Open Access Article
Open Access ArticleAmino acid based gallium-68 chelators capable of radiolabeling at neutral pH†
Thomas W.
Price
ab,
Juan
Gallo
 c,
Vojtěch
Kubíček
c,
Vojtěch
Kubíček
 d,
Zuzana
Böhmová
d,
Timothy J.
Prior
d,
Zuzana
Böhmová
d,
Timothy J.
Prior
 e,
John
Greenman
e,
John
Greenman
 a,
Petr
Hermann
a,
Petr
Hermann
 d and
Graeme J.
Stasiuk
d and
Graeme J.
Stasiuk
 *ab
*ab
aSchool of Life Sciences, Department of Biomedical Sciences, University of Hull, Cottingham Road, Hull, HU6 7RX, UK. E-mail: g.stasiuk@hull.ac.uk
bPositron Emission Tomography Research Centre, University of Hull, Cottingham Road, Hull, HU6 7RX, UK
cAdvanced (magnetic) Theranostic Nanostructures Lab, International Iberian Nanotechnology Laboratory, Av. Mestre José Veiga s/n, 4715-330 Braga, Portugal
dDepartment of Inorganic Chemistry, Faculty of Science, Charles University, Hlavova 2030, 12840, Prague 2, Czech Republic
eChemistry, School of Mathematical and Physical Sciences, University of Hull, Cottingham Road, Hull, HU6 7RX, UK
First published on 17th November 2017
Abstract
Gallium-68 (68Ga) has been the subject of increasing interest for its potential in the production of radiotracers for diagnosis of diseases. In this work we report the complexation of 68Ga by the amino acid based tripodal chelate H3Dpaa, and two bifunctional derivatives, H3Dpaa.dab and H4Dpaa.ga, under a range of conditions with particular emphasis on the rapid complexation of 68Ga at pH 7.4. 100 μM H3Dpaa achieved a radiochemical yield of 95% at pH 7.4 in 5 minutes at 37 °C. The bifunctional derivatives H4Dpaa.ga and H3Dpaa.dab achieved 94% and 84% radiochemical yields, respectively, under the same conditions. The resulting Ga(III) complexes show thermodynamic stabilities of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaDpaa = 18.53, log
KGaDpaa = 18.53, log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaDpaa.dab = 22.08, log
KGaDpaa.dab = 22.08, log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaDpaa.ga = 18.36. Unfortunately, the resulting radiolabelled species do not present sufficient serum stability for in vivo application. Herein we show a flexible synthesis for bifunctional chelators based on amino acids that rapidly complex 68Ga under physiological conditions.
KGaDpaa.ga = 18.36. Unfortunately, the resulting radiolabelled species do not present sufficient serum stability for in vivo application. Herein we show a flexible synthesis for bifunctional chelators based on amino acids that rapidly complex 68Ga under physiological conditions.
Introduction
Positron emission tomography (PET) is a highly sensitive imaging technique that has been widely applied in cancer diagnosis through the use of [18F]-fluorodeoxyglucose (FDG).1 However, FDG is taken up in all areas of enhanced metabolism.1 More specific, targeted, probes will provide improved diagnosis. Whilst incorporation of 18F and 11C into small organic molecules can provide a route to targeted probes, this route often requires multistep syntheses with harsh reaction conditions that are not compatible with many biomolecules.2,3 More rapid assessment of new targeting motifs may be achievable by exploiting the modular system created by conjugation of targeting motifs to bifunctional chelates that complex radiometals such as 68Ga, 64Cu or 89Zr.4,5Of these positron emitting metal isotopes, 68Ga is of particular interest.6–9 The generator source of 68Ga allows for local production at the site of use, opening the possibility of individual hospitals producing their own radiotracers instead of relying upon centralised production facilities. Comparisons can be drawn to the successful 99mTc single photon emission computed tomography (SPECT) isotope.10 The 68 minute half-life of 68Ga is amenable to imaging with peptides and other molecules with relatively short blood circulation times in vivo.11
Traditional macrocyclic chelators, including 1, 4, 7, 10-tetraazacyclododecane-1, 4, 7, 10-tetraacetic acid (DOTA), have been successfully applied to 68Ga complexation.15–17 Conjugated DOTA derivatives have been applied to imaging of neuroendocrine tumours – with [68Ga]-DOTATATE being recently approved for use by the FDA. However, radiolabeling of DOTA with 68Ga requires relatively aggressive conditions, a pH of 4 and heating to over 80 °C for efficient radiolabeling.15,18 These conditions limit the range of targeting motifs that can be used with [68Ga]-DOTA to acid and temperature stable compounds.
