Teaching energy in living systems to a blind student in an inclusive classroom environment†
Dilek
Teke‡
 and
Mustafa
Sozbilir
and
Mustafa
Sozbilir
 *
*
Department of Mathematics and Science Education (Chemistry Education Division), Kazım Karabekir Education Faculty, Atatürk University, 25240-Erzurum, Turkey. E-mail: sozbilir@atauni.edu.tr
First published on 19th August 2019
Abstract
This study aimed to identify the needs of a 10th grade congenitally blind student in an inclusive chemistry classroom and design and develop tactile materials to teach the ‘energy in living systems’ topic with particular emphasis on covering the symbolic language of the chemistry. A single case study design was used to carry out an in-depth and thorough investigation. The student's needs were identified through classroom observations and interviews. Based on the needs identified, 2D and 3D tactile instructional materials were designed and developed to cover the topic. Interview and observation data suggested that following the instructions the student developed an understanding of the concepts regarding the energy in living systems together with the symbolic representations used in chemistry. He showed an understanding of molecular structures when he was asked to recognize the 3D models of the molecular structures. He was able to recognize all of the structures and could respond to questions regarding the parts of these molecular structures. Although the results are limited to one congenitally blind student, there is evidence that a blind student can learn complex molecular structures if he/she was provided with materials which suit his/her needs. The results also suggest evidence of how to produce low cost instructional materials for a blind student in chemistry.
Introduction
Educational settings need to be adapted to the needs of students with impairments. There are two types of approaches in the education of students with special needs. One way is to separate students with special needs from their peers and educate them in special education classes and the second approach is providing education in inclusive classroom environments. In recent years, inclusion classes, where students with special needs are integrated into general education classes, have notably become prevalent (Batu et al., 2004).Inclusive education refers to special education practices based on the principle of providing education to individuals with special educational needs along with their peers in mainstream schools or through extra-curricular activities provided through non-formal education with additional educational support (Pavri and Luftig, 2000; Ministry of National Education [MoNE], 2015). These practices are possible for individuals with additional needs of all kinds at all levels (Cook and Semmel, 1999; Kırcaali-İftar and Uysal, 1999). The idea of inclusion assumes that every student has equitable access to education and that schools organize learning spaces that accommodate everyone (Gudjonsdottir and Oskarsdottir, 2016). Inclusion is an on-going process, a never ending quest, aiming at increased participation in education for everyone involved. Moreover, inclusion is fundamentally grounded in ideologies of social justice, democracy, human rights and full participation of all (Gudjonsdottir and Oskarsdottir, 2016, pp. 8–9).
On the other hand, integrated education is the process of responding to the barriers in the learning environments that increase participation in learning and reduce discrimination in education. In practice, integrated education refers to a wide range of strategies, activities and processes aimed at achieving access to education, which is the right of everyone. While integration is closely related to the right to education of children with disabilities, it is also the right to education for all children who are disadvantaged for different reasons (Düşkün, 2016).
The most important feature distinguishing inclusive education from integrated education is that the former is directly related to students with disabilities (Ulutaşdemir, 2007). While integrated education adapts to students (substitutive reaction), inclusive education adapts the lesson to relevant differences between students (active reaction) (Markic and Abels, 2016, p. 3).
Chemistry education for students with impairments
Chemistry knowledge is an important part of daily life for some people including those who are blind or have low-vision (BLV). Although efforts towards teaching symbolic representations in chemistry together with studies focussing on conceptual understanding have recently been directed towards addressing barriers and making chemistry more accessible for these students, there is still a lack of research. Most of the studies (i.e., Tombaugh, 1981; Supalo, 2005; Supalo et al., 2009; Boyd-Kimball, 2012; Supalo and Kennedy, 2014; Supalo et al., 2014; Grumman and Carroll, 2019) have focussed on having accessible laboratory environments, adaptation of available technology to make chemistry more accessible and conceptual understanding to teach symbolic representations in chemistry.Research focussing on access to chemistry laboratories for BLV students
Some of the studies (i.e., Flair and Setzer, 1990; Supalo et al., 2009) aimed at acquiring experimental laboratory skills in chemistry. Concerning the safety in laboratories as well as participation in experiments, it is important to have access to the labels on chemicals. In this context, Tombaugh (1981) developed a material that helps students with visual impairment to read labels consisting of the Braille alphabet so that they can work independently in chemistry laboratories. Besides, she also introduced materials to help blind students to assemble molecular models and speaking instruments to take measures and carry out an experiment independently. Moreover, with the purpose of making chemistry laboratories more accessible, Flair and Setzer (1990) designed a simple titration experiment focussing on the use of four different odours as olfactory indicators.Most of the research in the last decade has turned into adapting technology to make laboratories more accessible. Supalo et al. (2008) described available technologies to make chemistry laboratories more accessible such as carrying out volumetric measurements, use of light sensors for identifying colour changes, and balances for mass determination. In addition, Supalo et al. (2009) introduced a summer camp activity with the help of Independent Laboratory Access for the Blind (ILAB), an example of how technology adapted into chemistry laboratories, to let BLV students carry out chemistry and physics experiments independently or as a group. ILAB tools consisted of new combinations of software and hardware, specially designed tools, and pre-existing devices, both off-the-shelf and modified. The activities covered the content including the properties of gases, energy conversion and conservation, synthesis of biodiesel from vegetable oil, condensation reactions and the formation of an ester. Students were all able to participate in the activities with the help of accessible tools developed and showed enthusiasm to participate in post-experiment group discussions. The results also demonstrated increased enthusiasm towards STEM-related professions among the BLV students. In another study Supalo et al. (2014) introduced a STEM summer camp activity for BLV students. During the camp key stakeholders such as the science faculty, teachers of the visually impaired (TVI), BLV students, parents and legal guardians worked together to implement a plan for having a successful hands-on science learning experience. Students performed different chemistry activities with the help of adaptive technologies as described in the study, and a survey was administered to collect data about students’ interest in science and the possible impact of the activities on students’ future career choices towards STEM fields in college. The results indicated greater interest in science as well as active participation in the experiments with the help of technologies adapted to the needs of BLV students.
