One-step microwave synthesis of carbon dots for highly sensitive and selective detection of copper ions in aqueous solution†
Abstract
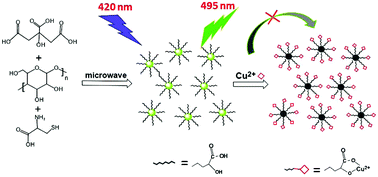
Highly fluorescent carbon dots (CDs) are prepared by a one-step microwave heating method using citric acid, L-cysteine and dextrin as the precursors. The average size of the CDs is 2.61 nm with good water dispersibility and photostability. Compared with undoped CDs, small amounts of nitrogen (N) and sulfur atom (S) co-doped CDs exhibit a red shift of the absorption spectra to the visible region and lead to a high quantum yield of 22%. Due to the large amount of carboxyl and hydroxyl groups (−COOH and −OH) on the surface of the CDs, Cu2+ is selectively detected in aqueous solution at pH 7.0 with a detection limit as low as 20 nM, which is much lower than the maximum concentration of Cu2+ in drinking water (∼20 μM) permitted by the US Environmental Protection Agency (EPA). Moreover, the application in tap water samples further demonstrates that the as-synthesized CDs are good candidates for the monitoring of Cu2+ in environmental water samples.


 Please wait while we load your content...
Please wait while we load your content...