Red-emission over a wide range of wavelengths at various temperatures from tetragonal BaCN2:Eu2+†
Yuji
Masubuchi
 *a,
Sayaka
Nishitani
b,
Akira
Hosono
*a,
Sayaka
Nishitani
b,
Akira
Hosono
 b,
Yuuki
Kitagawa
c,
Jumpei
Ueda
b,
Yuuki
Kitagawa
c,
Jumpei
Ueda
 c,
Setsuhisa
Tanabe
c,
Setsuhisa
Tanabe
 c,
Hisanori
Yamane
c,
Hisanori
Yamane
 d,
Mikio
Higuchi
a and
Shinichi
Kikkawa
d,
Mikio
Higuchi
a and
Shinichi
Kikkawa
 a
a
aFaculty of Engineering, Hokkaido University, N13 W8, Kita-ku, Sapporo 060-8628, Japan. E-mail: yuji-mas@eng.hokudai.ac.jp
bGraduate School of Chemical Science and Engineering, Hokkaido University, N13 W8, Kita-ku, Sapporo 060-8628, Japan
cGraduate School of Human and Environmental Studies, Kyoto University, Yohida-nihonmatsu-cho, Sakyo-ku, Kyoto 606-8501, Japan
dInstitute of Multidisciplinary Research for Advanced Materials, Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai 980-8577, Japan
First published on 25th April 2018
Abstract
A new polymorph of BaCN2 was obtained via a simple nitridation reaction of BaCO3 under an NH3 flow at 900 °C. The product was analyzed via single crystal X-ray diffraction and infrared spectroscopy, and it was found to have a tetragonal I4/mcm crystal structure (space group no. 140) with a = 0.60249(4) nm and c = 0.71924(5) nm. In this structure, each Ba2+ cation is situated in the square antiprism of N atoms of NCN2− anionic groups. Eu2+ doped BaCN2 can be excited by irradiation of blue and green light (from 400 to 550 nm), and it generates an intense red emission peak at 660 nm with a quantum efficiency of 42% in response to 465 nm excitation at room temperature. The peak emission wavelength varies over an extremely wide range of temperature, from 640 nm at 500 K to 680 nm at 80 K, and this red-shift with decreasing temperature is attributed to a unit cell shrinkage that results in significant crystal field splitting of the 5d energy levels of the Eu2+ ions.
Introduction
Many inorganic materials, including oxides, nitrides, sulfides, and chalcogenides, have been developed as host materials for phosphors.1–3 Divalent Eu-doped nitride phosphors are especially widely studied because of the superior thermal and chemical stabilities of their optical properties. High performance nitride phosphors are already used for commercial LED applications.4,5 The emission spectra of Eu2+-doped phosphors are typically characterized by the parity-allowed transition from the 4f7 to the 4f65d1 state. The energy of the 4f65d1 excited state is closely correlated with various structural parameters, such as covalency, bond length, and coordination number. These factors determine the crystal field strength and the nephelauxetic effect that in turn modify the emission properties. Therefore, the luminescence characteristics obtained from Eu2+ doping can be tailored by choosing a suitable host material.Metal cyanamides and carbodiimides are interesting inorganic materials that exhibit a nitrogen-related pseudo-oxide chemistry, since NCN2− anionic groups can replace O2− anions. The triatomic anionic groups in these compounds can have varying numbers of neighboring metal cations and thus form a characteristic host lattice depending on the cation. Many transition metal cyanamides have been prepared via solution processes. As an example, CuCN2, ZnCN2 and CdCN2 can be obtained from the reaction of the associated metal chlorides and cyanamides in aqueous media.6–8 Solid-state metathesis has also resulted in the formation of MnCN2 from MnCl2 and ZnCN2,9 while SrCN2 was synthesized via a flux route based on a combination of strontium halide, alkaline cyanide, and alkaline azide precursors.10 Calcium carbide has also been used as the starting material to form CaCN2 under a pressurized N2 atmosphere,11 and the high temperature nitridation reactions of CaCO3 and SrCO3 under an ammonia flow have also been applied to synthesize the respective cyanamides.12,13 The rhombohedral structure (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c) of BaCN2 with a = 1.5282(2) nm and c = 0.7013(2) nm has only been reported for a cyanamide prepared from the reaction of Ba3N2 and melamine under an Ar flow at temperatures between 740 °C and 850 °C.14 In this structure, six N atoms of NCN2− coordinate to each Ba2+ cation, forming a distorted octahedron. In addition, only the formation of BaCN2 has been reported in the reaction of BaCO3 with hydrogen cyanide or the nitridation reaction of BaCO3.12,15
c) of BaCN2 with a = 1.5282(2) nm and c = 0.7013(2) nm has only been reported for a cyanamide prepared from the reaction of Ba3N2 and melamine under an Ar flow at temperatures between 740 °C and 850 °C.14 In this structure, six N atoms of NCN2− coordinate to each Ba2+ cation, forming a distorted octahedron. In addition, only the formation of BaCN2 has been reported in the reaction of BaCO3 with hydrogen cyanide or the nitridation reaction of BaCO3.12,15
Alkaline earth cyanamides and related compounds such as metal cyanate (MOCN) and thiocyanate (MSCN) have been applied as host materials for Eu2+ doping. A blue emission peak at 457 nm is generated by Eu2+-doped Sr(OCN)2, while Eu2+-doped Sr(SCN)2 produces a green emission at 508 nm in response to excitation at 420 nm.16,17 A longer emission wavelength of 610 nm (corresponding to an orange color) has been reported for Eu2+-doped α-SrCN2.13 However, these characteristic emissions are only evident at low temperatures and are completely quenched at room temperature. Their thermal quenching temperatures (T1/2) at which the luminescence intensities are reduced by 50% are 65 K, 157 K and 90 K for Eu2+-doped Sr(OCN)2, Sr(SCN)2 and α-SrCN2, respectively. However, a room temperature orange emission at 603 nm has been reported for Eu2+-doped α-SrCN2 obtained from the reaction of SrI2, EuI2, CsN3 and CsCN in Ta ampules.10 This emission at room temperature has been attributed to the reduced defect concentration caused by direct doping with Eu2+I2. Ba(SCN)2 was also studied as a host material with regard to doping with Eu2+.18 Ba2+ was coordinated with eight SCN− anionic groups to form distorted square antiprism polyhedra. This material was found to generate an intense bright green emission peak at 511 nm at low temperature that was completely quenched at room temperature, with a T1/2 of 181 K.
