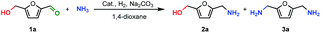Open Access Article
Open Access ArticleHighly selective synthesis of 2,5-bis(aminomethyl)furan via catalytic amination of 5-(hydroxymethyl)furfural with NH3 over a bifunctional catalyst†
Hangkong Yuana,
Bright T. Kusema*b,
Zhen Yanb,
Stéphane Streiffb and
Feng Shi *a
*a
aState Key Laboratory for Oxo Synthesis and Selective Oxidation, Lanzhou Institute of Chemical Physics (LICP), Chinese Academy of Sciences, Lanzhou 730000, China. E-mail: fshi@licp.cas.cn
bEco-Efficient Products and Processes Laboratory (E2P2L), UMI 3464 CNRS-Solvay, 3966 Jin Du Road, Xin Zhuang Ind. Zone, 201108 Shanghai, China. E-mail: bright.kusema@solvay.com
First published on 27th November 2019
Abstract
The development of facile protocols for the selective synthesis of biomass-derived diamine is a highly desirable pursuit in the field of heterogeneous catalysis. Herein, a simple and highly efficient bi-functional CuNiAlOx catalyst was developed for the one pot transformation of 5-(hydroxymethyl)furfural (5-HMF) into 2,5-bis(aminomethyl)furan (BAF) using a two-stage reaction process. Cu4Ni1Al4Ox was found to be the most effective catalyst for this reaction, affording BAF in 85.9% yield. Our results could promote controllable conversion and utilization of biomass resource.
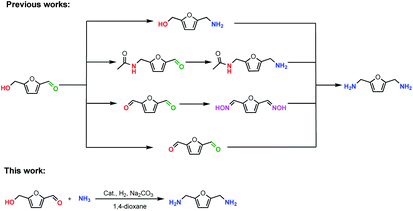
Amines are an important class of nitrogen-containing chemicals and are widely used in the synthesis of pharmaceuticals, agrochemicals, surfactants, dyes, polymers, and biologically active compounds.1 Among the different amines, primary diamines are some of the most useful monomers for the production of polyamides and polyureas which were widely employed in everyday life including automotive, aerospace, electrical and electronics, building, and biomedicine industry.2 Traditionally, they are produced from carbonyl and alcohol compounds which are derived from fossil resources. However, the depletion of fossil resources is a big concern as well as the environmental impact associated with their usage. Furthermore, there is a rising demand for biocompatible polymers to be used in industry, for example, in packaging and biomedicine. Therefore, the application of renewable resources to replace fossil resources for the production of primary diamines is highly desired.
Biomass as a renewable and ubiquitous resource is considered as the next generation feedstock for the chemical industry. Utilization of biomass as raw material has potential to replace diminishing fossil fuels with renewable feedstocks, thereby alleviating the dependence on petro-based materials, and reducing pollution.3 In addition, biomass-derived diamine monomers would meet the rising demand for biocompatible polymers. Therefore, the development of effective methods for the preparation of diamines from bio-based renewable materials is an attractive prospect in view of establishing the sustainable development of societies.4 As one of the most important platform molecules derived from biomass, 5-(hydroxymethyl)furfural (5-HMF) is attracting increasing attention owing to the unique and versatile chemical structure containing both aldehyde and hydroxyl groups.5 5-HMF could be used to synthesize 2,5-bis(aminomethyl)furan (BAF); however, selective synthesis of BAF from 5-HMF is a challenging task because the transformation is prone to form a series of by-products of secondary, tertiary and polymeric amine species.6 Thus, many multi-step routes were developed for the synthesis of BAF. Komanoya et al. developed a two-step method for the synthesis of BAF, in which 5-HMF was firstly converted into 5-(hydroxymethyl)furfurylamine (HMFA) over Ru/Nb2O5 catalyst and then converted into BAF over a homogenous [Ru(CO)ClH(PPh3)3] catalyst.7 Lin and co-workers have prepared BAF with 45.7% yield through the hydroxymethyl group of 5-HMF into amide group with CH3CN by Ritter reaction, followed by reductive amination of aldehyde groups with NH3 and hydrolysis of amide to primary amine8, Kim et al. reported the reductive amination of 2,5-diformylfuran to BAF over acid treated RANEY®-Ni catalysts with a yield of 42.6%.6 Xu and co-workers have demonstrated that BAF could be efficiently synthesized in 94.1% yield from hydrogenation of 2,5-diformylfuran dioxime which was generated from oximation of 2,5-diformylfuran (DFF).9 