Open Access Article
Open Access Article1,1-Difluoroethyl chloride (CH3CF2Cl), a novel difluoroalkylating reagent for 1,1-difluoroethylation of arylboronic acids†
Jianchang Liua,
Jida Zhanga,
Chaolin Wua,
Hefu Liua,
Hui Liu a,
Fenggang Suna,
Yueyun Li
a,
Fenggang Suna,
Yueyun Li a,
Yuying Liua,
Yunhui Donga and
Xinjin Li
a,
Yuying Liua,
Yunhui Donga and
Xinjin Li *ab
*ab
aCollege of Chemistry and Chemical Engineering, Shandong University of Technology, 266 West Xincun Road, Zibo, 255000, China. E-mail: lixj@sdut.edu.cn
bKey Laboratory of Organofluorine Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, 345 Lingling Road, Shanghai 200032, China
First published on 9th September 2019
Abstract
1,1-Difluoroethylated aromatics are of great importance in medicinal chemistry and related fields. 1,1-Difluoroethyl chloride (CH3CF2Cl), a cheap and abundant industrial raw material, is viewed as an ideal 1,1-difluoroethylating reagent, but the direct introduction of the difluoroethyl (CF2CH3) group onto aromatic rings using CH3CF2Cl has not been successfully accomplished. Herein, we disclose a nickel-catalyzed 1,1-difluoroethylation of arylboronic acids with CH3CF2Cl for the synthesis of (1,1-difluoroethyl)arenes.
Organic fluorine compounds have attracted extensive attention in recent years, since the introduction of fluorine atom(s) into organic molecules often results in dramatic changes in physical, chemical and biological properties.1,2 Among them, aromatic compounds containing the difluoroethyl (CF2CH3) group are of great importance, because it mimics the steric and electronic features of a methoxy group, which makes it a significant group for drug design.3 For instance, a triazolopyrimidine-based dihydroorotate dehydrogenase (DHODH) inhibitor4 shows remarkable advantage in terms of potency due to the replacement of a methoxy group by a difluoroethyl group (Scheme 1A). LSZ102,5 a clinical agent, is currently applied in phase I/Ib trials for the treatment of estrogen receptor alpha-positive breast cancer (Scheme 1A). Thus, the invention of reagents or methods for the synthesis of (1,1-difluoroethyl)arenes is a very appealing and pretty meaningful task.
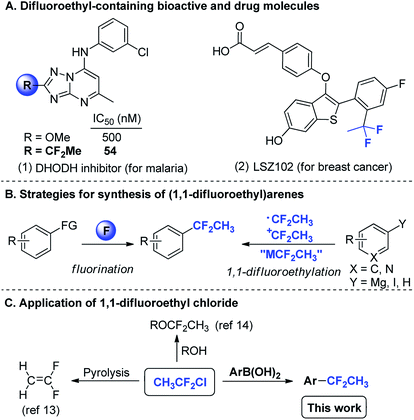 | ||
| Scheme 1 (A) Difluoroethyl-containing bioactive and drug molecules; (B) strategies for synthesis of (1,1-difluoroethyl)arenes. M = metal; (C) application of 1,1-difluoroethyl chloride. | ||
The synthesis of such compounds are generally accomplished by two strategies:6 one is the transformation of a functional group to a difluoromethylene (CF2) group or a CF2CH3 moiety, such as nucleophilic fluorination of ketones or their derivatives,7 dihydrofluorination of terminal arynes,8 and benzylic C–H fluorination;9 the other is the direct introduction of a CF2CH3 moiety onto aromatic rings, including nucleophilic,10 electrophilic11 and radical 1,1-difluoroethylation12 (Scheme 1B). In spite of these important accomplishments, it is still a key challenge to develop low-cost and easily available difluoroalkylating reagents for synthesis of (1,1-difluoroethyl)arenes.
