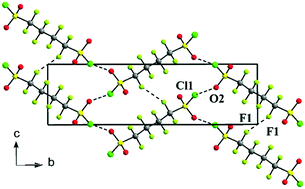
Cooperative intermolecular S–Cl⋯O and F⋯F associations in the crystal packing of α,ω-di(sulfonyl chloride) perfluoroalkanes, ClSO2(CF2)nSO2Cl, where n = 4, 6†
Abstract
Single-crystal, X-ray crystallographic studies of ClSO2(CF2)4SO2Cl and ClSO2(CF2)6SO2Cl reveal the first examples of S–Cl⋯O halogen bonding complemented by short F⋯F contacts between neighbouring chains that result in stabilized crystals. These compounds allow the study of intermolecular associations involving halogens where hydrogen bonding cannot be operative.
- This article is part of the themed collection: The halogen bond: a new avenue in recognition and self-assembly


 Please wait while we load your content...
Please wait while we load your content...