South African university students’ attitudes towards chemistry learning in a virtually simulated learning environment†
Mafor
Penn
 *a and
Umesh
Ramnarain
*a and
Umesh
Ramnarain
 *b
*b
aDepartment of Science and Technology Education, Faculty of Education, University of Johannesburg, South Africa. E-mail: mpenn@uj.ac.za
bScience Education, Science and Technology Education Faculty of Education, University of Johannesburg, South Africa. E-mail: uramnarain@uj.ac.za; Web: https://www.uj.ac.za/contact/Pages/Prof-Umesh-Ramnarain.aspx
First published on 17th May 2019
Abstract
This mixed method study investigated changes in 3rd year Bachelor of Education students’ attitudes towards chemistry after learning interventions with virtual chemistry simulations. After participant students identified certain concepts from their 3rd year chemistry module as being abstract and not easily comprehensible, these concepts were facilitated during a 5 week learning intervention using PhET simulations as an alternative to traditional laboratory experimentation. In the first quantitative phase of the study, a 30-item pre-attitude test was administered to assess students’ attitudes towards chemistry, followed by PhET chemistry simulation learning interventions. Thereafter, students wrote a post-attitude test. Findings of this phase revealed a significantly higher mean post-attitude test score, with students showing a positive attitude towards chemistry learning, post-intervention. The quantitative phase was followed by a qualitative phase which examined students’ experiences on the use of simulations through semi-structured interviews. Findings from the qualitative phase revealed that students experienced autonomy and enjoyment during engagement with the simulations. They also perceived that their experiences in the virtually simulated environment improved their visualisation of chemistry concepts, thereby improving conceptual understanding. However, the students acknowledged that simulations cannot replicate the realism and authenticity associated with practical work in an actual laboratory. The implications of these findings are that virtual simulations provide a complementary learning tool capable of improving students’ attitudes towards chemistry, and perceived by students to support their visualisation of abstract chemistry concepts. The findings of this study are particularly significant for chemistry learning at schools and universities in economically challenged countries such as South Africa where there is a shortage of well-equipped laboratories.
Introduction
Students’ attitudes towards science and the learning of science is one of the fundamental goals of science teaching and learning (Hofstein and Mamlok-Naaman, 2011; Can and Boz, 2012). Hofstein and Mamlok-Naaman (2011) identify three key factors which can be considered to enhance students’ attitudes and these include “the methods used to present the content, instructional techniques and gender issues” (p. 90). The nature of chemistry as a science subject is such that students primarily experience difficulties in visualising abstract micro-phenomena (Cai et al., 2014). These micro-complexities inherent in the nature of chemistry concepts, demand the incorporation of multiple tools in representing concepts, in order to enhance students’ conceptual understandings. Many studies which have examined students’ performance in chemistry have implicated abstraction, low imaginative power, poor concept representation and formation as some of the factors responsible for poor performance in chemistry both at high school and tertiary levels (Harle and Towns, 2011; Esquembre, 2015). These factors also correlate with poor attitudes towards chemistry learning (Chua and Karpudewan, 2017). Studies on challenges in chemistry learning have concluded that several facts including learning environment and cognitive ability have an influence on attitude towards science in general (Otor and Achor, 2013; Xu et al., 2013).The importance of attitude as an affective construct in chemistry learning and the role of a learning environment as an influencing factor stimulated the inquiry reported in this article. Taylor, Ramsey and Howe in (Hassan, 1985) pointed out that although attitude is regarded as a significant outcome of science teaching and a relevant variable in science learning, “little has been made toward specifying and determining the conditions that affect their dynamics and influence their development” (p. 4). This study investigated 3rd year Bachelor of Education students’ attitudes towards chemistry before and after a learning intervention with virtual simulations. In South Africa, there is a dearth of research on assessing the factors that affect university students’ attitudes towards chemistry (Ramnarain and Ramaila, 2018) especially amongst students who are aspiring to become chemistry teachers. Hence, the study was designed with the aim of exploring an alternative learning environment, namely, a virtually simulated environment, and how this may possibly impact students’ attitudes towards the subject.
In chemistry learning, experimentation in traditional school chemistry laboratories has been regarded as a means by which student visualisation of the abstract chemistry phenomena can be enhanced (Estapa and Nadolny, 2015; Hsu et al., 2017). Some of the advantages of experimental chemistry include, students’ active engagement in learning tasks, visualisation of learned chemical processes/concepts and exposure to processes that scientists follow to provide authentic real world solutions. Visualisation supports students to create their own mental representations of chemical processes and enables long-term retention of learned concepts (Mosotho and Mamontsi, 2014).
The South African context
In spite of the accrued benefits of experimental chemistry, several challenges are associated with the availability and use of traditional chemistry laboratories in schools and higher institutions of learning. Some of the challenges include, the dangers associated with handling chemicals, overcrowding of students, little time allocated for experimentation within syllabi and lack of physical laboratory resources for sustaining student practical experiences (Chiu et al., 2015; Faour and Ayoubi, 2018). In South Africa, these challenges are especially acute as Hattingh et al. (2007) suggest that the lack of resources for practical work is especially prevalent in developing world countries like South Africa. The previous Apartheid education system was comprised of separate education departments described as “Blacks”, “Whites”, “Coloureds”, and “Indians”, with inequitable distribution of resources between departments. Apartheid education was characterized by gross inequalities in the financing of education (Lelliott, 2014). Although this was reflected in all areas of school funding, the legacy of these policies is most visible in school infrastructure such as school laboratories. The racial inequities of the Apartheid system resulted in an unequal distribution of education resources, with township schools attended by Black learners having scant resources for practical work. In South Africa, the term “township” usually refers to underdeveloped urban areas that, from the late 19th century until the end of apartheid, were set aside for “non-whites” (Chisholm and Sujee, 2006). The lack of physical resources is exacerbated by large classes (Onwu and Stoffels, 2005). Although there have been incremental improvements since the advent of democracy in 1994, these have been insufficient to address the huge backlogs that continue to exist (Reddy et al., 2016). Township schools remain poorly resourced and have scant facilities for practical work in science. Therefore, one of the main challenges in the implementation of experimental chemistry has been the non-availability of physical resources such as apparati and chemicals at historically under-resourced schools (Onwu and Stoffels, 2005; Lelliott, 2014).In an effort to address this deficit in school science, the emphasis has largely been on the provision of new technologies in educational environments, which had thus far been resource-poor (Wallet, 2015). According to Isaacs (2012), “The dominant view seemed to be that Information and Communication Technology (ICT) itself would catalyse the much-needed changes in the education system” (p. 11). As a result, today at historically disadvantaged South African schools there is availability of smartboards, computers, tablets and free-wifi connectivity. In order to adequately prepare teachers to exploit ICTs in their teaching, South African teacher education institutions such as universities have prioritised the development technological pedagogical and content knowledge (TPACK) as a key learning area in the teacher education curricula. TPACK is defined by Koehler et al. (2013), as “the basis of effective teaching with technology requiring an understanding of the representation of concepts using technology” (p. 16). It is important for teachers to have the necessary TPACK to ensure that ICT integration in the classroom is effective. Further to this, Jen et al. (2016), explain that, the TPACK of teachers will allow the teaching of topics to be more understandable to the learners. It is against this background that this research investigated the use of virtual learning simulations in chemistry at a South African university.