In this paper, we present the crystal structure of a new polymorph of barium carbodiimide (BaCN2) prepared by a simple nitridation reaction of BaCO3. This polymorph crystallized in a tetragonal lattice with square antiprism coordination around the Ba2+ ions, and it was studied as a novel host material for Eu2+ ions. Eu2+-doped BaCN2 was found to produce a red emission over a wide range of wavelengths depending on temperature, and these emission characteristics were investigated in relation to the crystal structure.
Experimental
Synthesis procedures
The new polymorph of BaCN2 was prepared via the heating of BaCO3 (99.9% purity, Wako Pure Chemicals Co.) on an aluminum boat under a 50 mL min−1 flow of NH3 at 900 °C for 15 h. Eu2+-doped BaCN2 was also prepared from a mixture of BaCO3 and Eu acetylacetonate hydrate (Eu(acac)3nH2O where n = 1.8, 99.9%, Aldrich). These two powders were mixed at a Ba![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Eu ratio of 1 − x
Eu ratio of 1 − x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x (x = 0.002–0.05) in ethanol and subsequently nitrided under the same conditions as that for non-doped BaCN2. The nitrided products were found to be air-sensitive, so the characterization described below was performed under either a dry N2 atmosphere or vacuum.
x (x = 0.002–0.05) in ethanol and subsequently nitrided under the same conditions as that for non-doped BaCN2. The nitrided products were found to be air-sensitive, so the characterization described below was performed under either a dry N2 atmosphere or vacuum.
Structural analysis
Crystalline phases were characterized using powder X-ray diffraction (XRD: Rigaku, Ultima IV) with Cu Kα radiation. The products were contained in an air-tight sample holder. Temperature dependence of the powder XRD pattern was also measured using XRD (Rigaku, Ultima III) equipped with a low temperature chamber unit (Anton Paar, TTK 450) over the range of 138–500 K, under vacuum. Single-crystal XRD data were collected by using the ω/ϕ scans method with a diffractometer, employing Mo Kα radiation (Bruker, D8 QUEST). Data collection and unit-cell refinement were performed using the APEX2 software package.19 Absorption corrections were applied using the SADABS multiscan procedure,20 the structural parameters of the crystal were refined with the SHELXL-2014 program21 and the crystal structures were drawn using the VESTA program.22 The temperature dependence of the lattice parameters was also determined for a BaCN2 crystal over the range of 90 K to 290 K with the diffractometer equipped with a cryostream (Oxford Cryosystem, Cobura). The powder XRD pattern of BaCN2 was calculated using the Rietveld program RIETAN-FP23 based on the refined structural parameters.Chemical composition
The Ba content in the specimens was determined by inductively coupled plasma-atomic emission spectroscopy (ICP-AES: Shimadzu, ICPE-9000) after dissolving a 10 mg sample in diluted nitric acid. The C, H, and N levels were assessed via C/H/N analysis (Exeter Analytical, Inc., CE440) while the O content was also measured using a combustion analyzer (Horiba, EMGA-620).XANES
Eu LIII-edge X-ray absorption near edge structure (XANES) data were acquired in the transmission mode at the BL-9C beam line of the Photon Factory at the High Energy Accelerator Research Organization (KEK), Tsukuba, Japan, with the storage ring operating at 2.5 GeV. A small amount of the sample powder was mixed with BN powder and then pressed into a pellet for the measurements. EuCl2 and Eu2O3 were used as references.FT-IR spectroscopy
Fourier transform infrared spectroscopy (FT-IR) was performed with an FT-IR/4700 spectrometer (Jasco). Test specimens were prepared by sandwiching BaCN2 powder between KBr plates and pressing the sample into a pellet under a dry nitrogen atmosphere.Optical measurements
Photoluminescence spectra were acquired at room temperature with a fluorescence spectrometer (Jasco, FP-6500) equipped with a 150 W Xe lamp as an excitation source. The temperature dependence of luminescence was assessed over the range of 80–500 K using a multichannel spectrometer (Ocean Optics, QE65 Pro) with 460 nm excitation by an LED in conjunction with a band-pass filter. During the measurement, the phosphor powder was heated from 80 K to 500 K and then cooled to 80 K in 5 K intervals at a heating/cooling rate of 10 K min−1, under vacuum.The total radiant flux spectra of the pump-LED source and sample luminescence under direct- and indirect- excitation of the sample were measured with an integrating sphere (Labsphere, LMS-100). The measured spectra were calibrated with a standard halogen lamp (Labsphere, CLS-600) to obtain the precise spectral power distributions, from which photon emission rate of the pump-LED under direct- and indirect- excitation, Φex_direct (λ) and Φex_indirect (λ), and that of sample luminescence under direct- and indirect excitation, Φlumin_direct (λ) and Φlumin_indirect (λ), can be calculated. Quantum efficiency (QE) values were calculated using the following equation:
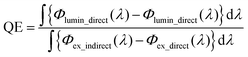 | (1) |
The fluorescence decay curves monitoring 650 nm luminescence were measured with a Quantaurus-Tau (Hamamatsu Photonics, C11367-01) under 365 nm picosecond LED excitation at different temperatures from 90 to 450 K. The temperature of the sample was controlled by a cryostat (Japan High Tech, 10035L). The obtained fluorescence decay curve was fitted after baseline subtraction using a single-exponential function,
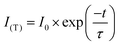 | (2) |
I 0 is initial intensity and τ is lifetime.