1,1-Difluoroethyl chloride (CH3CF2Cl; HCFC-142b; bp = −9.5 °C), a cheap and abundant industrial raw material used for vinylidene fluoride (VDF),13 is viewed as an ideal source to prepare difluoroethylated derivatives.14 We envisioned that the direct introduction of CF2CH3 onto aromatic rings could be through transition-metal-catalyzed 1,1-difluoroethylation of arylboronic acids with CH3CF2Cl (Scheme 1C). Although transition-metal-catalyzed difluoroalkylation of aromatics using activated difluoroalkyl halides (RCF2X, R = π system) has been successfully reported,15 the use of unactivated RCF2X (R = alkyl) for synthesis of difluoroalkylarenes was rarely reported.11,16 To the best of our knowledge, the use of CH3CF2Cl to prepare Ar–CF2CH3 compounds through a Suzuki-type cross-coupling reaction has not been reported and remains challenges because of the difficulties in activating the inert C–Cl bond of CH3CF2Cl and in suppressing homo-coupling and deboronation of arylboronic acids. Herein, we wish to disclose a nickel-catalyzed 1,1-difluoroethylation of arylboronic acids with CH3CF2Cl.
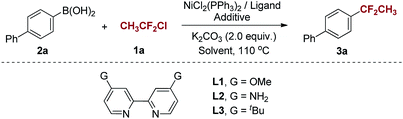
At the onset of our investigation, 4-biphenylboronic acid (2a) was chosen as the model substrate for the nickel-catalyzed 1,1-difluoroethylation reaction (Table 1). To our delight, the product 3a was obtained in 5% yield in the presence of NiCl2(PPh3)2 (5 mol%), 2,2′-bipyridine (5 mol%), K2CO3 (2.0 equiv.) in DME at 110 °C (Table 1, entry 1). The low yield should be due to the residual 2a and the formed homo-coupling and deboronation of 2a (determined by GC-MS). To improve the reaction efficiency, different diamine ligands were examined (Table 1, entries 2–5). As a result, diOMe-bpy (L1) improved the yield of 3a to 15% (Table 1, entry 2), while diNH2-bpy (L2) and ditBu-bpy (L3) gave poor yields of 3a (Table 1, entry 3 and 4). 1,10-Phenanthroline (phen), commonly used in copper-catalyzed reactions, led to a negative result in current reaction (Table 1, entry 5). With the optimal combination of Ni and ligand established (for details, see ESI†), we next investigated the influence of additives and solvent. As previously reported,16,17 pyridine derivatives could promote Ni-catalyzed Suzuki–Miyaura cross-coupling reactions. Thus, we attempted the current reaction in the presence of pyridine and its derivatives. As expected, the use of 4-(N,N-dimethylamino)pyridine (DMAP) afforded a higher yield of 3a (Table 1, entry 8), while pyridine (Py) and 4-cyanopyridine (4-CNPy) proved to be ineffective for the reaction (Table 1, entry 6 and 7). To improve the yield of 3a, we increased the loading of DMAP to 50 mol% and the yield was correspondingly enhanced to 49% (Table 1, entry 9). Consequently, the use of 70 mol% of DMAP provided the desired product in 61% yield (Table 1, entry 10). In addition, when 3 mol% of NiCl2(PPh3)2 and ligand were employed in the reaction, the yield of 3a was increased to 70% and did little change by prolonging reaction time (Table 1, entry 12). However, only trace of the product was formed in the absence of ligand L1 (Table 1, entry 13), indicating that the diamine ligand is essential in the Ni-catalyzed 1,1-difluoroethylation of arylboronic acids. For further insight the reactivity of other ligands in the presence of DMAP, we conducted the reactions with ligands L2 and L3 under the optimized conditions (Table 1, entry 14 and 15). The results showed that the combination of ligand L2 with DMAP offered a low yield of 24% due to the residue of 2a, and ligand L3 gave a yield of 61% because of a little more deboronation of 2a. For solvent screening (for details, see ESI†), it was found that 1,4-dioxane and DMF were inferior to DME (Table 1, entry 16 and 17).