Virtual learning simulations
As an alternative to traditional laboratory experimentation, virtual learning simulation laboratories provide a platform where students’ learning can be enhanced through the use of simulations, illustrations and demonstrations in 2-Dimensional (2D) and 3-Dimensional (3D) multi-media formats (Mayer, 2011; Makransky et al., 2017). Virtual reality software have been employed to promote students’ attitudes and conceptual understandings of concepts in surgical medicine, aeronautics and robotics in recent years (Wu et al., 2013; Hsu et al., 2017). These software have also been used extensively in science education to create sophisticated virtual laboratories in which students can learn science by experimentation, visualise abstract concepts and create mental representations which promote long term memory (Weiman et al., 2010; Wu et al., 2013; Hsu et al., 2017). Moore et al. (2013), also found that “implicitly scaffolded interactive simulations” could “provide environments that support guided inquiry learning” (p. 257).Simulations are interactive digital learning environments that imitate a real-life process or situation (Makransky et al., 2017). Virtual simulation laboratories employ the use of software to reproduce and imitate real physical and chemical processes that are investigated in a traditional laboratory. These simulations for chemistry concepts are embedded with 2D and 3D representations of micro-worlds like atoms, electrons, bonds, ions and molecules as is the case with particle chemistry. In low immersion virtual reality, also referred to as desktop simulations such as the PhET simulations, the interactions are not too complex as may be the case in high immersion virtual technologies (Makransky et al., 2017). The PhET Interactive simulations project at the University of Colorado Boulder, include several chemistry simulations, available freely at http://phet.colorado.edu. PhET simulations are supplemented with guidelines that direct students’ inquiry activities towards specific learning outcomes (Creating PhET Activities, 2013; Chamberlain et al., 2014).
Some of the key features of virtual learning environments which promote learning are underpinned by learning theories such as the cognitive and social constructivist theories. Cognitive constructivists postulate that, the stimulation of the mind enhances critical thinking while social constructivists emphasis on social interactions and mediation tools for enhancing learning (Bruner, 1990). All of these paradigms are exploited through learner-centred interactions, collaborative and individual learning activities, multiple-media, simulations and demonstrations that are embedded within the simulation labs (Cai et al., 2014). Students are therefore, able to see, manipulate, interrogate and discuss science concepts during simulation laboratory sessions. These diverse visual representations have been seen to better enhance students’ engagement and interest in learning more complex science concepts than traditional laboratories (Wu et al., 2013; Makransky et al., 2017). In education, virtual learning environments are also fuelled by a rapid increase in the technological advancements which have a low cost effect on global access to teaching and learning resources (Swallow and Olofson, 2017).
Despite the affordances of virtual learning, several factors need to be considered in its implementation for experimental work in chemistry. These include the loss of realism, poor development of science process skills, increased cognitive load and possible misconceptions that may arise from the 2D and 3D virtual representations when compared to the real chemical processes (Makransky et al., 2017). A secondary objective of this research was therefore to explore students’ perceptions of their experiences of learning in a virtual environment.
Attitude towards science
The emotional and mental entities which drive a person's action towards an object or subject are referred to as attitude (Perloff, 2016). It is a complex phenomenon which underscores the affective, cognitive and phsyco-social domains (Van Rensburg et al., 1999; Reddy et al., 2013). For science in particular, attitude can be described as a learned positive or negative feeling about science. It also summarises the way a person believes about science (Salta and Tzougraki, 2004). Factors implicated in students’ attitudes towards science learning include the levels of difficulty, the abstract nature of certain phenomena and the teaching strategies used in class to teach science concepts (Chua and Karpudewan, 2017; Kaur and Zhao, 2017). Statements like “I hate science” or “I don’t want to attend science class today” or “Science is very abstract” are some of the evidences of how students relate to the learning of science in general. These affective expressions tend to reflect how positive or negative a student's attitudes will be towards specific science subjects like chemistry and the related careers (Morrell and Lederman, 1998; Osborne et al., 2003). Teachers who employ traditional rote teaching methods characterised by memorisation, practice and drill, find that students are completely estranged from chemistry learning and tend to show negative attitudes towards chemistry (Chua and Karpudewan, 2017). The need to investigate attitude as a construct in learning is underlined by strong correlations that have been reported between attitude and other constructs like, motivation in science learning, achievement in science and students’ conceptual understandings (Otor and Achor, 2013; Chua and Karpudewan, 2015; Makransky and Petersen, 2019). For instance, students tend to show positive attitudes towards a subject when they find it easy to learn, form an active part of constructing their own knowledge about the subject, can relate to the concepts, and when the learning environment is student-centered (Hofstein and Lunetta, 1982).The study was hence guided by the following research questions:
(1) Is there a significant difference in 3rd year Bachelor of Education students’ attitudes towards chemistry pre and post learning intervention with virtual learning experiences?
(2) How do 3rd year Bachelor of Education students perceive the use of virtual simulations for chemistry learning and teaching?