Results and discussion
Crystal structure of the BaCN2 polymorph
The powder XRD pattern of the product is shown in Fig. S1 (ESI†), where it is compared with the pattern calculated for rhombohedral (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c) BaCN2 reported by W. Schnick.14 From these patterns, it is evident that the rhombohedral BaCN2 phase does not appear even as an impurity phase. The Ba concentration in the product was found to be 77.3(9) wt%, while C, H, and N contents were 6.83(2), <0.30 and 15.42(5) wt%, respectively. The H and O levels were below the detection limits of the C/H/N and the combustion analyses, respectively. From these data, the chemical formula for the compound was determined to be Ba1C1.01(1)N1.96(2), which is consistent with BaCN2. Thus, the product obtained by the nitridation of BaCO3 is a new polymorph of BaCN2.
c) BaCN2 reported by W. Schnick.14 From these patterns, it is evident that the rhombohedral BaCN2 phase does not appear even as an impurity phase. The Ba concentration in the product was found to be 77.3(9) wt%, while C, H, and N contents were 6.83(2), <0.30 and 15.42(5) wt%, respectively. The H and O levels were below the detection limits of the C/H/N and the combustion analyses, respectively. From these data, the chemical formula for the compound was determined to be Ba1C1.01(1)N1.96(2), which is consistent with BaCN2. Thus, the product obtained by the nitridation of BaCO3 is a new polymorph of BaCN2.
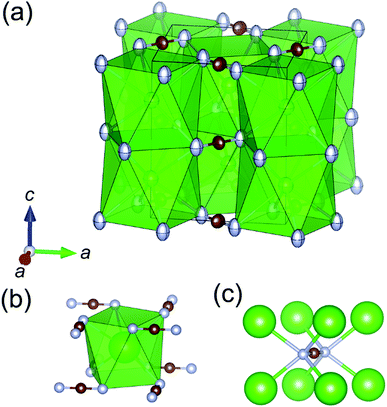
The crystal structure of the BaCN2 polymorph was analyzed by single crystal XRD. The structure was refined to give the tetragonal space group I4/mcm (no. 140) with a = 0.60249(4) nm and c = 0.71924(5) nm, and Z = 4 (Table 1). The atomic coordinates and anisotropic displacement parameters are given in Table 2. This structure is an isotype of the alkaline cyanates, AOCN (A = K, Rb, or Cs),24,25 in which each Ba2+ ion is coordinated with eight N atoms belonging to NCN2− groups (Fig. 1), and in the regular square antiprism of N. The NCN2− anion is in the square prism of Ba atoms (Fig. 1(c)). The Ba-centered square antiprism polyhedra are connected via face sharing along the c-axis and edge-sharing in the ab-plane. Along the c-axis, Ba2+ and NCN2− layers are stacked on one another along the c-axis to form a CsCl-type arrangement of both ions in conjunction with an ordered arrangement of the NCN2− groups. The C–N bond length is 0.1233(7) nm (Table 3), which corresponds to values reported for C–N double bonds in metal cyanamides.14 This value is respectively longer and shorter than the C–N triple and single bonds in PbCN2 (0.1156 nm for triple and 0.1297 nm for single).26 The Ba–N bond length in this new polymorph is 0.2928(5) nm, which is longer than the values reported for rhombohedral BaCN2 having the octahedral coordinated Ba2+ cations (0.2773–0.2867 nm).14 The face-sharing Ba2+ polyhedra along the c-axis result in a Ba–Ba distance of 0.35962(3) nm, whereas the distance in the ab-plane is 0.42602(2) nm and thus similar to that in rhombohedral BaCN2.
