 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Hydration or hydroxylation: direct synthesis of fullerenol from pristine fullerene [C60] via acoustic cavitation in the presence of hydrogen peroxide
Sadia Afreena,
Ken Kokubob,
Kasturi Muthoosamya and
Sivakumar Manickam *a
*a
aDepartment of Chemical & Environmental Engineering, The University of Nottingham Malaysia Campus, Jalan Broga, Semenyih, Selangor D.E. 43500, Malaysia. E-mail: Sivakumar.Manickam@nottingham.edu.my
bDivision of Applied Chemistry, Graduate School of Engineering, Osaka University, 2-1 Yamadaoka, Suita, Osaka 565-0871, Japan
First published on 21st June 2017
Abstract
A green and clean approach that requires low energy and avoids the use of any toxic or corrosive reagents/solvents for the synthesis of potential fullerenol moieties [C60(OH)n·mH2O] was proposed in this investigation, in which pristine fullerene (C60) in dil. H2O2 (30%) aqueous media was ultrasonicated (20 kHz, 200 W) at 30% amplitude for 1 h. The attachment of hydroxyl groups (–OH) was investigated via FTIR and the quantification of –OH groups attached to the C60 cage was conducted via elemental analysis. The number of secondary bound water molecules (mH2O) with each fullerenol molecule [C60(OH)n] was measured via TGA, and the estimated average structure of fullerenol was calculated to be C60(OH)8·2H2O. The synthesized fullerenol was moderately soluble in water and DMSO. Furthermore, the size of the synthesized C60(OH)8·2H2O particles determined by both AFM and DLS analysis was found to be in the range of 135–155 nm. The proposed ultrasound-assisted acoustic cavitation technique encompasses a one-step facile reaction strategy, requires less time for the reaction, and reduces the number of solvents required for the separation and purification of C60(OH)8·2H2O, which could be scalable for the commercial synthesis of fullerenol moieties in the future.
Introduction
The discovery of fullerene (C60) by Sir Harold Kroto and his group in 1985 1,2 pioneered the new chapter of fullerene chemistry in the domain of carbon allotropes and gradually this new area of chemistry has provided versatile fullerene (C60) derivatives3 with potential features that could be exploited in numerous technological applications. Fullerene C60, which is specifically known as Buckminster fullerene, is a carbon allotrope and has been incessantly reported as a useful potential carbon nanomaterial for various biological and metallurgical applications.4–6 However, owing to its insolubility in most organic and inorganic solvents,7,8 it is difficult to employ in many prospective studies. This tough to dissolve feature could be overcome by introducing various hydrophilic functional groups on the C60 cage.9–14 Fullerenol, which is also known as fullerol, polyhydroxylated fullerene and hydroxylated fullerene, is one of the mostly pronounced and water-soluble fullerene derivatives15 that has been derived by the hydroxylation of the C60 molecule in various ways (both solvent-associated and solvent-free methods) over the past few years. Ever since the first preparation of fullerenol, it has been a great challenge to increase the attachment of more hydroxyl groups (–OH) onto the C60 cage as well as to make the synthesis simpler and faster. The attachment of the largest number of –OH groups [C60(OH)44·8H2O] has been reported by Kokubo et al.16 Zhang et al.17 reported the synthesis of C60(OH)27.2 via mechanochemical means where potassium hydroxide was used as the hydroxylation reagent with C60 and the two mixed vigorously in a ball mill. Wang et al.18 reported another solvent-free reaction path to obtain C60(OH)16 using a dil. H2O2 (30%) and sodium hydroxide mixture. The use of alkali was very common in almost all the reported successful methods for the preparation of fullerenol together with other chemicals, e.g., sulfuric acid (H2SO4) and nitric acid (HNO3), various solvents e.g., toluene (C7H8), benzene (C6H6) and tetrahydrofuran (THF) and phase transfer catalysts (PTC) e.g., tetrabutylammonium hydroxide (TBAH).19–21 The methods proposed by Zhang et al.,22 Alves et al.,23 Kokubo et al.,24 Lu et al.,25 Zhang et al.26 and Wang et al.27 to prepare fullerenols with different numbers of –OH groups are also associated with the use of H2O2, NaOH and in some cases PTC. However, although the previous methods were proven to be successful for the synthesis of moderate to highly soluble fullerenols, it is difficult to remove the impurities obtained from NaOH and PTC which contaminate the synthesized fullerenol.27,28 In some cases the higher solubility of fullerenol was due to the presence of Na+ impurity introduced during the synthesis.23Also, the reaction time is much longer with these methods (from several hours to days) to generate and incorporate –OH groups onto the C60 cage. In this context, the development of simpler and faster approaches for the synthesis fullerenol, which are tailored by the use of minimal reagents and customized with easy purification and separation steps, is urgently required in fullerene chemistry. In this investigation, a facile method is demonstrated to overcome the above-mentioned barriers to a great extent via the direct ultrasonication of C60 in the presence of dil. H2O2 (30%).
Several studies29–31 evidence that ultrasonication in H2O2 associated aqueous media results in the formation of the hydroxyl radical (˙OH) which generates hydrated C60 as C60@(H2O)n.32–34 Alternatively, it will be advantageous if the formation of ˙OH radicals can be tuned to form potential fullerenol moieties as well rather than just leaving it as hydrated C60. Based on this, we explore an ultrasound induced acoustic cavitation strategy whereby with optimal ultrasonic variables (30% amplitude and 1 h sonication at pulse mode), pristine C60 is functionalized with –OH groups in aqueous media in the presence of dil. H2O2 (30%). Following the synthesis, quantitative analysis is conducted with the functionalized C60 to determine the average structure of fullerenol that could be potentially derived by this ultrasound assisted acoustic cavitation technique.
It is worth to mentioning that the synthesis of fullerenol using H2O2 as a hydroxylation reagent has been practiced before, but in association with other solvents and/or reagents and PTC as well.18,24 In this regard, herein, we propose a simpler technique which avoids the use of multiple reagents/solvents as well as PTC and thus produces fullerenol more easily and efficiently in comparison to the methods reported thus far. Fig. 1 represents the chronological development of the methods proposed for the synthesis of fullerenol over years and the salient features of the technique proposed in this study in comparison. Only dil. H2O2 (30%) is used as a hydroxylation reagent and no other supporting reagents and/or solvents or PTC are used for the synthesis. Besides, the reaction time is reduced to 1 h and unreacted C60 is only present as an impurity, the separation of which is easy after the reaction.