A range of chelators have been tested for their 68Ga complexation abilities.4,12–14 Recent trends in chelate design for 68Ga have been to improve the radiolabeling procedure by reducing the temperature required for efficient complexation of 68Ga and raising the pH at which this occurs.4,12,19,20 This is a challenge due to the formation of kinetically inert gallium hydroxide species above pH 4.5.6,10 Radiolabelling at pHs and temperatures close to physiological conditions is necessary to maintain the structure of peptides and aptamers. Furthermore, radiolabelling at neutral pH would reduce the formulation required after synthesis of the radiotracer, simplifying the tracer production procedure.
The smaller macrocycle, 1,4,7-triazacyclononane-1,4,7-triacetic acid (NOTA) has been applied to 68Ga complexation, and radiolabelling proceeds efficiently at room temperature, although acidic conditions are still required.16 Novel, non-macrocyclic chelates THP21 and DATA22 have been shown to rapidly complex 68Ga at higher pH values. While the conjugated DATA probe, DATA-TOC, has only been radiolabelled at pH 4–5,23 THP conjugates, THP-RGD and THP-TATE, were radiolabelled at pH 5–6.5.24,25 Despite this recent progress, rapid radiolabeling with 68Ga at pH 7 has not yet been widely realised, with few bifunctional chelators capable of achieving rapid complexation at neutral pH reported (structures of these bifunctional chelators are shown in Fig. S1†).26–28
Development of alternative chelates that can achieve this may allow for improved design of the imaging probe through different pharmacokinetic profiles/biodistributions.29 While THP-TATE can be radiolabelled at pH 6.5, its lipophilic nature results in a significantly different biodistribution when compared to DOTA-TATE, with longer renal and liver uptake. Balancing improved radiolabeling properties with ideal imaging properties requires the development of new chelates to optimize both properties.29
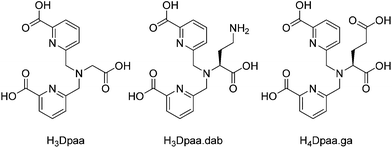
We report here the application of the chelate N,N-bis[(6-carboxypyridin-2-yl)methyl]glycine (H3Dpaa) and two bifunctional derivatives, H3Dpaa.dab and H4Dpaa.ga (Fig. 1) to 68Ga complexation. H3Dpaa is composed of two picolinic acid arms attached to a central glycine unit to produce a tripodal, hexadentate ligand. Picolinic acids have been demonstrated to be highly capable 68Ga coordinating arms.30 The aminebis(picolinic acid) motif has been applied to the complexation of a variety of metals with a number of different amines being used to form chelates with varying properties.31–42 The incorporation of a glycine residue into the chelate backbone provides both a carboxylic acid group that can bind strongly to Ga(III) due to a good hard acid/base match, and also a site which can be readily functionalised through application of other amino acids.31,33 H3Dpaa has previously been applied to complexation of lanthanide(III) ions such as gadolinium(III) (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGdDpaa = 10.6)32,33 and terbium(III) (log
KGdDpaa = 10.6)32,33 and terbium(III) (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KTbDpaa = 10.4).32 H3Dpaa has recently been applied to manganese(II) (log
KTbDpaa = 10.4).32 H3Dpaa has recently been applied to manganese(II) (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KMnDpaa = 13.2),34 lanthanum(III) (log
KMnDpaa = 13.2),34 lanthanum(III) (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KLaDpaa = 13.6)35 and gallium-67 (log
KLaDpaa = 13.6)35 and gallium-67 (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaDpaa = 18.7)35 complexation showing the versatility of this ligand for metal coordination. Herein we show a flexible synthesis for bifunctional chelators that rapidly complex 68Ga under physiological conditions with a radiochemical yield of up to 95%. Unfortunately, the radiolabelled species [68Ga][Ga(Dpaa)], [68Ga][Ga(Dpaa.dab)] and [68Ga][Ga(Dpaa.ga)] show poor stability in serum competition studies and are unsuitable for advancing to in vivo studies.