| R 1 = Σ||Fo| − |Fc||/Σ|Fo|. wR2 = [Σw(Fo2 − Fc2)2/Σ(wFo2)2]1/2, w = 1/[σ2(Fo2) + (aP)2 + bP], where Fo is the observed structure factor, Fc is the calculated structure factor, σ is the standard derivation of Fc2, and P = (Fo2 + 2Fc2)/3. S = [Σw(Fo2 − Fc2)2/(n − p)]1/2, where n is the number of reflections and p is the total number of parameters refined. | |
|---|---|
| Chemical formula | BaCN2 |
| Formula weight, Mr (g mol−1) | 177.37 |
| Crystal form, color | Granule, translucent colorless |
| Crystal size, mm3 | 0.064 × 0.052 × 0.036 |
| Radiation wavelength, λ (nm) | 0.071073 |
| Temperature, T (K) | 302 |
| Crystal system | Tetragonal |
| Space group | I4/mcm (No. 140) |
| Unit-cell dimensions, a (nm) | 0.60249(4) |
| c (nm) | 0.71924(5) |
| Unit-cell volume, V (nm3) | 0.26108(4) |
| Z | 4 |
| Calculated density, Dcal (Mg m−3) | 4.512 |
| Absorption coefficient, μ (mm−1) | 14.862 |
| Absorption correction | MULTI-SCAN (SADABS)20 |
| Limiting Indices | −7 ≤ h ≤ 7 |
| −7 ≤ k ≤ 7 | |
| −9 ≤ l ≤ 9 | |
| Number of reflections | 92 |
| Weight parameters, a, b | 0.0156, 4.0817 |
| Goodness-of-fit on F2, S | 1.392 |
| R 1, wR2 (I > 2σ(I)) | 0.0206, 0.0441 |
| R 1, wR2 (all data) | 0.0246, 0.0454 |
| Atom | Site | Occ. | x | y | z |
|---|---|---|---|---|---|
| U ij × 10−2 nm2 (= Å2). | |||||
| Ba | 4a | 1 | 0 | 0 | 0.25 |
| C | 4d | 1 | 0.5 | 0 | 0 |
| N | 8h | 1 | 0.3533(11) | 0.1447(11) | 0 |
| Atom | Site | U 11 = U22 | U 33 | U 12 | U 23 = U13 |
|---|---|---|---|---|---|
| Ba | 4a | 0.0106(3) | 0.0118(4) | 0 | 0 |
| C | 4d | 0.008(3) | 0.009(5) | 0.003(9) | 0 |
| N | 8h | 0.008(2) | 0.015(4) | −0.001(3) | 0 |
| C–N | ×2 | 0.1233(7) |
| Ba–N | ×8 | 0.2928(5) |
| Ba–Ba | ×2 | 0.35962(3) |
| ×4 | 0.42602(2) |
The FT-IR spectrum of this compound exhibits an asymmetric stretch peak (νas) at 1960 cm−1 and deformation vibration peaks (δ) at 670 and 680 cm−1 (Fig. 2). These peak positions are in good agreement with those generated by both rhombohedral BaCN2 and α-SrCN2, each of which are metal carbodiimides having two symmetric N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bonds.10,14 The tetragonal BaCN2 obtained in this work is therefore believed to contain carbodiimide anions ([N
N double bonds.10,14 The tetragonal BaCN2 obtained in this work is therefore believed to contain carbodiimide anions ([N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N]2−). To confirm the tetragonal structure, a powder XRD pattern was calculated using the Rietveld program based on the structural parameters in Tables 1 and 2, and a comparison of the observed and the calculated powder XRD patterns is presented in Fig. 3. The experimental diffraction lines are in good agreement with the calculated pattern, although several additional weak reflections are also present in the former. The impurity phase associated with these reflections has not yet been identified but might consist of a metal cyanamide having both triple and single bonds between C and N, for which there is some evidence in the FT-IR spectrum (Fig. 2).
N]2−). To confirm the tetragonal structure, a powder XRD pattern was calculated using the Rietveld program based on the structural parameters in Tables 1 and 2, and a comparison of the observed and the calculated powder XRD patterns is presented in Fig. 3. The experimental diffraction lines are in good agreement with the calculated pattern, although several additional weak reflections are also present in the former. The impurity phase associated with these reflections has not yet been identified but might consist of a metal cyanamide having both triple and single bonds between C and N, for which there is some evidence in the FT-IR spectrum (Fig. 2).
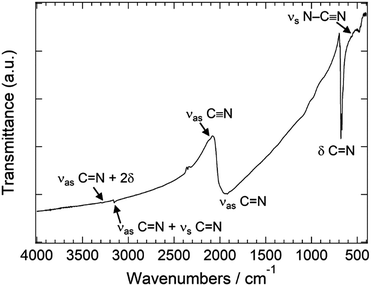 | ||
| Fig. 2 FT-IR spectrum of the nitrided product. The symbols νas, νs, and δ correspond to the absorptions for asymmetric stretch, symmetric stretch and deformation vibrations, respectively. | ||
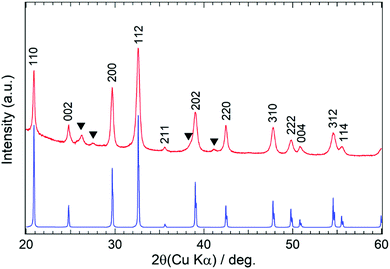 | ||
| Fig. 3 Powder XRD pattern of the nitrided product (red line) and the pattern calculated using the structural parameters in Tables 1 and 2 (blue line). Black triangles indicate diffraction lines attributed to an impurity phase. | ||
Thermogravimetric and differential thermal analyses (TG-DTA) for the tetragonal BaCN2 under a N2 flow were performed in our previous study and indicate an endothermic peak at approximately 910 °C without a weight loss corresponding to the melting of BaCN2.27 A solidified product of BaCN2 after annealing above the melting temperature contains rhombohedral BaCN2 instead of the tetragonal phase. We did not find an additional signal in the DTA curve indicating the phase transition, however we believe the transition temperature was obscured by the large endothermic peak of the melting, and the high temperature phase is the rhombohedral BaCN2. The phase transition from the tetragonal to rhombohedral BaCN2 is irreversible because there is no trace of the tetragonal phase in the XRD pattern of the annealed product. Density of tetragonal BaCN2 is 4.512 g cm−3, which is higher than that of rhombohedral BaCN2 (3.738 g cm−3).14 Interestingly, a low-temperature but high-symmetry phase (I4/mcm) transformed to a high-temperature but low-symmetry (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c) phase. The mechanism of the phase transition is still unknown, however this counterintuitive phase transition was also reported in SrCN2, in which the phase transition from a hexagonal high-symmetry β-phase to an orthorhombic low-symmetry α-phase occurs around 920 K and the phase transition is irreversible upon cooling, similar to the case of BaCN2.28
c) phase. The mechanism of the phase transition is still unknown, however this counterintuitive phase transition was also reported in SrCN2, in which the phase transition from a hexagonal high-symmetry β-phase to an orthorhombic low-symmetry α-phase occurs around 920 K and the phase transition is irreversible upon cooling, similar to the case of BaCN2.28