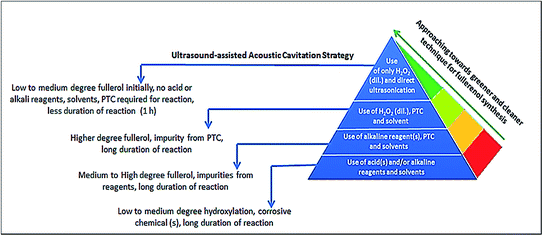 | ||
| Fig. 1 Applying the greener and cleaner ultrasonic cavitation strategy to synthesize fullerenol in a facile and faster way compared to other conventional methods. | ||
In the present method direct ultrasonication induces cavitation bubbles in the liquid H2O2 and C60 containing aqueous media. Continuous formation and then their collapse generate high energy transient hot spots inside the liquid media which dissociate water molecules into hydrogen and hydroxyl radicals. These hydroxyl radicals in turn combine and form H2O2. Further disassociation of H2O2 due to the effect of acoustic cavitation generates –OOH anions and/or ˙OH radicals which are exohedrally attached to the C60 cage by either nucleophilic attack or successive radical addition, respectively.35–39 Fig. 2 represents the experimental conditions for the synthesis of fullerenol.
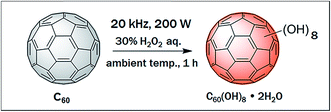 | ||
| Fig. 2 Synthesis of water soluble fullerenol via acoustic cavitation induced by ultrasound at ambient temperature, within 1 h reaction time and in the presence of dil. H2O2 (30%). | ||
The attachment of –OH groups onto the C60 cage was identified by Fourier transform infrared spectroscopy (FTIR) and the number of –OH groups and bound water molecules were determined by elemental analysis and thermogravimetric analysis (TGA). The common formula of fullerenol is C60(OH)n, where n is the number of –OH groups attached to each C60 cage which could vary from 2 to 44.16,24,40 However, the presence of –OH groups on the C60 cage also binds some water molecules, and the number of bound water molecules increases with an increase in the number of –OH groups attached to each C60 moiety. Therefore, the most accurate formula of the fullerenol molecule that could be obtained practically is C60(OH)n·mH2O,24,39 where m is the number of secondary bound water molecules associated with each fullerenol moiety. Elemental analysis together with TGA clearly support that the average structure of the synthesized fullerenol obtained by the present ultrasound-assisted technique is C60(OH)8·2H2O.
Experimental
Materials & equipment
Pristine C60 (98%) was purchased from Sigma Aldrich (USA) and used as the starting material to synthesize fullerenol. Hydrogen peroxide (H2O2) aqueous solution (30% reagent grade) from R&M chemicals (UK) was used as the hydroxylation reagent. Type II pure water (TOC < 50 ppb) was obtained from a Milli-Q system (Merck Millipore Integral 5, France). A Bandelin Sonoplus (UW 3200, 20 kHz, 200 W, Germany) with a titanium horn sonotrode (MS 73) was employed to introduce ultrasound. A graduated centrifuge tube (50 mL, angle 60° conical bottom) was used as the reactor or treatment vessel. A refrigerated circulator water bath (Julabo F34-ED, Germany) was used to maintain the reaction temperature close to ambient temperature during ultrasonication. Toluene (AR grade) was obtained from R&M Chemicals (Malaysia) for the separation and purification of unreacted C60 from C60(OH)8·2H2O. Dimethyl sulfoxide (DMSO) was obtained from Wako Pure Chemical Industries, Ltd (Japan) to check the solubility of synthesized fullerenol. After separation and purification, the C60(OH)8·2H2O dispersion was dried in a freeze dryer (Christ Alpha 1-2 LDplus, Germany).Characterization
The formation and attachment of –OH groups onto the C60 cage was identified by Fourier transform infrared spectroscopy (FTIR) (JASCO FT/IR-4100). Quantification of the attached –OH groups was attained by elemental analysis using a Yanaco, CHN Corder MT-6. Thermogravimetric analysis (TGA) was performed on a Mettler Toledo instrument (TGA/DSC 1/LF/1100, Switzerland) to measure the amount of secondary bound water molecules with C60(OH)8. The particle size of C60(OH)8·2H2O in solution was measured using a Photal, FPAR-1000HR. The thickness of the C60(OH)8·2H2O particles was examined via a 5500 Agilent Technologies AFM (USA) using an ultra-sharp tip (non-contact high resonance frequency, nanosensor probe). The morphological study was carried out using a Quanta 400 (USA) field emission scanning electron microscope (FE-SEM).Synthesis of C60(OH)n·mH2O
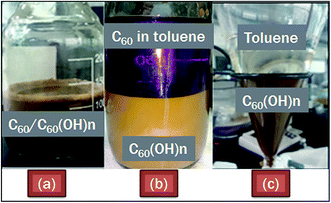
Pure C60 (200 mg) was added to 30% H2O2 (20 mL) and subjected to ultrasonication (30% amplitude, 200 W, pulse mode) for 1 h at ambient temperature. To avoid a rapid increase in the temperature owing to ultrasound dissipation through the liquid media, the reactor was fitted with a refrigerated circulator water bath which maintained the temperature inside the reactor close to ambient temperature. Initially, C60 was immiscible in aqueous H2O2 and was a colorless heterogeneous mixture which turned light brown after 30 min of ultrasonication. Subsequently, in the next 30 min of ultrasonication it turned into a completely dark brown dispersion (Fig. 3a).Separation and purification of C60(OH)n·mH2O
Since pure C60 was used as the starting material to synthesize fullerenol and no other reagents were used except 30% H2O2 for hydroxylation, after the reaction it was easier to separate the impurity, i.e. unreacted C60, than the reported methods. After washing the dark brown dispersion with an equal volume of toluene 10 times, unreacted C60 was separated from C60(OH)n·mH2O. After adding toluene in the dispersion, two separated layers were formed immediately; the bottom layer was dark brown and the upper layer was initially dark purple due to the dissolution of unreacted C60 particles into the toluene layer (pristine C60 is soluble in toluene and gives a purple colored solution) (Fig. 3b). Washing with toluene was repeated until the dark purple top toluene layer turned colorless, which indicated the complete removal of unreacted C60 from the brown layer (Fig. 3c). The dark brown dispersion containing C60(OH)n·mH2O was then separated from the toluene layer and dried in a freeze dryer for 30 h (−40 °C, 0.12 mbar).Results and discussion
Identification of –OH groups
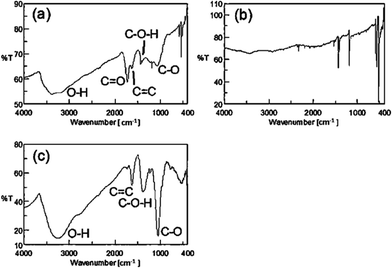
To identify the functional group(s), the dried C60(OH)n·mH2O was analyzed via FTIR (Fig. 4a). The clear broad peak at 3395 cm−1 within the range of 3600–3100 cm−1 indicates the characteristic O–H stretching, which does not appear in the IR spectrum of pristine C60 (Fig. 4b) but has been reported to be present also in the IR spectrum of pristine C60(OH)12 (Fig. 4c),24 thus this initially confirms the attachment of –OH groups onto the C60 cage after functionalization.This peak was not intense when C60 was ultrasonicated in type II pure water (H2O) under the same experimental conditions but in the absence of any H2O2 (Fig. 5b), which indicates that the use of H2O2 in aqueous media is a more efficient approach to introduce –OH groups onto the C60 cage rather than only using H2O for the synthesis of fullerenol in this ultrasound-assisted technique.