Compared to the above two or multi-steps methods, the direct amination of easily available 5-HMF to BAF would be advantageous with respect to atom efficiency and step economy. To date, however, only few catalytic systems dealing with direct transformation of 5-HMF into the BAF have been established. In 2018, an elegant method for the direct amination of 5-HMF with ammonia to BAF with 85% yield was achieved in the presence of homogeneous Ru/phosphine ligand complex.10 Recently, Wei and co-workers presented an effective method for direct conversion of 5-HMF into BAF with 60.7% yield using a RANEY®-Ni catalyst.11 However, the use of the noble metal and organic ligand as well as the low product yield is unsatisfactory. Therefore, it is still desirable for the development of an efficient heterogeneous catalytic system for the conversion of 5-HMF to BAF.
According to a mechanistic investigation, the hydrogen-borrowing reaction of hydroxyl group with NH3 is considered the rate-determining step of direct amination of 5-HMF to BAF.11 The key point to realize this reaction is to develop highly active and selective sites for hydrogen-borrowing reaction of hydroxyl with NH3, in which catalytic dehydrogenation of hydroxyl to the corresponding C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and catalytic hydrogenation of resulted imine are involved.12 It is well-known that nickel has an excellent catalytic hydrogenation ability in hydrogenation reaction and copper possesses a good catalytic dehydrogenation ability in borrowing-hydrogen reactions.13 Thus, a bifunctional catalyst combining both excellent catalytic dehydrogenation properties of copper and good catalytic hydrogenation properties of nickel might be a good choice for this hydrogen-borrowing reaction. In addition, it has been reported that the formation of a series of by-products, i.e., secondary amines, oligomer and undesired hydrogenated by-products, resulted mainly from condensation of reactive di-aldehyde and diamine products during the amination of 5-HMF with NH3.6 If the two-stage consecutive reaction process was adopted, in which hydrogen-borrowing reaction of hydroxyl with NH3 occurred after the completion of reductive amination of aldehyde with NH3, it would prevent formation of reactive di-aldehyde and thus it would reduce the by-products. Therefore, we speculated that the combination of bifunctional Cu–Ni catalyst and employing a two-stage reaction process in one-pot might realize highly selective synthesis of BAF from 5-HMF. Herein, we describe the controllable diamination of 5-HMF with NH3 to BAF in 85.9% yield over a bifunctional CuNiAlOx catalyst using a two-stage reaction process (Scheme 1). The route in this work is atom efficient and an environmentally friendly process as water is the only byproduct generated.
O and catalytic hydrogenation of resulted imine are involved.12 It is well-known that nickel has an excellent catalytic hydrogenation ability in hydrogenation reaction and copper possesses a good catalytic dehydrogenation ability in borrowing-hydrogen reactions.13 Thus, a bifunctional catalyst combining both excellent catalytic dehydrogenation properties of copper and good catalytic hydrogenation properties of nickel might be a good choice for this hydrogen-borrowing reaction. In addition, it has been reported that the formation of a series of by-products, i.e., secondary amines, oligomer and undesired hydrogenated by-products, resulted mainly from condensation of reactive di-aldehyde and diamine products during the amination of 5-HMF with NH3.6 If the two-stage consecutive reaction process was adopted, in which hydrogen-borrowing reaction of hydroxyl with NH3 occurred after the completion of reductive amination of aldehyde with NH3, it would prevent formation of reactive di-aldehyde and thus it would reduce the by-products. Therefore, we speculated that the combination of bifunctional Cu–Ni catalyst and employing a two-stage reaction process in one-pot might realize highly selective synthesis of BAF from 5-HMF. Herein, we describe the controllable diamination of 5-HMF with NH3 to BAF in 85.9% yield over a bifunctional CuNiAlOx catalyst using a two-stage reaction process (Scheme 1). The route in this work is atom efficient and an environmentally friendly process as water is the only byproduct generated.
