Chemical composition and bioactive properties of the wild mushroom Polyporus squamosus (Huds.) Fr: a study with samples from Romania
Andrei
Mocan
ab,
Ângela
Fernandes
a,
Lillian
Barros
 a,
Gianina
Crişan
b,
Marija
Smiljković
c,
Marina
Soković
a,
Gianina
Crişan
b,
Marija
Smiljković
c,
Marina
Soković
 c and
Isabel C. F. R.
Ferreira
c and
Isabel C. F. R.
Ferreira
 *a
*a
aCentro de Investigação de Montanha (CIMO), Instituto Politécnico de Bragança, Campus de Santa Apolónia, 5300-253 Bragança, Portugal. E-mail: iferreira@ipb.pt; Fax: +351-273-325405; Tel: +351-273-303219
bDepartment of Pharmaceutical Botany, “Iuliu Haţieganu” University of Medicine and Pharmacy, 23 Gheorghe Marinescu Street, 400337 Cluj-Napoca, Romania
cUniversity of Belgrade, Institute for Biological Research “Siniša Stanković”, Department of Plant Physiology, Bulevar Despota Stefana 142, 11000 Belgrade, Serbia
First published on 8th November 2017
Abstract
In Eastern Europe, wild mushrooms are widely collected in mountain areas and used for their medicinal properties or as healthy foods. This study aimed at determining the chemical composition (nutritional value, free sugars, organic acids, phenolic compounds, fatty acids and tocopherols) and bioactive properties (antioxidant, antimicrobial and antiquorum sensing) of wild Polyporus squamosus (Huds.) Fr from Romania. The results indicate that the fruiting bodies of P. squamosus are rich in carbohydrates (74.22 g per 100 g dw) and proteins (18.7 g per 100 g dw). Trehalose was the main free sugar, while malic acid was the organic acid detected in the highest amount (2.21 g per 100 g dw), and p-hydroxybenzoic acid was the main phenolic compound. Among tocopherols, β-tocopherol was the most abundant form (114.7 μg per 100 g dw). Additionally, regarding the fatty acids’ pattern, polyunsaturated acids represent more than 57% of all fatty acids, followed by monounsaturated fatty acids (24.96%). The highest measured antioxidant effect of P. squamosus extract was found using the TBARS inhibition assay (EC50 = 0.22 mg mL−1), followed by the β-carotene/linoleate assay (EC50 = 1.41 mg mL−1). A minimal inhibitory concentration of the tested extracts was obtained between 0.61–20.4 mg mL−1, while the bactericidal effect was achieved between 1.2–40.8 mg mL−1. Antibiofilm potential was obtained at all tested concentrations, and subinhibitory concentrations of the extract exhibited an antiquorum effect and reduced the formation of P. aeruginosa pili, which all together influenced the virulence of this bacterium. Due to the investigated bioactivities and compounds of P. squamosus and its well-balanced nutritional profile, this mushroom can be further used as a medicinal ingredient based on its antioxidative and antimicrobial potential.
1. Introduction
Over the last decade dietary habits have progressively changed, at a global scale, with nutraceuticals, functional foods and food supplements receiving an increasing demand from consumers, and consequently gaining scientific interest from academia.1 Nowadays considering the extensive evidence of health-promoting abilities of different food classes, great attention is given by the consumers and food industry to natural foods, especially to wild foods coming from unpolluted areas (i.e. mountains).2,3 Wild edibles include annual and perennial herbs, forbs, vines, sedges and rushes, grasses, broadleaved and needle-like or scale-like leaved shrubs, trees, ferns, as well as mushrooms, algae and lichens.2,4Mushrooms possess an important nutritional value since they are quite rich in proteins, having a high content of essential amino acids and fibers, and being poor in fat. Moreover, edible mushrooms represent important sources of vitamins (B1, B2, B12, C, D, and E).5–8 Additionally, they represent an important source of different bioactive compounds such as unsaturated fatty acids, phenolic compounds, terpenoids, and carotenoids, and can be ultimately developed into functional foods, nutraceuticals or pharmaceutical products, taking advantage of their unique chemical features and the advantage of the additive and synergistic effects of all their panel of bioactive compounds.5,8–13
The usage of wild edible mushrooms in cooking, traditional medicine, and religious rituals is a very well established and appreciated practice which has been maintained and developed in different cultures and civilizations around the world, particularly in South and Central-Eastern Europe, China, Mexico and Latin America.14–17 Moreover, the consumption of wild growing mushrooms is currently preferred to eating of cultivated fungi in many countries of Central and Eastern Europe.18 Collecting them is very popular in numerous regions in the mountain areas, representing an important component of the human diet, and the main proportion amongst other wild foods.19 However, despite a lack of accurate data about the use of wild mushrooms, it is estimated that their consumption may reach several kilograms per person per year.
The several changes in the environmental factors have an important impact on human health currently, which is reflected in the production of free radical species which are linked to the damage of vital biomolecules or cell structures (DNA, proteins and membranes).20 Furthermore, this physiological condition is associated with the development of serious ailments such as cardiovascular and neurodegenerative diseases, and different forms of cancers. Besides their nutritional value, compounds from mushrooms can act as antioxidant, antitumor, antiviral, antimicrobial, and immunomodulatory agents.11,21–27 Moreover, they have been shown to be modulators of the immune system and to exert hypoglycemic, antithrombotic, antibiotic, antiviral, antihypertensive, and antilipidemic properties, as well as anti-inflammatory and antimicrobial actions.28–30
Several vitamins and minerals such as ascorbic acid, niacin, riboflavin, thiamine and some essential micronutrients were described in species of the genus Polyporus. Polyporus squamosus (Huds.) Fr is an edible mushroom at an early and tender stage, used frequently as a spice, and being widely distributed in Europe, Asia, North America and Australia.24Polyporus species are well used medicinal mushrooms being described to possess diuretic, cytotoxic, nephroprotective, hepatoprotective, immuno-enhancing, anti-inflammatory, antioxidative, hair-growing, and antimicrobial activities.31–35 Particularly, P. squamosus was shown to possess antioxidant,36–38 immunomodulating,39 antibacterial,37 antifungal24 properties as well as antibiofilm and anti-quorum sensing activities.24 Moreover, there are some studies reporting the chemical composition of P. squamosus fruiting bodies from Portugal, Serbia, and Turkey.24,36,40,41
Different mushrooms are investigated by the scientific community, in searching for new therapeutic alternatives, and many results proved their bioactive properties as well as their medicinal value.11,13,21,25,28,29,34,39,42,43 The mountainous area of Romania, due to climatic conditions and flora diversity, is one of the European regions with high wild edible mushroom diversity, many of them being medicinal as well. Despite of their great popularity, data regarding the chemical composition and bioactive properties of the wild mushrooms available in the region as well as their nutritional value are very meager. Therefore, the aim of the present study was to provide a comprehensive study on the chemical composition of P. squamosus collected from Romania as well as on some of its biological activities (antioxidant, antimicrobial, and antiquorum sensing).