Research design and methodology
The study followed a sequential explanatory mixed-method design (Creswell and Creswell, 2017). This design enabled the researchers to “collect both quantitative and qualitative data, merge the data, and use the results to best understand a research problem” (Creswell, 2002, p. 564). In the first phase of the study, quantitative data was collected by means of an attitude test that was administered before and after students were engaged in chemistry virtual simulations. Fifty (n = 50) students were randomly selected from a population of 68 3rd year Bachelor of Education (BEd) students majoring in physical sciences education at a South African university. For the qualitative phase of the study, a purposive stratified sampling technique was used to select ten students from high (4 students), medium (3 students) and low (3 students) post-attitude score strata. Two of the students selected from the high score stratum had the same post-attitude score, hence the rationale for 4 students rather than 3 as in the other strata. Semi-structured interviews were conducted individually with each of the participants in this phase to understand their perceptions of stimulated learning of chemistry concepts. Seven questions were posed during the interview sessions as can be seen in Table 5 below. Further elaborations were elicited by the interviewer, where the provided response needed clarity. Samples of the students’ transcripts can be found in Appendices S2 and S3 (ESI†) provided with the paper.Sample description
The participants in this study were 3rd year Bachelor of Education (BEd) Physical Sciences students, 22 (44%) females and 28 (56%) males. In the teacher education programme, BEd physical sciences students take both methodology and pure sciences modules such as chemistry and physics. The pure science modules are not at the same academic level as those taken for a Bachelor of Science (BSc) degree, but are pitched at a lower level. These chemistry and physics modules are focussed to a large extent on topics that are covered in the school curriculum. In terms of funding mechanism, 20% of the BEd 3rd year students were self-sponsored students, while 78% of them were sponsored by the state through the National Student Financial Aid Scheme (NSFAS) and the Funza Lushaka bursary (a national bursary scheme made available to enable students completing a full teaching qualification in a national priority area). Only 1 student (2%) had private sponsorship. At the end of their qualification, a majority of the students who are funded by the state will be deployed as science teachers to poorly resourced schools, where traditional resources for science teaching are limited. It is therefore important they are equipped with the relevant virtual learning resources and techniques that could be useful for their practice in this context.The instrument
Quantitative data were collected by means of the adapted Attitude towards Chemistry (ATC) questionnaire (Appendix S1, ESI†). This instrument was first developed, validated and used with Greek students by Salta and Tzougraki (2004), in assessing grade 11 students’ attitudes towards chemistry. The ATC questionnaire consists of 30 items which assess 4 constructs with regards to the attitudes towards chemistry learning on a 5-point Likert scale. On the Likert scale, item options were ordered from “strongly disagree” to “strongly agree” with “neither disagree nor agree” as the neutral point of the scale. Moving from “strongly disagree” to “strongly agree,” positive items were scored from 1 to 5, respectively, while negative items were scored in the reverse order, making the total attitude score attainable by the participant to range from 30–150, with 30 being the lowest possible attitude score, and 150 being the highest possible score for each participant. The four constructs factored in the 30-item questionnaire are represented on Table 1 below.| Construct | Questionnaire items | Example of items |
|---|---|---|
| The importance of chemistry module | 5, 12, 13, 15 and 20 | Item 12: The progress of chemistry improves the quality of our lives. |
| The difficulty of chemistry module | 2, 7, 17, 18, 24 and 26 | Item 18: I find the use of chemical symbols easy like walk-over. |
| Interest in the chemistry module | 1, 3, 9, 10, 16, 19, 21, 23 and 25 | Item 25: I find the chemistry module very interesting. |
| Usefulness of chemistry for future careers | 14, 22 and 30 | Item 14: My future career is independent from chemistry knowledge. |
As shown on the table above, the attitude test/questionnaire assessed students’ attitudes towards the chemistry module by degree of importance, the difficulty of the module, interest in the chemistry module and the usefulness of chemistry in their future careers. The questionnaire was piloted with a different group of 40 3rd year BSc, to ascertain the readability of items. The Cronbach's alpha for the 4 constructs within the ATC during the pilot study was quite acceptable at 0.7 as the sample size was relatively smaller. Minor modifications to the ATC questionnaire were made, for example, the word “course” was changed to “module” to suit the context of participant students. The adapted ATC was then adopted for the main study, and administered before and after the virtual simulation intervention. The internal reliability between the 4 constructs within the adapted ATC questionnaire for the main study was calculated using SPSS 25 with α = 0.88, which was considered to be acceptable (Pallant, 2010; Taber, 2016).
Data analysis
Quantitative data generated from the pre- and post-test administration of ATC questionnaires were captured and analysed using the Statistical Package for the Social Sciences (SPSS) version 25. Descriptive and parametric statistics were employed to calculate and compare the mean attitude scores for the pre and post-test. A paired sample t-test was used to establish whether the observed mean differences were statistically significant or not. Interview data yielded from the qualitative phase of the study, was transcribed and analysed using computer-aided qualitative data analysis software, Atlas.ti version 8. Data were then coded and classified (Mouton, 2001) through a process guided by the trends and patterns which emerged during qualitative content analysis. Content analysis helps a researcher to identify the key meanings within the data provided and interpret the content of the textual data according to these meanings and patterns generated (Leedy and Ormrod, 2014).The intervention
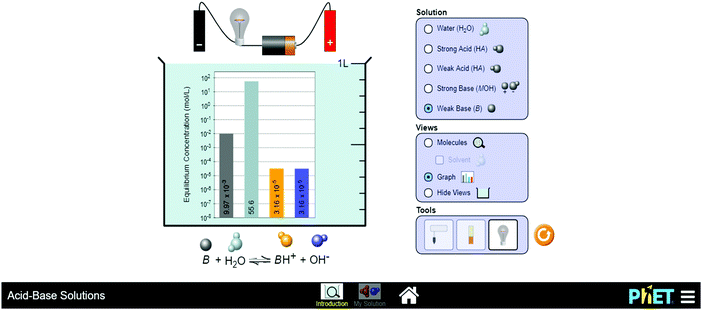
The PhET simulation learning intervention was conducted over a 5 week period. Prior to learning interventions, an online survey was conducted with 3rd year BEd students in order to identify chemistry concepts in their module that they considered as being abstract. 90% and 78% of the students indicated chemical reactions and spectrophotometry were the most abstract concepts respectively. Indeed other studies such as a study by Cakmakci et al. (2006), show that these concepts are difficult due to complex interaction of micro-worlds.The learning intervention was covered during tutorial sessions that followed formal lectures on the chemistry concepts. These tutorials were 1 hour sessions that took place twice a week. Though the participant students were in the third year of their teacher education they are a unique sample in that unlike the students from the pure and applied chemistry their priority modules are the methodology of teaching modules, which are aimed at preparing them for science teaching. All the participant students consented to the study and the university's ethical clearance was obtained. Firstly, all participants were guided in downloading free open online learning PhET simulations to their laptops and had internet access for simulations that were not available off-line. During the learning intervention, a corporative learning approach between the instructor and the students was employed. Structured activities for simulations promoted student to student interactions and student to instructor interactions in order to accomplish self and peer learning goals (Johnson and Johnson, 2009). Students firstly engaged in instructor-directed learning, then self and peer-learning (2 or 3 students per group) of chemical reactions and spectrophotometry concepts which they had identified as abstract and confusing. We directed them on the use and manipulation of the PhET virtual simulations in investigating certain chemical reactions, writing and balancing chemical equations, and measuring absorbance and transmittance through various molar concentrations of solutions. In the first set of learning interventions, the 3rd year students engaged with different aspects of chemical reactions taught to them at the school grade level (9–12) for which they are being trained to teach after the BEd qualification. For example, within the grade 9 South African curriculum for Natural Sciences students are expected to learn and understand chemical equations, reactions of metals and non-metals with oxygen, acid–base reactions, pH values and the applications of chemical reactions (Department of Basic Education [DBE], 2011). At the grade 12 level, students are expected to measure the rate of chemical reactions and the mechanism of reactions and catalyst. In addressing to these curricular expectations, the researchers planned and scaffolded learning activities (some borrowed from the PhETs and some constructed from the module content) during the virtual simulation tutorials. The diagram in Fig. 1 below shows a sample screenshot of one of the PhET simulation laboratories engaged in.