To validate our hypothesis, the one-pot amination of 5-HMF (1a) to BAF (3a) was initially investigated with NH3 in the presence of CuNiAlOx catalyst, Na2CO3 co-catalyst, under 4.5 MPa of H2 in 1,4-dioxane at 90 °C for 6 h, then at 210 °C for 18 h (Table 1). The catalyst used here was prepared by a co-precipitation method by adding an aqueous solution of Na2CO3 and NaOH to a Cu(NO3)2, Ni(NO3)2 and Al(NO3)3 solution. The precipitates were washed with deionized water, dried in air at 100 °C for 12 h and reduced under H2 flow at 450 °C for 3 h. The CuNiAlOx catalysts are denoted as CumNinAlyOx (m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n
n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) y
y![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x = molar ratios of Cu, Ni, Al and O).
x = molar ratios of Cu, Ni, Al and O).
| Entry | Catalysts | Conv.b [%] | Yieldb [%] | Mass balance [%] | |
|---|---|---|---|---|---|
| 1a | 2a | 3a | |||
| a Reaction conditions: 5-HMF (10 g, 79.4 mmol), NH3 (23 g, 1352.9 mmol), catalyst (2.0 g), Na2CO3 (0.915 g, 8.6 mmol, 11 mol% to 5-HMF), H2 (4.5 MPa), 1,4-dioxane (50 mL), 90 °C for 6 h then 210 °C for 18 h.b Determined by GC-FID using biphenyl as the internal standard material.c 90 °C for 9 h then 210 °C for 18 h.d The catalyst was recovered and reused for the 3rd run.e The catalyst was the mixture of Cu1Al1Ox (3.38 g) and Ni1Al4Ox (1.51 g). | |||||
| 1 | Cu1Ni4Al4Ox | 100 | 18.7 | 46.1 | 64.8 |
| 2 | Cu1Ni1Al1.6Ox | 100 | 5.8 | 56.3 | 62.1 |
| 3 | Cu4Ni1Al4Ox | 100 | 8.1 | 75.2 | 83.3 |
| 4 | Cu6Ni1Al5.6Ox | 100 | 5.6 | 60.4 | 66.0 |
| 5 | Cu19Ni1Al16Ox | 100 | 13.9 | 36.6 | 50.5 |
| 6 | Cu1Al1Ox | 100 | 8.1 | 36.9 | 45.0 |
| 7 | Ni1Al4Ox | 100 | 26.0 | 45.5 | 71.5 |
| 8c | Cu4Ni1Al4Ox | 100 | 9.5 | 85.9 | 95.4 |
| 9d | Cu4Ni1Al4Ox | 100 | 5.9 | 63.7 | 69.6 |
| 10e | Cu1Al1Ox/Ni1Al4Ox | 100 | 12.6 | 60.2 | 72.8 |
Clearly, with the increase of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni ratios in these catalysts, the yield of BAF and mass balance of the reaction were gradually improved, and a 75.2% yield of BAF with 83.3% mass balance was achieved when Cu4Ni1Al4Ox was used as a catalyst (Table 1, entry 3). However, both the yield of BAF and the mass balance were obviously decreased when the Cu
Ni ratios in these catalysts, the yield of BAF and mass balance of the reaction were gradually improved, and a 75.2% yield of BAF with 83.3% mass balance was achieved when Cu4Ni1Al4Ox was used as a catalyst (Table 1, entry 3). However, both the yield of BAF and the mass balance were obviously decreased when the Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni ratio was further increased. Possibly, the increasing of Cu