2. Materials and methods
2.1. Standards and reagents
The following chemicals were of HPLC grade and obtained from Lab-Scan (Lisbon, Portugal): acetonitrile 99.9%, n-hexane 95% and ethyl acetate 99.8%. All other chemicals including methanol were of analytical grade and obtained from common providers. Fatty acid methyl ester (FAME) reference standard mixture 37 (standard 47885-U) was purchased from Sigma (St Louis, MO, USA), as well as sugar standards, organic acids, phenolic acids, and Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid). Tocol and tocopherol standards were purchased from Matreya (Pleasant Gap, PA, USA). 2,2-Diphenyl-1-picrylhydrazyl (DPPH) was obtained from Alfa Aesar (Ward Hill, MA, USA). Water was treated in a Milli-Q water purification system (TGI Pure Water Systems, USA). Mueller–Hinton agar (MH) and malt agar (MA) were obtained from the Institute of Immunology and Virology, Torlak (Belgrade, Serbia).2.2. Mushroom samples
Fruiting bodies (tender stage) of P. squamosus were collected from the Apuseni mountain region, Sartăş, Alba County, Transylvania, Romania in May 2017, and authenticated by Dr Andrei Mocan, Senior Lecturer at the Department of Pharmaceutical Botany, “Iuliu Haţieganu” University of Medicine and Pharmacy, Cluj-Napoca, Romania, and a voucher specimen was deposited at the herbarium of the Department of Pharmaceutical Botany, “Iuliu Haţieganu” University of Medicine and Pharmacy. Fresh and cleaned fruiting bodies were randomly divided and dried using a laboratory plant dryer at 25 °C until reaching a constant mass. Afterwards, the mushroom material was ground into a fine powder using a laboratory mill, mixed to obtain a homogeneous sample, and kept at 4 °C, for further analyses. All assays were carried out three times (three separate samplings) and in triplicate, and the values reported are represented as average and standard deviation (SD).2.3. Extraction procedure
The ground powder mushroom material (1.5 g) was stirred with methanol (30 mL) at 25 °C at 150 rpm for 1 h, and further filtered through Whatman no. 4 paper. Additionally, the residue was further extracted with an additional portion of methanol. After combining the two fractions, the methanolic extracts were evaporated using a rotary evaporator (Büchi R-210; Flawil, Switzerland) under reduced pressure, re-dissolved in methanol at a concentration of 40 mg mL−1 (stock solution), and stored at 4 °C for further analyses (HPLC-DAD-ESI/MS and antioxidant assays).42 For the evaluation of antimicrobial activity, the dry extract was dissolved in 5% DMSO (1 mg mL−1), following the procedure mentioned in section 2.6.2.4. Chemical characterization
2.5. Evaluation of antioxidant activity
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, v/v with water) and sodium carbonate (75 g L−1, 4 mL). The tubes were vortexed for 15 s and allowed to incubate for 30 min at 40 °C, followed by the absorbance measurement at 765 nm (Analytikjena spectrophotometer; Jena, Germany). The commercially available gallic acid was used as a standard. The reduction of the Folin–Ciocalteu reagent was expressed in mg of gallic acid equivalents (GAE) per g of extract.
10, v/v with water) and sodium carbonate (75 g L−1, 4 mL). The tubes were vortexed for 15 s and allowed to incubate for 30 min at 40 °C, followed by the absorbance measurement at 765 nm (Analytikjena spectrophotometer; Jena, Germany). The commercially available gallic acid was used as a standard. The reduction of the Folin–Ciocalteu reagent was expressed in mg of gallic acid equivalents (GAE) per g of extract.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 w/v brain tissue homogenate.42 The porcine brain homogenate was centrifuged for 10 min at 3000g, and afterwards an 100 μL aliquot of the supernatant was incubated for 1 h at 37 °C with 200 μL samples of a range of concentrations of the extract in the presence of FeSO4 (10 mM; 100 μL) and ascorbic acid (0.1 mM; 100 μL). After the incubation time, the reaction was stopped by adding 500 μL trichloroacetic acid (28% w/v), followed by 380 μL thiobarbituric acid (TBA, 2%, w/v). The mixture was then incubated again for 20 min at 80 °C. Afterwards, the samples were centrifuged at 3000g for 10 min to remove the precipitated proteins, and the absorbance of the malondialdehyde (MDA)–TBA complex in the supernatant was measured at 532 nm. Using the following equation the inhibition ratio (%) was calculated: Inhibition ratio (%) = [(A − B)/A] × 100%, where A and B were the absorbance of the control and the sample solution, respectively.50
2 w/v brain tissue homogenate.42 The porcine brain homogenate was centrifuged for 10 min at 3000g, and afterwards an 100 μL aliquot of the supernatant was incubated for 1 h at 37 °C with 200 μL samples of a range of concentrations of the extract in the presence of FeSO4 (10 mM; 100 μL) and ascorbic acid (0.1 mM; 100 μL). After the incubation time, the reaction was stopped by adding 500 μL trichloroacetic acid (28% w/v), followed by 380 μL thiobarbituric acid (TBA, 2%, w/v). The mixture was then incubated again for 20 min at 80 °C. Afterwards, the samples were centrifuged at 3000g for 10 min to remove the precipitated proteins, and the absorbance of the malondialdehyde (MDA)–TBA complex in the supernatant was measured at 532 nm. Using the following equation the inhibition ratio (%) was calculated: Inhibition ratio (%) = [(A − B)/A] × 100%, where A and B were the absorbance of the control and the sample solution, respectively.50
2.6. Evaluation of the antimicrobial activity
The bacterial cell suspension was adjusted with sterile saline to a concentration of approximately 1.0 × 105 in a final volume of 100 μL per well. The extract was dissolved in 5% DMSO (1 mg mL−1) and added into a Tryptic Soy broth (TSB) medium (100 μL) with bacterial inoculum (1.0 × 104 CFU per well) to achieve the desired concentration. The microplates were incubated using a rotary shaker (160 rpm) for 24 h at 37° C. The following day, 30 μL of 0.2 mg mL−1 solution of INT (p-iodonitrotetrazolium violet) was added, and the plates were returned to the incubator for at least one-half hour to ensure an adequate colour reaction.51,52 The inhibition of growth was indicated by a clear solution or a definite decrease in color reaction. The lowest concentrations without visible growth (using the binocular microscope) were defined as concentrations that completely inhibited bacterial growth (MICs). The MBCs were determined by serial sub-cultivation of 2 μL of the extract into microtitre plates containing 100 μL of broth per well and further incubation for 24 h. The lowest concentration with no visible growth was defined as the MBC, indicating 99.5% killing of the original inoculum. The optical density of each well was measured at a wavelength of 655 nm by microplate manager 4.0 (Bio-Rad Laboratories) and compared with a blank and the positive control. The antibiotics streptomycin and ampicillin were used as positive controls (1 mg mL−1 in sterile physiological saline). Three independent experiments were performed in triplicate.