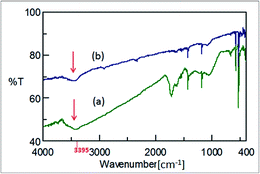 | ||
| Fig. 5 IR spectra of C60(OH)n·mH2O obtained via ultrasonication (a) in the presence of dil. H2O2 (30%) and (b) in type II pure H2O without any H2O2. | ||
The peaks at 1625, 1427 and 1057 cm−1 (Fig. 4a and 5a) could possibly be attributed to the bond stretching of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–O–H and C–O, respectively.41,42 Indeglia et al.42 emphasized that the presence of C–O bond stretching is inevitable in all the fullerenols which perhaps indicates the formation of hemiketal groups prior to the hydroxylation of the C60 cage. In contrast, in the sample sonicated only with water, these significant peaks, which display the characteristic bond stretching of fullerenol, were absent in the IR spectrum (Fig. 5b), and thus also support that to synthesize fullerenol moieties via this ultrasound strategy the presence of H2O2 plays an important role in intensifying the hydroxylation. The additional peaks at 575 and 525 cm−1 in the finger print region (<1000 cm−1) in the IR spectra of C60(OH)n·mH2O (Fig. 4a and 5a) are the characteristic peaks of pure C60, therefore these peaks are not attributed to any potential functional group(s). However, there could have been a trace amount of unreacted C60 remaining in C60(OH)n·mH2O during separation and purification, which is possibly responsible for these peaks in the IR spectra of C60(OH)n·mH2O. We cannot rule out this possibility especially when we scale-up this method for the mass production of C60(OH)n·mH2O.
C, C–O–H and C–O, respectively.41,42 Indeglia et al.42 emphasized that the presence of C–O bond stretching is inevitable in all the fullerenols which perhaps indicates the formation of hemiketal groups prior to the hydroxylation of the C60 cage. In contrast, in the sample sonicated only with water, these significant peaks, which display the characteristic bond stretching of fullerenol, were absent in the IR spectrum (Fig. 5b), and thus also support that to synthesize fullerenol moieties via this ultrasound strategy the presence of H2O2 plays an important role in intensifying the hydroxylation. The additional peaks at 575 and 525 cm−1 in the finger print region (<1000 cm−1) in the IR spectra of C60(OH)n·mH2O (Fig. 4a and 5a) are the characteristic peaks of pure C60, therefore these peaks are not attributed to any potential functional group(s). However, there could have been a trace amount of unreacted C60 remaining in C60(OH)n·mH2O during separation and purification, which is possibly responsible for these peaks in the IR spectra of C60(OH)n·mH2O. We cannot rule out this possibility especially when we scale-up this method for the mass production of C60(OH)n·mH2O.
Estimation of the number of –OH groups and the structure of fullerenol
IR spectra alone are not enough to determine and confirm the –OH groups, their numbers and the structure of fullerenol. Therefore, elemental analysis was conducted to determine the composition and average structure of C60(OH)n·mH2O. The number of bound water molecules (m) within the C60(OH)n structure was calculated via TGA. After the ultrasound-assisted functionalization of pure C60, the average composition of C and O (C: 82.6 wt% and O: 17.2 wt%) in C60(OH)n was first obtained from SEM-EDS analysis. In pure C60, no trace of oxygen (C: 100%) was detected before the reaction which predicts the formation and presence of some oxygen containing functional group(s) in the functionalized C60. However, EDS cannot analyze the presence and composition of hydrogen present in a sample. The composition and structure of C60(OH)n was finally deduced from elemental analysis (Table 1).| % C | % H | H2Oa (wt%) | |
|---|---|---|---|
| a Measured by TGA, difference between exp. and calc. should be within ±1%. | |||
| Experimentally obtained | 80.52 | 0.96 | 5.58 |
| Estimated average structure calculated for- | |||
| C60(OH)2·8H2O | 80.18 | 2.02 | 16.0 |
| C60(OH)4·6H2O | 80.36 | 1.80 | 12.1 |
| C60(OH)6·4H2O | 80.54 | 1.58 | 8.1 |
| C60(OH)8·2H2O | 80.72 | 1.35 | 4.0 |
| C60(OH)10·1H2O | 79.30 | 1.33 | 2.0 |
| C60(OH)10·0H2O | 80.91 | 1.13 | 0 |
In the elemental analysis of fullerenols if the product is a pure single isomer and can be purified totally, the difference should be within 0.4%, but generally the product fullerenol is a mixture of many isomers and it is very difficult to separate the isomers from each other. Therefore, from our many synthetic experiences, even with reaction conditions completely the same as much possible, the difference in elemental analysis is somewhat large even though the chemical and physical properties of the fullerenol are essentially the same. Due to this fact, we always judge the average molecular formula of fullerenol within 1% error of elemental analysis [Tables 1 and 2]. From elemental analysis it became evident that the number of –OH groups attached to each C60 cage is n = 8. The composition (C: 80.52%, H: 0.96%) obtained from elemental analysis is similar to that calculated theoretically for the structure of C60(OH)8, thus the structure of C60(OH)n synthesized by the present ultrasound strategy was calculated as C60(OH)8 (Table 1). Similarly, elemental analysis was conducted to estimate the number of –OH groups that could possibly be attached when pristine C60 was sonicated in only type II pure H2O without the addition of H2O2. By this method the number of –OH groups that could be attached to the C60 cage is only 2 (n = 2) (Table 2), which again supports the role of H2O2 in intensifying the hydroxylation.