Research focussing on instructional aids to make chemistry accessible for BLV students
Due to the barriers in learning spaces of schools, BLV students fear from science and mathematics and therefore develop low interest (Maguvhe, 2015). Barriers to accessing science are grouped under three categories. They are: (i) inadequate preparations of teachers, (ii) limited access to facilities and equipment, and (iii) acceptance by educators (Harshman et al., 2013). Accommodating BLV students in science has been studied for a long time. However, most of the research to date has addressed broad accommodation (Harshman et al., 2013) rather than developing or adapting instructional materials or methods for specific chemistry topics and testing them in terms of whether they meet the unique challenges of BLV students in chemistry or not.Harshman et al. (2013) reported a case study to identify instructional techniques that support or impede the representation of information for BLV students in the context of a typical gas laws unit in a college chemistry course for science majors. This study focussed on the gas laws of chemistry to identify specific challenges and potential solutions. Findings indicated the problems associated with three communication modalities of interest: verbal, written, and tactile representations of information to aid learning. Based on the findings of this study and many other studies (i.e.Supalo, 2005, 2016; Boyd-Kimball, 2012; Wedler et al., 2012; Melaku et al., 2016; Grumman and Carrol, 2019) it can be said that proper accommodation requires the use of tactile and audible materials to make chemistry accessible. Smothers and Goldston (2010) taught four basic chemistry concepts such as dissolution, chemical change, expansion and condensation. On the other hand, Lewis and Bodner (2013) used embossed tactile materials supported with Braille printing to teach the periodic table to visually impaired students. Besides, Supalo and Kennedy (2014) worked on teaching organic chemistry to blind students with tactile materials. Other recent studies focussing on teaching basic chemistry concepts with the help of activities based on the use of tactile materials include those conducted by Kızılaslan (2016), who focussed on teaching the states of matter, and Zorluoğlu (2017), who focused on the particulate nature of matter. Common findings of all of these studies are that the use of tactile materials developed with embossed drawings supplemented with Braille typing enhances learning in chemistry for BLV students. Moreover, Wedler et al. (2012) utilized tactile drawings, molecular model kits, existing software, Bash and Perl scripts written in-house, and three-dimensional printing in a process that allows a blind or visually impaired student to learn structure–function relationships.
BLV students also benefit from the use of audible materials in learning science. For instance, the study by Vitoriano et al. (2016) described the development of a digital sound thermometer for students with visual impairment to enhance inclusive chemistry teaching for the concept of temperature. In another study, Gupta and Singh (1998) designed an audible electronic thermometer and calorimeter to help students with visual impairment to understand temperature measurements and heat changes more easily. In addition, audible sensors were added to the designed material. Moreover, Boyd-Kimball (2012) aimed to help students with visual impairments in writing chemical reactions, to calculate unit conversions and concentrations, to draw Lewis's point structures, to understand structural symbols of molecules with three-dimensional models, and to design and develop topics related to functional groups in organic chemistry voice and tactile materials. All these studies provided evidence for the possibility of access to chemistry content which had been seen rather abstract for students with blindness.
In chemistry, it is common to use symbols such as letters for elements, and drawings to represent cases or processes in chemical events to make them common for all people working in chemistry regardless of any particular language. This is often called symbolic representation. However, it might be challenging for students to acquire and use it properly and it may be found inaccessible by BLV students. For instance, Micklos Lewis and Bodner (2013) examined the understanding of chemical equations developed by three students with blindness by an interview study. Their results suggested that BLV students had views of the symbolic representations of chemical reactions that differed from those of practicing chemists. In addition, Boyd-Kimball (2012) developed materials for students with blindness to help them understand chemical reactions and to calculate concentrations, and Lewis dot structures. Moreover, Hospital (2016) developed a material and a handbook about the elements and atomic numbers of the elements in the table using the Braille alphabet to learn the periodic table for visually impaired students. Finally, Fantin, Sutton et al. (2016) created a free and accessible electronic periodic table for blind and low vision students to understand the symbols in chemistry more freely.
The rationale of the study
Given the relevant literature, there are studies on teaching chemistry to BLV students and these studies focused on accessibility in a laboratory, developing and/or adapting accessible instructional materials with the help of technology. Although there are studies that investigated the effectiveness of material development and/or adaptation in teaching science and particularly chemistry elsewhere, there is a lack of research in Turkey regarding teaching chemistry to BLV students (i.e., Sözbilir, 2016; Kizilaslan et al., 2019; Okcu and Sozbilir, 2019). On the other hand, a majority of the instructional materials and technologies are universal and could easily be adapted to classroom settings regardless of language; it is not always easy to use in different contexts due to either teachers’ lack of readiness or accessibility of technology and/or materials in different languages. It is also known that individual needs may be quite different in different cases. Therefore, it is important to study further by focussing on identifying students' special needs in learning chemistry at high school level and developing or adapting materials with the local circumstances. For instance, designing and printing molecular structures in 3D is a recent technology (Grumman and Carroll, 2019), and is still not available everywhere although it is developing very rapidly. Therefore, we aimed to show that even under very difficult conditions it is possible to develop 3D tactile molecular models to teach complex chemical structures. Moreover, it was hypostasized that integrating tactile materials into the instructional process will enhance students’ access to the symbolic representations and lead to better learning of the macro and sub-micro aspects of chemistry. This would entail both equal accesses to the chemistry content for students with blindness and help to learn chemistry from the aspects of macro, sub-micro and symbolic representations.This study was done with the intention that the materials designed and developed would contribute to the understanding of the smallest building blocks of foods and the chemical events covered in the ‘energy in living systems’ topic in 10th grade chemistry for a student with blindness. This study was based on the identification of the needs of the student in an inclusive classroom environment in chemistry and designing and developing tactile materials to teach the topic together with covering the symbolic representations used. The research questions that guided the study are:
• What are the individual learning needs of a 10th grade congenitally blind student in teaching the topic ‘energy in living systems’ in terms of the symbolic representations used?
• What should be the characteristics of instructional materials to be designed by considering individual learning needs of a 10th grade congenitally blind student in teaching the topic ‘energy in living systems’ in terms of the symbolic representations used?
• Do the newly developed materials facilitate learning the topic?