Bacterial strains, growth media and culture conditions. P. aeruginosa PA01 (ATCC 27853) used in this study is from the collection of the Mycoteca, Institute for Biological Research “Sinisa Stankovic”, Belgrade, Serbia. Bacteria were routinely grown in Luria–Bertani (LB) medium (1% w/v NaCl, 1% w/v tryptone, 0.5% w/v yeast extract) with shaking (220 rpm) and cultured at 37 °C.
Biofilm formation. The effect of different concentrations of extract (ranging from 0.5 to 0.125 of MIC, MIC was 0.2 mg mL−1) on biofilm forming ability was tested on polystyrene flat-bottomed microtitre 96 well plates as described in ref. 53 and 54 with some modifications. Briefly, 100 μL of overnight culture of P. aeruginosa (inoculum size was 1 × 108 CFU mL−1) was added to each well of the plates in the presence of 100 μL subinhibitory concentrations (subMIC) of extract (0.5, 0.25 and 0.125 MIC) or 100 mL medium (control). After incubation for 24 h at 37 °C, each well was washed twice with sterile PBS (pH 7.4), dried, and stained for 10 min with 0.1% crystal violet in order to determine the biofilm mass. After drying, 200 μL of 95% ethanol (v/v) was added to solubilize the dye that had stained the biofilm cells. The excess stain was washed off with dH2O. After 10 min, the content of the wells was homogenized and the absorbance at λ = 625 nm was read on a Sunrise™ Tecan ELISA reader (Mannedorf, Switzerland). The experiment was done in triplicate and repeated two times and values were presented as mean values ± SE.
Inhibition of biofilm formation of P. aeruginosa. P. aeruginosa was cultured overnight at 37 °C in LB medium and adjusted to a concentration of 1.0 × 108 CFU mL−1 for final inoculum. Filter paper discs (Whatman; 4 mm in diameter) were impregnated with a solution of P. squamosus extract (0.125, 0.25, 0.5 mg per disc), streptomycin and ampicillin (0.125, 0.25, 0.5 mg per disc). Discs were dried at room temperature (3 h, protected from light), and aseptically placed onto the plates prior inoculated with P. aeruginosa (1 × 108 CFU mL−1). Petri dishes were placed for incubation in a thermostat at 37 °C for 24 h. After incubation, whether inhibition or antiquorum zones were obtained was recorded. Minimal inhibitory concentrations were determined as a diameter of the growth clear inhibition zones around the discs (no growth at all), while antiquorum, i.e., antibiofilm zones were determined as transparent zones around the discs (growth with different characteristics) behind the margin of the inhibition zone.55 The experiment was done in triplicate and repeated two times and values were presented as mean values ± SE.
Inhibition of twitching and flagellar motility of P. aeruginosa. After growth in the presence or absence of P. squamosus extract (subMIC 0.15 mg mL−1), streptomycin and ampicillin (subMIC 0.15 mg mL−1), the cells of P. aeruginosa PA01 were washed twice with sterile PBS and re-suspended in PBS at 1 × 108 CFU mL−1 (OD of 0.1 at 660 nm). Briefly, the cells were stabbed into a nutrient agar plate with a sterile toothpick and incubated overnight at 37 °C. The plates were then removed from the incubator and incubated at room temperature for two more days. Colony edges and the zone of motility were measured with a light microscope.55,56 Fifty microlitres of P. squamosus extract was mixed into 10 mL of molten MH (Mueller–Hinton) agar medium and poured immediately over the surface of a solidified LB agar plate as an overlay. The plate was point inoculated with an overnight culture of PAO1 once the overlaid agar had solidified and incubated at 37 °C for 3 days. The extent of swimming was determined by measuring the area of the colony. The experiment was done in triplicate and repeated two times. The colony diameters were measured three times in different directions and the values were presented as mean values ± SE.
Inhibition of synthesis of P. aeruginosa PA01 pyocyanin. Overnight culture of P. aeruginosa PA01 was diluted to OD600 nm 0.2. Then, P. squamosus extract (250 μL), 0.125 MIC (0.025 mg mL−1), 0.25 MIC (0.05 mg mL−1) and 0.5 MIC (0.10 mg mL−1) were added to P. aeruginosa (4.75 mL) and incubated at 37 °C for 24 h. The treated culture was extracted with chloroform (3 mL), followed by mixing the chloroform layer with 0.2 M HCl (1 mL). Absorbance of the extracted organic layer was measured at 520 nm using a Shimadzu UV1601 spectrophotometer (Kyoto, Japan).57 The experiment was done in triplicate and repeated two times. The values for OD were presented as mean values ± SE.
