Open Access Article
Open Access ArticleRevival of the nearly extinct fluorescence of coumarin 6 in water and complete transfer of energy to rhodamine 123†
Rajashree
Banerjee
and
Pradipta
Purkayastha
 *
*
Department of Chemical Sciences, Indian Institute of Science Education and Research (IISER) Kolkata, Mohanpur 741246, WB, India. E-mail: ppurkayastha@iiserkol.ac.in
First published on 3rd August 2017
Abstract
The nearly extinct fluorescence of coumarin 6 in water due to microcrystal formation is revived by micelles. Practically complete transfer of energy from coumarin 6 to rhodamine 123 through resonance energy transfer could be achieved.
Introduction
An antenna system for light harvesting is considered to be efficient if it has a very high molar extinction coefficient, commendable photostability, and an ability to transfer its energy. Based on this concept many artificial light harvesting systems have been developed using dendrimers,1–3 biopolymers,4–6 porphyrin aggregates,7–9 nanocrystalline materials,10,11etc. The antenna materials absorb photons and transfer the excited state energy to an acceptor through Förster resonance energy transfer (FRET) as one of the options. This non-radiative excited state energy transfer phenomenon depends on the distance between donor and acceptor molecules and the overlap between the emission spectrum of the donor and the absorbance spectrum of the acceptor.12,13On several occasions coumarins have been used in polymers to harvest and transfer solar radiation energy.14–16 Systems were developed using fluorescent coumarin dyes (donors) covalently linked through an amide spacer to inorganic Ru(II) complexes (acceptors) where the peripheral coumarin antenna units sensitize the production of the metal-to-ligand charge transfer (MLCT) states of the complex.17 New coumarin derivatives were designed to produce light harvesting devices for constructing stable dye-sensitized solar cells.18 Ismail et al. included coumarin 6 (C6) (Fig. 1) in an active layer of a poly(3-hexylthiophene)(P3HT):[6,6]-phenyl-C61 butyric acid methyl ester (PCBM) to provide bulk heterojunction solar cell.19 They varied the amount of C6 in the active layer blends and recorded the optical absorption and photocurrent of the device.
C6, however, has a tendency to form microcrystals through H-type aggregation in solution.20–22 Early reports suggested the generation of C6 microcrystals through the reprecipitation method using a concentrated solution of the dye in acetone at room temperature with a large volume of water.20 Later, our group established that even at micromolar concentration and in an aqueous medium, C6 quickly forms microcrystals (Fig. 2).21 The C6 microcrystals formed in water lead to quick loss in C6 fluorescence and hence their applications become limited. In some of our previous studies, we have reported ways to retrieve C6 fluorescence through guest–host chemistry in an aqueous medium.21,23 Herein, we state that C6 fluorescence can be revived by using micelles in water followed by nearly complete transfer of the energy absorbed from light to an acceptor through Förster resonance energy transfer (FRET).
Results and discussion
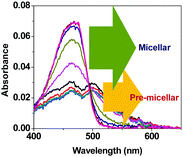
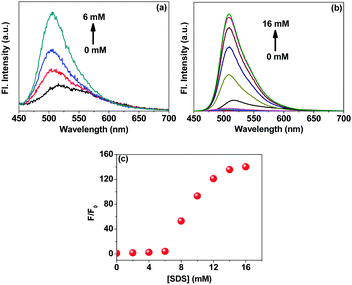
The aggregated C6 molecules (microcrystals) were treated with sodium dodecyl sulphate (SDS) in an aqueous medium. The absorption spectrum of C6 shows bands at around 460 and 500 nm in water after the microcrystal formation (Fig. 3). Absorbance at 500 nm, due to the aggregates,20–22 remains structurally unchanged upon addition of SDS up to ∼6 mM, indicating the existence of the aggregates under pre-micellar conditions. The spectrum changes abruptly at and above 8 mM SDS concentration where the absorption band due to the aggregates starts disappearing. Thus, it is evident that the C6 microcrystals begin to disaggregate in the presence of SDS micelles. This is understandable as C6 is a hydrophobic dye; hence the hydrophobic core of the micelle will attract the dye molecules. The predominating hydrophobic effect over the π–π stacking interaction leads to complete disintegration of the microcrystals.Although the absorption spectrum of C6 did not show much change in structure at the pre-micellar concentration of SDS, the fluorescence spectrum shows noticeable sharpening with a hypsochromic shift (Fig. 4a). This is probably due to the SDS molecules surrounding the C6 microcrystals providing a relatively less polar environment in the vicinity. The fluorescence intensity of C6 increases substantially upon formation of the SDS micelles due to revival of the molecular characteristics of C6 (Fig. 4b and c). Time-resolved emission decay data of the excited state of C6 in water take a biexponential fit. The faster and slower components decay in 526 ps and 2.53 ns, respectively. The slower one is reported to be for the C6 molecule itself.24 The faster component is for the intramolecular charge transfer (ICT) in C6.21Table 1 provides the excited state lifetimes of C6 under different conditions in an aqueous environment. In water, the C6 microcrystals have π–π stacking interactions and hence a fast decay component is observed. However, upon encapsulation in SDS micelles, a growth component suggests the occurrence of ICT in the C6 molecules in the excited state. This is possible as the C6 molecules regain their molecular identity upon being disaggregated by the micelles.