Luminescence properties
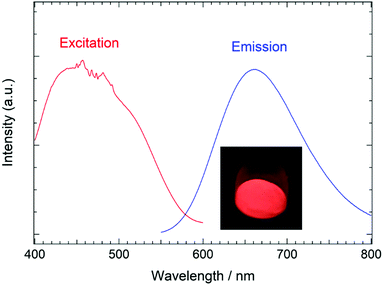
Eu2+-doped tetragonal BaCN2 was obtained via the nitridation of a mixture of BaCO3 and Eu(acac)3nH2O under a flow of NH3. The lattice parameters of the tetragonal BaCN2 were found to decrease with increasing Eu2+ content, eventually reaching constant values at 1 at% Eu2+, as shown in Fig. S2 (ESI†). This result indicates that the Eu2+ ions were incorporated into the tetragonal BaCN2 lattice. The photoluminescence properties of the 1 at% Eu2+-doped tetragonal BaCN2 were assessed, and a single broad red emission peak at 660 nm (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 152 cm−1) was observed in response to excitation at 435 nm. The full width at half maximum of this peak was 120 nm (2777 cm−1), as can be seen in Fig. 4. An excitation band ranging from 400 to 550 nm with two maximums at 435 nm (22
152 cm−1) was observed in response to excitation at 435 nm. The full width at half maximum of this peak was 120 nm (2777 cm−1), as can be seen in Fig. 4. An excitation band ranging from 400 to 550 nm with two maximums at 435 nm (22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 989 cm−1) and 500 nm (20
989 cm−1) and 500 nm (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1) was observed upon monitoring the emission at 660 nm. The presence of Eu2+ in the phosphor was supported by an absorption peak at 6972 eV in the Eu LIII-edge XANES data, which is in good agreement with the absorption energy for EuCl2. Both the excitation and emission of this compound are attributed to the transitions between the 4f7 and 4f65d1 energy levels of the Eu2+ ions. The red emission could be excited by irradiation at approximately 450 nm, which is the most commonly used wavelength in LEDs. Varying the Eu2+ concentration did not change the emission peak position significantly, indicating that the Eu2+ ion underwent low fluctuations in the chemical environment with increasing the Eu2+ concentration. The QE of the red emission with excitation at 465 nm was found to be 42% at room temperature.
000 cm−1) was observed upon monitoring the emission at 660 nm. The presence of Eu2+ in the phosphor was supported by an absorption peak at 6972 eV in the Eu LIII-edge XANES data, which is in good agreement with the absorption energy for EuCl2. Both the excitation and emission of this compound are attributed to the transitions between the 4f7 and 4f65d1 energy levels of the Eu2+ ions. The red emission could be excited by irradiation at approximately 450 nm, which is the most commonly used wavelength in LEDs. Varying the Eu2+ concentration did not change the emission peak position significantly, indicating that the Eu2+ ion underwent low fluctuations in the chemical environment with increasing the Eu2+ concentration. The QE of the red emission with excitation at 465 nm was found to be 42% at room temperature.
The luminescence of Eu2+ ions in a host material is influenced by structural parameters, including covalency, bond-length and coordination number, and the 4f–5d transition energy of phosphors doped with Eu2+ can be estimated by using an empirical equation.29,30 The equation indicates that an increase in the coordination number and the size of the substituted cation will induce a blue-shift in the emission wavelength of a Eu2+-doped phosphor. As noted, red emission at 660 nm was observed in the case of the Eu2+-doped tetragonal BaCN2. By comparing its crystal structure with that of Ba(SCN)2:Eu2+, which emits green light at 511 nm, it is evident that the tetragonal BaCN2 had a shorter Ba–N bond length (0.2928(5) nm) compared to the Ba–N (and S) bonds in Ba(SCN)2, for which the average bond length is 0.3086 nm.18 The Ba2+ ions in BaCN2 were coordinated in a regular square antiprism (D4d point symmetry) environment having identical Ba–N bond lengths, while Ba(SCN)2 is based on a distorted square antiprism coordination (C2 point symmetry) in association with a wide range of Ba–N and Ba–S bond lengths. The greater negative charge of the NCN2− anions forming the symmetric environment in BaCN2 will tend to stabilize the d orbitals of the Eu2+ ions, in contrast to the distorted coordination of the monovalent SCN− anions. As a result, increased crystal field splitting is expected in the BaCN2 host material, leading to the red emission of the Eu2+-doped BaCN2, because of the reduced energy difference between the 4f7 ground state and the 4f65d1 excited state. Conversely, this explanation does not support the shorter emission wavelength of Eu2+-doped SrCN2, in which each cation is coordinated with six NCN2− anions in a distorted octahedral environment.13,17 The empirical equation noted above is, of course, not applicable to all phosphors. As an example, the emission peak is shifted toward longer wavelengths upon changing the M site cation from Ca → Sr → Ba in M2Si5N8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Eu2+ (Ca2Si5N8: 610 nm, Sr2Si5N8: 630 nm, Ba2Si5N8: 640 nm).31 The equation does not take into account the effect of site symmetry on the emission peak shift, so the symmetric square antiprism coordination in BaCN2 might increase the crystal field splitting to a greater extent than distorted octahedral coordination, such that the energy position of the 5d excited state is reduced.
Eu2+ (Ca2Si5N8: 610 nm, Sr2Si5N8: 630 nm, Ba2Si5N8: 640 nm).31 The equation does not take into account the effect of site symmetry on the emission peak shift, so the symmetric square antiprism coordination in BaCN2 might increase the crystal field splitting to a greater extent than distorted octahedral coordination, such that the energy position of the 5d excited state is reduced.