3. Results and discussion
3.1. Chemical composition of Polyporus squamosus
Research on the chemical composition of P. squamosus was conducted to target different classes of bioactive compounds. To the best of our knowledge, this is the first study referring to the chemical composition of P. squamosus fruiting bodies originating from Romania. Results concerning the nutritional value and individual sugars are presented in Table 1. Carbohydrates were the most abundant class of macronutrients with 74.22 g per 100 g dw, followed by proteins (18.7 g per 100 g dw), ash (5.7 g per 100 g dw), and fat (1.46 g per 100 g dw), having an energetic value of 384.7 kcal per 100 g dw. The fruiting body of P. squamosus represents a very good low-fat medicinal food which can be used on a regular basis. The nutritional composition of P. squamosus from the present study was similar to one of the specimens collected from Northeast Portugal (Bragança) and Northern Serbia, where carbohydrates and proteins were also described as being the most abundant macronutrients.24 Additionally, our results are in line with the findings of Akata et al. who reported a similar trend in macronutrient composition: carbohydrates 65.24%, proteins 13.32%, fat 3.98%, and ash 7.14%.36| Polyporus squamosus | |
|---|---|
| dw – dry weight. | |
| Fat (g per 100 g dw) | 1.46 ± 0.02 |
| Protein (g per 100 g dw) | 18.7 ± 0.1 |
| Ash (g per 100 g dw) | 5.7 ± 0.1 |
| Carbohydrates (g per 100 g dw) | 74.22 ± 0.01 |
| Energetic value (kcal per 100 g dw) | 384.7 ± 0.4 |
| Sugars (g per 100 g dw) | |
| Fructose | 0.730 ± 0.004 |
| Mannitol | 1.05 ± 0.02 |
| Trehalose | 6.81 ± 0.03 |
| Sum | 8.59 ± 0.06 |
Regarding sugar composition, trehalose was the most abundant compound (6.81 g per 100 g dw), while fructose was found in the smallest amount (0.730 g per 100 g dw). In a previous study conducted by our team on P. squamosus fruiting bodies originating from Portugal and Serbia, trehalose was also found as being the most abundant sugar, while rhamnose, not detected in the herein sample, was found in the smallest amount, for both samples.24 Among organic acids, it was possible to identify oxalic, quinic, malic and fumaric acids (Table 2). Malic acid was detected in the highest amount (2.21 g per 100 g dw), whilst fumaric and oxalic acids were detected in much lower amounts (0.0003 g per 100 g dw, and 0.076 g per 100 g dw). In a similar manner to our results, malic acid was described as being the most abundant organic acid in samples of P. squamosus originating from Portugal and Serbia, while quinic acid was not detected.24 An important fact is that the presence of these compounds in foods not only influences their flavor, but also the stability, acceptability and shelf-life.59 Moreover, organic acids present in natural matrices are involved in the reduction of oxidative stress.60
| dw – dry weight. | |
|---|---|
| Organic acids (g per 100 g dw) | |
| Oxalic acid | 0.076 ± 0.001 |
| Quinic acid | 1.40 ± 0.02 |
| Malic acid | 2.21 ± 0.01 |
| Fumaric acid | 0.0003 ± 0.0001 |
| Total organic acids | 3.69 ± 0.04 |
| Phenolic acids (mg per 100 g dw) | |
| p-Hydroxybenzoic acid | 2.03 ± 0.05 |
| p-Coumaric acid | 0.272 ± 0.002 |
| Total phenolic compounds | 2.34 ± 2.26 |
| Related compounds (mg per 100 g dw) | |
| Cinnamic acid | 0.037 ± 0.001 |
Phenolic compounds which contribute directly to antioxidative protection and play an important role in stabilizing lipid peroxidation were also determined in the studied sample,11 being identified as p-hydroxybenzoic acid and p-coumaric acid (Table 2). Similarly, Fernandes et al.24 described p-hydroxybenzoic acid as the main phenolic acid in samples of P. squamosus of different origins, while cinnamic acid, a phenolic-related compound, was also found in small amounts.
Lipophilic compounds, including fatty acids and tocopherols, were also characterized (Table 3), and twenty-three fatty acids were identified, both saturated and unsaturated. The fatty acids determined in high percentages were linoleic (C18:2n6), oleic (C18:1n9) and palmitic (C16:0) acids. Moreover, polyunsaturated fatty acids, which are known to possess several health beneficial effects, were found to be predominant in P. squamosus (57.37%), followed by monounsaturated fatty acids (24.96%), and saturated fatty acids (17.68%). In the previous report of Fernandes et al.24 a similar fatty acid pattern was described in P. squamosus from different origins, being C18:2n6, C18:1n9 and C16:0 the main fatty acids.
| Polyporus squamosus | |
|---|---|
| SFA – saturated fatty acids; MUFA – monounsaturated fatty acids; PUFA – polyunsaturated fatty acids. | |
| C6:0 | 0.033 ± 0.001 |
| C10:0 | 0.025 ± 0.001 |
| C12:0 | 0.049 ± 0.002 |
| C14:0 | 0.246 ± 0.001 |
| C14:1 | 0.011 ± 0.001 |
| C15:0 | 1.09 ± 0.01 |
| C16:0 | 13.40 ± 0.07 |
| C16:1 | 0.83 ± 0.01 |
| C17:0 | 0.123 ± 0.002 |
| C18:0 | 1.20 ± 0.02 |
| C18:1n9 | 22.27 ± 0.01 |
| C18:2n6 | 56.55 ± 0.04 |
| C18:3n3 | 0.441 ± 0.005 |
| C20:0 | 0.132 ± 0.001 |
| C20:1 | 0.151 ± 0.001 |
| C20:2 | 0.161 ± 0.003 |
| C20:3n3 + C21:0 | 0.16 ± 0.01 |
| C20:5n3 | 0.059 ± 0.006 |
| C22:0 | 1.04 ± 0.03 |
| C22:1n9 | 0.36 ± 0.02 |
| C23:0 | 0.10 ± 0.01 |
| C24:0 | 0.235 ± 0.004 |
| C24:1 | 1.34 ± 0.02 |
| Total SFA (% of total FA) | 17.68 ± 0.04 |
| Total MUFA (% of total FA) | 24.96 ± 0.01 |
| Total PUFA (% of total FA) | 57.37 ± 0.03 |
| Tocopherols (μg per 100 g dw) | |
| α-Tocopherol | 1.54 ± 0.05 |
| β-Tocopherol | 114.7 ± 0.8 |
| γ-Tocopherol | 5.3 ± 0.5 |
| δ-Tocopherol | 3.7 ± 0.4 |
| Total tocopherols | 125.2 ± 0.6 |
Tocopherols are known to be powerful natural antioxidants, and are present in natural sources usually as a mixture of four isomers (namely α-, β-, γ-, and δ-tocopherol).10,49 α-, β-, γ- and δ-Tocopherols were found in the analyzed sample, and β-tocopherol was determined in the highest amount (114.7 μg per 100 g dw). Interestingly, β-tocopherol was also described as the dominant tocopherol in P. squamosus originating from Portugal, while γ-tocopherol was predominate for the Serbian originating sample. According to Fernandes et al.24 these differences could be ascribed to natural variability inherent to samples grown under different environmental conditions.
