Preactivation-based, iterative one-pot synthesis of anticoagulant pentasaccharide fondaparinux sodium†
Abstract
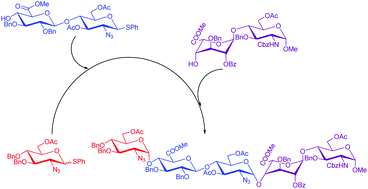
A preactivation-based, one-pot glycosylation method with exclusive stereoselectivity was accomplished for the rapid synthesis of fondaparinux. Two convergent routes for assembling three glucosamine building blocks and two uronic acid donors were developed, and these routes greatly simplified the preparation procedures and improved the synthetic efficiency. Both disaccharide acceptors were obtained efficiently by glycosylation. This strategy offers another possibility for the rapid synthesis of fondaparinux.


 Please wait while we load your content...
Please wait while we load your content...