoic acid, 2-hexenyl ester, (E)- | 1.54 | P |
| 46 | Butanoic acid, 3-methyl-, 2-hexenyl ester, (E)- | 1.53 | N |
| 47 | Octadecanal, 2-bromo- | 1.53 | P |
| 48 | (3-Fluorophenyl) methanol, 3-methylbutyl ether | 1.52 | P |
| 49 | 2,7-Octadiene-1,6-diol, 2,6-dimethyl- | 1.52 | N |
| 50 | 7-Oxabicyclo[4.1.0]heptane, 1-methyl-4-(2-methyloxiranyl)- | 1.52 | N |
| 51 | E-10-Methyl-11-tetradecen-1-ol propionate | 1.52 | P |
| 52 | 17-Octadecynoic acid | 1.50 | N |
| 53 | Cyclohexene, 1-(2-nitro-2-propenyl)- | 1.50 | P |
| 54 | Acetamide, N-(4,6,6-trimethylbicyclo[3.1.1]hept-3-en-2-yl)-, (1-alpha. 2-beta, 5-alpha)- | 1.49 | P |
| 55 | (2,2,6-Trimethyl-bicyclo[4.1.0]hept-1-yl)-methanol | 1.48 | P |
| 56 | 10-Heptadecen-8-ynoic acid, methyl ester, (E)- | 1.48 | N |
| 57 | tert-Hexadecanethiol | 1.46 | P |
| 58 | Undec-10-ynoic acid | 1.45 | P |
| 59 | 2,6-Octadiene-1,8-diol, 2,6-dimethyl- | 1.44 | P |
| 60 | Hexanal | 1.44 | P |
| 61 | Geranyl isovalerate | 1.41 | N |
| 62 | 3-Isopropylidene-5-methyl-hex-4-en-2-one | 1.37 | N |
| 63 | 2,4-Di-t-butyladamantane-2,4-diol | 1.36 | N |
| 64 | 7-Propylidene-bicyclo[4.1.0]heptane | 1.36 | P |
| 65 | 2,4-Nonadienal, (E,E)- | 1.35 | P |
| 66 | 3,7-Octadiene-2,6-diol, 2,6-dimethyl- | 1.35 | N |
| 67 | (−)-Myrtenol | 1.35 | P |
| 68 | 4-(2,5-Dihydro-3-methoxyphenyl)butylamine | 1.35 | N |
| 69 | 2-Pentadecanone, 6,10,14-trimethyl- | 1.34 | P |
| 70 | N-Benzyl-2-aminocinnamate, methyl ester | 1.34 | N |
| 71 | 2,2-Difluoroethanol, tert-butyldimethylsilyl ether | 1.33 | P |
| 72 | Fumaric acid, 2-methylcyclohex-1-enylmethyl pentadecyl ester | 1.33 | N |
| 73 | 2,5-Dimethylcyclohexanol | 1.32 | N |
| 74 | 3-Decanynoic acid | 1.31 | P |
| 75 | 2,4-Heptadienal, (E,E)- | 1.31 | P |
| 76 | 2(4H)-Benzofuranone, 5,6,7,7a-tetrahydro-4,4,7a-trimethyl-, (R)- | 1.30 | N |
| 77 | Dodecane, 2-methyl- | 1.29 | N |
| 78 | Benzaldehyde | 1.29 | N |
| 79 | 1b,4a-Epoxy-2H-cyclopenta[3,4]cyclopropa[8,9]cycloundec[1,2-b]oxiren-5(1aH)-one, 2,7,9,10-tetrakis(acetyloxy)decahydro-3,6,8,8,10a-pentamethyl- | 1.26 | N |
| 80 | Pyrazine, 2-ethyl-3,5-dimethyl- | 1.26 | N |
| 81 | Butanoic acid, 2-methyl-, hexyl ester | 1.26 | N |
| 82 | Limonen-6-ol, pivalate | 1.25 | P |
| 83 | Oct-3-ene-1,5-diyne, 3-t-butyl-7,7-dimethyl- | 1.23 | N |
| 84 | Naphthalene, 1,2,3,5,6,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-, (1S-cis)- | 1.21 | N |
| 85 | Tricyclo[2.2.1.0(2,6)]heptan-3-ol, 4,5,5-trimethyl- | 1.16 | N |
| 86 | 7-Methyl-Z-tetradecen-1-ol acetate | 1.16 | N |
| 87 | Tricyclo[4.2.2.0(1,5)]decan-7-ol | 1.14 | P |
| 88 | 1,5,5-Trimethyl-6-methylene-cyclohexene | 1.12 | N |
| 89 | Mesitylene | 1.11 | N |
| 90 | cis-3-Hexenyl-, alpha.-methylbutyrate | 1.10 | N |
| 91 | 3-Cyclohexen-1-carboxaldehyde, 3,4-dimethyl- | 1.10 | N |
| 92 | 2-Methyl-4-(2,6,6-trimethylcyclohex-2-enyl)but-3-en-2-ol | 1.09 | N |
| 93 | 1,2-15,16-Diepoxyhexadecane | 1.08 | N |
| 94 | cis-, beta, -Farnesene | 1.07 | P |
| 95 | Naphthalene, 1,2-dihydro-1,1,6-trimethyl- | 1.02 | P |
| 96 | Caryophyllene | 1.02 | N |
| 97 | Cedrane, 8-propoxy- | 1.02 | P |
| 98 | Ethyl iso-allocholate | 1.02 | P |
| 99 | 10-Hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-eicosahydropicene-2-carboxylic acid | 1.01 | N |