| Conditions | τ 1 (ps) | τ 2 (ns) | χ 2 |
|---|---|---|---|
| Monitored at 506 nm | |||
| C6 in H2O | 526 (18.68%) | 2.53 (81.32%) | 1.13 |
| C6 in SDS micelles | 680 (−5.49%) | 2.62 (105.49%) | 1.02 |
| [Rh123] (μM) | |||
| 1 | 416 (−4.97%) | 2.69 (104.97%) | 1.03 |
| 2.4 | 405 (−4.35%) | 2.75 (104.35%) | 1.04 |
| 3.4 | 367 (−3.8%) | 2.79 (103.8%) | 1.08 |
| 5.4 | 358 (−3.05%) | 2.88 (103.05%) | 1.12 |
| 6.6 | 352 (−1.73%) | 2.93 (101.73%) | 1.17 |
| Monitored at 540 nm | |||
| C6 in H2O | 648 (38%) | 2.22 (62%) | 1.11 |
| C6 in SDS micelles | 790 (−4.75%) | 2.62 (104.75%) | 0.98 |
| [Rh123] (μM) | |||
| 1 | — | 3.21 | 1.13 |
| 2.4 | — | 3.63 | 1.17 |
| 3.4 | — | 3.85 | 1.13 |
| 5.4 | — | 4.14 | 1.20 |
| 6.6 | — | 4.29 | 1.19 |
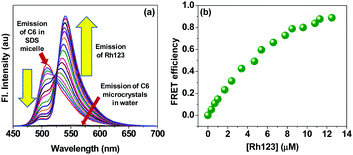
Now as the nearly extinct fluorescence from the C6 microcrystals could be revived by applying micelles from SDS in an aqueous environment, we further intended to investigate execution of energy transfer by C6 in an aqueous medium, which is otherwise impossible due to microcrystal formation and loss in fluorescence yield. Upon revival of fluorescence in water, C6 retains its molecular identity and is fit to act as an antenna material for energy harvesting. The spectroscopic characteristics of C6 and rhodamine 123 (Rh123) make them an appropriate FRET pair as in SDS micelles. The emission spectrum of C6 has an appreciable overlap with the absorption spectrum of Rh123 (Fig. S1, ESI†). We excited C6 in SDS micelles at 430 nm and gradually added Rh123 that accepts the emitted energy from C6 and emits at 540 nm (Fig. 5a). The calculated FRET efficiency shows nearly 100% transfer of energy from C6 in micelles to Rh123 (Fig. 5b). Hence, in totality, the concept fulfils the aspects for an efficient energy harvesting system in an aqueous environment.
The emission from C6 is due to excited state intramolecular charge transfer (ICT) that is confirmed by the time resolved emission study. After exciting the donor (C6) using a suitable excitation source (405 nm), emissions at 506 nm (emission of C6) and 540 nm (emission of Rh123) were monitored. The decay profiles are shown in Fig. 6 and the lifetimes calculated from the fits are tabulated in Table 1. With gradual addition of the acceptor (Rh123), excited state ICT in C6 (growth component) becomes faster as the energy is donated by C6 to the acceptor. Monitoring the emission of Rh123 shows the stability of the acceptor as its excited state lifetime gradually increases.