3.2. Antioxidant activity of Polyporus squamosus extract
The maintenance of equilibrium between free radical production and antioxidant defenses is an essential condition for normal organism functioning, and an eventual imbalance is reflected through the accumulation of damaged cell structures (cell membranes, proteins). Compounds from wild mushrooms have been described as excellent natural antioxidants.11 In the present study, the antioxidant activity of P. squamosus was evaluated through five assays involving different mechanisms: reducing power, radical scavenging activity and lipid peroxidation inhibition (Table 4). For the radical scavenging (DPPH scavenging activity), lipid peroxidation inhibition (TBARS, β-carotene/linoleate) and reducing power activities (ferricyanide/Prussian blue assay) the results are presented in EC50 values, indicating that higher values correspond to lower reducing potential (ferricyanide/Prussian blue) or antioxidant potential, whereas for the Folin–Ciocalteu assay (which measures the reducing power of the extract in relation to the molybdenum ion present in the Folin–Ciocalteu reagent), higher values indicate higher reducing power.61 As seen in Table 4, the highest measured antioxidant effect of P. squamosus extract was found in the TBARS inhibition assay (EC50 = 0.22 mg mL−1), followed by β-carotene/linoleate (EC50 = 1.41 mg mL−1), ferricyanide/Prussian blue reducing activity (EC50 = 2.35 mg mL−1), and DPPH scavenging activity (EC50 = 8.2 mg mL−1). A similar trend was observed in the study of Fernandes et al.,24 in which the highest antioxidant effect of P. squamosus extracts from different origins was also measured for the TBARS inhibition assay. Nevertheless, other authors have previously reported higher DPPH scavenging and reducing power activities in P. squamosus from Turkey.36,38| Reducing power | Radical scavenging activity | Lipid peroxidation inhibition | ||
|---|---|---|---|---|
| Folin–Ciocalteu (mg GAE per g extract) | Ferricyanide/Prussian blue (EC50; mg mL−1) | DPPH scavenging activity (EC50; mg mL−1) | β-Carotene/linoleate (EC50; mg mL−1) | TBARS (EC50; mg mL−1) |
| Concerning the Folin–Ciocalteu assay, higher values mean higher reducing power; for the other assays, the results are presented in EC50 values, which means that higher values correspond to lower reducing power or antioxidant potential. EC50: extract concentration corresponding to 50% of antioxidant activity or 0.5 of absorbance for the ferricyanide/Prussian blue assay. Trolox EC50 values: 0.041 mg mL−1 (reducing power), 0.042 mg mL−1 (DPPH scavenging activity), 0.018 mg mL−1 (β-carotene bleaching inhibition) and 0.023 mg mL−1 (TBARS inhibition). | ||||
| 17.9 ± 0.1 | 2.35 ± 0.02 | 8.2 ± 0.1 | 1.41 ± 0.01 | 0.22 ± 0.01 |
3.3. Antimicrobial activity of Polyporus squamosus extract
The results of the antimicrobial activity indicate that P. squamosus extract possesses a good antibacterial activity on E. cloacae and B. cereus, while the extract was not active on the tested fungi. Additionally, according to the previous findings of Fernandes et al., S. aureus was the most sensitive strain to P. squamosus extract with a MIC value of 0.20 mg mL−1 and a MBC value of only 0.39 mg mL−1, which are much more lower as the results presented herein for the same strain.24 Moreover, the bacteriostatic concentration of P. squamosus extract for P. aeruginosa was determined by the microdilution method to be 0.6–20.4 mg mL−1 and the bactericidal concentration is from 1.2–40.8 mg mL−1 (Table 5).| Micrococcus flavus | Salmonella typhimurium | Escherichia coli | Enterobacter cloacae | Listeria monocytogenes | Bacillus cereus | Staphylococcus aureus | Pseudomonas aeruginosa | ||
|---|---|---|---|---|---|---|---|---|---|
| MIC – Minimum inhibitory concentrations; MBC – minimum bactericidal concentrations. | |||||||||
| P. squamosus | MIC | 10.2 ± 0.02 | 20.4 ± 0.09 | 20.4 ± 0.05 | 0.61 ± 0.02 | 20.4 ± 0.02 | 1.2 ± 0.02 | 20.4 ± 0.05 | 20.4 ± 0.08 |
| MBC | 20.4 ± 0.02 | 40.8 ± 0.06 | 40.8 ± 0.06 | 1.2 ± 0.03 | 40.8 ± 0.20 | 2.4 ± 0.03 | 40.8 ± 0.60 | 40.8 ± 0.80 | |
| Ampicillin | MIC | 0.13 ± 0.03 | 0.13 ± 0.03 | 0.18 ± 0.03 | 0.17 ± 0.02 | 0.20 ± 0.03 | 0.17 ± 0.02 | 0.10 ± 0.01 | 0.40 ± 0.03 |
| MBC | 0.15 ± 0.05 | 0.20 ± 0.03 | 0.27 ± 0.02 | 0.20 ± 0.06 | 0.33 ± 0.05 | 0.20 ± 0.06 | 0.20 ± 0.06 | 0.67 ± 0.08 | |
| Streptomycin | MIC | 0.07 ± 0.06 | 0.17 ± 0.01 | 0.13 ± 0.03 | 0.03 ± 0.00 | 0.22 ± 0.06 | 0.03 ± 0.00 | 0.13 ± 0.02 | 0.13 ± 0.03 |
| MBC | 0.17 ± 0.03 | 0.27 ± 0.00 | 0.20 ± 0.05 | 0.07 ± 0.00 | 0.40 ± 0.08 | 0.07 ± 0.00 | 0.30 ± 0.03 | 0.23 ± 0.02 | |
The effect of P. squamosus extract on biofilm formation of P. aeruginosa was tested with 10.0 mg (0.5 MIC), 5.0 mg (0.25 MIC) and 2.5 mg mL−1 (0.125 MIC). Under the concentration of 0.5 MIC of tested extract only 36.0% of P. aeruginosa biofilm was inhibited, ampicillin inhibited 30.9% and streptomycin 50.7% of biofilm formation. P. squamosus extract at 0.25 MIC inhibited 35.5% of biofilm formation, and ampicillin and streptomycin 43.5% and 29%, respectively. The lowest concentration of extract, 0.125 MIC inhibited biofilm formation of 4%, ampicillin of 7.8% and streptomycin 11.4% (Table 6).