| Entry | Ligand (mol%) | Additive (mol%) | Solvent | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 2a (0.2 mmol, 1.0 equiv.), 1a (2.0–2.6 mmol), NiCl2(PPh3)2 (5 mol%), K2CO3 (2.0 equiv.), solvent (2 mL), 110 °C, N2, 5 h.b Isolated yield.c NiCl2(PPh3)2 (3 mol%).d The reaction was conducted for 12 h. bpy = 2,2′-bipyridine, phen = 1,10-phenanthroline, DME = 1,2-dimethoxyethane, DMAP = 4-(N,N-dimethylamino)pyridine. | ||||
| 1 | bpy (5) | — | DME | 5 |
| 2 | L1 (5) | — | DME | 15 |
| 3 | L2 (5) | — | DME | 7 |
| 4 | L3 (5) | — | DME | 7 |
| 5 | Phen (5) | — | DME | Trace |
| 6 | L1 (5) | Py (10) | DME | Trace |
| 7 | L1 (5) | 4-CNPy (10) | DME | Trace |
| 8 | L1 (5) | DMAP (10) | DME | 30 |
| 9 | L1 (5) | DMAP (50) | DME | 49 |
| 10 | L1 (5) | DMAP (70) | DME | 61 |
| 11 | L1 (5) | DMAP (100) | DME | 59 |
| 12c | L1 (3) | DMAP (70) | DME | 70 (69)d |
| 13c | — | DMAP (70) | MME | Trace |
| 14c | L2 (3) | DMAP (70) | DME | 24 |
| 15c | L3 (3) | DMAP (70) | DME | 61 |
| 16c | L1 (3) | DMAP (70) | 1,4-Dioxane | 31 |
| 17c | L1 (3) | DMAP (70) | DMF | 21 |
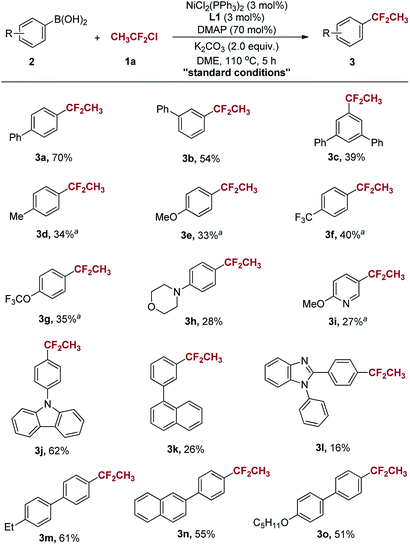
With the optimised conditions in hand, we further explored the substrate scope of the 1,1-difluoroethylation of arylboronic acids with CH3CF2Cl (Scheme 2). The reaction can tolerate a variety of functional groups, such as methyl, methoxyl, trifluoromethyl, trifluoromethoxy and substituted morpholine (Scheme 2, 3d–3h). Generally, the substituent group of arylboronic acids with π-system showed good reactivity toward CH3CF2Cl, affording the desired product in moderate to good yields (Scheme 2, 3a, 3b, 3m–3o). It was found that the reaction afforded slightly low yields with sterically hindered arylboronic acids under the standard conditions (Scheme 2, 3c, 3k, 3l). In addition, this transformation was also applicable to 4-(9H-carbozol-9-yl)phenylboronic acid and the 1,1-difluoroethylated product was obtained in 62% yield (Scheme 2, 3j).
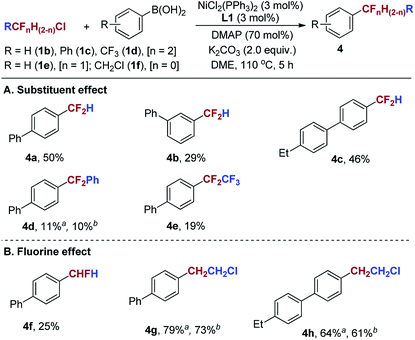
To investigate the substituent effect of fluoroalkyl chlorides on the Suzuki-type reaction, we conducted comparative experiments using HCF2Cl (1b), PhCF2Cl (1c) and CF3CF2Cl (1d) under the standard conditions (Scheme 3A). When arylboronic acids and 1b were subjected to the standard conditions, the reaction provided difluoromethylated products (4a–4c) in moderate yields. However, the use of 1c and 1d gave poor yields of the corresponding products 4d and 4e, respectively. These results indicate that the reactivity of RCF2Cl in the reaction with 2a decreases in the following order: CH3CF2Cl > HCF2Cl > CF3CF2Cl ≈ PhCF2Cl. Next, we intended to explore the fluorine effect on the reaction. Comparative experiments by the use of H2CFCl (1e) and ClCH2CH2Cl (1f) were conducted under the standard conditions (Scheme 3B). It was found that the reaction of 1e with 2a afforded monofluoromethylated product 4f in a low yield, while β-chloroethylarenes (4g, 4h) were obtained in good yields under the standard conditions. These results indicate that the reactivity of RCl in the reaction with 2a decreases in the following order: ClCH2CH2Cl ≈ CH3CF2Cl > H2CFCl.