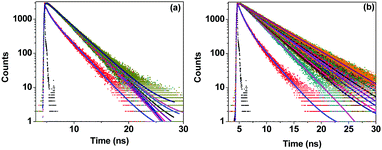 | ||
| Fig. 6 Time resolved decay traces for C6 in water, SDS micelles and added Rh123 monitored at (a) 506 nm and (b) 540 nm. The solid lines are fits to the raw data. The sample was excited at 405 nm. | ||
Conclusion
In summary, we could device a simple way to revive the fluorescence of C6 that disappears due to microcrystal formation in water through self-aggregation. Application of SDS micelles leads to disintegration of the aggregation by hydrophobic interaction and hence revives the lost fluorescence of C6. The energy from C6 could be transferred to Rh123 that acted as an acceptor in the FRET process. This work provides an efficient energy harvesting device in an aqueous environment under normal temperature and pressure.Acknowledgements
The authors gratefully acknowledge the financial help from the Department of Science and Technology, Science and Engineering Research Board, Government of India, through project number EMR/2015/000950. RB acknowledges IISER Kolkata for her fellowship.References
- G. Kodis, Y. Terazono, P. A. Liddell, J. Andreasson, V. Garg, M. Hambourger, T. A. Moore, A. L. Moore and D. Gust, J. Am. Chem. Soc., 2006, 128, 1818 CrossRef CAS PubMed.
- Y. H. Jeong, M. Son, H. Yoon, P. Kim, D. H. Lee, D. Kim and W. D. Jang, Angew. Chem., Int. Ed., 2014, 53, 7089 CrossRef.
- X. Zhang, Y. Zeng, T. Yu, J. Chen, G. Yang and Y. Li, J. Phys. Chem. Lett., 2014, 5, 2340 CrossRef CAS PubMed.
- R. A. Miller, A. D. Presley and M. B. Francis, J. Am. Chem. Soc., 2007, 129, 3104 CrossRef CAS PubMed.
- C. V. Kumar and M. R. Duff, J. Am. Chem. Soc., 2009, 131, 16024 CrossRef CAS PubMed.
- K. J. Channon, G. L. Devlin and C. E. MacPhee, J. Am. Chem. Soc., 2009, 131, 12520 CrossRef CAS PubMed.
- N. Aratani, D. Kim and A. Osuka, Acc. Chem. Res., 2009, 42, 1922 CrossRef CAS PubMed.
- Y. Ishida, T. Shimada, D. Masui, H. Tachibana, H. Inoue and S. Takagi, J. Am. Chem. Soc., 2011, 133, 14280 CrossRef CAS PubMed.
- M. R. Wasielewski, Acc. Chem. Res., 2009, 42, 1910 CrossRef CAS PubMed.
- A. O. Govorov, Adv. Mater., 2008, 20, 4330 CrossRef CAS.
- I. Robel, V. Subramanian, M. Kuno and P. V. Kamat, J. Am. Chem. Soc., 2006, 128, 2385 CrossRef CAS PubMed.
- G. D. Scholes and G. R. Fleming, J. Phys. Chem. B, 2000, 104, 1854 CrossRef CAS.
- R. van Grondelle and V. I. Novoderezhkin, Phys. Chem. Chem. Phys., 2006, 8, 793 RSC.
- A. Adronov, D. R. Robello and J. M. J. Fréchet, J. Polym. Sci., Part A: Polym. Chem., 2001, 39, 1366 CrossRef CAS.
- S. L. Gilat, A. Adronov and J. M. J. Fréchet, J. Org. Chem., 1999, 64, 7474 CrossRef CAS.
- J. Serin, X. Schultze, A. Adronov and J. M. J. Fréchet, Macromolecules, 2002, 35, 5396 CrossRef CAS.
- D. S. Tyson and F. N. Castellano, Inorg. Chem., 1999, 38, 4382 CrossRef CAS PubMed.
- Z. S. Wang, Y. Cui, K. Hara, Y. Dan-oh, C. Kasada and A. Shinpo, Adv. Mater., 2007, 19, 1138 CrossRef CAS.
- Y. A. M. Ismail, T. Soga and T. Jimbo, Sol. Energy. Mater. Sol. Cells, 2010, 94, 1406 CrossRef CAS.
- S. Fery-Forgues, R. El-Ayoubi and J.-F. Lamère, J. Fluoresc., 2008, 18, 619 CrossRef CAS PubMed.
- P. Ghosh, T. Das, A. Maity, S. Mondal and P. Purkayastha, RSC Adv., 2015, 5, 4214 RSC.
- X. Miao, Y. Li, X. Wang, S. M.-Y. Lee and Y. Zheng, ACS Appl. Mater. Interfaces, 2016, 8, 12620 CAS.
- R. Banerjee, S. Mondal and P. Purkayastha, RSC Adv., 2016, 6, 105347 RSC.
- A. S. Kristoffersen, S. R. Erga, B. Hamre and Ø. Frette, J. Fluoresc., 2014, 24, 1015 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details and spectra of the donor and the acceptor used in FRET. See DOI: 10.1039/c7sm01198a |
| This journal is © The Royal Society of Chemistry 2017 |