Research design
This study was designed taking into consideration the situations, context, and needs of a single student with congenital blindness. Therefore, a single case study design (Yin, 2014) was utilized. In a single case study, an average case and an extreme case were investigated in detail. By investigating such a case in-depth, a detailed and elaborate description of the phenomenon was reached (Bleijenbergh, 2010, p. 61). At this point, the study was carried out descriptively. At all stages, ethical guidelines outlined by the Atatürk University Committee on Ethics were followed.The case
This study was carried out as a case study with Bilal (pseudonym), a tenth-grade congenitally blind student in a public high school in an inner-city located in Eastern Anatolia of Turkey. Bilal has an illness called retinoblastoma, which is a rarely seen tumour on the retina, generally seen in those younger than 5 years (Özkan, 2016). Bilal is literate in the Braille alphabet. This makes him access printed materials in Braille. He has no other special needs than blindness. Bilal follows the classes with his peers in a regular classroom with extra support given through a Support & Training Room (STR), which is a support system provided for students with special needs in school for extracurricular activities. Bilal was provided extra support through the STR in three different aspects of chemistry, namely, macro, sub-micro and symbolic dimensions in teaching the topic ‘energy in living systems’.While understanding that the sub-micro dimension is related to mental images, understanding the macro dimension is related to observable events, experiments and experiences. On the other hand, understanding symbolic representations covers pictorial and algebraic-structural formulas such as graphs and chemical equations (Johnstone, 1991; Meijer, 2011).
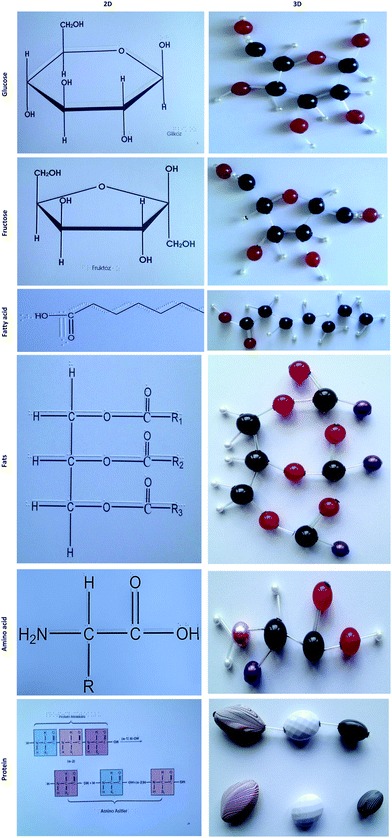
In this study, the molecular and structural formulas of carbohydrates, fats and proteins covered in the ‘energy in living systems’ topic are taught using tactile materials to enhance understanding symbolic representations. Tactile senses enhance haptic perception, which is the human ability to experience and interpret things based on touch and movement. Haptic materials in this study are divided into two groups:
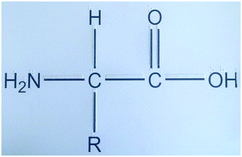
1. 2D embossed representations: Molecular and structural formulas are commonly shown in 2D. As the participant was blind, 2D embossed representations of the molecular structures supported with Braille prints of the letters representing the molecules were used. In these representations both the overall structural representations and symbols of the molecules are represented in Braille and normal typesetting to make them accessible for sighted and non-sighted people. See a sample representation in Fig. 1.
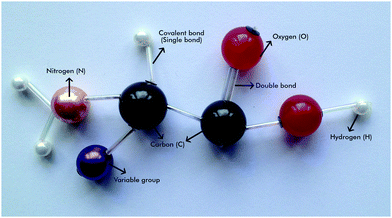
2. 3D representations: 3D representations of organic molecular structures are commonly represented by using molecular model sets. In these 3 models, atoms are represented by plastic polygons or spheres and the bonds are represented by rods. Different atoms are generally shown in the same size other than hydrogen but in different colours. However, these uniform spheres give students the impression that atoms are all the same size other than hydrogen and the molecular volume is confined to the space occupied by the plastic pieces. Moreover, a blind person cannot differentiate the atoms in different colours. There should be an approach to representing different atoms not just in different colours but in different sizes to make them accessible through touching. Therefore, in this study, we used beads purchased from bead shops and peddlers instead of regular molecular model sets. In the model we used beads in different sizes and colours. While different sizes of beads facilitate differentiating the different atoms for a BLV student, the difference in the colours also helps to differentiate the different atoms for sighted people such as teachers, peers and parents. In this way, the tactile materials developed become accessible for both sighted and non-sighted people.
Data collection tools
Data in this study were collected via interviews and observations. The interviews were carried out by the first author to gain an understanding of the student's background knowledge of the ‘energy in living systems’ topic. A semi-structured interview technique was utilised. An interview protocol was developed consisting of 21 questions (the set of questions is given in Appendix 1) focussing on the student's background knowledge on the topic as well as his individual needs regarding learning symbolic representations in chemistry. The same interview protocol was used before and after teaching. Each interview took around half an hour and was tape-recorded. The interviews were transcribed verbatim and analysed. The language of the documents is Turkish. All analysis was done in the Turkish language and only required parts were translated into English for this paper.In addition to the interviews, Bilal was observed in his regular classroom environment for a two months’ interval (2 hours in each week and a total of 8 weeks). He attended a classroom with his sighted peers. The classroom had 35 students and had no special arrangements for extra support that a blind student may need. The school had no special resources for a blind student but a chemistry book printed in Braille was provided by the MoNE. The Braille printed book covers only Braille print of the normal texts in a regular chemistry book. There is no representation of any kind of figure or molecular structure as well as mathematical expressions. Therefore, Bilal preferred listening to his teacher instead of using the book. However, the chemistry teacher had no experience working with a blind student and therefore the teacher taught neither the symbolic representations in chemistry nor the mathematical calculations in algebraic problems.
The observations were carried out during teaching. Each teaching session was recorded with a camcorder with the consent of Bilal. All observations were unstructured in nature. Because the researcher was also taking part in teaching, she took notes immediately after the instruction with the purpose of reflection. All these notes were also used in data analysis as supplementary information.
As a result of the pre-interview and classroom observations, the student's individual learning needs as well as the level of knowledge of the topic were determined, and based on these findings, the features of the instructional material to be designed were established. 2D and 3D instructional materials supplemented with information sheets printed in Braille and Turkish were developed to allow Bilal's chemistry teacher, peers and parents have access to the materials if needed. All materials developed were tested. The implementation was carried out in the STR with the help of the first author in six sessions; each session was 45 minutes with 15 minutes break in between, for three weeks (2 sessions in each week).