The activities in Fig. 1 required students to manipulate the PhET simulation to establish reactant and resultant products in acid–base reactions, and measure the degree of acidity or alkalinity of the resultants in relation to the electricity generated by free ions. Samples of learning activities could be accessed on https://phet.colorado.edu/en/contributions/view/3660 and also attached in Appendix S4 (ESI†).
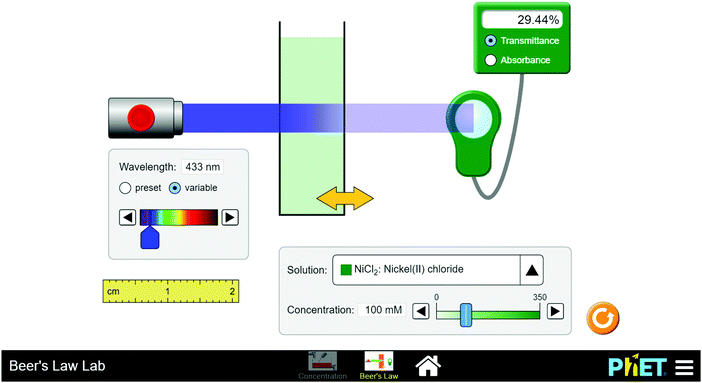
Fig. 2 below also shows a sample of the PhET virtual simulation lab for the investigation of Beer's law.
In learning spectrophotometry and Beer's law which is a topic embedded in the chemistry 3rd year module, students were expected to use the PhET simulations in navigating through simple as well as complex problems during the intervention. For example a simple problem given was as follows;
A solution containing 40 ppm of X had an absorbance of 0.43 in a 1 cm cell at 690 nm. If 5 mL of this solution was diluted with water to 100 mL, what will be the absorbance of the new solution at 690 nm?
In solving this problem, students individually manipulated the cells and the associated concentration of solution X to establish the absorbance of the new solution. In addition to this question, more demanding questions were included, and these required students to solve authentic real life problems associated with the medical applications of the related concepts, for example measuring the concentration of haemoglobin in blood, the bone marrow density of a patient and many others. The students worked on these authentic problem in groups (other questions used during this intervention are included in Appendix S5, ESI†). Also important to be mentioned is the fact that tutorial sessions did not involve the use of direct guidance but rather the students were given the opportunity to explore concepts through the simulations.
Results
Students’ attitudes towards chemistry pre- and post-intervention
Data captured from the pre- and post-attitude tests was analysed through descriptive statistics and parametric testing within SPSS 25, after the normality of the data had been established. Table 2 below shows the means and standard deviations of the pre and post attitude test scores.| Mean | N | Std deviation | Std error mean | |
|---|---|---|---|---|
| Pretest | 58.92 | 50 | 11.65 | 1.65 |
| Posttest | 82.58 | 50 | 6.17 | 0.87 |
Table 2 above shows that the mean post-test attitude score (M = 82.58, S.D. = 6.17) was higher than the pre-test score (M = 58.92, S.D. = 11.65). A further breakdown of the questionnaire items into specific constructs revealed that, post-mean differences for individual constructs as featured in the ATC questionnaire were different. Table 3 shows that, even though the mean attitude scores for the post-test was higher for all four constructs in the ATC questionnaire, the highest post-test mean differences were recorded for the constructs “difficulty of chemistry module” and “interest in the chemistry module”.
| Mean | N | Std deviation | Std error mean | ||
|---|---|---|---|---|---|
| Pair 1 | Difficulty-post | 22.60 | 50 | 2.277 | 0.322 |
| Difficulty-pre | 15.38 | 50 | 3.386 | 0.479 | |
| Pair 2 | Interest-post | 31.66 | 50 | 2.946 | 0.417 |
| Interest-pre | 21.96 | 50 | 4.944 | 0.699 | |
| Pair 3 | Usefulness-post | 11.50 | 50 | 1.705 | 0.241 |
| Usefulness-pre | 8.06 | 50 | 1.900 | 0.269 | |
| Pair 4 | Importance-post | 16.82 | 50 | 2.647 | 0.374 |
| Importance-pre | 13.52 | 50 | 3.466 | 0.490 | |
Table 3 above shows that, the large variation in the post attitude test scores was largely accounted for by 2 constructs, namely “difficulty of the subject” (M = 22.60) and “interest in the subject” (M = 31.66). This indicates that, prior to the virtual learning interventions, participants perceived the chemistry module to be difficult and did not show much interest in it.
After establishing that the data had a normal distribution, a paired sample t-test for pre and post-test scores was conducted. The results are captured on Table 4 below.
| Paired differences | t | df | Sig. (2-tailed) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Std deviation | Std error mean | Lower | Upper | |||||
| Pair | Post-test–pre-test | 23.660 | 8.385 | 1.186 | 21.277 | 26.043 | 19.952 | 49 | 0.000 |
As seen on Table 4, the post-test means were significantly higher than the pre-test means, with students showing a significantly more positive attitudes towards chemistry, t(50) = 19.95, p < 0.01 at 95% confidence interval. These results suggest that BEd 3rd year chemistry students showed an improvement in their attitudes towards chemistry post intervention with virtual learning using PhET simulations.
Despite the results obtained with the attitude test scores, more had to be done in establishing the rationale for the observed changes in attitude scores. We then proceeded to the next phase of the study and conducted semi-structured interviews with 10 of the surveyed students to gain further insights into their learning experiences in the virtual learning environment.