The relative contents of ‘p-Xylene’ (VIP 2.52), ‘alpha.-Methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol’ (VIP 2.49) and ‘Hydrazinecarboxylic acid, phenylmethyl ester’ (VIP 2.32) increased from approximate 0–1% to 4–6%, and the highest values occurred at the moderate or sufficient roasting levels for all the tea cultivar groups. ‘p-Xylene’ is reported to be a major aromatic constituent of another oolong tea called Fenghuang Dancong,37 it has plastic, green or pungent ordor and its threshold in water is 1000 μg L−1. The compound ‘alpha.-Methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol’ is abundant in 19 tea varieties suitable for producing green tea38 and also appear in Osmanthus fragrans and strawberry.39,40 As far as we known, ‘Hydrazinecarboxylic acid, phenylmethyl ester’ was first discovered in tea aroma.
Pentadecane and 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-3-buten-2-one, ranked top two according to the VIP value among the volatiles which were negatively correlated with roasting degree. The former compound's threshold in water is 1.3 × 107 μg L−1, indicating it may contribute little to tea aroma. The latter has violet, sweet and floral fragrance, and its threshold in water is 76 μg L−1.35 Its decline trend is consistent with lessen of flowery scent during roasting.
The above mentioned six compounds, including ‘Naphthalene, 1,2,3,4-tetrahydro-1,6,8-trimethyl-’, ‘1,1,5-trimethyl-1,2-dihydronaphthalene’, ‘p-Xylene’, ‘alpha.-methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol’, ‘Hydrazinecarboxylic acid, phenylmethyl ester’, and ‘3-buten-2-one, 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-’ might be the major characteristic markers for roasting process of Wuyi rock tea.
Other 94 compounds with VIP values greater than 1.00, didn't show consistent trend or were not detected in all the tea groups. Many of these compounds have been found in tea and aromatic plants, which assumes that they are also key aroma of Wuyi rock tea but have little change during roasting process. Some of them have floral and fruity or chemical odor, like D-limonene with fruity aroma, hexanal with tallowy and fruity smell, pyrazine, 2-ethyl-3,5-dimethyl- with nutty and roasted odor, threshold valves of them are 34, 4.5, and 0.04 μg L−1, respectively.35 Three compounds showed steady in most of our samples.
The OPLS model gave the overall result that 55 and 39 volatiles were correlated with the roasting degree, negative or positive respectively (Table 4). Volatiles could provide rich chemical fingerprinting, and characterize the special flavor of tea, fruits, and oils via multivariate statistical analysis.41,42 M. Zhang (2020) successfully discriminate five similar oolong tea varieties based on aromatic profiles analyzed by HS-SPME/GC-MS.41 It was found that the major volatiles of oolong tea in northern Thailand were significantly correlated with altitude.43 Chen et al. classified old (more than one-year storage) and new (less than one-year storage) Tieguanyin oolong teas correctly according to aroma pattern.44 In this work, aroma fingerprint was competent to distinguish Wuyi rock tea with different roasting grades, indicating that it was a promising objective method to classify roasting levels which were independent on experiences of tea processing and sensory evaluation.