Analysis of data
Data were collected through interviews, observations and field notes. The data obtained were analysed descriptively (Lazar et al., 2017, pp. 26–27, p. 305). As there was only one participant, the data analysis was mainly based on the description and interpretation of the data with supporting quotations and pictures.A sample text extracted from the data set was re-coded (re-analysed) by the first author with a three months’ interval and both analyses were compared by the second author. Minor differences were discussed and discrepant views were agreed. As there were no significant differences between the two sets analysis, the analysis was considered as reliable.
Results
In this section, the findings from the observations, field notes and interviews together with the interpretations based on these findings were presented according to the research questions.Findings based on the observations and the pre-interview
Evidence gathered from the observations indicated that Bilal was not able to carry out algorithmic problems as he was not trained on developing mathematical skills and not taught about use of symbolic representations; therefore he did not know the symbolic representations in chemistry. But he was good at acquiring knowledge provided verbally. In addition, in the anecdotal evidence gathered through personal communication with him, it was stated that he could not complete tasks that involve mathematical calculations in chemistry as well as molecular structures and equations representing chemical reactions. He did not know about how a chemical reaction is represented as an equation including the writing of chemical formulas. Furthermore, his teacher had no knowledge of how to teach equations to a blind student representing either mathematical or chemical equations. All these findings are similar to what has been reported before (Harshman et al., 2013; Maguvhe, 2015). The findings indicated a strong need for designing and developing tactile or audible educational materials to facilitate his learning for his access to symbolic representations used in chemistry.The pre-interview aimed to determine the level of Bilal's knowledge in the concepts related to the subject. The questions asked in the pre-interview covered the concepts related to the learning outcomes in the ‘energy in living systems’ topic. The following quotation taken from the pre-interview demonstrates Bilal's level of knowledge in the topic:
Interviewer (I): Could you tell me what an element is?
Student (S): Pure substances containing the same atoms …
I: Can you give an example of the elements which are frequently encountered in daily life?
S: … chlorine, which is represented by C. Namely, more precisely, I know the first 20 elements by their names and symbols. Flor by F … chlorine by C … Carbon is represented by the symbol C. Calcium is represented by Ca. Potassium is represented by P. Nitrogen is represented by N. Another … Oxygen is represented by O. There is boron represented by B. Is it enough?
As seen from the quotation, Bilal repeats some knowledge by heart, which is most probably acquired by memorising the definitions given in the classroom by his teacher or having read the Braille book. Bilal was also able to tell symbols of some elements by heart with some mistakes but he could not write them. The following excerpt taken from the same pre-interview illustrates his level of knowledge of molecular structures of fats and proteins as well as their definitions.
I: Could you tell me how the general structure of the fats is?
S: I do not know very well right now.
I: How about proteins? What is protein?
S: Well… what can I say? It is a sort of structural thing. There is amino acid. I suppose there is an amino acid, nitrogen…eee. They are linked by a peptide bond.
…
I: Do you know anything about how is the general structure of the proteins?
S: …eee. I know nothing.”
Classroom observations showed also several problems that Bilal encountered in a classroom with a class size of 35 students. Bilal mostly relied on listening to the teacher. He attended a residential school for the blind in primary and middle school from grade 1 to 8. In the residential school class sizes are quite small, up to a maximum of 10 students and in most cases less than six or seven students, compared to a regular size in his current inclusive class in a high school. Small class sizes in the residential school for the blind made it easy for them to follow the teacher. In addition, teachers in the residential school were more experienced in helping blind students. However, Bilal was having trouble in following what his teacher was telling in a noisy environment. Another main problem was his chemistry teacher's ignorance of Bilal among the 35 students. Anecdotal evidence during conversations with the teacher suggests that the teacher was focussing on the rest of the classroom assuming that Bilal was able to follow him. On the other hand, the teacher felt guilty because he was not able to help Bilal as he had no experience whatsoever before in teaching a blind student. Moreover, neither school had any supporting material to teach chemistry nor was the teacher using any hands-on materials during teaching. The teaching was in the form of exposition via lecture. Furthermore, the expectations from Bilal were not high regarding his capacity to learn chemistry. The teacher was convinced that blind students were exempt from learning algebraic calculations as well as graphical and symbolic representations. Therefore, the only knowledge Bilal had on these matters was what he had acquired from his teacher's talks by heart. Finally, during the exams, Bilal was separated from the rest of the class by asking him to answer only those questions that do not require the use of any algebraic calculation or do not include any representation as figures or formulas for chemical reactions. There was neither Braille printing facility in the school nor the teacher had any knowledge of Braille printing. Therefore, during the exams, Bilal was taken to the STR, the teacher read aloud the questions and he answered verbally. His responses were evaluated and marked by the teacher. As Bilal was mastered in answering questions that require declarative knowledge by heart, he was receiving high marks. On the records, Bilal was seen a high achiever in chemistry.
These findings indicate a clear need for designing appropriate instructional materials to help Bilal in accessing the knowledge of structural representations of molecules.
Materials designed for energy in living systems
The instructional materials were designed by the first author taking Bilal's individual learning needs into account based on five learning outcomes stated in the 10th grade chemistry curriculum, which are stated as below:1. Relates the transformation of nutrients to energy to digestion and respiratory processes.
2. Explains the formation and use of simple sugars, which are the primary energy sources for organisms.
3. Relates the ways of storing the excess of energy in the human body to energy consumption.
4. Relates the combustion and hydrolysis properties of fats to their use in the body.
5. Relates the structure and functions of proteins to amino acids (MoNE, 2013, p. 18).
Three different types of materials were designed and developed for supporting instruction. The first type was 3D tactile representations of molecules made from beads. The second type was 2D tactile prints. The molecular structures of the molecules covered were drawn with the help of ChemDraw and these drawings were transferred to an embosser (SpotDot Emprint Colour Ink and Braille Embosser). The third type of material is information sheets which were printed in Braille with the help of a Braille printer (Everest-D V4). These information sheets are designed to provide summary information including the key aspects of the topic. They were printed both in normal scripts and in Braille. In this way, the teacher, Bilal's peers and his parents can have access to the information provided on the sheets. Samples of three types of materials are shown in Fig. 1 (2D representation), Fig. 2 (3D representation) and Fig. 3 (information sheet). The total number of instructional materials developed included nine 3D materials representing carbohydrates, fats and proteins, 12 2D embossed printed representations of the 3D materials as well as a chemical reaction mechanism, and, finally, eight information sheets printed in Braille and normal text.
 | ||
| Fig. 3 A sample of an information sheet covering the definitions of major nutrients (carbohydrates, fats and proteins) printed in normal typesetting (in Turkish) and Braille. | ||
For designing a representation of a molecular structure in 3D, six different beads were used. The largest and black bead represents the carbon (C) atom. Normally, there are two holes in the beads, but, as the carbon atom forms 4 bonds, two additional holes are made in the black beads. The red bead represents the oxygen (O) atom. The hydrogen (H) atom was represented by the white bead, which is the smallest. While the orange beads were used to represent the nitrogen (N) atom, the purple beads represent variable groups (R). The narrow, long and transparent rods were used to represent the bonds between the atoms and the groups (Fig. 2). A thick and strong rope was used for connecting the beads in forming 3D representations for molecules of carbohydrates, fats, and proteins (see Fig. 4).