Students’ perceptions of virtual learning experiences
Table 5 below shows some of the generated categories from the process of content analysis performed on the transcribed semi-structured interview textual data. These categories formed the basis for assertions generated and discussed in the section below. The students were labelled S1–S10 as pseudonyms (find attached samples of the transcribed textual data from S1 and S7 in Appendices S2 and S3 (ESI†) respectively).| Semi-structured interview questions | Some quotations | Categories |
|---|---|---|
| How did you find the PhET simulation laboratories and the activities? Provide a brief explanation. | ∘ The simulations were very engaging and fun just to try | There is a game element with the simulations |
| ∘ Felt like I was playing a game | ||
| ∘ I enjoy the simulations as I could do them on my laptop at home | ||
| After the intervention classes, did you need help with the activities associated with the PhET simulation laboratories? Explain your reasoning. | ∘ I did not need anyone to explain the tasks to me | Improved ability to self-teach |
| ∘ I found that I could self-direct my learning of the chemistry concepts and that was the best thing for me | ||
| ∘ There is a sense of accomplishment in knowing that I can understand certain concepts without asking for the lecturer's help | ||
| ∘ I could read and understand the instructions which boosted my ability to single-handedly solve chemistry problems | ||
| What were some of the gains you observed with using PhET simulations? | ∘ I could do tasks without fear of spilling acid solutions or other reactants | Danger factor eliminated |
| ∘ You know with chemistry experiments there is always a danger factor. The simulations eliminated this fear | ||
| ∘ I could repeat task over and over without worrying about finishing the reagents | Trial and error is possible | |
| ∘ I kept practicing | ||
| ∘ With the PhETs the good thing is that if you don’t get it then you do it over and over again | ||
| ∘ I have no fear of making errors with the PhETs. I manipulated the effects of different variables in several of the chemical reactions | Increased confidence in problem solving | |
| ∘ You try different things in the virtual laboratories without fear of spoiling anything | ||
| ∘ The ability to do several different activities increased my confidence to deal with the problems from chemistry class | ||
| ∘ I found that I had more learning material than before, but more interesting from the comfort of my own space | Anytime and anywhere (convenience) | |
| ∘ All you needed was data, I did not have to be on campus to engage with my chemistry experiments | ||
| ∘ There were no time constraints for the laboratory session, since everything is online | ||
| ∘ The simulation laboratories are always available and do not close at the end of the school day like our laboratories at Varsity | ||
| What were some of the disadvantages of using the PhET simulation laboratories? | ∘ The simulations create some misconceptions | Misconceptions |
| ∘ Misconceptions can be promoted and one needs to be careful when using simulations | Loss of realism | |
| ∘ I find the PhETs can be addictive | All curriculum learning outcomes are not covered | |
| ∘ I could stay on the computer and forget the time | Lack authenticity | |
| ∘ I am not sure if the PhETs cover our specific South African curriculum (CAPS) needs | ||
| ∘ I don’t think learners can learn science process skills by using these PhETS | ||
| ∘ The PhETs are not real, they are just so fake and you can’t touch anything or smell the fumes of any chemicals | ||
| ∘ They are all in a machine which is just…not tangible when I think of it | ||
| Did the PhET simulations improve your conceptual understandings of the learned chemistry concepts in any way? | ∘ Yes very much, because I could try different experiments that enabled me to critically analyse some of the concepts in spectrophotometry | Improvement of conceptual understandings |
| ∘ For me yes because it was easy to do different difficulty level activities over and over until they I understood | ||
| In your practice as an aspiring chemistry teacher will you consider incorporating PhET simulations when teaching certain abstract concepts? | ∘ Definitely yes | Open to try PhETs in teaching chemistry concepts |
| ∘ It is a yes for me since I find that these will capture my learners’ attention | ||
| ∘ I will most certainly incorporate them in all lessons were they can be beneficial | ||
| ∘ I may not use them for certain concepts as the learning outcomes may be missed | ||
| ∘ It is an excellent tool for complementing chemistry learning. So I will actually blend the PhET sims in my teaching | ||
| ∘ They are the perfect tool for a blended learning approach | ||
| Do you have any advice on how simulations can be improved to meet learning needs for South African classrooms? | ∘ Yes, I believe that if we can make these simulations context-specific our students will really benefit more | Teachers and students should be equipped to design curriculum specific simulations |
| ∘ I think the simulations should target specific learning outcomes for the module or subject | ||
| ∘ Opportunity should be given to teachers and learners to formulate their own simulations to cater for diverse learning needs | ||
Based on content analysis of interview data, the following assertions were made by the researchers.
Assertion 1: Students perceive virtual reality simulations as fun and enjoyable
During follow-up interviews, the first question asked required that the students elaborate on their overall experience of virtual learning using simulations. All ten students indicated that learning using simulations was fun and it had a ‘game’ feel to it. For instance, student S1 stated “I really enjoyed investigating my chemistry concepts with the simulations. It felt like I was playing a game with multiple levels of difficulty”. Student S2 added “the simulations were very engaging and fun just to try”. For this assertion, 8 out of the 10 participant students indicated that simulations were fun while the remaining 2 interviewees indicated that, the simulations had a ‘game’ feel to them. These and other responses revealed that the 3rd year student associated a feeling of enjoyment and fun in using the simulations for their learning.
Assertion 2: Students assumed more autonomy for their learning when using the virtual reality simulations
When asked to explain if they needed help with running the simulations, one student S3 indicated, “I found the instructions for engaging with the simulations so simple that I could carry out the tasks without asking for help”. S4 maintained that “I could read and understand the instructions which boosted my ability to single-handedly solve chemistry problems.” While S6 also said that, “I did not need anyone to explain the tasks to me as I always did in the chem lab at Varsity”. In total, 7 out of the 10 participants indicated that their autonomy in solving chemistry problems had improved, while the remaining 3 indicated that they had always needed little or no assistance when solving problems in chemistry prior to this intervention. We then concluded that a strong sense of autonomy was seen to be one of the benefits yielded from using the virtual environment for learning chemistry concepts.
Assertion 3: Students showed increased confidence in chemistry investigations
When asked about the possible gains that came out of the virtual learning experiences, student S9 indicated that “I have no fear of making errors with the simulations as I usually do when I go to the lab”. S8 also added that “I could do a task without fear of spilling acid solutions or other reactants. You know with chemistry experiments there is always a danger factor.” A related sub-assertion is the convenience students associated with using these simulations. All the 10 participants indicated that using the simulations were more convenient for supporting their learning because they could access the experiments and activities anytime from anywhere. This convenience enable them to practice tasks, leading to increased confidence in their ability to solve chemistry problems and carry out chemistry investigations in a real chemistry laboratory. In other words words, students used virtual experimentation as a platform for trial and error, in preparing for traditional lab sessions.