Ni ratio was further increased. Possibly, the increasing of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni ratio would increase the generation of di-aldehyde, which might result in the formation of oligomers and lower yield of BAF. When Cu1Al1Ox and Ni1Al4Ox were prepared by using the same procedure as Cu4Ni1Al4Ox, and subsequently used as the catalysts in direct amination of HMF, the yields of BAF were <50% (Table 1, entries 6 and 7). It should be noted that the lower yield of 60.3% was obtained with physical mixtures of Cu1Al1Ox (3.38 g) and Ni1Al4Ox (1.51 g) as catalyst. These results suggested that the synergetic effect of Cu and Ni species might be crucial to realize this transformation. Then, the effect of the catalyst loadings on amination of 5-HMF with NH3 was studied (Table S1, ESI†). The yield of BAF was decreased to 65.9% with 76.2% mass balance when the amount of Cu4Ni1Al4Ox was reduced to 1.5 g. Additionally, increasing the amount of Cu4Ni1Al4Ox to 2.5 g gave the similar result as the 2.0 g Cu4Ni1Al4Ox. The influence of the amounts of Na2CO3 as co-catalyst and NH3 on the catalytic reaction was also investigated, and it was discovered that 8.6 mmol Na2CO3 and 1352.9 mmol NH3 are the optimal amounts (Tables S2 and S3, ESI†). Na2CO3 act as a base to promote the hydrogen transfer reaction. It should be noted that the addition of excess NH3 is indispensable to increase the selectivity towards the primary amine, thus achieving good BAF yield and mass balance. Furthermore, the impact of H2 pressure was studied (Table S4, ESI†). Lower BAF yield and mass balance were obtained when H2 pressure was reduced from 4.5 MPa to 3.0 MPa. H2 shifts the equilibrium from imine to amine product, and also maintains the catalyst activity by avoiding carbonaceous deposits. Next, temperature screening of the second stage of the reaction showed that reducing the reaction temperature to 190 °C lowered the yield of BAF and substantially increased the yield of HMFA (2a), while raising the reaction temperature to 220 °C decreased mass balance (Table S5, ESI†). This observation suggests that 210 °C is the suitable temperature for the reaction. Investigation of the reaction time of first and second stage demonstrated that higher yield of 85.9% with 95.4% mass balance was afforded by prolonging the reaction time of first stage from 6 h to 9 h (Table 1, entry 8 and Tables S6 and S7, ESI†). It should be noted that HMFA was only produced after the first reaction stage, and the yield of BAF gradually increased to 75.2% after 18 h at the second stage, and remained constant even after prolonging the reaction time to 34 h. The above results clearly indicated that the reaction proceeded via reductive amination of aldehyde functional group of 5-HMF with NH3 at 90 °C, followed by hydrogen-borrowing reaction of hydroxyl group with NH3 at 210 °C. Finally, the Cu4Ni1Al4Ox is easily recovered by simple filtration and it could be used for three catalytic cycles without significant decline in catalytic performance, indicating the good stability of the catalyst (Table 1, entry 9). In order to figure out whether the reaction takes place homogeneously or heterogeneously, a leaching experiment was performed (Fig. S4†). After 2 h of reaction of HMF with NH3 over Cu4Ni1Al4Ox at 210 °C and separating the solid catalysts, further reaction was operated on the residual liquids for another 16 h. Clearly, the reaction stopped after removing the solid catalyst. This observation demonstrates that the leached Cu and Ni should not be the active species.