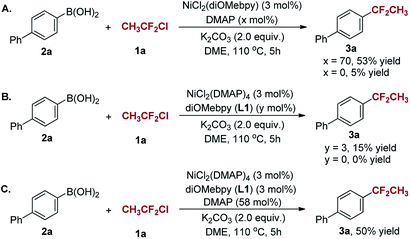
To examine the role of DMAP in the reaction, we prepared nickel complexes NiCl2(diOMebpy) and NiCl2(DMAP)4. Both of them could serve as precatalysts and offered 3a in 50% and 15% yield, respectively (Scheme 4A and B). However, NiCl2(diOMebpy) provided 3a in a low yield (5%) in the absence of DMAP (Scheme 4A) and the use of NiCl2(DMAP)4 resulted in no product without diOMebpy (Scheme 4B). It was noted that the additional DMAP did enhance the yield from 15% to 50% (Scheme 4C). These results demonstrate that one of the roles of DMAP may function as a co-ligand in the Ni-catalyzed reaction. Apparently, the combination of diOMebpy as a bidentate ligand with 70 mol% of DMAP facilitates the Ni-catalyzed 1,1-difluoroethylation of arylboronic acids with CH3CF2Cl.
In order to obtain some insight into the mechanism of the current reaction, radical inhibition and radical clock experiments were conducted (Scheme 5). In the presence of 2,2,6,6-tetramethylpiperidine-1-oxy (TEMPO) as a radical scavenger, the reaction was readily inhibited and compound 5 was detected by 19F NMR and GC-MS. Furthermore, when diallyl ether was added to the reaction under the standard conditions, a ring-closing product 6 was formed (determined by 19F NMR and GC-MS), along with 9% yield of 3a (for details, see ESI†). These results demonstrate that a 1,1-difluoroethyl radical is indeed generated in the reaction.
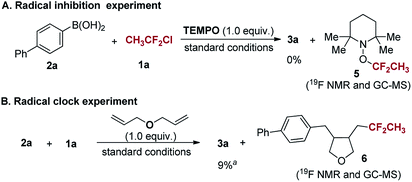 | ||
| Scheme 5 Radical trapping experiments. aThe yield was determined by 19F NMR spectroscopy using PhCF3 as an internal standard. | ||
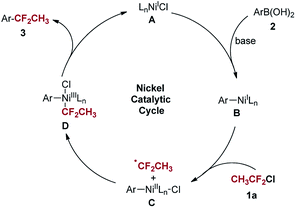
On the basis of these results and previous reports,16,18 a plausible mechanism involving Ni(I)/Ni(III) catalytic cycle was proposed for the 1,1-difluoroethylation reaction (Scheme 6). The LnNi(I)Cl intermediate (A) is supposed to be generated via the comproportionation of initially formed LnNi(0) species and the remaining LnNi(II)Cl2,19 followed by transmetalation with arylboronic acids. The formed LnNi(I)Ar species (B) reacts with CH3CF2Cl through a SET pathway to produce 1,1-difluoroethyl radical and LnNi(II)(Ar)(Cl) species (C). Subsequently, the resulting LnNi(III)(Ar)(CF2CH3)(Cl) species (D) undergoes reductive elimination to give the coupling product 3 and regenerates LnNi(I)Cl to complete the catalytic cycle. It is noted that DMAP may not only function as a co-ligand to coordinated to the nickel center,16,20 but also activate the arylboronic acids to facilitate the transmetalation.17a