4 Conclusions
Roasting had remarkable effects on the non-volatiles and volatiles of Wuyi rock tea. The both content of catechins and amino acids reduced, and the ratio of polyphenols/amino acids obviously increased with the rise of roasting degree, moderate roasting reduced bitterness & astringency taste and formed unique, soft flavor, while high-level roasting not only decreased the fresh and brisk taste of tea infusion, but also lowered the contents of catechins and theanine related to the health benefits of tea. A model for roasting-level discrimination was successfully established based on 315 volatiles with OPLS-DA. A total of 99 volatiles were found to be key for this OPLS model, and might account for the diverse aroma flavor of Wuyi rock tea with different roasting grades. ‘Naphthalene, 1,2,3,4-tetrahydro-1,6,8-trimethyl-’, ‘1,1,5-trimethyl-1,2-dihydronaphthalene’, ‘p-Xylene’, ‘alpha.-methyl-.alpha.-[4-methyl-3-pentenyl]oxiranemethanol’, ‘Hydrazinecarboxylic acid, phenylmethyl ester’, and ‘3-buten-2-one, 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)-’ are the key characteristic markers for roasting process of Wuyi rock tea. This work contributed to the scientific elucidation of overall quality and chemical changes of Wuyi rock tea with roasting procedure.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This research was funded by Collaborative Innovation Center of Chinese Oolong Tea Industry (Grant No. 2015-75), Science and Technology Innovation Platform Project of Fujian Provincial Department of Science and Technology (No. 2018N2004), Key Research and Development Program of Zhejiang Province (Grant No. 2018C02012), National Key Research and Development Program of China (Grant No. 2017YFD0400800), National Natural Science Foundation of China (Grant No. 31501474), Natural Science Foundation of Zhejiang Province (Grant No. LY15C200007), Fundamental Research Funds for the Central Universities, Guiding project of Fujian Science and Technology Department (Grant No. 2018N0030), and the 2016 Project of Funding Municipal Project. We thank Mr Baoshun Liu for kindly providing Wuyi rock tea samples.Notes and references
- B. Li, S. B. Vik and Y. Tu, J. Nutr. Biochem., 2012, 23, 953–960 CrossRef CAS PubMed.
- W. X. Bai, C. Wang, Y. J. Wang, W. J. Zheng, W. Wang, X. C. Wan and G. H. Bao, J. Agric. Food Chem., 2017, 65, 2999–3005 CrossRef CAS PubMed.
- S. Chen, M. Li, G. Zheng, T. Wang, J. Lin, S. Wang, X. Wang, Q. Chao, S. Cao and Z. Yang, Molecules, 2018, 23, 104 CrossRef PubMed.
- F. Zhao, H.-T. Lin, S. Zhang, Y.-F. Lin, J.-F. Yang and N.-X. Ye, J. Agric. Food Chem., 2014, 62, 2772–2781 CrossRef CAS PubMed.
- Y.-Q. Xu, P.-P. Liu, J. Shi, Y. Gao, Q.-S. Wang and J.-F. Yin, Food Chem., 2018, 249, 176–183 CrossRef CAS PubMed.
- T. Bertuzzi, E. Martinelli, A. Mulazzi and S. Rastelli, Food Chem., 2020, 303, 125372 CrossRef CAS PubMed.
- W. Schlörmann, S. Zetzmann, B. Wiege, N. Haase, A. Greiling, S. Lorkowski, C. Dawczynski and M. Glei, Food Chem., 2020, 307, 125548 CrossRef PubMed.
- S. Chen, H. Liu, X. Zhao, X. Li, W. Shan, X. Wang, S. Wang, W. Yu, Z. Yang and X. Yu, Food Res. Int., 2020, 128, 108778 CrossRef CAS PubMed.
- L. Wu, X. Huang, S. Liu, J. Liu, Y. Guo, Y. Sun, J. Lin, Y. Guo and S. Wei, Food Chem., 2020, 310, 125941 CrossRef CAS PubMed.
- T. Xia, Manufacture of tea, China Agricalture Press, Beijing, 2014 Search PubMed.
- S. Gong, C. Lu, X. Liu, Y. Zhao, H. Shen, Y. Gou, J. Wu, L. Zhao and Z. Gu, Methodology of sensory evaluation of tea, Standards Press of China, 2018 Search PubMed.
- Y. Xu, Y. Jin, Y. Wu and Y. Tu, J. Liq. Chromatogr. Relat. Technol., 2010, 33, 1791–1801 CrossRef CAS.
- L. Zhong, Physicochemical quality analysis of tea leaf, Shanghai Science and Technology Press, Shanghai, 1989 Search PubMed.
- W. Zhou, J. Xu and L. Xu, Determination of total polyphenols and catechins content in tea, Standards Press of China, 2018 Search PubMed.
- Q. Wang, C. Peng and J. Gong, J. Sci. Food Agric., 2011, 91, 2412–2418 CrossRef CAS PubMed.
- R. M. Kazan, H. A. Seddik, Z. M. Marstani, M. M. Elsutohy and N. G. Yasri, Microchem. J., 2019, 150, 104103 CrossRef CAS.
- N. Donlao and Y. Ogawa, LWT--Food Sci. Technol., 2019, 116, 108567 CrossRef CAS.
- R. Wang, J. Xue, L. Meng, J.-W. Lee, Z. Zhao, P. Sun, L. Cai, T. Huang, Z. Wang and Z.-K. Wang, Joule, 2019, 3, 1464–1477 CrossRef CAS.
- S. Chan and E. Garcia, Manila Journal of Science, 2011, 7, 19–23 Search PubMed.