Assertion 4: Student perceive virtual simulations to significantly improve conceptual understanding of abstract chemistry concepts
When asked if virtual learning activities improved conceptual understandings, all participants indicated that there was a significant improvement in their conceptual understanding of all aspects of chemical reaction and spectrophotometry. This assertion could explain the high attitude score for the construct “difficulty of the chemistry module” in the ATC post-test. Students indicated that concepts they previously found very difficult were now comprehensible. When probed on the qualities of virtual learning which enhanced their understandings, S2 indicated that, “I could visualise some of the abstract phenomena, especially with absorbance and transmittance.” S10 also said, “For me I find that it was the learning activities that enhanced my understandings coupled with the 2D representations of reactants and products in chemical reactions. Also, Activities had different levels of difficulty which kept me motivated to get to the next level.” In total 8 students indicated that their conceptual understandings were significantly improved, while the remaining two showed indifference
Assertion 5: Students perceived that simulations misrepresented chemistry bonding structures
When asked of the possible disadvantages experienced in this learning environment, student S7 indicated that “The PhET simulation 2D bonding structures could create misconceptions in the way they are represented.” when asked to elaborate on this, he stated that “the rigidity of the represented bonds in compounds and molecules within the stimulations hindered one‘s ability to visualise inter and intra-molecular bond interactions in a chemical reaction between two reactant. I feel generally representation of chemical interactions is problematic”. This elaboration was noted as many of the misconceptions that arise in chemistry stem from inadequate molecular level models used either in real or virtual laboratories. Student S10 indicated that “when balancing the chemical equations the number “1” is also placed in front of a single molar entity which is not the conventional way of representation when balancing chemical equations. But I think it is our place as educators to discuss these misconceptions when using the PhET simulations as an instructional tool”. Despite the fact that for this assertion, only 4 out of the 10 participants in this phase, referred to some misconceptions that could arise when using simulations, we considered it important to report their perceptions. This is because misconceptions can be very difficult to correct once created. Based on this assertion we concluded that, when using simulations as a teaching and learning tool, one has to be aware of possible misconceptions that can arise as a result of molecular level representations within the simulations and make these explicit to the learners upfront, before they engage in tasks.
Assertion 6: Students found that virtual experimentation lacked authenticity
With one of the goals of science learning being the ability to develop science process skills, all 10 students indicated that, virtual learning environments did not support the enhancement of skills such as measuring precise molar quantities of reactants and products, accurately titrating a base against an acid, or applying the use of one's senses (touch and smell). Student S3 said “I don’t think learners can learn science process skills by using these PhETs.” S6 also said “The PhETs are not real, they are just so fake and you can’t touch anything or smell the fumes of any chemicals.” S5 added that “they are all in a machine which is just…not tangible when I think of it”. The students further indicated there is an inherent loss of realism associated with virtual learning, and that this may misrepresent the nature of science to learners.
The integration of findings from the quantitative and qualitative phases of this study suggest that students had a favourable experience of learning chemistry in a virtual environment, and this resulted in a positive shift in their attitude towards chemistry. However, the students relayed that some caution needs to be exercised with simulations as they can lead to misconceptions and may not promote authentic learning. These findings are now discussed.
Discussion
The findings from this study reveal that, virtual laboratory-learning interventions have a positive effect on 3rd year BEd chemistry students’ attitudes towards chemistry. Other factors like the nature of instruction and student–instructor interactions that might have affected participant's attitudes were not assessed for this study. Reverting to literature, the findings are similar in many ways to those from other recent studies that investigated the effects of virtual laboratory learning interventions (e.g., Merchant et al., 2014; Estapa and Nadolny, 2015; Chua and Karpudewan, 2017; Hsu et al., 2017). In these studies virtual laboratory learning (immersive or non-immersive virtual reality and augmented reality), were reported to have a positive impact on students’ attitudes and motivation towards science learning in general. Aravind and Heard (2010), also reported that the use of virtual laboratories simplified complex physics concepts and changed student's negative perceptions of the physics course. Similarly, Tüysüz (2010) found that, students who were comfortable in using virtual laboratories, showed a more positive attitude towards learning chemistry concepts. Contrary to these findings, Faour and Ayoubi (2018) reported no attitude differences in a study in which they assessed grade 10 student's attitudes towards physics post intervention with virtual laboratory.From the semi-structured interview phase, pre-service teachers indicated that they derived several benefits from using the virtual simulation laboratories for chemistry learning. Gains included, access to simulation from anywhere anytime, increased confidence in handling chemistry problems, a feeling of enjoyment and gaming while learning, more autonomy in doing learning tasks and the elimination of the fear factor associated with the dangers of chemistry experiments. These perceptions validate the theoretical underpinnings of this study in that learning can be fostered where there is a multiplicity of content representation (Mayer, 2011). Furthermore, the chemistry students indicated that, improved conceptual understandings was among some of the gains from using the simulations provided by PhET. These gains in conceptual understandings were associated with the opportunity to visualize concepts or phenomena, several opportunities for trial and error, and embedded learning activities with different levels of difficulty, which enhanced self-assessment. On the other hand, the students raised concerns that the simulations lacked authenticity and did not depict the realism associated with doing experiments in the laboratory. Similar to the findings in this phase, Hsu et al. (2017) reported that, students did not connect with the authenticity of virtual laboratory spaces.