Implementation of instructional materials
Teaching took place in a STR with the help of the first author in six sessions (each ended in 45 minutes with an additional 15 minute break) in three weeks (2 sessions each week). The teaching was started with an introduction of the particular topic with information sheets. The student was allowed to read the information given on the information sheet, and a discussion took place about the knowledge given. For instance, the first class started by handing out “Information Sheet 1”, which covers the definition of energy and the derivation of energy through the digestion and respiration of fats and proteins. Then Information Sheet 2, which covers monosaccharides, was given. The questions were answered and then the symbolic representations were introduced, starting with the 2D materials for glucose and fructose. Guidance was given about how to use the representations. Once the student showed a sign of acquiring the representations for glucose and fructose, then 3D materials made with beads were given with guidance on how to use 3D models made with beads and what each bead represents. In the second stage of the instruction, Bilal was instructed on how to match the 2D and 3D representations for the same molecule. This helped the student to understand different representations of the same molecule. The same approach was repeated at the following stages of the teaching to cover lactose, sucrose and maltose.Bilal came across such representations for the first time. Therefore, it took a while to get him to familiarise himself. He needed guidance to compare the representations in 2D and 3D. He managed to work on them while one hand was on the 2D and the other on the 3D one with the help of his teacher. However, the main difficulty in using the 3D model was that, as it was made from beads and rope, it did not have a fixed shape. Therefore it might cause misunderstanding of the representations. Another difficulty that Bilal experienced was the explanation of the geometry of the molecules. In 2D prints, it was hard for him to understand which part was on and below the planar surface. 3D representations were more helpful initially to give this idea. However, later on, Bilal started to understand this better with 3D representations as they were more versatile to play with. However, the main problem with the 3D models was that they were loose and not rigid. It took a while for Bilal to first recognise the beads and differentiate them according to their sizes and then started to recognise which part was connected to which beads as it was loose. However, after a while, Bilal became a master in playing with the models made with beads and he was successful in correctly identifying the representation (see Fig. 5). Finally, Bilal's views were gathered about the usefulness of 2D and 3D materials. He pointed out that 3D representations were more understandable than 2D ones. This is because he could move the beads around in his hand as well as he laid down the beads on a surface and also recognised the differences between them.
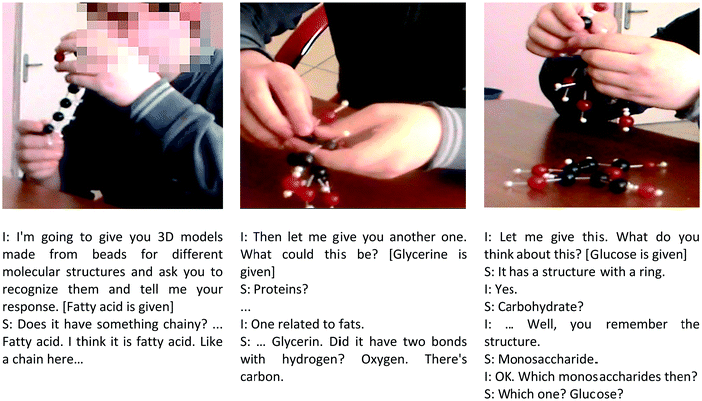 | ||
| Fig. 5 The student is working with the 3D models made from beads and recognising what they represent. | ||
Findings based on the post-interview
During the pre-interview Bilal gave correct answers to questions Q1, Q2, Q3, Q4, Q5, Q8, Q10, Q15, Q16 and Q18. Some of these questions were in fact asked to him to see whether he knew the declarative knowledge which is based on memorisation. As his responses showed, he could tell the correct definitions of the terms they were excluded from the post-interviews as we focussed on improvement. Bilal was a quiet person and therefore taciturn initially. However, as time went by, he got used to the first author and started to collaborate much openly. Instead, the questions with no answer, incorrect answers or partially correct answers were asked (see Fig. 6). However, the post-interview displays encouraging results regarding his understanding of the symbolic representations in chemistry. As seen from Fig. 6, while Bilal was not able to answer questions Q7, Q11, Q12, Q17, Q19, and Q21 at all in the pre-interview, after the intervention, he was able to fully respond to questions Q7, Q17, Q19 and Q21 and also gave partly correct answers to questions Q11 and Q12. On the other hand, there were questions (Q9, Q13, Q14 and Q20) for which partially correct answers were given in the pre-interview but Bilal's responses showed correct understanding in the post-interview. However, he was not able to show any improvement for question Q6 that was partially correct in both the pre- and post-interviews. Some of the quotations taken from the post-interview to illustrate his responses are given below: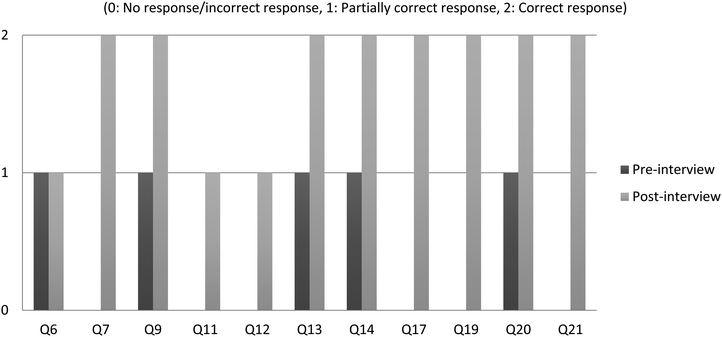 | ||
| Fig. 6 Pre- and post-interview results based on the questions (the questions that Bilal answered correctly in the pre-interview were excluded in order to ease the readability). | ||
I: What is digestion?