| 1/2 MIC | 1/4 MIC | 1/8 MIC | |
|---|---|---|---|
| P. squamosus | 36.0 ± 1.50 | 35.5 ± 1.80 | 4.0 ± 0.08 |
| Ampicillin | 30.9 ± 2.40 | 43.5 ± 2.10 | 7.8 ± 0.06 |
| Streptomycin | 50.7 ± 3.80 | 29.0 ± 0.90 | 11.4 ± 0.09 |
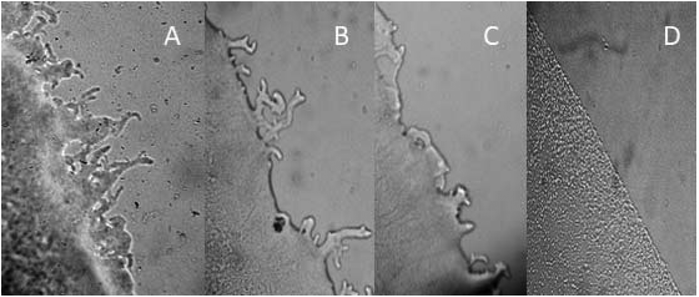
The extract of P. squamosus was tested for the twitching motility effect on the P. aeruginosa colony and producing of pili. The untreated colonies of P. aeruginosa were flat with a rough appearance displaying irregular colony edges (Fig. 1A) and a hazy zone surrounding the colony. It can be noted that extract applied in 0.25 MIC on P. aeruginosa colony influenced the reduction of twitching and bacteria produced almost round, smooth, irregular colony edges, and the pili were reduced both in size and in numbers, (Fig. 1B). The reduction of pili and changes in their shape and size were more obvious at a concentration of extract in 0.5 MIC, (Fig. 1C). Streptomycin reduced the pili completely (Fig. 1D).
The concentrations of 0.5 MIC P. squamosus extract were tested for antipyocyanin production by P. aeruginosa. At the tested concentrations of the extract the green pigment content was not decreased. Ampicillin (19%) and streptomycin (21%) also possessed very low potential in reduction of pyocyanin production (Table 7).
| % of inhibition | |
|---|---|
| No – no effect. | |
| P. squamosus | No |
| Ampicillin | 19.0 ± 0.80 |
| Streptomycin | 21.0 ± 0.60 |
4. Conclusions
An in-depth chemical analysis of several classes of compounds revealed fruiting bodies of P. squamosus from Romania as a rich source of bioactive molecules with potent in vitro activities. According to the obtained results, P. squamosus represents a source of polyunsaturated fatty acids and β-tocopherol. Moreover, the biological properties of P. squamosus indicate that it might be further explored as a medicinal mushroom in the composition of different pharmaceuticals as well as a dietary supplement. Nonetheless, further studies are needed to elucidate the different mechanisms of action involved as well as the specific compounds responsible for the tested biological activities.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors are grateful to the Foundation for Science and Technology (FCT, Portugal) and FEDER under Programme PT2020 for financial support to CIMO (UID/AGR/00690/2013), L. Barros research contract and A. Feitor (SFRH/BPD/114753/2016) grant. The authors are also grateful to the Interreg España-Portugal for financial support through the project 0377_Iberphenol_6_E. The authors are grateful to the Serbian Ministry of Education, Science and Technological Development for financial support (grant number 173032).References
- B. Bigliardi and F. Galati, Innovation trends in the food industry: The case of functional foods, Trends Food Sci. Technol., 2013, 31, 118–129 CrossRef CAS.
- A. M. Carvalho and A. M. Barata, in Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications, ed. Isabel C. F. R. Ferreira, P. Morales and L. Barros, John Wiley & Sons, Ltd., 2017, pp. 159–198 Search PubMed.
- N. Martins and I. C. F. R. Ferreira, Mountain food products: A broad spectrum of market potential to be exploited, Trends Food Sci. Technol., 2017, 67, 12–18 CrossRef CAS.
- J. Pinela, M. Carocho, M. I. Dias, C. Caleja, L. Barros and I. C. F. R. Ferreira, in Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications, ed. I. C. F. R. Ferreira, P. Morales and L. Barros, John Wiley & Sons, Ltd., 2017, pp. 315–351 Search PubMed.
- F. S. Reis, L. Barros, A. Martins and I. C. F. R. Ferreira, Chemical composition and nutritional value of the most widely appreciated cultivated mushrooms: An inter-species comparative study, Food Chem. Toxicol., 2012, 50, 191–197 CrossRef CAS PubMed.
- S. A. Heleno, L. Barros, M. J. Sousa, A. Martins and I. C. F. R. Ferreira, Tocopherols composition of Portuguese wild mushrooms with antioxidant capacity, Food Chem., 2010, 119, 1443–1450 CrossRef CAS.
- L. Barros, M. J. Ferreira, B. Queirós, I. C. F. R. Ferreira and P. Baptista, Total phenols, ascorbic acid, β-carotene and lycopene in Portuguese wild edible mushrooms and their antioxidant activities, Food Chem., 2007, 103, 413–419 CrossRef CAS.
- L. Barros, T. Cruz, P. Baptista, L. M. Estevinho and I. C. F. R. Ferreira, Wild and commercial mushrooms as source of nutrients and nutraceuticals, Food Chem. Toxicol., 2008, 46, 2742–2747 CrossRef CAS PubMed.
- L. Barros, P. Baptista and I. C. F. R. Ferreira, Effect of Lactarius piperatus fruiting body maturity stage on antioxidant activity measured by several biochemical assays, Food Chem. Toxicol., 2007, 45, 1731–1737 CrossRef CAS PubMed.
- L. Barros, D. M. Correia, I. C. F. R. Ferreira, P. Baptista and C. Santos-Buelga, Optimization of the determination of tocopherols in Agaricus sp. edible mushrooms by a normal phase liquid chromatographic method, Food Chem., 2008, 110, 1046–1050 CrossRef CAS PubMed.
- I. C. F. R. Ferreira, L. Barros and R. M. V. Abreu, Antioxidants in wild mushrooms., Curr. Med. Chem., 2009, 16, 1543–1560 CrossRef CAS PubMed.
- E. Pereira, L. Barros, A. Martins and I. C. F. R. Ferreira, Towards chemical and nutritional inventory of Portuguese wild edible mushrooms in different habitats, Food Chem., 2012, 130, 394–403 CrossRef CAS.
- O. Taofiq, S. A. Heleno, R. C. Calhelha, M. J. Alves, L. Barros, A. M. González-Paramás, M. F. Barreiro and I. C. F. R. Ferreira, The potential of Ganoderma lucidum extracts as bioactive ingredients in topical formulations, beyond its nutritional benefits, Food Chem. Toxicol., 2017, 108, 139–147 CrossRef CAS PubMed.