Limitations
In this study one of the main caveats lie in the fact that selection of an exclusive control group in an experimental design was not possible as this is daunting for education studies (Schanzenbach, 2012), where all student must be taught in the same way. Significant differences in the attitudes towards chemistry indicate that other factors which were not investigated in this study might have had an effect on attitude changes. The particular chemistry concepts selected were based on a specific chemistry module which excludes other topics that might have been covered in organic chemistry and physics. However, the primary aim of the intervention was to ensure that students are introduced to virtual learning and also understand how to use it in their own teaching of chemistry concepts, which had not been the case prior to this intervention. We also acknowledge that other factors including the method of instruction, time for laboratory sessions and the learning environments might have influenced the students’ negative attitudes before the intervention.Conclusion
The implications, which emanated from the findings of this study, are directly linked to the positive impact of virtual learning environments on students’ attitudes towards chemistry. Virtual laboratories provided a platform for the participants to experiment with and visualise abstract chemistry concepts. Even though virtual laboratories cannot replace real chemistry laboratories, they are capable of complimenting them when learning abstract chemistry concepts and where resources are limited as is the case with several under-resourced South African high schools. Furthermore, benefits like autonomous learning, lab safety, lower cost of equipment and chemicals and catering for individual learning differences suggest that virtual simulations could play a significant role in enhancing the quality of science learning experiences. There is therefore a need for investing in further development of simulation software that would accommodate a broad spectrum of chemistry topics and cater for curriculum-specific desired learning outcomes.The current study reports the findings of a study with a relatively small sample size and a low predictive power of factors that could possibly affected attitude scores. Based on these short comings, we cannot make any generalisations from the findings. Added to this is the fact that only a few chemistry concepts were exploited for the study and therefore we cannot assume that the experiences will be the same for all science subjects and concepts. We therefore, recommend that larger scale research be conducted to assess student attitudes towards virtual learning spaces in science teaching and learning. Pedagogical approaches with virtual simulations also merit further attention. Scientific inquiry has been advocated as a common curriculum goal in school science education in South Africa. Future research could investigate the affordances of virtual simulations in supporting inquiry-based learning.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
We sincerely appreciate the Physics education research team of the University of Colorado for providing an open virtual learning platform with PhET Interactive Simulations University of Colorado Boulder https://phet.colorado.edu, for all learners globally.References
- Aravind V. R. and Heard J. W., (2010), Physics by simulation: teaching circular motion using applets, Latin Am. J. Phys. Educ., 4(1), 35–39, retrieved from http://www.journal.lapen.org.mx.
- Bruner J. S., (1990), Acts of meaning, Cambridge, MA: Harvard University Press.
- Cai S., Wang X. and Chiang F.-K., (2014), A case study of Augmented Reality simulation system application, Comput. Hum. Behav., 37, 31–40, DOI:10.1016/j.chb.2014.04.018.
- Cakmakci G., Leach J. and Donnelly J., (2006), Students’ ideas about reaction rate and its relationship with concentration or pressure, Int. J. Sci. Educ., 28(15), 1795–1815, DOI:10.1080/09500690600823490.
- Can H. B. and Boz Y., (2012), A cross-age study on high school students’ attitudes toward chemistry, International Journal on New Trends in Education and Their Implications, 3(3), 82–89, retrieved from http://www.ijonte.org/FileUpload/ks63207/File/08.can1.pdf.
- Chamberlain J. M., Lancaster K., Parson R. and Perkins K. K., (2014), How guidance affects student engagement with an interactive simulation, Chem. Educ. Res. Pract., 15(4), 628–638, 10.1039/c4rp00009a.
- Chisholm L. and Sujee M., (2006), Tracking racial desegregation in South African schools, J. Educ., 40, 141–159, retrieved from http://hdl.handle.net/20.500.11910/6479.
- Chiu J. L., DeJaegher C. J. and Chao J., (2015), The effects of augmented virtual science laboratories on middle school students' understanding of gas properties, Comput. Educ., 85, 59–73, DOI:10.1016/j.compedu.2015.02.007.
- Chua K. H. and Karpudewan M., (2015), The interaction effects of gender and grade level on secondary school students’ attitude towards learning chemistry, Eurasia J. Math., Sci. Technol. Educ., 10(5), 219–227.
- Chua K. H. and Karpudewan M., (2017), The role of motivation and perceptions about science laboratory environment on lower secondary students’ attitude towards science, Asia-Pacific Forum on Science Learning and Teaching, 18(2), retrieved from https://www.eduhk.hk/apfslt/download/v18_issue2_files/karpudewan.pdf.
- Creating PhET Activities Using Guiding Inquiry Strategies. Creating PhET Activities Using Guiding Inquiry Strategies. PhET Interactive Simulations, 25 Apr. 2008. Web. 24 Oct. 2013, http://phet.colorado.edu/en/for-teachers/activity-guide.
- Creswell J. W., (2002), Educational research: Planning, conducting, and evaluating quantitative and qualitative research, Upper Saddle River, NJ: Merrill Prentice Hall.
- Creswell J. W. and Creswell J. D., (2017), Research design: qualitative, quantitative and mixed methods, Thousand Oaks, CA: Sage, 5th edn.
- Department of Basic Education, (2011), Curriculum and assessment policy statement: grades 7–9 Natural Sciences, Pretoria: Government Printer.
- Esquembre F., (2015), Facilitating the Creation of Virtual and Remote Laboratories for Science and Engineering Education, IFAC-Papers Online, 48–29, 049.
- Estapa A. and Nadolny L., (2015), The Effect of an Augmented Reality Enhanced Mathematics Lesson on Student Achievement and Motivation, J. STEM Educ., 16(3), retrieved from https://www.learntechlib.org/p/151963/.
- Faour M. A. and Ayoubi Z., (2018), The effect of using virtual laboratory on grade 10 students’ conceptual understanding and their attitudes towards physics, J. Educ. Sci., Environ. Health, 4(1), 54–68, DOI:10.21891/jeseh.387482.
- Harle M. and Towns M., (2011), A review of spatial ability literature, its connection to chemistry and implications for instruction, J. Chem. Educ., 88(3), 351–360, DOI:10.1021/ed900003n.
- Hassan O. E., (1985), An investigation into factors affecting attitude towards science of secondary school students in Jordan, Sci. Educ., 69(1), 3–18.
- Hattingh A., Aldous C. and Rogan J. M., (2007), Some factors influencing the quality of practical work in science classrooms, Afr. J. Res. Sci. Math. Technol. Educ., 11(1), 75–90, DOI:10.1080/10288457.2007.10740613.
- Hofstein A. and Lunetta V. N., (1982), The Role of the Laboratory in Science Teaching: Neglected Aspects of Research, Rev. Educ. Res., 52(2), 201–217, DOI:10.2307/1170311.
- Hofstein A. and Mamlok-Naaman R., (2011), High-school students’ attitudes toward and interest in learning chemistry, Educ. Quím., 22(2), 90–102, DOI:10.12691/education-5-2-6.
- Hsu Y. S., Lin Y.-H. and Yang B., (2017), Impact of augmented reality lessons on students’ STEM interest, Res. Pract. Technol. Enhanced Learn., 12(2), 1–14, DOI:10.1186/s41039-016-0039-z.
- Isaacs S., (2012), Mobile learning for teachers in Africa and the Middle East: Exploring the potential of mobile technologies to support teachers and improve practice, Paris: UNESCO.
- Jen T.-H., Yeh Y.-F., Hsu Y.-S., Wu H.-K. and Chen K.-M., (2016), Science teachers’ TPACK-Practical: Standard-setting using an evidence-based approach, Comput. Educ., 95, 45–62, DOI:10.1016/j.compedu.2015.12.009.