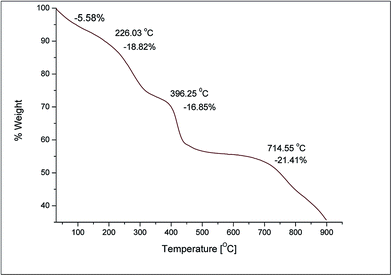
The formation and attachment of –OH groups were further confirmed by TGA (Fig. 6). The weight loss (wt%) of C60(OH)8·2H2O was observed from room temperature to 900 °C at a rate of 10 °C min−1 under N2 flow at 20 mL min−1.
An initial weight loss (5.58 wt%) was observed from room temperature to 100 °C which indicates the loss of bound water molecules. Since the number of –OH groups attached to the C60 cage is less than 10, the weight loss (5.58 wt%) for secondary bound water in C60(OH)8 could be observed from room temperature to 100 °C.16 From this percentage of weight loss, the number of bound water molecules associated with each C60(OH)8 molecule was calculated to be 2 (m = 2) which is shown in Table 1 as well the estimated complete structure of the synthesized fullerenol.
After the decomposition of bound water the degradation continued to around 226 °C, which could be due to some of the intermediates such as epoxy or hemiketal oxygen and/or carbonyl oxygen generated during the ultrasound-assisted reaction.41–43 These intermediates may be present in C60(OH)8·2H2O in trace amounts but possibly will not hinder the characteristic physical and chemical properties of C60(OH)8·2H2O. However a detailed understanding of these intermediates present in fullerenol is not yet fully accomplished which encourages further studies. Dehydration of the –OH groups (16.85 wt%) attached to the C60 molecular cage mostly occurred in the second step of TGA at around 396 °C, the value of which is very close to that theoretically calculated (15.2%) for the dehydration of 8 –OH groups. The degradation observed at around 714 °C is due to the sublimation of C60 molecules. Together with the elemental analysis, the TGA result manifests that C60 could actually be successfully functionalized to fullerenol via ultrasound-assisted hydroxylation in the presence of aq. H2O2 and the average structure of the fullerenol derived from these empirical studies is C60(OH)8·2H2O.
In applying this technique for the production of fullerenol it is also necessary to explore the yield of the prepared C60(OH)8·2H2O. In this work, the yield was verified by repeating the experiment three times. The yield of C60(OH)8·2H2O was investigated based on both the amount of C60(OH)8·2H2O obtained after drying and the amount of unreacted C60 separated after reaction. The yield was found to vary between 2.18 and 4.04%. There is always a possibility of material loss during the process of drying, especially directly from the liquid state to solid state, which should be considered in any future work when reproducing the proposed method to prepare C60(OH)8·2H2O. The yield achieved is not high on the laboratory scale; however by optimizing the reaction conditions, selecting different solvents for separation and purification, improving the drying method to avoid any loss of the material, the yield of C60(OH)8·2H2O could be increased using the proposed ultrasound method.
Particle size measurements
Usually the particles of fullerenols having a fewer number of –OH groups have been reported to be aggregative and the particle size may vary in the range of 50–300 nm.41 DLS analysis and AFM scanning were carried out to investigate the size and morphology of the C60(OH)8·2H2O particles, respectively. For the particle size measurements, C60(OH)8·2H2O was dissolved in DMSO (0.33 mg mL−1). As a polar aprotic solvent, DMSO can dissolve both polar and nonpolar compounds. C60(OH)8·2H2O in DMSO initially formed a suspension which was then centrifuged (TOMY, LC-200) for 5 min at 7500 rpm to obtain a clear solution of C60(OH)8·2H2O in DMSO. Both the suspension and the solution (collected as supernatant after centrifugation) were analyzed for particle size measurements via the DLS method. The average particle size of C60(OH)8·2H2O in the suspension was found to be larger (312 nm) (Fig. 7b) than that in the solution (120 nm) (Fig. 7a).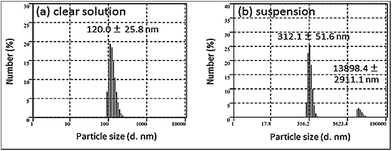 | ||
| Fig. 7 Particle size measurements: (a) C60(OH)8·2H2O/DMSO solution (collected as supernatant after centrifugation) and (b) C60(OH)8·2H2O/DMSO suspension (0.33 mg mL−1). | ||
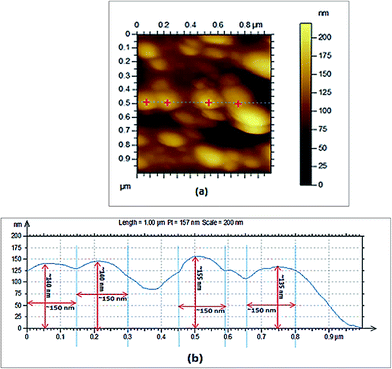
Also, larger sized particles of about 13.9 μm could be seen in the suspension (Fig. 7b) which could either be due to the highly aggregative nature of C60(OH)8·2H2O along with some intermediates possibly present as described in the earlier section of this study or due to the presence of trace amounts of unreacted pristine C60 which remained in the sample after the separation process. Hence, we infer that C60(OH)8·2H2O when dispersed in DMSO contains particles of a wider size range and thus could be considered as a polydispersed suspension, which after centrifugation provides a clear solution of uniform sized particles of C60(OH)8·2H2O of about 120 nm. The particle size was further verified using the topography vs. distance chart (Fig. 8b) obtained from the AFM analysis of C60(OH)8·2H2O.
The cross section of the AFM image shows that the width of the particles is around 150 nm and their height varies from 135 to 155 nm (Fig. 8b), which indicates that the synthesized C60(OH)8·2H2O particles could be considered spherical in shape with a diameter in the range of 135–155 nm. The average width and height of the particles obtained from the AFM analysis are congruent with the particle sizes (120 ± 25.8 nm and 312 ± 51.6 nm) obtained by DLS analysis for the saturated solution of C60(OH)8·2H2O in DMSO (Fig. 7a) and suspension of C60(OH)8·2H2O in DMSO (Fig. 7b), respectively. C60(OH)8·2H2O is considered as the first member of the polyhydroxylated fullerene group to show solubility in water at a low concentration and at the same time forms aggregates when dispersed in water or DMSO. Therefore, some bigger particles are observed in the suspension of C60(OH)8·2H2O/DMSO. This aggregation is observed in both the AFM and SEM images. The image (Fig. 8a) and height profile (Fig. 8b) obtained from the AFM analysis reveal that the individual particles of C60(OH)8·2H2O are actually not finely separated from each other, rather they are assembled in the form of nearly spherical shaped aggregates with a range of sizes.