Temperature dependence of luminescence
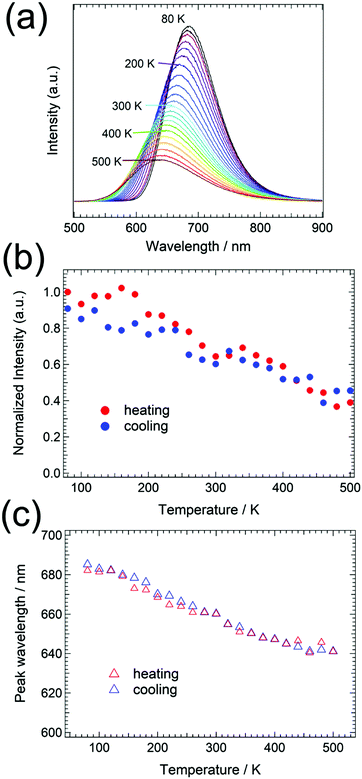
The effects of temperature on emission were examined between 80 K and 500 K. As shown in Fig. 5, the emission peak wavelength shifts from 680 nm at 80 K to 640 nm at 500 K, and the thermal quenching temperature (T1/2), at which the emission intensity is reduced by 50%, was approximately 400 K. Both emission intensity and peak wavelength were recovered again by decreasing the temperature (Fig. 5(b) and (c)). This strong red emission at ambient temperature is in contrast to that of Eu2+-doped Ba(SCN)2, which is a green phosphor with a lower T1/2 of 181 K. Reduced thermal quenching is usually associated with a small Stokes shift. As an example, Eu2+-doped SrLiAl3N4 shows narrow band red emission with minimal thermal quenching and has an estimated Stokes shift of 956 cm−1.32 Minimal thermal quenching is also observed for α-Sialon:Eu2+ and CaAlSiN3:Eu2+ phosphors, which have Stokes shifts of 5011 cm−1 and 2000 cm−1, respectively.33,34 According to P. Dorenbos, the energy position of the lowest excited 4f65d1 state can be estimated at about 20% of the excitation maximum to be 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 762 cm−1.35 This leads to a Stokes shift of about 2610 cm−1, which is comparable to that of the nitride phosphors having excellent thermal stability. A larger Stokes shift in a Eu2+-doped phosphor has been reported to occur in a more asymmetric activator site geometry, because of a significant reorganization of anions around the Eu2+ emission center under photoexcitation.36,37 In contrast, the presence of alkaline earth ions having larger ionic radii in the host lattice results in a smaller Stokes shift because of restricted structural relaxation around the activator in its excited state.38 The Ba2+ sites in BaCN2 are coordinated in a symmetric environment, each of which has an identical activator-N bond length. So, the smaller Stokes shift in the Eu2+-doped BaCN2 is related with the symmetric geometry of the Ba site and large bond length.
762 cm−1.35 This leads to a Stokes shift of about 2610 cm−1, which is comparable to that of the nitride phosphors having excellent thermal stability. A larger Stokes shift in a Eu2+-doped phosphor has been reported to occur in a more asymmetric activator site geometry, because of a significant reorganization of anions around the Eu2+ emission center under photoexcitation.36,37 In contrast, the presence of alkaline earth ions having larger ionic radii in the host lattice results in a smaller Stokes shift because of restricted structural relaxation around the activator in its excited state.38 The Ba2+ sites in BaCN2 are coordinated in a symmetric environment, each of which has an identical activator-N bond length. So, the smaller Stokes shift in the Eu2+-doped BaCN2 is related with the symmetric geometry of the Ba site and large bond length.
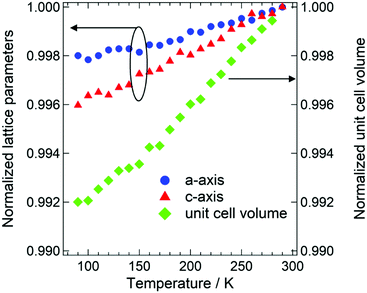
Thermally stable phosphors such as the nitrides mentioned above do not exhibit significant changes in emission wavelength with increasing temperature (typically less than 1 nm on going from room temperature to 423 K and above), which is indicative of the high thermal stability of the chromatic behavior.32–34 This thermal stability is caused by the rigid structure constructed from [SiN4] and/or [AlN4] tetrahedra. However, a large shift in the emission wavelength (from 680 nm at 80 K to 640 nm at 500 K) of BaCN2:Eu2+ is evident in Fig. 5(c). In the case of nitride phosphors, the emission wavelength is usually tailored by substitution between alkaline earth ions that modifies the environment around the activator ions, such as the crystal field strength, symmetry and polyhedron volume. As an example, a blue shift in the emission spectrum is observed for CaAlSiN4:Eu2+ upon substituting Sr for Ca.39 The emission peak shifts from 650 nm for CaAlSiN4:Eu2+ to 610 nm for SrAlSiN4:Eu2+, which has a 4% larger unit cell volume. In addition, a red shift of the emission peak of the Sr2Si5N8:Eu2+ phosphor is achieved by replacing Sr with Ca, accompanied by a reduction in the unit cell volume.40 To assess these effects in the present material, the temperature dependence of the lattice parameters of BaCN2 was examined between 90 K and 290 K. The normalized lattice parameters and the unit cell volume values are plotted in Fig. 6. The lattice shrinkage along the c-axis was estimated to be 0.4% over this temperature range, which was two times that along the a-axis (0.2%) because of the stacking structure of the Ba2+ and NCN2− ions along the c-axis. The unit cell volume was reduced by 0.8% over this same temperature range, which would be associated with a reduction in the Ba–N bond length resulting in significant crystal field splitting of the 5d states. The reduced 5d energy position in turn decreases the energy difference between the ground 4f7 state and the excited 4f65d1 state and induces a large red-shift in the emission wavelength.