In conclusion, the first transition-metal-catalyzed 1,1-difluoroethylation of arylboronic acids with the cheap and easily available CH3CF2Cl has been successfully developed. This method can tolerate methyl, methoxyl, trifluoromethyl and heteroarenes, affording 1,1-difluoroethylated products in moderate to good yields. The reactivity of different alkyl chlorides in the reaction was also investigated. Initial mechanism study showed the nickel-catalyzed 1,1-difluoroethylation probably involves a NiI/III process. Current efforts are to develop catalytic system for improving yields and novel reactions with CH3CF2Cl as cheap fluorine source.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by Natural Science Foundation of Shandong Province (No. ZR2017BB052), the Doctoral Scientific Research Foundation of Shandong University of Technology (No. 4041-416021) and open project foundation of Key Laboratory of Organofluorine Chemistry, Chinese Academy of Sciences. We thank Professor Jinbo Hu (SIOC) for helpful discussions.Notes and references
- (a) J.-P. Bégué and D. Bonnet-Delpon, Bioorganic and Medicinal Chemistry of Fluorine, Wiley, Hoboken, NJ, 2008 CrossRef; (b) K. Uneyama, Organofluorine Chemistry, Blackwell, Oxford, U.K., 2006 CrossRef; (c) D. A. Nagib and D. W. C. MacMillan, Nature, 2011, 480, 224 CrossRef CAS; (d) T. Furuya, A. S. Kamlet and T. Ritter, Nature, 2011, 473, 470 CrossRef CAS PubMed; (e) Y. Fujiwara, J. A. Dixon, F. O'Hara, E. D. Funder, D. D. Dixon, R. A. Rodriguez, R. D. Baxter, B. Herlé, N. Sach, M. R. Collins, Y. Ishihara and P. S. Baran, Nature, 2012, 492, 95 CrossRef CAS PubMed.
- For reviews, see: (a) M. Schlosser, Angew. Chem., Int. Ed., 2006, 45, 5432 CrossRef CAS PubMed; (b) D. O'Hagan, Chem. Soc. Rev., 2008, 37, 308 RSC; (c) T. Furuya, J. E. M. N. Klein and T. Ritter, Synthesis, 2010, 1804 CAS; (d) O. A. Tomashenko and V. V. Grushin, Chem. Rev., 2011, 111, 4475 CrossRef CAS PubMed; (e) X.-F. Wu, H. Neumann and M. Beller, Chem.–Asian J., 2012, 7, 1744 CrossRef CAS PubMed; (f) F.-L. Qing, Chin. J. Org. Chem., 2012, 32, 815 CrossRef CAS; (g) C. Ni, M. Hu and J. Hu, Chem. Rev., 2015, 115, 765 CrossRef CAS PubMed.
- (a) P. Kirsch, Modern fluoroorganic chemistry: synthesis, reactivity, applications, Wiley-VCH, Weinheim, 2013 CrossRef; (b) G. M. Blackburn, D. E. Kent and F. J. Kolkmann, J. Chem. Soc., Perkin Trans. 1, 1984, 1119 RSC; (c) D. B. Berkowitz and M. Bose, J. Fluorine Chem., 2001, 112, 13 CrossRef CAS; (d) D. O'Hagan, Y. Wang, M. Skibinski and A. M. Z. Slawin, Pure Appl. Chem., 2012, 84, 1587 Search PubMed.
- M. O. Anderson, J. Zhang, Y. Liu, C. Yao, P.-W. Phuan and A. S. Verkman, J. Med. Chem., 2012, 55, 5942 CrossRef CAS.
- G. S. Tria, T. Abrams, J. Baird, H. E. Burks, B. Firestone, L. A. Gaither, L. G. Hamann, G. He, C. A. Kirby, S. Kim, F. Lombardo, K. J. Macchi, D. P. McDonnell, Y. Mishina, J. D. Norris, J. Nunez, C. Springer, Y. Sun, N. M. Thomsen, C. Wang, J. Wang, B. Yu, C.-L. Tiong-Yip and S. Peukert, J. Med. Chem., 2018, 61, 2837 CrossRef CAS PubMed.
- For a review, see: X. Li, J. Liu, X. Li, H. Liu, H. Liu, Y. Li, Y. Liu and Y. Dong, J. Fluorine Chem., 2018, 216, 102 CrossRef CAS.