Ni ratio would increase the generation of di-aldehyde, which might result in the formation of oligomers and lower yield of BAF. When Cu1Al1Ox and Ni1Al4Ox were prepared by using the same procedure as Cu4Ni1Al4Ox, and subsequently used as the catalysts in direct amination of HMF, the yields of BAF were <50% (Table 1, entries 6 and 7). It should be noted that the lower yield of 60.3% was obtained with physical mixtures of Cu1Al1Ox (3.38 g) and Ni1Al4Ox (1.51 g) as catalyst. These results suggested that the synergetic effect of Cu and Ni species might be crucial to realize this transformation. Then, the effect of the catalyst loadings on amination of 5-HMF with NH3 was studied (Table S1, ESI†). The yield of BAF was decreased to 65.9% with 76.2% mass balance when the amount of Cu4Ni1Al4Ox was reduced to 1.5 g. Additionally, increasing the amount of Cu4Ni1Al4Ox to 2.5 g gave the similar result as the 2.0 g Cu4Ni1Al4Ox. The influence of the amounts of Na2CO3 as co-catalyst and NH3 on the catalytic reaction was also investigated, and it was discovered that 8.6 mmol Na2CO3 and 1352.9 mmol NH3 are the optimal amounts (Tables S2 and S3, ESI†). Na2CO3 act as a base to promote the hydrogen transfer reaction. It should be noted that the addition of excess NH3 is indispensable to increase the selectivity towards the primary amine, thus achieving good BAF yield and mass balance. Furthermore, the impact of H2 pressure was studied (Table S4, ESI†). Lower BAF yield and mass balance were obtained when H2 pressure was reduced from 4.5 MPa to 3.0 MPa. H2 shifts the equilibrium from imine to amine product, and also maintains the catalyst activity by avoiding carbonaceous deposits. Next, temperature screening of the second stage of the reaction showed that reducing the reaction temperature to 190 °C lowered the yield of BAF and substantially increased the yield of HMFA (2a), while raising the reaction temperature to 220 °C decreased mass balance (Table S5, ESI†). This observation suggests that 210 °C is the suitable temperature for the reaction. Investigation of the reaction time of first and second stage demonstrated that higher yield of 85.9% with 95.4% mass balance was afforded by prolonging the reaction time of first stage from 6 h to 9 h (Table 1, entry 8 and Tables S6 and S7, ESI†). It should be noted that HMFA was only produced after the first reaction stage, and the yield of BAF gradually increased to 75.2% after 18 h at the second stage, and remained constant even after prolonging the reaction time to 34 h. The above results clearly indicated that the reaction proceeded via reductive amination of aldehyde functional group of 5-HMF with NH3 at 90 °C, followed by hydrogen-borrowing reaction of hydroxyl group with NH3 at 210 °C. Finally, the Cu4Ni1Al4Ox is easily recovered by simple filtration and it could be used for three catalytic cycles without significant decline in catalytic performance, indicating the good stability of the catalyst (Table 1, entry 9). In order to figure out whether the reaction takes place homogeneously or heterogeneously, a leaching experiment was performed (Fig. S4†). After 2 h of reaction of HMF with NH3 over Cu4Ni1Al4Ox at 210 °C and separating the solid catalysts, further reaction was operated on the residual liquids for another 16 h. Clearly, the reaction stopped after removing the solid catalyst. This observation demonstrates that the leached Cu and Ni should not be the active species.
The prepared catalytic samples were characterized by X-ray diffraction (XRD), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and N2 adsorption–desorption to reveal their structures. At first, XRD patterns of the different CuNiAlOx catalysts were obtained to elucidate the crystal structures of the samples (Fig. 1).