S: This is the disintegration of nutrients with the help of enzymes, probably disintegration due to the enzymes. For example, chemical digestion in the digestive tract, stomach and intestine, and in the small intestine. What was that? These nutrients … presumably, mixed with blood. They’re broken down into their monomers.
…
I: What is breathing?
S: For example, you know, we mention organic nutrients. Well, for energy, breaking down organic nutrients through oxygen. Breaking down through oxygen. Oxygen, let's say, breaking down through oxygen. Oxygen needed is taken up from the air. Oxygen needed. For example, generally, (oxygen) is carried by the hemoglobin in red blood cells.
…
I: How is the structure of carbohydrates?
S: Glucose's thing (structure) is, well, C6H12O6. That is, well, monosaccharides are single sugar form; disaccharides are made by monosaccharides. The removal of H2O. C12H22O11 (general molecular structure of disaccharides).
….
I: How is the general structure of the fats?
S: It consisted of fatty acids and glycerol. Fatty acids and glycerol. You know, R group with 17 carbons…
…
I: What is a protein?
S: Ok. Protein synthesizes the things needed for growth, growth, repair well repair. That is, (protein) as building blocks of the body.
I: How is the general structure of the proteins?
S: Amino acids there is, on the side, amino acids group, and also a carboxyl group…. –NH2……. There is carboxyl group, COOH. Also, variable R groups, hydrogen.
The above quotations suggest that Bilal demonstrated an improvement in learning the concepts regarding the energy in living systems as well as an understanding of the symbolic representations developed to help him understand the molecular structures. However, these findings cannot be used to argue as evidence of deep learning of the concepts. To test students’ understanding of the molecular structure, a further test was done by asking him to recognise particularly the 3D models of the structures in different orders. He successfully recognised all structures and was able to respond to any question regarding the parts of these representations (see the dialogues given in Fig. 5).
Discussion
The results identified several difficulties associated with the accessibility to the chemistry knowledge particularly, which requires the use of vision such as mastering mathematical calculations; writing formulas of chemical compounds and molecules, and chemical equations; and understanding molecular structures. For an inexperienced teacher, it is not easy to meet these kinds of needs of a blind student. The literature (i.e.Boyd-Kimball, 2012) provides evidence of successful teaching of writing and balancing chemical reactions, mathematical calculations, Lewis dot structures, molecular geometries and structural formulas. However, the majority of this information is written in academic journals and they are not accessible by teachers. Moreover, there are no materials available for a chemistry teacher on how to teach a blind student in Turkish although there are some general guidelines for adaptation. Therefore, teachers tend to exempt those students from the topics including representations and more attention is given to the declarative knowledge. BLV students are also exempt from answering questions that include illustrations in the university entrance exam in Turkey. Therefore, this also discourages teachers from looking for an appropriate solution. On the other hand, research on this issue is limited in Turkish and studies recently done focus on lower grades (see Kızılaslan, 2016; Zorluoğlu, 2017; Kızılaslan et al., 2019). Thus, there is no research available to the teachers in Turkish focussing on teaching particular chemistry topics.Regarding the materials design principles to be taken into consideration particularly for meeting Bilal's needs, we focussed on three things: information sheets in Braille to provide a summary of basic information, 2D tactile representations and 3D models made from everyday materials. Information sheets prepared in Braille are found to be helpful as they provide information in summary and could be used whenever the student needs. The student may use them for repeating the topic as well as preparing for the exams. 2D tactile drawings were found to be helpful in terms of grasping the representation on two dimensions. There are several techniques available for developing 2D tactile materials with drawings. However, all are dependent on the use of technology which is rarely available. In our case, we used a Braille embosser (Emprint SpotDot) to print drawings representing molecular structures. This embosser can print both Braille text and lines. However, these kinds of printers are rarely available for teachers in schools. Therefore, the teacher may prefer using everyday materials such as silicone guns to raise the lines and make tactile materials. Further to 3D models, we used beads. They are readily available and cheap but it is time-consuming to make them. On the other hand, they are not rigid. Their loose structures cause confusion and may lead students to misunderstand. Moreover, they are not versatile or suitable for further uses. Currently, 3D printing technology is providing better solutions to this problem. However, designing materials requires knowledge of 3D designing as well as having 3D printers readily available. On the other hand, although 3D printers are developing very fast and promising good practices (i.e.Grumman and Carrol, 2019), unfortunately many schools still have either no access or limited access to them. However, 3D printers clearly have a lot to offer in terms of material development to meet the individual needs of BLV students.
According to the findings obtained, it can be argued that the 2D and 3D materials developed facilitated learning the knowledge of molecular structures covered under the topic. The findings confirm the previous literature findings that the use of tactile materials in teaching chemistry to visually impaired students has a significant influence on their understanding (Supalo, 2005; Lewis and Bodner, 2013).
Conclusions and recommendations
In this study, the problems experienced by a blind student in learning the symbolic representations used in chemistry are determined. Based on the pre-interview and observation data it was concluded that blind students’ needs are ignored in this inclusive classroom environment; therefore, he was not able to learn the symbolic representations in chemistry. This is an important point to raise as the student was experiencing some exclusion within inclusion as described by Johnson (2018). Although there are several developments in technology as well as knowledge how to work with BLV students, it takes time to transfer the new knowledge developed into practice. On the other hand, there is limited or no access to the facilities or equipment for both students and teachers. Therefore, the teacher felt guilty and tended to give the student high marks by excluding him from some part of the chemistry and thus only testing their declarative knowledge. The student was able to learn verbal information covered in the classroom either through teacher description or by reading from the book on his own through a Braille copy of the chemistry book. The main reason for having difficulties in understanding the molecular structures in chemistry is actually not the student's learning capacity but not providing any kind of opportunity for him. Our results also provided evidence that if a blind student is provided with materials which suit his/her needs he/she can develop a better understanding.The results are based on a single case study. It is not possible to make generalisations from such a single case. However, the following suggestions can be made based on the experience we gained through this study:
• BLV students need extra support. However, teachers are busy teaching several students with different needs and expectations; therefore, it is not realistic to expect everything from the teachers. If researchers in chemistry education turn their attention to this field and devote their time and effort to develop instructional methods and instructional materials, teachers would be able to use them with confidence. Otherwise, each teacher has to try each case and perhaps could not succeed each time.