The SEM image (Fig. 9) provides further insight into the aggregation of the synthesized C60(OH)8·2H2O particles when they are in the powder form. In the powder form, the C60(OH)8·2H2O particles are much more aggregative and even display sizes bigger than 300 nm, but when they are dispersed in solvent(s), aggregation is less effective. Also, this aggregation nature decreases with an increase in the number of –OH groups attached to each C60 molecule.16 Even though C60(OH)8·2H2O exhibits amphiphilic behavior, it is moderately polyhydroxylated; as a result the interaction potential between the particles becomes more effective than the intermolecular hydrogen bond potential which ultimately causes Brownian aggregation, and results in variable sizes of self-assembled C60(OH)8·2H2O particles in the suspension.26,44
Color and solubility
C60(OH)8·2H2O obtained after separation, purification and drying was not completely black, rather it was nearly brown (Fig. 10a), and when dispersed in DMSO it gave a dark brown color suspension (Fig. 10b). Fullerenol having more than 10 –OH groups is observed to be dark brown in color, which gradually shifts from dark brown to yellow with an increase in the number of –OH groups (Fig. 10c), as previously reported.24 | ||
| Fig. 10 (a) C60(OH)8·2H2O after drying, (b) C60(OH)8·2H2O in DMSO (0.33 mg mL−1) and *(c) colors of different fullerenols previously reported [*reprinted from Kokubo et al. (ref. 24)]. | ||
The solubility of C60(OH)8·2H2O was examined both in water and in organic solvents, i.e. DMSO, toluene and benzene (Table 3).
It is noteworthy to mention that C60(OH)8·2H2O moderately dissolves in water at a lower concentration owing to its amphiphilic nature. It was found to be soluble in DMSO but did not show any solubility in toluene and benzene. On the other hand, pure C60 dissolves both in toluene and benzene but does not show any solubility in water and DMSO.
Reaction pathways
Acoustic cavitation generated from ultrasonication results in chemical reactions inside liquid media.45 When acoustic cavitation is induced throughout liquid media (30% H2O2 in this case) it produces cavitation bubbles and upon continuous ultrasonication these bubbles form and collapse randomly. The collapse of these bubbles produces transient local hot spots with intense local heat and pressure inside the liquid media which assist in high-energy chemical reactions among the molecules either trapped inside the cavitation bubbles or present in the liquid media.46,47 In this investigation, due to ultrasound induced acoustic cavitation, radicals such as ˙OH, ˙OOH and ˙H originate from H2O and H2O2 molecules.31,48,49 Especially, the formation of ˙OH radicals due to the thermal decomposition of aqueous media has been found to be evident by electron spin resonance (ESR) and spin trapping29,30,50–52 studies. H2O2 is thermodynamically unstable and dissociates into H2O and O2 under thermal decomposition. During ultrasonic cavitation, H2O and H2O2 molecules are trapped inside microbubbles, and when these bubbles collapse with the enormous amount of heat (several thousand degrees K) and pressure (hundreds of atmospheres)53,54 the molecules decompose to ˙OH and ˙OOH55,56 radicals. The reaction may progress in two pathways simultaneously (Fig. 11). ˙OH radicals as reactive oxygen species (ROS) attach onto the C60 cage to give fullerenol (Path I), and/or –OH and ˙OOH radicals attack the electron deficient C60 double bonds in a nucleophilic reaction and this leads to the formation of fullerene epoxide [C60On] as an intermediate in the first stage (Path II) which is similar to the mechanism of the Bingel reaction.37,57 Further, the repeated attack of ˙OH (or ˙OOH) on C60O via an SN2 reaction results in polyhydroxylated fullerene or fullerenol.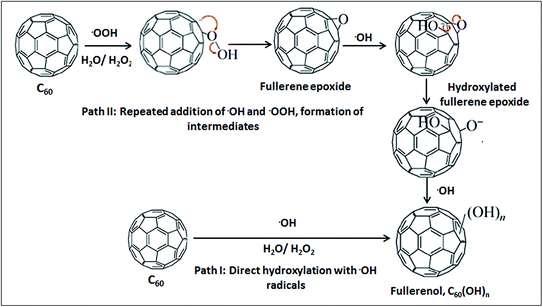 | ||
| Fig. 11 Possible reaction paths in the ultrasound-assisted synthesis of fullerenol in the presence of dil. H2O2 (30%). | ||
Repeated epoxidation may take place which produces successive epoxide groups e.g., C60O2 and C60O3. These epoxide groups could be possible candidates to generate other intermediates e.g. hydroxylated fullerene epoxide [C60(OH)xOy]16,58 during sonolysis. Additionally, the subsequent ring opening of C60(OH)xOy with ˙OH can result in the formation of fullerenol.59 The formation of these intermediates during the sonolysis of H2O2 or H2O in the presence of C60 is inevitable, and their presence in the final fullerenol (although in a trace amount) cannot go unnoted. However, because they are only present in trace amounts in the fullerenol they are not expected to cause any significant impact.
Future prospects
To explore the potential applications of fullerenols, it is indeed essential to produce high quality fullerenol which means not only higher water solubility but also free of any impurities. The presence of impurities, which generally come from the preparation process, makes fullerenol undesirable for any specific biological and metallurgical applications. More importantly, the commercial value of fullerenol depends on the presence and percentage of impurities. Moreover, a faster approach is desirable to facilitate the commercial production of fullerenol. The proposed technique for the preparation of hydroxylated C60 by ultrasonication in the presence of H2O2 is free from the use of additional hydroxylating reagents, i.e. NaOH, H2SO4, and PTC (causes impurities in fullerenol), which is a cleaner approach to produce fullerenol in an easier and a faster way. Previously, C60(OH)12 was used as a starting material to synthesize highly soluble fullerenols [C60(OH)36, C60(OH)40] by vigorously stirring with dil. H2O2 for several days.24 Similarly C60(OH)8·2H2O synthesized by this method could be used as a starting material to further produce fullerenols containing a greater number of hydroxyl groups, e.g. C60(OH)24, C60(OH)36 and C60(OH)40. Moreover, compounds that express specific biochemical functions, which are required for diagnostics as well as drug therapy studies, can be derivatized from C60(OH)8·2H2O by conjugating it with other potential functional groups or biomolecules. The conjugation of folic acid with C60(OH)8·2H2O produced via this method is currently under investigation as an extended study of this work with the view to develop a highly sensitive biosensor for early stage cancer detection. Further potential applications for C60(OH)8·2H2O synthesized by the proposed method of ultrasonication include as an antioxidant since it offers higher antioxidant activities compared to the fullerenols that have more hydroxyl groups, i.e., C60(OH)24, C60(OH)26 and C60(OH)36;39 an electrochemically active nanomediator since based on density functional theory (DFT) it has also been found that fullerenols having less hydroxyl groups are thermodynamically more stable than those containing more hydroxyl groups due to the symmetric orientation of the –OH groups around the C60 molecular cage;60,61 a light harvesting material in solar cell applications62 and the preparation of rich carbon structures of different shapes, sizes and isomeric orientations recently termed as Janus particles for various other applications.44It is anticipated that there must be a substantial difference between the levels of energy generated during continuous ultrasonication and pulse mode ultrasonication which should be also addressed in future investigations. In addition, the duration of ultrasonication may cause a remarkable difference in the structure of fullerenol. Besides the variables of ultrasonication (time and power input), it is equally important to optimize the other parameters in future studies, i.e. temperature, size and geometry of the treatment vessel, nature and concentration of any dissolved gas, concentration of H2O2, solute to reagents ratio (C60: 30% H2O2, mg mL−1) and height of the mixture in the treatment vessel, where all of them alone or together can play vital roles in producing fullerenols possessing different combinations of –OH groups and bound H2O molecules in addition to increasing the yield of C60(OH)8·2H2O while applying the proposed ultrasound technique for the synthesis of fullerenols.