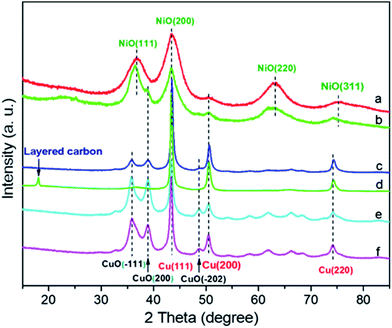 | ||
| Fig. 1 XRD patterns of fresh and reused CuNiAlOx catalysts. (a) Cu1Ni4Al4Ox, (b) Cu1Ni1Al1.6Ox, (c) Cu4Ni1Al4Ox, (d) reused Cu4Ni1Al4Ox, (e) Cu6Ni1Al5.6Ox, (f) Cu19Ni1Al16Ox. | ||
The Cu1Ni4Al4Ox catalyst shows diffraction peaks at 36.9°, 43.5°, 63.2°, and 75.4°, which can be ascribed to the diffraction peaks of NiO(111), NiO(200), NiO(220), and NiO(311) (JCPDS card no: 47-1049). For the Cu1Ni1Al1.6Ox, except for the diffraction peaks of the NiO, additional reflections are also visible at 38.9°, and 50.4°, which can be assigned to CuO(200) and Cu(200), respectively (CuO, JCPDS card no: 89-5899; Cu, JCPDS card no: 89-2838). With the increase of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al ratios in these catalysts, characteristic Cu(111), Cu(200), Cu(220), CuO(−111), CuO(200), and CuO(−202) reflections appearing at 43.2°, 50.4°, 74.1°, 35.6°, 38.9°, and 48.9°, respectively, were detected for Cu4Ni1Al4Ox, Cu6Ni1Al5.6Ox, and Cu19Ni1Al16Ox (CuO, JCPDS card no: 89-5899; Cu, JCPDS card no: 89-2838). Moreover, there were no peaks observed for Ni species for Cu4Ni1Al4Ox, Cu6Ni1Al5.6Ox, and Cu19Ni1Al16Ox, which implied that the nickel species might be amorphous state or highly dispersed. Compared with fresh Cu4Ni1Al4Ox, a new diffraction peak located at 18.0° indicating formation of the layered carbon (JCPDS card no: 50-1363) and disappearance of CuO phase were observed in Cu4Ni1Al4Ox catalyst reused for three times. Thus, we suppose that the Cu in CuNiAlOx catalysts might promote the borrowing-hydrogen reaction but the role of CuO is still unclear14 and speculate that a slight loss of the catalytic activity could be attributed to formation of the layered carbon on the surface of Cu4Ni1Al4Ox catalyst.
Al ratios in these catalysts, characteristic Cu(111), Cu(200), Cu(220), CuO(−111), CuO(200), and CuO(−202) reflections appearing at 43.2°, 50.4°, 74.1°, 35.6°, 38.9°, and 48.9°, respectively, were detected for Cu4Ni1Al4Ox, Cu6Ni1Al5.6Ox, and Cu19Ni1Al16Ox (CuO, JCPDS card no: 89-5899; Cu, JCPDS card no: 89-2838). Moreover, there were no peaks observed for Ni species for Cu4Ni1Al4Ox, Cu6Ni1Al5.6Ox, and Cu19Ni1Al16Ox, which implied that the nickel species might be amorphous state or highly dispersed. Compared with fresh Cu4Ni1Al4Ox, a new diffraction peak located at 18.0° indicating formation of the layered carbon (JCPDS card no: 50-1363) and disappearance of CuO phase were observed in Cu4Ni1Al4Ox catalyst reused for three times. Thus, we suppose that the Cu in CuNiAlOx catalysts might promote the borrowing-hydrogen reaction but the role of CuO is still unclear14 and speculate that a slight loss of the catalytic activity could be attributed to formation of the layered carbon on the surface of Cu4Ni1Al4Ox catalyst.
The CuNiAlOx catalysts were further characterized by TEM. Fig. S1† shows TEM micrographs of CuNiAlOx. TEM analysis (Fig. 2) of the best Cu4Ni1Al4Ox catalyst showed the formation of mainly Cu and Ni nanoparticles with sizes of 10–30 nm. The crystal lattices of Cu(111) and CuO(111) can be observed clearly in the magnified TEM micrographs. Compared with fresh Cu4Ni1Al4Ox catalyst, the HRTEM images of the catalyst which was used three times showed that the crystal lattices of Cu(111) was maintained and the crystal lattices of CuO(111) was not observable, which are in good agreement with XRD results.
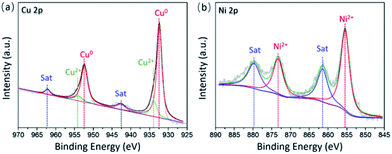
Furthermore, the surface composition of the CuNiAlOx catalysts was analyzed by XPS. The XPS spectra of Cu 2p of fresh Cu4Ni1Al4Ox is shown in Fig. 3a.