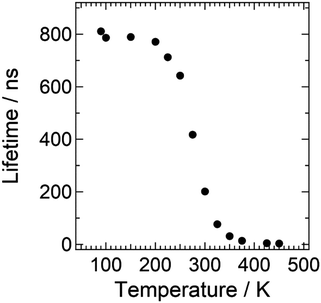
No crystalline phase transition of the BaCN2:Eu phosphor was observed in the powder XRD patterns between 138 K and 500 K, as shown in Fig. S3 (ESI†). The tetragonal BaCN2 crystal structure was also confirmed at 90 K by using single crystal XRD (Tables S1 and S2, ESI†). To further investigate the emission property depending on the temperature, fluorescence decay curves upon changing temperature were measured by monitoring 650 nm luminescence, and they are shown in Fig. S4 (ESI†). The decay curves were fitted using a single exponential function to estimate the lifetime. Fig. 7 shows the temperature dependence of the lifetime for the 5d level of Eu2+ doped in the BaCN2 host lattice. From the lifetime, the quenching temperature T1/2,![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lifetime of 277 K was obtained and found to be different from the T1/2 value estimated from the temperature dependence of emission intensity. It is already known that photoluminescence intensity as a function of temperature can be influenced by many factors such as the changes in absorption strength with temperature and additional intensity due to thermoluminescence.
lifetime of 277 K was obtained and found to be different from the T1/2 value estimated from the temperature dependence of emission intensity. It is already known that photoluminescence intensity as a function of temperature can be influenced by many factors such as the changes in absorption strength with temperature and additional intensity due to thermoluminescence.
Both the radiative rate constant (kr) and nonradiative rate constant (knr) were estimated by the combination of the lifetime and estimated QE at different temperatures. Variation of kr upon changing the temperature shown in Fig. S5 (ESI†) implies a change in crystal field splitting of the 5d states of Eu2+ with temperature,42,43 which was induced by the significant change of the lattice parameters.
Interestingly, this wide variation in the emission spectrum upon changing the temperature could have applications in temperature sensing devices.41 Optical temperature sensing has been previously investigated using various molecules and phosphors having a multi emission-center.44–46 The pronounced temperature dependence of the emission wavelength of the BaCN2:Eu2+ phosphor is attributed to changes in the crystal field strength resulting from the varying distance between Eu and N atoms. BaCN2 was also found to have large thermal expansion coefficients, αa = 1.5 × 10−5 K−1 and αc = 2.3 × 10−5 K−1 at 290 K, as estimated from the lattice parameters. These values are almost one order of magnitude larger than that of Si3N4 (3.0 × 10−6 K−1).47 The relatively “soft” host lattice of BaCN2 thus leads to a wide variation in the emission wavelength with temperature, induced by changes in the crystal field splitting of the 5d energy levels of Eu2+ ions. BaCN2:Eu obtained in this work is highly air-sensitive. Recently, we found the product is stable in organic solvents such as hexane and acetone and silicone resin. Moisture resistive coating in organic solution should be further investigated to realize applications as a temperature sensing phosphor.
Conclusion
A new form of BaCN2 with a tetragonal structure was prepared by a simple nitridation reaction of BaCO3 under an NH3 flow. The crystal structure is an isotype of alkaline cyanates (AOCN, A = K, Rb, Cs), in which Ba2+ ion is coordinated with eight N atoms in [N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N]2− groups to construct a regular square antiprism environment. Eu2+-doped tetragonal BaCN2 exhibited a strong red emission peak at 660 nm in response to irradiation with blue or green light and showed relatively small thermal quenching compared with other cyanate and thiocyanate phosphors. The red emission shifts toward longer wavelengths (up to 680 nm) upon decreasing the measurement temperature, as a result of shrinkage of the host lattice. The present results demonstrated that Eu2+-doped BaCN2 is a promising new red-emitting phosphor to be utilized in temperature sensing devices.
N]2− groups to construct a regular square antiprism environment. Eu2+-doped tetragonal BaCN2 exhibited a strong red emission peak at 660 nm in response to irradiation with blue or green light and showed relatively small thermal quenching compared with other cyanate and thiocyanate phosphors. The red emission shifts toward longer wavelengths (up to 680 nm) upon decreasing the measurement temperature, as a result of shrinkage of the host lattice. The present results demonstrated that Eu2+-doped BaCN2 is a promising new red-emitting phosphor to be utilized in temperature sensing devices.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was partly supported by the JSPS Grants-in-Aid for Scientific Research on Innovative Areas “Mixed Anion” (grant numbers JP16H06439 and JP16H06441). This work was also performed under the Cooperative Research Program of “Network Joint Research Center for Materials and Devices.”Notes and references
- R. J. Xie and H. T. Hintzen, J. Am. Ceram. Soc., 2013, 96, 665 CrossRef.
- L. Chen, C. C. Lin, C. W. Yeh and R. S. Liu, Materials, 2010, 3, 2172 CrossRef.
- Z. Xia, Z. Xu, M. Chen and Q. Liu, Dalton Trans., 2016, 45, 11214 RSC.
- R. J. Xie and N. Hirosaki, Sci. Technol. Adv. Mater., 2007, 8, 588 CrossRef.
- R. J. Xie, N. Hirosaki, K. Sakuma and N. Kimura, J. Phys. D: Appl. Phys., 2008, 41, 144013 CrossRef.
- M. Krott, X. Liu, B. P. T. Fokwa, M. Speldrich, H. Lueken and R. Dronskowski, Inorg. Chem., 2007, 46, 2204 CrossRef PubMed.