KGaDpaa = 18.7)35 complexation showing the versatility of this ligand for metal coordination. Herein we show a flexible synthesis for bifunctional chelators that rapidly complex 68Ga under physiological conditions with a radiochemical yield of up to 95%. Unfortunately, the radiolabelled species [68Ga][Ga(Dpaa)], [68Ga][Ga(Dpaa.dab)] and [68Ga][Ga(Dpaa.ga)] show poor stability in serum competition studies and are unsuitable for advancing to in vivo studies.
Results and discussion
Synthesis and characterisation
In this paper we report the application of N,N-bis[(6-carboxypyridin-2-yl)methyl]glycine (H3Dpaa) and two bifunctional derivatives to 68Ga complexation. The ligand H3Dpaa was synthesized in two steps as described by Mazzanti et al.32 from ethyl 6-(chloromethyl)picolinate and glycine ethyl ester hydrochloride followed by deprotection under acidic conditions. The bifunctional chelates, (S)-6,6′-(((3-amino-1-carboxypropyl)azanediyl)bis(methylene))dipicolinic acid (H3Dpaa.dab) and N,N-bis((6-carboxypyridin-2-yl)methyl)-L-glutamic acid (H4Dpaa.ga), were synthesised in a similar fashion from the relevant protected amino acid analogues, H-dab(Boc)-OMe hydrochloride33 and diethyl glutamate hydrochloride as shown in Scheme 1. Et4Dpaa.ga was obtained in a 72% yield as a yellow oil. Deprotection of this proligand via acid hydrolysis yielded H4Dpaa.ga as an off white solid in an 82% yield. Crystals of H4Dpaa.ga suitable for single crystal X-ray diffraction were obtained by precipitation from water. The obtained structure‡ (Fig. S22†) shows a distinct asymmetry caused by the chirality of the amino acid used. The molecule displays a configuration with one picoline ring on top of the other at a separation at little over 3 Å. This arrangement facilitates an intramolecular hydrogen bond between the glycine carboxylic acid and the pyridine (N2–H2⋯O6). The second picolinic acid is present with the nitrogen protonated and free carboxylate (N1–H1⋯O7i, where i = x − ½, ½ − y, z + ½). This arrangement facilitates an R22(8) embrace to the terminal carboxylic acid group of another molecule. In the solid there is an extensive hydrogen bond network.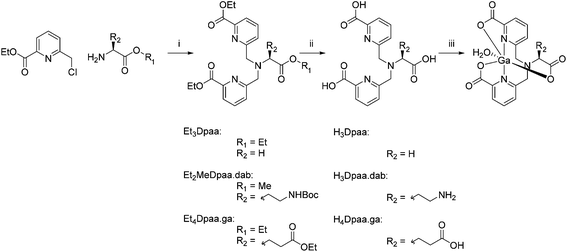 | ||
| Scheme 1 Synthetic scheme for the synthesis of ligands reported in this paper. (i) K2CO3, KI, MeCN, 60 °C, 12 h. (ii) 6 M HCl, reflux, 16 h, (iii) GaCl3, H2O, pH 4. | ||
Ga(III) complexes were synthesized by addition of GaCl3 to an aqueous solution of the ligand. The resulting complexes precipitated out of solution and were collected.
Evidence for complexation can be seen through the distinct NMR resonances of the two protons in the CH2 environment between the picolinate arms and the central amine. While these protons are equivalent (δH3Dpaa = 3.92, δH3Dpaa.dab = 4.41, δH4Dpaa.ga = 4.40) on the NMR time scale in the free ligands, in the Ga(III) complexes they are inequivalent (δ[Ga(Dpaa)] = 4.62 and 4.48, δ[Ga(Dpaa.dab)] = 4.56–4.33 and 4.07, δ[Ga(Dpaa.ga)] = 4.66 and 4.33) and show a strong geminal coupling to one another (2JHH = 16–17 Hz).