The XPS spectra of fresh Cu4Ni1Al4Ox display two main peaks at 932.4 and 952.3 eV that can be attributed to Cu 2p3/2 and Cu 2p1/2 binding energies of Cu0 or Cu1+. Considering that metallic copper was observed from XRD patterns and HRTEM image of the Cu4Ni1Al4Ox catalyst, we suppose that binding energy of 932.4 eV might be assigned to the metallic copper. Other binding energies at 933.9 eV for Cu 2p3/2 and 953.9 eV for Cu 2p1/2 are ascribed to Cu2+. In addition to these peaks, shake-up satellite peaks were observed in the region of 940–945 eV (Cu 2p3/2) and 959-965 eV (Cu 2p1/2), also indicating the presence of Cu2+ species in Cu4Ni1Al4Ox catalyst. According to XPS-peak-differentiation-imitating analysis, the ratio of Cu0/Cu2+ is 4.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Fig. 3b shows Ni 2p spectra of Cu4Ni1Al4Ox catalysts. The peaks of the Ni 2p XPS spectrum at 855.4 eV (Ni 2p3/2) and 856.7 eV (Ni 2p1/2) were in accordance with NiO. Besides these peaks, shake-up satellite peaks were observed in the region of 857–865 eV (Ni 2p3/2) and 877–884 eV (Ni 2p1/2). Additional spectra for the other catalysts can be found in the ESI (Fig. S2†).
1. Fig. 3b shows Ni 2p spectra of Cu4Ni1Al4Ox catalysts. The peaks of the Ni 2p XPS spectrum at 855.4 eV (Ni 2p3/2) and 856.7 eV (Ni 2p1/2) were in accordance with NiO. Besides these peaks, shake-up satellite peaks were observed in the region of 857–865 eV (Ni 2p3/2) and 877–884 eV (Ni 2p1/2). Additional spectra for the other catalysts can be found in the ESI (Fig. S2†).
The N2 adsorption–desorption tests (Fig. S3, ESI†) showed that the BET surface areas of the Cu1Ni4Al4Ox, Cu1Ni1Al1.6Ox, Cu4Ni1Al4Ox, Cu6Ni1Al5.6Ox, Cu19Ni1Al16Ox, Cu1Al1Ox and Ni1Al4Ox were 202.39, 169.75, 101.16, 83.35, 68.16, 79.77, and 326.21 m2 g−1, respectively (Table S8, ESI†). Therefore, the high selectivity of Cu4Ni1Al4Ox should not be attributed solely to its high surface area. It can be inferred that the good catalytic performance of Cu4Ni1Al4Ox catalyst in the direct amination of HMF to desired BAF product is attributed to bulk Cu and highly dispersed Ni species for the dehydrogenation and hydrogenation properties, respectively.
In conclusion, we have successfully developed an efficient methodology for highly selective synthesis of biomass-derived BAF via direct amination of 5-HMF with NH3 over bifunctional CuNiAlOx catalyst using two-stage reaction process. By tuning the Cu/Ni molar ratio, Cu4Ni1Al4Ox exhibited excellent catalytic performance in the direct amination of 5-HMF to BAF, affording the desired product in 85.9% yield. Experimental results suggested that the reaction proceeded via reductive amination of aldehyde group of 5-HMF with NH3 at 90 °C, followed by hydrogen-borrowing reaction of hydroxyl of 5-HMF with NH3 at 210 °C. This work offers an effective methodology for the controlled synthesis of biomass-derived diamines monomers.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
Financial supports from NSFC (21802147), the Youth Innovation Promotion Association (2019409), “Light of West China” Program and Fujian Innovation Academy of CAS are gratefully acknowledged. Solvay is gratefully acknowledged for funding.Notes and references
- (a) E. Karsten, H. Erhard, R. Roland and H. Hartmut, Amines, Aliphatic in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005 Search PubMed; (b) A. Ricci, Amino group chemistry: from synthesis to the life sciences, John Wiley & Sons, 2008 Search PubMed.