Conclusion
Herein, we have proposed a facile and fast approach to prepare fullerenol via the ultrasound-assisted hydroxylation of C60 only in dil. H2O2 (30%) which acts as a hydroxylating reagent and we have quantified the possible structure of fullerenol that could be derived by this technique. It appears that during the ultrasonication of pure C60 in aqueous media, even only in the presence of H2O2, not only leads to the hydration of C60 in the reaction media but also results in the generation of potential fullerenol candidate(s), which upon quantitative analysis has been identified as C60(OH)8·2H2O. Since no alkali, acids or PTC have been used for the synthesis, the proposed method offers a greener and cleaner approach towards the hydroxylation of the C60 cage compared to existing methods. Quantitative studies reveal that this hydroxylation technique assisted by ultrasonication in the presence of H2O2 can lead to the formation of fullerenol possessing an average structure of C60(OH)8·2H2O and with an average yield of 2%. C60(OH)8·2H2O was found to be amphiphilic and thus moderately soluble in water at a low concentration and it could further be exploited as a starting material to prepare highly water soluble fullerenol moieties. The presence of aq. H2O2 intensifies the hydroxylation and enhances the number of hydroxyl groups (n = 8) on the C60 cage in comparison to that obtained (n = 2) while applying the same ultrasonication but only in the presence of pure water. This indicates that H2O2 plays a vital role in the hydroxylation which could have potential to obtain fullerenol moieties, where the yield could be increased by varying the concentration of H2O2. The proposed technique encompasses a one-step reaction strategy, requires a short time for the reaction, offers a green and clean approach with a low energy requirement, avoids the use of any toxic or corrosive reagents for the synthesis, and reduces the number of solvents required for the separation and purification of C60(OH)8·2H2O. Hence, this potential approach should further be investigated to for the scale-up mass production of fullerenol moieties for a wider range of technological applications.Author contributions
The manuscript was written through contributions of all authors.Funding sources
Authors would like to thank Fundamental Research Grant Scheme (FRGS) for the funding support (FRGS/1/2013/SG05/UNIM/01/1).Conflict of interest
The authors declare no competing financial interest.Abbreviations
| AFM | Atomic force microscopy |
| C60 | Fullerene |
| C60(OH)n·mH2O | Fullerenol |
| DMSO | Dimethyl sulfoxide |
| DLS | Dynamic light scattering |
| FE-SEM | Field emission scanning electron microscopy |
| FTIR | Fourier transform infrared spectroscopy |
| –OH | Hydroxyl group |
| PTC | Phase transfer catalyst |
| SEM-EDS | Scanning electron microscopy with energy dispersive X-ray spectroscopy |
| TGA | Thermogravimetric analysis |
Acknowledgements
KK thanks for the funding support by the Program for Creating Future Wisdom, Osaka University, selected in 2014. Authors would like to express their gratitude to Dr Huang Nay Ming, University of Malaya, Malaysia for carrying out AFM analysis.References
- H. W. Kroto, J. R. Health, S. C. O'Brien, R. F. Curl and R. E. Smalley, C60: Buckminsterfullerene, Nature, 1985, 318, 162–163 CrossRef CAS.
- R. E. Smalley, Discovering the Fullerenes, Rev. Mod. Phys., 1997, 69(3), 723–730 CrossRef CAS.
- S. Bosi, T. D. Ros, G. Spalluto and M. Prato, Fullerene Derivatives: an Attractive Tool for Biological Applications, Eur. J. Med. Chem., 2003, 38(11–12), 913–923 CrossRef CAS PubMed.
- M. Prato, [60]Fullerene Chemistry for Materials Science Applications, J. Mater. Chem., 1997, 7(7), 1097–1109 RSC.
- T. D. Ros and M. Prato, Medicinal Chemistry with Fullerenes and Fullerene Derivatives, Chem. Commun., 1999, 663–669 RSC.
- N. Tagmatarchis and H. Shinohara, Fullerenes in Medicinal Chemistry and their Biological Applications, Mini-Rev. Med. Chem., 2001, 1(4), 339–348 CAS.
- A. Hirsch, The Chemistry of the Fullerenes: An Overview, Angew. Chem., Int. Ed. Engl., 1993, 32(8), 1138–1141 CrossRef.
- N. Martın, New Challenges in Fullerene Chemistry, Chem. Commun., 2006, 20, 2093–2104 RSC.
- I. Lamparth and A. Hirsch, Water-Soluble Malonic Acid Derivatives of C60 with a Defined Three-Dimensional Structure, J. Chem. Soc., Chem. Commun., 1994, 14, 1727–1728 RSC.
- I. C. Wang, L. A. Tai, D. D. Lee, P. P. Kanakamma, C. K.-F. Shen, T.-Y. Luh, C. H. Cheng and K. C. Hwang, C60 and Water-Soluble Fullerene Derivatives as Antioxidants Against Radical-Initiated Lipid Peroxidation, J. Med. Chem., 1999, 42(22), 4614–4620 CrossRef CAS PubMed.
- C. F. Richardson, D. I. Schuster and S. R. Wilson, Synthesis and Characterization of Water-Soluble Amino Fullerene Derivatives, Org. Lett., 2000, 2(8), 1011–1014 CrossRef CAS PubMed.
- V. K. Periya, I. Koike, Y. Kitamura, S.-i. Iwamatsu and S. Murata, Hydrophilic [60]Fullerene Carboxylic Acid Derivatives Retaining the Original 60π Electronic System, Tetrahedron Lett., 2004, 45, 8311–8313 CrossRef CAS.