- S. Esteghlal, H. H. Gahruie, M. Niakousari, F. J. Barba, A. E.-D. Bekhit, K. Mallikarjunan and S. Roohinejad, Foods, 2019, 8, 262 CrossRef CAS PubMed.
- A. Mao, H. Su, S. Fang, X. Chen, J. Ning, C. Ho and X. Wan, Int. J. Food Sci. Technol., 2018, 53, 2586–2594 CrossRef CAS.
- Z. Yu and Z. Yang, Crit. Rev. Food Sci. Nutr., 2020, 60, 844–858 CrossRef CAS PubMed.
- X. Guo, C. Song, C. T. Ho and X. Wan, Food Chem., 2018, 263, 18–28 CrossRef CAS PubMed.
- S. Roshanak, M. Rahimmalek and S. A. H. Goli, J. Food Sci. Technol., 2016, 53, 721–729 CrossRef CAS PubMed.
- X. Guo, C. T. Ho, W. Schwab, C. Song and X. Wan, Food Chem., 2019, 280, 73–82 CrossRef CAS PubMed.
- R. Joshi and A. Gulati, Food Chem., 2015, 167, 290–298 CrossRef CAS PubMed.
- J.-M. Chen, J.-Y. Song, J. He, X.-R. Gu and X. Zhang, Acta Hortic. Sin., 2016, 12, 2461–2472 Search PubMed.
- X. Feng, L. Pan, Q. Wang, Z. Liao, X. Wang, X. Zhang, W. Guo, E. Hu, J. Li and J. Xu, PLoS One, 2020, 15, e0233094 CrossRef CAS PubMed.
- Y. Liao, L. Zeng, H. Tan, S. Cheng, F. Dong and Z. Yang, J. Agric. Food Chem., 2020, 68, 1397–1404 CrossRef CAS PubMed.
- L. S. Karaffa, The Merck index: an encyclopedias of chemicals, drugs, and biologicals, RSC Publishing, Cambridge, UK, 2013 Search PubMed.
- S. Jin, L.-W. Han, N.-X. Ye, W. Wang, H.-s. Huang and W. Liu, Chinese Journal of Tropical Crops, 2019, 40, 576–582 Search PubMed.
- E. Sheibani, S. E. Duncan, D. D. Kuhn, A. M. Dietrich, J. J. Newkirk and S. F. O'Keefe, Food Sci. Nutr., 2016, 4, 456–468 CrossRef CAS PubMed.
- M. Wang, Y. Zhu, Y. Zhang, J. Shi and Z. Lin, Shipin Kexue, 2019, 40, 219–228 Search PubMed.
- X. Guo, C. T. Ho, X. Wan, H. Zhu, Q. Liu and Z. Wen, Food Chem., 2021, 341, 128230 CrossRef CAS PubMed.
- C. Wang, S. Lv, Y. Wu, X. Gao, J. Li, W. Zhang and Q. Meng, SpringerPlus, 2016, 5, 576 CrossRef PubMed.
- F. Shang, Y. Wang, J. Wang, L. Zhang, P. Cheng and S. Wang, Microchem. J., 2019, 146, 957–964 CrossRef CAS.
- Z.-K. Yan, Y.-L. Li and D.-H. Zhuang, Food Res. Dev., 2015, 36, 78–81 CAS.
- Q. He, D.-Y. Huang, C. Lu, D.-Q. Qi, S.-H. Tian, J. Lin, Y.-B. Zhou and C.-W. Shen, Acta Agric. Boreali-Occident. Sin., 2017, 26, 1363–1378 Search PubMed.
- Y. H. Kim, K. H. Kim, J. E. Szulejko and D. Parker, Sensors, 2013, 13, 7939–7978 CrossRef CAS PubMed.
- C. Hu, Y. Liang, M. Zeng, L. Zhang and Q. Min, Chem. Reagents, 2010, 32, 231–234 CAS.
- M. Zhang, L. Li, G. Song, H. Wang, H. Wang and Q. Shen, LWT--Food Sci. Technol., 2020, 120, 108922 CrossRef CAS.
- J. Lin, P. Zhang, Z. Pan, H. Xu, Y. Luo and X. Wang, Food Chem., 2013, 141, 259–265 CrossRef CAS PubMed.
- P. Pripdeevech, J. Rothwell, P. E. D'Souza and P. Panuwet, Int. J. Food Prop., 2017, 20, S2450–S2462 CrossRef CAS.
- L. Chen, Q.-X. Lin, Y.-G. Zhang, J. Chen, L.-L. Wang, W.-Q. Yu and Z.-M. You, J. Tea Sci., 2018, 38, 253–262 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/d0ra09703a |
| ‡ These authors are considered as co-first authors. |
| This journal is © The Royal Society of Chemistry 2021 |