- C. V. Toledo, C. Barroetaveñ, Â. Fernandes, L. Barros and I. C. F. R. Ferreira, Chemical and antioxidant properties of wild edible mushrooms from native nothofagus spp. forest, Argentina, Molecules, 2016, 21, 1–15 CrossRef PubMed.
- C. Barroetaveña and C. V. Toledo, in Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications, ed. I. C. F. R. Ferreira, P. Morales and L. Barros, John Wiley & Sons, Ltd., 2017, pp. 65–81 Search PubMed.
- E. Boa, Wild edible fungi: A global overview of their use and importance to people, FAO, Rome, Italy, 2004 Search PubMed.
- Y.-C. Dai, Z.-L. Yang, B.-K. Cui, C.-J. Yu and L.-W. Zhou, Species Diversity and Utilization of Medicinal Mushrooms and Fungi in China (Review), Int. J. Med. Mushrooms, 2009, 11, 287–302 CrossRef.
- P. Kalac, Chemical composition and nutritional value of European species of wild growing mushrooms: A review, Food Chem., 2009, 113, 9–16 CrossRef CAS.
- M. Gąsecka, M. Siwulski and M. Mleczek, Evaluation of bioactive compounds content and antioxidant properties of soil-growing and wood-growing edible mushrooms, J. Food Process. Preserv., 2017, e13386 CrossRef.
- V. Lobo, A. Patil, A. Phatak and N. Chandra, Free radicals, antioxidants and functional foods : Impact on human health, Pharmacogn. Rev., 2010, 4, 118–126 CrossRef CAS PubMed.
- I. C. F. R. Ferreira, J. A. Vaz, M. H. Vasconcelos and A. Martins, Compounds from Wild Mushrooms with Antitumor Potential, Anticancer Agents Med. Chem., 2010, 10, 424–436 CrossRef CAS PubMed.
- F. S. Reis, A. Martins, M. H. Vasconcelos, P. Morales and I. C. F. R. Ferreira, Functional foods based on extracts or compounds derived from mushrooms, Trends Food Sci. Technol., 2017, 66, 48–62 CrossRef CAS.
- F. Souilem, Â. Fernandes, R. C. Calhelha, J. C. M. Barreira, L. Barros, F. Skhiri, A. Martins and I. C. F. R. Ferreira, Wild mushrooms and their mycelia as sources of bioactive compounds: Antioxidant, anti-inflammatory and cytotoxic properties, Food Chem., 2017, 230, 40–48 CrossRef CAS PubMed.
- Â. Fernandes, J. Petrović, D. Stojković, L. Barros, J. Glamočlija, M. Soković, A. Martins and I. C. F. R. Ferreira, Polyporus squamosus (Huds.) Fr from different origins: Chemical characterization, screening of the bioactive properties and specific antimicrobial effects against Pseudomonas aeruginosa, LWT–Food Sci. Technol., 2016, 69, 91–97 CrossRef.
- O. Taofiq, A. Martins, M. F. Barreiro and I. C. F. R. Ferreira, Anti-inflammatory potential of mushroom extracts and isolated metabolites, Trends Food Sci. Technol., 2016, 50, 193–210 CrossRef CAS.
- L. Barros, B. A. Venturini, P. Baptista, L. M. Estevinho and I. C. F. R. Ferreira, Chemical Composition and Biological Properties of Portuguese Wild Mushrooms : A Comprehensive Study Chemical Composition and Biological Properties of Portuguese Wild Mushrooms, J. Agric. Food Chem., 2008, 56, 3856–3862 CrossRef CAS PubMed.
- L. Barros, R. C. Calhelha, J. A. Vaz, I. C. F. R. Ferreira, P. Baptista and L. M. Estevinho, Antimicrobial activity and bioactive compounds of Portuguese wild edible mushrooms methanolic extracts, Eur. Food Res. Technol., 2007, 225, 151–156 CrossRef CAS.
- M. Soković, A. Ćirić, J. Glamočlija and D. Stojković, in Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications, ed. I. C. F. R. Ferreira, P. Morales and L. Barros, John Wiley & Sons, Ltd., 2017, pp. 83–122 Search PubMed.
- M. J. Alves, I. C. F. R. Ferreira, J. Dias, V. Teixeira and A. Martins, A Review on Antifungal Activity of Mushroom Extracts and Isolated Compounds.Pdf, Planta Med., 2012, 78, 1707–1718 CrossRef CAS PubMed.
- D. S. Stojković, N. Kovačević-Grujičić, F. S. Reis, S. Davidović, L. Barros, J. Popović, I. Petrović, A. Pavić, J. Glamočlija, A. Ćirić, M. Stevanović, I. C. F. R. Ferreira and M. Soković, Chemical composition of the mushroom Meripilus giganteus Karst. and bioactive properties of its methanolic extract, LWT–Food Sci. Technol., 2017, 79, 454–462 CrossRef.
- Y. Zhao, R. Xie, X. Chao, Y. Zhang, R. Lin and W. Sun, Bioactivity-directed isolation, identification of diuretic compounds from Polyporus umbellatus, J. Ethnopharmacol., 2009, 126, 184–187 CrossRef CAS PubMed.
- T. Ohsawa, M. Yukawa, C. Takao, M. Murayama and H. Bando, Studies on constituents of fruit body of Polyporus umbellatus and their cytotoxic activity, Chem. Pharm. Bull., 1992, 40, 143–147 CrossRef CAS PubMed.
- D. Y. Uan, J. M. Ori, K. K. Omatsu, T. M. Akino and Y. K. Ano, An Anti-aldosteronic Diuretic Component (Drain dampness) in Polyporus Sclerotium, Biol. Pharm. Bull., 2004, 27, 867–870 Search PubMed.
- Y. Zhao, Traditional uses, phytochemistry, pharmacology, pharmacokinetics and quality control of Polyporus umbellatus (Pers.) Fries : A review, J. Ethnopharmacol., 2013, 149, 35–48 CrossRef CAS PubMed.
- Y. Zhao, X. Chao, Y. Zhang, R. Lin, U. Pierre, M. Curie-paris and I. P. De Chimie, Cytotoxic Steroids from Polyporus umbellatus, Planta Med., 2010, 76, 1755–1758 CrossRef CAS PubMed.
- I. Akata, B. Ergönül and F. Kalyoncu, Chemical Compositions and Antioxidant Activities of 16 Wild Edible Mushroom Species Grown in Anatolia.pdf, Int. J. Pharmacol., 2012, 8, 134–138 CAS.