- R. Partha and J. L. Conyers, Biomedical Applications of Functionalized Fullerene-Based Nanomaterials, Int. J. Nanomed., 2009, 4, 261–275 CrossRef CAS.
- K. N. Semenov, N. A. Charykov and V. N. Keskinov, Fullerenol Synthesis and Identification. Properties of the Fullerenol Water Solutions, J. Chem. Eng. Data, 2011, 56(2), 230–239 CrossRef CAS.
- A. Đorđević and G. Bogdanović, Fullerenol – a New Nanopharmaceutic?, Arch. Oncol., 2008, 16(3–4), 42–45 Search PubMed.
- K. Kokubo, S. Shirakawa, N. Kobayashi, H. Aoshima and T. Oshima, Facile and Scalable Synthesis of a Highly Hydroxylated Water-Soluble Fullerenol as a Single Nanoparticle, Nano Res., 2011, 4(2), 204–215 CrossRef CAS.
- P. Zhang, H. Pan, D. Liu, Z.-X. Guo, F. Zhang and D. Zhu, Effective Mechanochemical Synthesis of [60]Fullerols, Synth. Commun., 2003, 33(14), 2469–2474 CrossRef CAS.
- S. Wang, P. He, J.-M. Zhang, H. Jiang and S.-Z. Zhu, Novel and Efficient Synthesis of Water-Soluble [60]Fullerenol by Solvent-Free Reaction, Synth. Commun., 2005, 35(13), 1803–1808 CrossRef CAS.
- J. Li, A. Takeuchi, M. Ozawa, X. Li, K. Saigo and K. Kitazawa, C60 Fullerol Formation Catalysed by Quaternary Ammonium Hydroxides, J. Chem. Soc., Chem. Commun., 1993, 23, 1784–1785 RSC.
- L. Y. Chiang, L.-Y. Wang, J. W. Swirczewski, S. Soled and S. Cameron, Efficient Synthesis of Polyhydroxylated Fullerene Derivatives via Hydrolysis of Polycyclosulfated Precursors, J. Org. Chem., 1994, 59(14), 3960–3968 CrossRef CAS.
- L. Y. Chiang, J. B. Bhonsle, L. Wang, S. F. Shu, T. M. Chang and J. R. Hwu, Efficient One-Flask Synthesis of Water-Soluble [60]Fullerenols, Tetrahedron, 1996, 52(14), 4963–4972 CrossRef CAS.
- J.-M. Zhang, W. Yang, P. He and S.-Z. Zhu, Efficient and Convenient Preparation of Water-Soluble Fullerenol, Chin. J. Chem., 2004, 22(9), 1008–1011 CrossRef CAS.
- G. C. Alves, L. O. Ladeira, A. Righi, K. Krambrock, H. D. Calado, R. P. F. Gil and M. V. B. Pinheiro, Synthesis of C60(OH)18–20 in Aqueous Alkaline Solution Under O2-Atmosphere, J. Braz. Chem. Soc., 2006, 17(6), 1186–1190 CrossRef CAS.
- K. Kokubo, K. Matsubayashi, H. Tategaki, H. Takada and T. Oshima, Facile Synthesis of Highly Water-Soluble Fullerenes More Than Half-Covered by Hydroxyl Groups, ACS Nano, 2008, 2(2), 327–333 CrossRef CAS PubMed.
- Y. Lu, K. Feng, P. Qiyun and Y. Xinlin, An Improved Method for Fullerol Preparation Based on Dialysis, Chin. J. Chem. Eng., 2010, 18(5), 876–879 CrossRef.
- G. Zhang, Y. Liu, D. Liang, L. Gan and Y. Li, Facile Synthesis of Isomerically Pure Fullerenols and Formation of Spherical Aggregates from C60(OH)8, Angew. Chem., Int. Ed., 2010, 49(31), 5293–5295 CrossRef CAS PubMed.
- F. F. Wang, N. Li, D. Tian, G. F. Xia and N. Xiao, Efficient Synthesis of Fullerenol in Anion Form for the Preparation of Electrodeposited Films, ACS Nano, 2010, 4(10), 5565–5572 CrossRef CAS PubMed.
- K. Matsubayashi, K. Kokubo, H. Tategaki, S. Kawahama and T. Oshima, One-step Synthesis of Water-soluble Fullerenols Bearing Nitrogen-containing Substituents, Fullerenes, Nanotubes, Carbon Nanostruct., 2009, 17(4), 440–456 CrossRef CAS.
- K. Makino, M. M. Mossoba and P. Riesz, Chemical Effects of Ultrasound on Aqueous Solutions. Formation of Hydroxyl Radicals and Hydrogen Atoms, J. Phys. Chem., 1983, 87(8), 1369–1377 CrossRef CAS.
- X. Fang, G. Mark and C. Sonntag, OH Radical Formation by Ultrasound in Aqueous Solutions Part I: the Chemistry Underlying the Terephthalate Dosimeter, Ultrason. Sonochem., 1996, 3(1), 57–63 CrossRef CAS.
- Y. Hu, Z. Zhang and C. Yang, Measurement of Hydroxyl Radical Production in Ultrasonic Aqueous Solutions by a Novel Chemiluminescence Method, Ultrason. Sonochem., 2008, 15(5), 665–672 CrossRef CAS PubMed.
- W.-B. Ko, J.-Y. Heo, J.-H. Nam and K.-B. Lee, Synthesis of a Water-Soluble Fullerene [C60] Under Ultrasonication, Ultrasonics, 2004, 41(9), 727–730 CrossRef CAS PubMed.
- R. Rivelino, A. M. Maniero, F. V. Prudente and L. S. Costa, Theoretical Calculations of the Structure and UV-Vis Absorption Spectra of Hydrated C60 Fullerene, Carbon, 2006, 44(14), 2925–2930 CrossRef CAS.
- J. Labille, A. Masion, F. Ziarelli, J. Rose, J. Brant, F. Villiéras, M. Palletier, D. Borschneck, M. R. Wiesner and J.-Y. Bottero, Hydration and Dispersion of C60 in Aqueous Systems: The Nature of Water–Fullerene Interactions, Langmuir, 2009, 25(19), 11232–11235 CrossRef CAS PubMed.
- W.-B. Ko and K.-N. Baek, The Oxidation of Fullerenes (C60, C70) with Various Oxidants under Ultrasonication, Phys. Solid State, 2002, 44(3), 424–426 CrossRef CAS.
- T. H. Goswami, B. Nandan, S. Alam and G. N. Mathur, A Selective Reaction of Polyhydroxy Fullerene with Cycloaliphatic Epoxy Resin in Designing Ether Connected Epoxy Star Utilizing Fullerene as a Molecular Core, Polymer, 2003, 44(11), 3209–3214 CrossRef CAS.