• Materials and activities which can address different senses can be used to learn more effectively. Students with learning needs, particularly visually impaired, mostly rely on senses other than vision. However, teachers are inclined to use more commonly sound in the absence of vision, but this is not enough. Visually impaired students use haptic sense quite effectively. Designing tactile materials is therefore essential for BLV students.
• In printed materials prepared for blind students, normal text and Braille printing can be used together. The use of normal text is necessary for Braille illiterate teachers, peers and parents to understand the relevant material and to provide support to the student.
• For the use of tactile materials in the classroom for BLV students, firstly, we started with 2D embossed drawings, then the use of 3D models made from beads representing the 2D representations. This sequencing is commonly used in teaching for students with vision. However, blind students may not necessarily be happy with this sequencing. This point is not examined in detail and further research is needed. Therefore, we may suggest that researchers investigate this further.
• None of the teachers teaching chemistry is receiving training on inclusive education at the teacher education stage. It was also the case for the teacher involved in this study. Therefore, if teachers have students with special needs in their classroom, extra training support should be available to develop skills they may need. In addition, subject teachers would be guided by a special education teacher when needed to better understand the needs of a student with special needs and find viable solutions.
Conflicts of interest
There are no conflicts to declare.Appendix 1. The questions asked during both the pre- and post-interviews
1. What is an element?2. Can you give examples of frequently encountered elements in daily life?
3. What is a compound?
4. What is an atom?
5. What are the subatomic particles?
6. What are models representing an atom?
7. What kind of interaction is available between chemical species?
8. What is an ionic bond?
9. What is a covalent bond?
10. How are physical and chemical changes defined?
11. What are the types of chemical reactions?
12. What is the law of conservation of mass?
13. What is digestion?
14. What is breathing?
15. What are the important energy sources for living things?
16. What are the primary energy sources for living things?
17. How is the structure of carbohydrates?
18. How is the excess energy stored in the body?
19. What is the general structure of fats?
20. What is a protein?
21. How is the general structure of proteins?
Acknowledgements
We would like to thank the student and the teacher, who allowed us to work closely with them. We are also grateful for the reviewers of this paper for their constructive comments which enhanced the paper significantly.References
- Batu S., Kırcaali-İftar G. and Uzuner Y., (2004), Özel gereksinimli öğrencilerin kaynaştırıldığı bir kız meslek lisesindeki öğretmenlerin kaynaştırmaya ilişkin görüş ve önerileri [Teachers’ views on inclusive education working in a special school for girl's vocational education including students with special needs], Ankara Üniversitesi Eğitim Bilimleri Fakültesi Özel Eğitim Dergisi [Journal of Special Education Ankara University College of Education], 5(2), 33–50, Retrieved from http://dergiler.ankara.edu.tr/dergiler/39/52/488.pdf.
- Bleijenbergh I., (2010), Case selection, in Mills A. J., Durepos G. and Wiebe E. (ed.), Encyclopedia of case study research, London: Sage, pp. 61–63.
- Boyd-Kimball D., (2012), Adaptive instructional aids for teaching a blind student in a nonmajors college chemistry course. J. Chem. Educ., 89(11), 1395–1399 DOI:10.1021/ed1000153.
- Cook B. G. and Semmel M. I., (1999), Peer acceptance of included students with disabilities as a function of severity of disability and classroom composition, J. Spec. Educ., 33(1), 50–61 DOI:10.1177/002246699903300105.
- Düşkün Y., (2016), Türkiye’de ortaöğretimde kapsayıcı eğitim durum analizi [An analysis for inclusive education for secondary level in Turkey], İstanbul, Turkey: Eğitim Reformu Girişimi, Retrieved from http://www.egitimreformugirisimi.org/wp-content/uploads/2017/03/ERG_KapsayiciEgitim_DurumAnalizi.pdf.
- Fantin D., Sutton M., Daumann L. J. and Fischer K. F., (2016), Evaluation of existing and new periodic tables of the elements for the chemistry education of blind students, J. Chem. Educ., 93(6), 1039–1048 DOI:10.1021/acs.jchemed.5b00636.
- Flair M. N. and Setzer W. N., (1990), An olfactory indicator for acid-base titrations – a laboratory technique for the visually impaired, J. Chem. Educ., 67(9), 795–796 DOI:10.1021/ed067p795.
- Grumman A. S. and Carroll F. A. (2019). 3D-printing electron density isosurface models and high-resolution molecular models based on van der Waals radii, J. Chem. Educ., 96(6), 1157–1164, DOI:10.1021/acs.jchemed.8b00597.
- Gudjonsdottir H. and Oskarsdottir E., (2016), Inclusive education, pedagogy and practice, in Markic S. and Abels S. (ed.), Science education towards inclusion, New York: Nova Science Publishers, pp. 7–22.
- Gupta H. O. and Singh R., (1998), Low cost science teaching equipment for visually impaired children, J. Chem. Educ., 75(5), 610–612, DOI:10.1021/ed075p610.
- Harshman J., Bretz L. and Yezierski E., (2013), Seeing chemistry through the eyes of the blind: a case study examining multiple gas law representations, J. Chem. Educ., 90(6), 710–716, DOI:10.1021/ed3005903.
- Hospital L., (2016), Adapting the APH periodic table to indicate major element groups, Retrieved from http://www.perkinselearning.org/accessible-science/activities/adapting-aph-periodic-table-indicate-major-element-groups.
- Johnson L., (2018), Inclusion “insight”, in Koomen M., Kahn S., Atchison C. L. and Wild T. A. (ed.), Towards inclusion of all learners through science teacher education, Boston: Brill Sense, pp. 5–14, DOI:10.1163/9789004368422_002.
- Johnstone A. H., (1991), Why is science difficult to learn? Things are seldom what they seem, J. Comput. Assisted Learn., 7(2), 75–83, DOI:10.1111/j.1365-2729.1991.tb00230.x.
- Kırcaali-İftar G. and Uysal A., (1999), Zihin özürlü öğrencilere özel eğitim danışmanlığı aracılığıyla uygulanan resimli fişlerle okuma-yazma öğretiminin etkililiği [An investigation on the effectiveness of an activity for reading and writing under the guidance of an expert on education for mentally disabled students], Özel Eğitim Dergisi [Ankara University Special Education Journal], 2(3), 3–13, Retrieved from http://dergiler.ankara.edu.tr/dergiler/39/46/391.pdf.