- (a) D. K. Chattopadhyay and K. V. S. N. Raju, Prog. Polym. Sci., 2007, 32, 352–418 CrossRef CAS; (b) J. M. García, F. C. García, F. Serna and L. D. L. P. José, Prog. Polym. Sci., 2010, 35, 623–686 CrossRef.
- (a) A. Corma, S. Iborra and A. Velty, Chem. Rev., 2007, 107, 2411–2502 CrossRef CAS PubMed; (b) D. Li, W. Ni and Z. Hou, Chin. J. Catal., 2017, 38, 1784–1793 CrossRef CAS.
- (a) I. Delidovich, P. J. Hausoul, L. Deng, R. Pfutzenreuter, M. Rose and R. Palkovits, Chem. Rev., 2016, 116, 1540–1599 CrossRef CAS PubMed; (b) V. Froidevaux, C. Negrell, S. Caillol, J. P. Pascault and B. Boutevin, Chem. Rev., 2016, 116, 14181–14224 CrossRef CAS PubMed; (c) M. Pelckmans, T. Renders, S. Van de Vyver and B. F. Sels, Green Chem., 2017, 19, 5303–5331 RSC; (d) M. Pera-Titus and F. Shi, ChemSusChem, 2014, 7, 720–722 CrossRef CAS PubMed.
- (a) P. Pal and S. Saravanamurugan, ChemSusChem, 2019, 12, 145–163 CrossRef CAS PubMed; (b) H. Yuan, J.-P. Li, F. Su, Z. Yan, B. T. Kusema, S. Streiff, Y. Huang, M. Pera-Titus and F. Shi, ACS Omega, 2019, 4, 2510–2516 CrossRef CAS PubMed; (c) F. Gao, J. Chen, Z. Huang and C. Xia, J. Mol. Catal., 2018, 32, 276–293 CAS.
- N.-T. Le, A. Byun, Y. Han, K.-I. Lee and H. Kim, Green Sustainable Chem., 2015, 05, 115–127 CrossRef CAS.
- T. Komanoya, T. Kinemura, Y. Kita, K. Kamata and M. Hara, J. Am. Chem. Soc., 2017, 139, 11493–11499 CrossRef CAS PubMed.
- X. Wang, W. Chen, Z. Li, X. Zeng, X. Tang, Y. Sun, T. Lei and L. Lin, J. Energy Chem., 2018, 27, 209–214 CrossRef.
- Y. Xu, X. Jia, J. Ma, J. Gao, F. Xia, X. Li and J. Xu, Green Chem., 2018, 20, 2697–2701 RSC.
- D. Pingen, J. B. Schwaderer, J. Walter, J. Wen, G. Murray, D. Vogt and S. Mecking, ChemCatChem, 2018, 10, 3027–3033 CrossRef CAS.
- K. Zhou, H. Liu, H. Shu, S. Xiao, D. Guo, Y. Liu, Z. Wei and X. Li, ChemCatChem, 2019, 11, 2649–2656 CrossRef CAS.
- K.-I. Shimizu, Catal. Sci. Technol., 2015, 5, 1412–1427 RSC.
- X. Cui, X. Dai, Y. Deng and F. Shi, Chem.–Eur. J., 2013, 19, 3665–3675 CrossRef CAS PubMed.
- (a) J. Aston, T. Peterson and J. Holowchak, J. Am. Chem. Soc., 1934, 56, 153–154 CrossRef CAS; (b) T. Yamakawa, I. Tsuchiya, D. Mitsuzuka and T. Ogawa, Catal. Commun., 2004, 5, 291–295 CrossRef CAS; (c) Y. Wu, H. Huang, X. Dai and F. Shi, ChemSusChem, 2019, 12, 3185–3191 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c9ra08560b |
| This journal is © The Royal Society of Chemistry 2019 |