- (a) L. N. Markovskij, V. E. Pashinnik and A. V. Kirsanov, Synthesis, 1973, 787 CrossRef; (b) G. S. Lal, G. P. Pez, R. J. Pesaresi and F. M. Prozonic, Chem. Commun., 1999, 215 RSC; (c) C. York, G. K. S. Prakash and G. A. Olah, Tetrahedron, 1996, 52, 9 CrossRef CAS.
- O. E. Okoromoba, J. Han, G. B. Hammond and B. Xu, J. Am. Chem. Soc., 2014, 136, 14381 CrossRef CAS PubMed.
- (a) J.-B. Xia, C. Zhu and C. Chen, J. Am. Chem. Soc., 2013, 135, 17494 CrossRef CAS; (b) P. Xu, S. Guo, L. Wang and P. Tang, Angew. Chem., Int. Ed., 2014, 53, 5955 CrossRef CAS; (c) J. Ma, W. Yi, G. Lu and C. Cai, Org. Biomol. Chem., 2015, 13, 2890 RSC; (d) A. M. Hua, D. N. Mai, R. Martinez and R. D. Baxter, Org. Lett., 2017, 19, 2949 CrossRef CAS PubMed.
- X. Li, J. Zhao, Y. Wang, J. Rong, M. Hu, D. Chen, P. Xiao, C. Ni, L. Wang and J. Hu, Chem.–Asian J., 2016, 11, 1789 CrossRef CAS PubMed.
- (a) Y. Ohtsuka and T. Yamakawa, J. Fluorine Chem., 2016, 185, 96 CrossRef CAS; (b) L. An, Y.-L. Xiao, S. Zhang and X. Zhang, Angew. Chem., Int. Ed., 2018, 57, 6921 CrossRef CAS PubMed.
- (a) Q. Zhou, A. Ruffoni, R. Gianatassio, Y. Fujiwara, E. Sella, D. Shabat and P. S. Baran, Angew. Chem., Int. Ed., 2013, 52, 3949 CrossRef CAS PubMed; (b) J. Rong, L. Deng, P. Tan, C. Ni, Y. Gu and J. Hu, Angew. Chem., Int. Ed., 2016, 55, 2743 CrossRef CAS PubMed.
- F. B. Downing, A. F. Benning, and R. C. McHarness, US Pat., US 2551573, 1945.
- A. L. Henne and M. A. Smook, J. Am. Chem. Soc., 1950, 72, 4378 CrossRef CAS.
- (a) Z. Feng, Q.-Q. Min, Y.-L. Xiao, B. Zhang and X. Zhang, Angew. Chem., Int. Ed., 2014, 53, 1669 CrossRef CAS PubMed; (b) Q.-Q. Min, Z. Yin, Z. Feng, W.-H. Guo and X. Zhang, J. Am. Chem. Soc., 2014, 136, 1230 CrossRef CAS PubMed; (c) Y.-L. Xiao, W.-H. Guo, G.-Z. He, Q. Pan and X. Zhang, Angew. Chem., Int. Ed., 2014, 53, 9909 CrossRef CAS PubMed.
- Y.-L. Xiao, Q.-Q. Min, C. Xu, R.-W. Wang and X. Zhang, Angew. Chem., Int. Ed., 2016, 55, 5837 CrossRef CAS PubMed.
- (a) L. An, Y.-L. Xiao, Q.-Q. Min and X. Zhang, Angew. Chem., Int. Ed., 2015, 54, 9079 CrossRef CAS PubMed; (b) X.-P. Xia, Y.-L. Xiao and X. Zhang, Chin. J. Chem., 2018, 36, 143 CrossRef.
- S. L. Zultanski and G. C. Fu, J. Am. Chem. Soc., 2013, 135, 624 CrossRef CAS PubMed.
- J. Cornella, E. Gómez-Bengoa and R. Martin, J. Am. Chem. Soc., 2013, 135, 1997 CrossRef CAS PubMed.
- C. Xu, W.-H. Guo, X. He, Y.-L. Guo, X.-Y. Zhang and X. Zhang, Nat. Commun., 2018, 9, 1170 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed experimental procedures and spectroscopic data. See DOI: 10.1039/c9ra06406k |
| This journal is © The Royal Society of Chemistry 2019 |