Crystal structure of Ga(III) complexes
Single crystals of [Ga(Dpaa)] and [Ga(Dpaa.ga)] of a suitable quality for X-ray diffraction were obtained from acidic aqueous solutions. In each of the two structures,§,¶ Ga(III) is in a six coordinate environment, with five coordination sites occupied by the ligand and the final site by water (Fig. 2). The bonds from the Ga(III) ion to nitrogen atoms are rather longer than those to the oxygen atoms of the carboxylates (as shown in Table 1). This is a consequence of the strain in the ligand and may also reflect the preference of Ga(III) for the hard oxygen donors. The central amine atom (N2) in each structure is rather too distant from the Ga(III) to suggest a bond.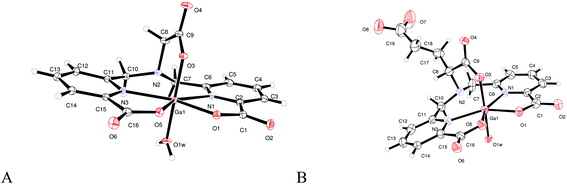 | ||
| Fig. 2 ORTEP representations of molecular structures of (A) [Ga(H2O)(Dpaa)]§ and (B) [Ga(H2O)(Dpaa.ga)]¶ obtained by single crystal X-ray crystallography (drawn with 30% certainty, solvent molecules omitted for clarity). | ||
| Bond | Bond length/Å | |
|---|---|---|
| GaDpaa | GaDpaa.ga | |
| O1–Ga1 | 2.0441(10) | 2.001(6) |
| N1–Ga1 | 2.2354(11) | 2.191(7) |
| N2⋯Ga1 | 2.4880(11) | 2.513(8) |
| N3–Ga1 | 2.2017(12) | 2.180(7) |
| O5–Ga1 | 2.0229(10) | 2.034(6) |
| O3–Ga1 | 1.9173(10) | 1.942(6) |
| O1W–Ga1 | 1.9109(10) | 1.952(5) |
Each structure features a distorted octahedral coordination of the Ga(III) as a consequence of the geometry of the ligand. The greatest distortion is obvious in the plane of the picolinic acids (Tables 1 and 2). In each case the N1–Ga–N3 angle is greater than 133° and consequently the N–Ga–O angles are much smaller than the ideal 90° expected for undistorted octahedral geometry.
| Angle | Bond angle/° | |
|---|---|---|
| GaDpaa | GaDpaa.ga | |
| O1–Ga1–N1 | 73.97(4) | 76.1(3) |
| N1–Ga1–N3 | 135.16(4) | 133.7(3) |
| N3–Ga1–O5 | 74.95(4) | 75.2(3) |
| O5–Ga1–O1 | 75.92(4) | 75.5(3) |
| O3–Ga1–O1 | 90.08(4) | 93.8(3) |
| O3–Ga1–N1 | 88.09(4) | 86.0(2) |
| O3–Ga1–N3 | 91.36(4) | 96.4(3) |
| O3–Ga1–O5 | 92.23(4) | 90.7(2) |
| O3–Ga1–O1W | 175.40(4) | 172.8(2) |
[Ga(Dpaa)] is relatively symmetric and there is a pseudo-mirror plane (through O3, N2, and O1W) present in the complex. The picolinate arms are close to planar; the angle subtended by the two mean planes of the picolinates is 9.53(3)°. In contrast the glutamic acid backbone introduces a twist in [Ga(Dpaa.ga)] removing the pseudo-mirror plane and pushing the picolinate rings further out of the same plane such that the angle between their mean planes is 15.85(3)°. It is important to note that the pendant carboxylate arm of the glutamic acid is not involved in Ga(III) coordination. Full crystallographic data can be found in the ESI.†
Potentiometry
Protonation constants of the studied compounds were determined by potentiometry (Table 3, distribution diagrams are shown in Fig. S2†). The determined protonation constants of H3Dpaa are in very good agreement with those previously reported.32,35 The first protonation constant (pK1 = 7.38) is assigned to the central amine group. The two remaining constants are ascribed to protonation of the picolinate arms. The protonation constant of the pendant acetic acid group could not be determined due to its highly acidic nature.| H3Dpaa | H3Dpaa.dab | H4Dpaa.ga | |
|---|---|---|---|
| a ([L] = 0.004 M, T = 25 °C, I = 0.1 M (NMe4)Cl). | |||
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K1 K1 |
7.38 | 11.35 | 7.17 |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K2 K2 |
3.73 | 5.39 | 4.67 |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K3 K3 |
2.82 | 3.77 | 3.92 |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K4 K4 |
— | 2.69 | 2.75 |
The additional carboxylate group in H4Dpaa.ga introduces an additional protonation constant in the weakly acidic region (pK2 = 4.67). However, it does not alter significantly the protonation constants of the ligand core (pK1 = 7.33) or the picolinate arms. The terminal amino group in H3Dpaa.dab is protonated above pH 11 (pK1 = 11.35). Presence of the additional protonated amino group significantly decreases basicity of the central amino group in the ligand core (pK2 = 5.39).
