Double signal amplification through a functionalized nanoporous Au–Ag alloy microwire and Au nanoparticles: development of an electrochemical ˙OH sensor based on a self-assembled layer of 6-(ferrocenyl)hexanethiol†
Abstract
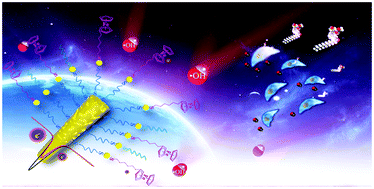
Novel electrochemical sensors were developed based on a FcHT functionalized NPAMW and AuNPs for the analysis of ˙OH released from live cells. Self-assembled layers of FcHT can be selectively attacked by ˙OH, enabling simultaneous recognition and quantification of ˙OH, while the NPAMW and AuNPs provide a 3D multiplexed conductive structure rendering dual signal amplification.


 Please wait while we load your content...
Please wait while we load your content...