- Kızılaslan A., (2016), Teaching concepts related “Phases of Matter and Heat” unit to 8th grade visually impaired primary students, (Unpublished Doctoral Thesis), Erzurum, Turkey: Atatürk University, Graduate School of Educational Sciences.
- Kizilaslan A. Sozbilir M. and Zorluoglu S. L., (2019), Making science accessible to students with visual impairments: insulation-materials investigation, J. Chem. Educ., 96(7), 1383–1388, DOI:10.1021/acs.jchemed.8b00772.
- Lazar J., Feng J. H. and Hochheiser H., (2017), Research methods in human-computer interaction, Cambridge, MA: Morgan Kaufmann Publishers.
- Maguvhe M., (2015), Teaching science and mathematics to students with visual impairments: reflections of a visually impaired technician, Afr. J. Disabil., 4(1), Art.#194, 6 pages, DOI:10.4102/ajod.v4i1.194.
- Markic S. and Abels S., (2016), Science education meets inclusion, in Markic S. and Abels S. (ed.), Science education towards inclusion, New York: Nova Science Publishers, pp. 1–6.
- Meijer M. R., (2011), Macro-meso-micro thinking with structure-property relations for chemistry education: An explorative design-based study, (Unpublished Doctoral Thesis), Utrecht, Nederland: Utrecht University.
- Melaku S., Schreck J. O., Griffin K. and Dabke R. B., (2016), Interlocking toy building blocks as hands-on learning modules for blind and visually impaired chemistry students, J. Chem. Educ., 93(6), 1049–1055, DOI:10.1021/acs.jchemed.5b00252.
- Micklos Lewis A. L. and Bodner G. M., (2013), Chemical reactions: what understanding do students with blindness develop? Chem. Educ. Res. Pract., 14(4), 625–636, 10.1039/C3RP00109A.
- Ministry of National Education (MoNE), (2013), Ortaöğretim kimya dersi öğretim programı [Chemistry curriculum for secondary schools], Ankara: Ministry of National Education.
- Ministry of National Education (MoNE), (2015), Millî eğitim istatistikleri: örgün eğitim (1) [National education statistics: formal education (1)], Ankara: Ministry of National Education.
- Okcu B. and Sozbilir M., (2019), Designing a bulb to teach electric circuits to visually impaired students, Phys. Teach., 57(99), 99–101, DOI:10.1119/1.5088470.
- Özkan A., (2016), Retinablastom: Kanserli çocuk [Retinablastoma: child with cancer], Retrieved from http://www.kanserlicocuk.com/Page.asp?Konu_ID=21.
- Pavri S. and Luftig R., (2000), The social face of inclusive education: are students with learning disabilities really ıncluded in the classroom? Prev. Sch. Failure, 45 (1), 8–14, DOI:10.1080/10459880109599808.
- Smothers S. M. and Goldston M. J., (2010), Atoms, elements, molecules, and matter: an investigation into the congenitally blind adolescents’ conceptual frameworks on the nature of matter, Sci. Educ., 94(3), 448–477, DOI:10.1002/sce.20369.
- Sözbilir M., (2016), Practical work in science with visually impaired students, in Eilks I., Markic S. and Ralle B. (ed.), Science education research and practical work, Aachen: Shaker Verlag, pp. 169–179.
- Supalo C. A., (2005), Techniques to enhance instructors’ teaching effectiveness with chemistry students who are blind or visually impaired, J. Chem. Educ., 82(10), 1513–1518, DOI:10.1021/ed082p1513.
- Supalo C. A., (2016), ConfChem conference on interactive visualizations for chemistry teaching and learning: concerns regarding accessible interfaces for students who are blind or have low vision, J. Chem. Educ., 93(6), 1156–1159, DOI:10.1021/acs.jchemed.5b00603.
- Supalo C. A. and Kennedy S. H., (2014), Using commercially available techniques to make organic chemistry representations tactile and more accessible to students with blindness or low vision, J. Chem. Educ., 91(10), 1745–1747, DOI:10.1021/ed4005936.
- Supalo C. A., Mallouk T. E., Rankel L., Amorosi C. and Graybil C. M., (2008), Low-cost laboratory adaptations for precollege students who are blind or visually impaired, J. Chem. Educ., 85(2), 243–247, DOI:10.1021/ed085p243.
- Supalo C. A., Mallouk T. E., Amorosi C., Lanouette J., Wohlers H. D. and McEnnis K., (2009), Using adaptive tools and techniques to teach a class of students who are blind or low-vision, J. Chem. Educ., 86(5), 587–591 DOI:10.1021/ed086p587.
- Supalo C. A., Isaacson M. D. and Lombardi M. V., (2014), Making hands-on science learning accessible for students who are blind or have low vision, J. Chem. Educ., 91(2), 195–199 DOI:10.1021/ed3000765.
- Tombaugh D., (1981), Chemistry and the visually impaired, J. Chem. Educ., 58(3), 222–226 DOI:10.1021/ed058p222.
- Ulutaşdemir N., (2007), Engelli çocukların eğitimi [Education of disabled students], Fırat Sağlık Hizmetleri Dergisi, 2(5), 119–130.
- Vitoriano F. A., Teles V. L. G., Rizzatti I. M. and Pesssoa de Lima R. C., (2016), Promoting inclusive chemistry teaching by developing an accessible thermometer for students with visual disabilities, J. Chem. Educ., 93(12), 2046–2051 DOI:10.1021/acs.jchemed.6b00162.
- Wedler H. B., Cohen S. R., Davis R. L., Harrison J. G., Siebert M. R., Willenbring D., Tantillo D. J. et al., (2012), Applied computational chemistry for the blind and visually impaired, J. Chem. Educ., 89(11), 1400–1404 DOI:10.1021/ed3000364.
- Yin R. K., (2014), Case study research: design and methods, 5th edn, London: Sage Publications.
- Zorluoğlu S. L., (2017), Teaching the concepts in particulate nature of matter to 6th grade visually impaired students, (Unpublished Doctoral Thesis), Erzurum, Turkey: Atatürk University, Graduate School of Educational Sciences.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c9rp00002j |
| ‡ This paper is produced from the first author's MSc thesis. |
| This journal is © The Royal Society of Chemistry 2019 |