- K. Morita, G. Mera, K. Yoshida, Y. Ikuhara, A. Klein, H. J. Kleebe and R. Riedel, Solid State Sci., 2013, 23, 50 CrossRef.
- G. Baldinozzi, B. Malinowska, M. Rakib and G. Durand, J. Mater. Chem., 2002, 21, 268 RSC.
- X. Liu, M. Krott, P. Muller, C. Hu, H. Lueken and R. Dronskows, Inorg. Chem., 2005, 44, 3001 CrossRef PubMed.
- M. Krings, G. Montana, R. Dronskowski and C. Wickleder, Chem. Mater., 2011, 23, 1694 CrossRef.
- A. Cochet, Angew. Chem., 1931, 44, 367 CrossRef.
- A. Perret and A. M. Krawezynski, Helv. Chim. Acta, 1932, 15, 1009 CrossRef.
- S. Yuan, Y. Yang, F. Chevire, F. Tessier, X. Zhang and G. Chen, J. Am. Ceram. Soc., 2010, 93, 3052 CrossRef.
- U. Berger and W. Schnick, J. Alloys Compd., 1994, 206, 179 CrossRef.
- K. G. Strid, Z. Anorg. Allg. Chem., 1968, 360, 205 CrossRef.
- S. Pagano, G. Montana, C. Wickleder and W. Schnick, Chem. – Eur. J., 2009, 15, 6186 CrossRef PubMed.
- C. Wickleder, Chem. Mater., 2005, 17, 1228 CrossRef.
- C. Wickleder, J. Alloys Compd., 2004, 374, 10 CrossRef.
- B. A. I., APEX2, Madison, W1, USA, 2014.
- B. A. I., SADABS v. 2014/5, Madison, W1, USA, 2015.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3 CrossRef PubMed.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272 CrossRef.
- F. Izumi and K. Momma, Solid State Phenom., 2007, 130, 15 CrossRef.
- S. B. Hendricks and L. Pauling, J. Am. Chem. Soc., 1925, 47, 2904 CrossRef.
- T. C. Waddington, J. Chem. Soc., 1959, 0, 2499 RSC.
- X. Liu, A. Decker, D. Schmiz and R. Dronskowski, Z. Anorg. Allg. Chem., 2000, 626, 103 CrossRef.
- A. Hosono, Y. Masubuchi, T. Endo and S. Kikkawa, Dalton Trans., 2017, 46, 16837 RSC.
- M. Krings, M. Wessel, W. Wilsmann, P. Muller and R. Dronskowski, Inorg. Chem., 2010, 49, 2267 CrossRef PubMed.
- L. G. van Uitert, J. Lumin., 1984, 29, 1 CrossRef.
- R. J. Xie, N. Hirosaki, T. Takeda and T. Suehiro, ECS J. Solid State Sci. Technol., 2013, 2, R3031 CrossRef.
- Y. Q. Li, J. E. J. van Steen, J. H. W. wan Krevel, G. Botty, A. C. A. Delsing, F. J. DiSalvo, G. de With and H. T. Hintzen, J. Alloys Compd., 2006, 417, 273 CrossRef.
- P. Pust, V. Weiler, C. Hecht, A. Tücks, A. S. Wochnik, A. K. Henb, D. Wiechert, C. Scheu, P. J. Schmidt and W. Schnick, Nat. Mater., 2014, 13, 891 CrossRef PubMed.
- K. Shioi, N. Hirosaki, R. J. Xie, T. Takeda and Y. Q. Li, J. Mater. Sci., 2010, 45, 3198 CrossRef.
- K. Uheda, N. Hirosaki, Y. Yamamoto, A. Naito, T. Nakajima and H. Yamamoto, Electrochem. Solid-State Lett., 2006, 9, H22 CrossRef.
- P. Dorenbos, J. Phys.: Condens. Matter, 2003, 15, 575 CrossRef.
- G. J. Dirksen and G. Blasse, J. Solid State Chem., 1991, 92, 591 CrossRef.
- A. Diaz and D. A. Keszler, Mater. Res. Bull., 1996, 31, 147 CrossRef.
- A. Meijerink and G. Blasse, J. Lumin., 1989, 43, 283 CrossRef.
- H. Watanabe and N. Kijima, J. Alloys Compd., 2009, 475, 434 CrossRef.
- Y. Q. Li, G. de With and H. T. Hintzen, J. Solid State Chem., 2008, 181, 515 CrossRef.
- X. D. Wang, O. S. Wolfbeis and R. J. Meier, Chem. Soc. Rev., 2013, 42, 7834 RSC.
- Y. Hasegawa, S. Tsuruoka, T. Yoshida, H. Kawai and T. Kawai, J. Phys. Chem. A, 2008, 112, 803 CrossRef PubMed.
- T. Arakawa, M. Takakuwa and J. Shiokawa, Inorg. Chem., 1985, 24, 3807 CrossRef.
- P. Low, B. Kim, N. Takama and C. Bergaud, Small, 2008, 4, 908 CrossRef PubMed.
- K. Miyata, Y. Konno, T. Nakanishi, A. Kobayashi, M. Kato, K. Fushimi and Y. Hasegawa, Angew. Chem., Int. Ed., 2013, 52, 6413 CrossRef PubMed.
- F. Venturini, R. Bürgi, S. M. Borisov and I. Klimant, Sens. Actuators, A, 2015, 223, 324 CrossRef.
- S. Iwai and A. Yasunaga, Die Naturwissenschaften, 1959, 46, 473 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: Crystallographic Data (CSD 434273) in CIF format, powder XRD, lattice parameters. See DOI: 10.1039/c8tc01289j |
| This journal is © The Royal Society of Chemistry 2018 |