- M. Dimitrijevic, S. Jovanovic, J. Cvetkovic and T. Mihajilov-, Analytical Methods antiradical activities of twelve selected Serbian wild, Anal. Methods, 2015, 7, 4181–4191 RSC.
- M. Elmastas, O. Isildak, I. Turkekul and N. Temur, Determination of antioxidant activity and antioxidant compounds in wild edible mushrooms, J. Food Compos. Anal., 2007, 20, 337–345 CrossRef CAS.
- A. A. Babakhin, N. Y. Logina, H. Nolte and L. M. DuBuske, Immunomodulating Activity of the Extract from High Mycelium Fungus Polyporus Squamosus, J. Allergy Clin. Immunol., 1996, 97, 229 CrossRef.
- N. Dursun, M. M. Öycan, G. KaŞık and C. Öztürk, Mineral contents of 34 species of edible mushrooms growing wild in Turkey, J. Sci. Food Agric., 2006, 86, 1087–1094 CrossRef CAS.
- P. G. Ergönül, I. Akata, F. Kalyoncu and B. Ergönül, Fatty Acid Compositions of Six Wild Edible Mushroom Species, Sci. World J., 2013, 2013, 163964 Search PubMed.
- M. Kostic, M. Smiljkovic, J. Petrovic, J. Glamocilija, L. Barros, I. C. F. R. Ferreira, A. Ciric and M. Sokovic, Chemical, nutritive composition and wide-broad bioactive properties of honey mushroom Armillaria mellea (Vahl: Fr.) Kummer, Food Funct., 2017, 8, 3239–3249 CAS.
- F. S. Reis, D. Sousa, L. Barros, A. Martins, P. Morales, I. C. F. R. Ferreira and M. H. Vasconcelos, Leccinum vulpinum Watling induces DNA damage, decreases cell proliferation and induces apoptosis on the human MCF-7 breast cancer cell line, Food Chem. Toxicol., 2016, 90, 45–54 CrossRef CAS PubMed.
- AOAC, Official Methods of Analysis of AOAC International, 20th edn, 2016 Search PubMed.
- C. Grangeia, S. A. Heleno, L. Barros, A. Martins and I. C. F. R. Ferreira, Effects of trophism on nutritional and nutraceutical potential of wild edible mushrooms, Food Res. Int., 2011, 44, 1029–1035 CrossRef CAS.
- D. S. Stojković, L. Barros, R. C. Calhelha, J. Glamočlija, A. Ćirić, L. J. L. D. van Griensven, M. Soković and I. C. F. R. Ferreira, A detailed comparative study between chemical and bioactive properties of Ganoderma lucidum from different origins, Int. J. Food Sci. Nutr., 2014, 65, 42–47 CrossRef PubMed.
- L. Barros, M. Dueñas, I. C. F. R. Ferreira, P. Baptista and C. Santos-Buelga, Phenolic acids determination by HPLC-DAD-ESI/MS in sixteen different Portuguese wild mushrooms species, Food Chem. Toxicol., 2009, 47, 1076–1079 CrossRef CAS PubMed.
- L. Barros, C. Pereira and I. C. F. R. Ferreira, Optimized analysis of organic acids in edible mushrooms from portugal by ultra fast liquid chromatography and photodiode array detection, Food Anal. Methods, 2013, 6, 309–316 CrossRef.
- S. A. Heleno, L. Barros, M. J. Sousa, A. Martins and I. C. F. R. Ferreira, Tocopherols composition of Portuguese wild mushrooms with antioxidant capacity, Food Chem., 2010, 119, 1443–1450 CrossRef CAS.
- F. S. Reis, S. A. Heleno, L. Barros, M. J. Sousa, A. Martins, C. Santos-Buelga and I. C. F. R. Ferreira, Toward the Antioxidant and Chemical Characterization of Mycorrhizal Mushrooms from Northeast Portugal, J. Food Sci., 2011, 76, 824–830 CrossRef PubMed.
- Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, Clinical and Laboratory Standards Institute, Wayne, PA, 8th edn, 2009, vol. 29 Search PubMed.
- T. Tsukatani, H. Suenaga, M. Shiga, K. Noguchi, M. Ishiyama, T. Ezoe and K. Matsumoto, Comparison of the WST-8 colorimetric method and the CLSI broth microdilution method for susceptibility testing against drug-resistant bacteria, J. Microbiol. Methods, 2012, 90, 160–166 CrossRef CAS PubMed.
- A. L. Spoering and K. I. M. Lewis, Biofilms and Planktonic Cells of Pseudomonas aeruginosa Have Similar Resistance to Killing by Antimicrobials, J. Bacteriol., 2001, 183, 6746–6751 CrossRef CAS PubMed.
- E. Drenkard and F. M. Ausubel, Pseudomonas biofilm formation and antibiotic resistance are linked to phenotypic variation, Nature, 2002, 416, 740–743 CrossRef CAS PubMed.
- C. O'May and N. Tufenkji, The swarming motility of Pseudomonas aeruginosa is blocked by cranberry proanthocyanidins and other tannin-containing materials, Appl. Environ. Microbiol., 2011, 77, 3061–3067 CrossRef PubMed.
- G. A. O'Toole and R. Kolter, Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development, Mol. Microbiol., 1998, 30, 295–304 CrossRef.
- S. S.-M. Yeo and F.-Y. Tham, Anti-quorum sensing & antimicrobial activities of some traditional Chinese medicinal plants commonly used in South-East Asia, Malays. J. Microbiol., 2012, 8, 11–20 Search PubMed.
- A. Espinel-Ingroff, Comparison of the E-test with the NCCLS M38-P method for antifungal susceptibility testing of common and emerging pathogenic filamentous fungi, J. Clin. Microbiol., 2001, 39, 1360–1367 CrossRef CAS PubMed.
- A. Begic-Akagi, N. Spaho, F. Gasi, P. Drkenda, A. Vranac and M. Meland, Sugar and organic acid profiles of the traditional and international apple cultivars for processing, J. Hyg. Eng. Des., 2014, 7, 190–196 Search PubMed.
- X. Wu, J. Cheng and X. Wang, Dietary Antioxidants: Potential Anticancer Agents, Nutr. Cancer, 2017, 69, 521–533 CrossRef CAS PubMed.
- D. Huang, O. U. Boxin and R. L. Prior, The chemistry behind antioxidant capacity assays, J. Agric. Food Chem., 2005, 53, 1841–1856 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2018 |