- W.-W. Chang, Z.-J. Li, W.-W. Yang and X. Gao, Reactions of Anionic Oxygen Nucleophiles with C60 Revisited, Org. Lett., 2012, 14(9), 2386–2389 CrossRef CAS PubMed.
- S.-E. Zhu, F. Li and G.-W. Wang, Mechanochemistry of Fullerenes and Related Materials, Chem. Soc. Rev., 2013, 42, 7535–7570 RSC.
- Z. Wang, S. Wang, Z. Lu and X. Gao, Syntheses, Structures and Antioxidant Activities of Fullerenols: Knowledge Learned at the Atomistic Level, J. Cluster Sci., 2015, 26(2), 375–388 CrossRef CAS.
- M. S. Meier and J. Kiegiel, Preparation and Characterization of the Fullerene Diols 1,2-C60(OH)2, 1,2-C70(OH)2, and 5,6-C70(OH)2, Org. Lett., 2001, 3(11), 1717–1719 CrossRef CAS PubMed.
- L. O. Husebo, B. Sitharaman, K. Furukawa, T. Kato and L. J. Wilson, Fullerenols Revisited as Stable Radical Anions, J. Am. Chem. Soc., 2004, 126(38), 12055–12064 CrossRef CAS PubMed.
- P. A. Indeglia, A. Georgieva, V. B. Krishna and J.-C. J. Bonzongo, Physicochemical Characterization of Fullerenol and Fullerenol Synthesis By-Products Prepared in Alkaline Media, J. Nanopart. Res., 2014, 16(2599), 1–15 CAS.
- L. Y. Chiang, R. B. Upasani, J. W. Swirczewski and S. Soled, Evidence of Hemiketals Incorporated in the Structure of Fullerols Derived from Aqueous Acid Chemistry, J. Am. Chem. Soc., 1993, 115(13), 5453–5457 CrossRef CAS.
- Y. Liu, G. Zhang, L. Niu, L. Gan and D. Liang, Assembly of Janus Fullerenol: a Novel Approach to Prepare Rich Carbon Structures, J. Mater. Chem., 2011, 21, 14864–14868 RSC.
- J. Raso, P. Mañas, R. Pagán and F. J. Sala, Influence of Different Factors on the Output Power Transferred into Medium by Ultrasound, Ultrason. Sonochem., 1999, 5(4), 157–162 CrossRef CAS PubMed.
- K. S. Suslick and G. J. Price, Applications of Ultrasound to Materials Chemistry, Annu. Rev. Mater. Sci., 1999, 29, 295–326 CrossRef CAS.
- J. Rooze, E. V. Rebrov, J. C. Schouten and J. T. F. Keurentjes, Dissolved Gas and Ultrasonic Cavitation – a Review, Ultrason. Sonochem., 2013, 20(1), 1–11 CrossRef CAS PubMed.
- L. Villeneuve, L. Alberti, J.-P. Steghens, J.-M. Lancelin and J.-L. Mestas, Assay of Hydroxyl Radicals Generated by Focused Ultrasound, Ultrason. Sonochem., 2009, 16(3), 339–344 CrossRef CAS PubMed.
- T. J. Mason, Some Neglected or Rejected Paths in Sonochemistry – a Very Personal View, Ultrason. Sonochem., 2015, 25, 89–93 CrossRef CAS PubMed.
- G. R. Buettner, Spin Trapping: ESR Parameters of Spin Adducts 1474 1528V, Free Radical Biol. Med., 1987, 3(4), 259–303 CrossRef CAS PubMed.
- C. L. Christman, A. J. Carmichael, M. M. Mossoba and P. Riesz, Evidence for Free Radicals Produced in Aqueous Solutions by Diagnostic Ultrasound, Ultrasonics, 1987, 25(1), 31–34 CrossRef CAS PubMed.
- P. Riesz and T. Kondo, Free Radical Formation Induced by Ultrasound and its Biological Implications, Free Radical Biol. Med., 1992, 13(3), 247–270 CrossRef CAS PubMed.
- C. Leonelli and T. J. Mason, Microwave and Ultrasonic Processing: Now a Realistic Option for Industry, Chem. Eng. Process., 2010, 49(9), 885–900 CrossRef CAS.
- M. Kohno, T. Mokudai, T. Ozawa and Y. Niwano, Free Radical Formation from Sonolysis of Water in the Presence of Different Gases, J. Clin. Biochem. Nutr., 2011, 49(2), 96–101 CrossRef CAS PubMed.
- C. Gong and D. P. Hart, Ultrasound Induced Cavitation and Sonochemical Yields, J. Acoust. Soc. Am., 1998, 104(5), 2675–2682 CrossRef.
- G. Mark, A. Tauber, R. Laupert, H.-P. Schuchmann, D. Schulz, A. Mues and C. Sonntag, OH-Radical Formation by Ultrasound in Aqueous Solution – Part II: Terephthalate and Fricke Dosimetry and the Influence of Various Conditions on the Sonolytic Yield, Ultrason. Sonochem., 1998, 5(2), 41–52 CrossRef CAS PubMed.
- A. Hirsch, Principles of Fullerene Reactivity. Fullerenes and Related Structures, Springer, Berlin, Heidelberg, 1999, pp. 1–65 Search PubMed.
- T. H. Goswami, R. Singh, S. Alam and G. N. Mathur, Thermal analysis: a unique method to estimate the number of substituents in fullerene derivatives, Thermochim. Acta, 2004, 419(1), 97–104 CrossRef CAS.
- L. Tianbao, L. Xinhai, H. Kexiong, J. Hanying and L. Jing, Synthesis and Characterization of Hydroxylated Fullerene Epoxide—an Intermediate for Forming Fullerol, J. Cent. South Univ. Technol., 1999, 6(1), 35–36 CrossRef.
- X. J. Li, X. H. Yang, L. M. Song, H. J. Ren and T. Z. Tao, A DFT study on structure, stability, and optical property of fullerenols, Struct. Chem., 2013, 24(4), 1185–1192 CrossRef CAS.
- J. Zhuo, T. Wang, G. Zhang, L. Liu, L. Gan and M. Li, Salts of C60(OH)8 electrodeposited onto a glassy carbon electrode: surprising catalytic performance in the hydrogen evolution reaction, Angew. Chem., Int. Ed., 2013, 52(41), 10867–10870 CrossRef CAS PubMed.
- S. P. Singh, Light Harvesting Nanomaterials, Bentham Science Publishers, 2015 Search PubMed.
| This journal is © The Royal Society of Chemistry 2017 |


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×).
000×).