- Johnson D. W. and Johnson F., (2009), Joining together: group theory and group skills, Boston: Allyn and Bacon, 10th edn.
- Kaur D. and Zhao Y., (2017), Development of Physics Attitude Scale (PAS): An Instrument to Measure Students’ Attitudes toward Physics (Article), Asia-Pacific Educ. Res., 26(5), 291–304, DOI:10.1007/s40299-017-0349-y.
- Koehler M. J., Mishra P. and Cain W., (2013), What is Technological Pedagogical Content Knowledge (TPACK)? Journal of Education, 193(3), 13–19, retrieved from https://www.learntechlib.org/p/159628/.
- Leedy P. D. and Ormrod J. E., (2014), Practical Research: Planning and Design, New York: Pearson, 10th edn.
- Lelliott A. D., (2014), Scientific Literacy and the South African School Curriculum, African J. Res. Math., Sci. Technol. Educ., 18(3), 311–323, DOI:10.1080/10288457.2014.967935.
- Makransky G., Terkildsena T. S. and Mayer R. E., (2017), Adding immersive virtual reality to a science lab simulation causes more presence but less learning, Learning and Instruction, 60, 225–236, DOI:10.1016/j.learninstruc.2017.12.007.
- Makransky G. and Petersen G. B., (2019), Investigating the process of learning with desktop virtual reality: a structural equation modeling approach, Comput. Educ., 134, 15–30, DOI:10.1016/j.compedu.2019.02.002.
- Mayer R. E., (2011), Instruction based on visualizations, in Mayer R. E. and Alexander P. A. (ed.), Handbook of Research on Learning and Instruction, New York: Routledge.
- Merchant Z., Goetz E. T., Cifuentes L., Kenney-Kennicutt W. and Davis T. J., (2014), Effectiveness of virtual reality-based instruction on students' learning outcomes in K-12 and higher education, Comput. Educ., 70, 29–40, DOI:10.1016/j.compedu.2013.07.033.
- Moore E. B., Herzog T. A. and Perkins K. K., (2013), Interactive simulations as implicit support for guided-inquiry, Chem. Educ. Res. Pract., 14(3), 257–268, 10.1039/c3rp20157k.
- Morrell P. D. and Lederman N. G., (1998), Students’ attitudes toward school and classroom science: are they independent phenomena? Sch. Sci. Math., 98(2), 76–84.
- Mosotho J. G. and Mamontsi K., (2014), Exploration of the Potential of using a Virtual Laboratory for Chemistry Teaching at Secondary School Level in Lesotho, S. Afr. J. Chem., 67, 113–117, retrieved from http://journals.sabinet.co.za/sajchem/.
- Mouton J., (2001), How to succeed in your Master's and Doctoral studies, A South African Guide and Resource Book, Pretoria.
- Onwu G. and Stoffels N., (2005), Instructional functions in large, under-resourced science classes: perspectives of South African teachers, Perspect. Educ., 23(3), 79–91.
- Osborne J., Simon S. and Collins S., (2003), Attitudes towards science: a review of the literature and its implications, Int. J. Sci. Educ., 25(9), 1049–1079, DOI:10.1080/0950069032000032199.
- Otor E. E. and Achor E. E., (2013), Effect of concept mapping strategy on students' attitude in difficult chemistry concepts, Eur. J. Educ. Sci., 1(3), 116–124.
- Pallant J., (2010), SPSS survival manual: a step by step guide to data analysis using SPSS, Maidenhead: Open University Press/McGraw-Hill.
- Perloff R. M., (2016), The Dynamics of Persuasion: Communication and Attitudes in the Twenty-First Century, Routledge: London.
- Ramnarain U. and Ramaila S., (2018), The relationship between chemistry self-efficacy of South African first year university students and their academic performance, Chem. Educ. Res. Pract., 19(1), 60–67, 10.1039/C7RP00110J.
- Reddy V., Gastrow M., Juan A. and Roberts B., (2013), Public attitudes to science in South Africa, S. Afr. J. Sci., 109(1/2), 1–8, DOI:10.1590/sajs.2013/1200.
- Reddy V., Visser M., Winnaar L., Arends F., Juan A. and Prinsloo C. H., (2016), TIMSS 2015: Highlights of Mathematics and Science Achievement of Grade 9 South African Learners, Human Sciences Research Council.
- Salta K. and Tzougraki C., (2004), Attitudes toward Chemistry among 11th Grade Students in High Schools in Greece, Wiley InterScience (http://www.interscience.wiley.com), DOI:10.1002/sce.10134.
- Schanzenbach D. W., (2012), Limitations of Experiments in Education Research, Educ. Finance and Policy, 7(2), 219–232, DOI:10.1162/EDFP_a_00063.
- Swallow M. J. C. and Olofson M. W., (2017), Contextual Understandings in the TPACK Framework, J. Res. Technol. Educ., 49(3–4), 228–244, DOI:10.1080/15391523.2017.1347537.
- Taber K. S., (2016), The Use of Cronbach's Alpha when developing and reporting research instruments in Science Education, Res. Sci. Educ., DOI:10.1007/s11165-016-9602-2.
- Tüysüz C., (2010), The effect of the virtual laboratory on students’ achievement and attitude in chemistry, Int. Online J. Educ. Sci., 2(1), 37–53, DOI:10.4236/ce.2013.410A002.
- Van Rensburg S., Ankiewicz P. and Myburgh C., (1999), Assessing South African learners’ attitudes towards Technology using the PATT (Pupils’ Attitude owards Technology) questionnaire, Int. J. Technol. Des. Educ., 9, 137–151.
- Wallet P., (2015), Information and Communication Technology (ICT) in Education in sub-Saharan Africa: a comparative analysis of basic e-readiness in schools, Quebec: UIS, DOI:10.15220/978-92-9189-178-8-en.
- Weiman C. E., Adams W., Loeblein P. and Perkins K. K., (2010), Teaching Physics Using PhET Simulations, Phys. Teach., 48(4), 225–227, DOI:10.1119/1.3361987.
- Wu H. K., Lee S. W. Y., Chang H. Y. and Liang J. C., (2013), Status, opportunities and challenges of augmented reality in education, Comput. Educ., 62, 41–49, DOI:10.1016/j.compedu.2012.10.024.
- Xu X., Villafane S. M. and Lewis J. E., (2013), College students’ attitudes toward chemistry, conceptual knowledge and achievement: structural equation model analysis, Chem. Educ. Res. Pract., 14(2), 188–200, 10.1039/C3RP20170H.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c9rp00014c |
| This journal is © The Royal Society of Chemistry 2019 |