Complexation of Ga(III), Cu(II) and Zn(II) ions by the three ligands was studied by potentiometry. These metal–ligand systems were chosen due to their importance for the potential application of these ligands to nuclear medicine. The results are summarized in Table 4.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K) obtained for complexes
K) obtained for complexes
| Metal ion | H3Dpaa | H3Dpaa.dab | H4Dpaa.ga |
|---|---|---|---|
a Determined by UV-VIS titration [L] = [M] = 0.1 mM, T = 25 °C, pH = 2–7.
b Determined by potentiometric titration ([L] = [M] = 0.004 M, T = 25 °C, I = 0.1 M (NMe4)Cl).
c Constant (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaHL) describing equilibrium Ga(III) + (HL)2− KGaHL) describing equilibrium Ga(III) + (HL)2− ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) [Ga(HL)]+ where the amine group deprotonation and hydroxido species formation are not considered.
d Determined by UV-VIS titration ([L] = [M] = 0.01 mM, T = 25 °C, pH = 0–2). [Ga(HL)]+ where the amine group deprotonation and hydroxido species formation are not considered.
d Determined by UV-VIS titration ([L] = [M] = 0.01 mM, T = 25 °C, pH = 0–2).
|
|||
| Ga(III) | 18.53a | 22.08b | 18.36b |
| 16.13b,c | |||
| Cu(II) | 10.85b | 19.1b,d | 14.52b,d |
| Zn(II) | 11.93b | 15.8b,d | 13.38b,d |
The study of the Ga(III)-Dpaa system was not straightforward due to low solubility of the uncharged [Ga(Dpaa)] species. Therefore, UV-VIS titration was performed at significantly lower concentration (Fig. S4†). Nitrogen atoms of the ligands are weakly basic and this leads to complexation of metal ions even in strongly acidic solutions. As a consequence, some of the complexes were fully formed in the beginning of potentiometric titrations. Thus, Cu(II) and Zn(II) systems with H4Dpaa.ga and H3Dpaa.dab were also studied by UV-VIS spectrophotometry at pH 0–2 (Fig. S6 and S8†). Spectrophotometry was not employed in Ga(III) systems for these ligands as stability constants could be determined from competition with hydroxide ions in the alkaline region (i.e. formation of [Ga(OH)4]−).
The stability constants of [Ga(L)] species are similar for both H3Dpaa and H4Dpaa.ga. This indicates a negligible role of the distant carboxylate in complexation reactions of H4Dpaa.ga in agreement with crystallographic data. For [Ga(Dpaa.ga)], the first protonation constant ([M(L)] + H ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) [M(HL)], log
[M(HL)], log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = 4.04, Table S2†) is comparable to that of free ligand, further supporting that the distant carboxylate group is not coordinated. In both systems, hydroxido species, [Ga(OH)(Dpaa)]− and [Ga(OH)(Dpaa.ga)]2−, are formed already in acidic region through a formal aqua ligand dissociation with corresponding pKa values of 4.41 and 5.27, respectively.
K = 4.04, Table S2†) is comparable to that of free ligand, further supporting that the distant carboxylate group is not coordinated. In both systems, hydroxido species, [Ga(OH)(Dpaa)]− and [Ga(OH)(Dpaa.ga)]2−, are formed already in acidic region through a formal aqua ligand dissociation with corresponding pKa values of 4.41 and 5.27, respectively.
This points to an unsaturated coordination sphere of the metal ion in these complexes in which some of the ligand donor groups remain uncoordinated. The unsaturated coordination sphere is corroborated by the crystal structures obtained in which this site is occupied by a bound water molecule. Complexes in which the coordination sphere of Ga(III) is not fully satisfied by a chelate with six coordinating atoms have been reported previously, with modelling suggesting either water of chloride bound in the vacant site.43,44 Stability constants of the studied Ga(III) complexes are significantly higher than those reported for complexes of H3Dpaa with lanthanide(III) ions (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = 10.6 and 10.4 for [Gd(Dpaa)] and [Tb(Dpaa)], respectively).32 This indicates that the ligands better suits “hard” and small metal ions such as Ga(III).
K = 10.6 and 10.4 for [Gd(Dpaa)] and [Tb(Dpaa)], respectively).32 This indicates that the ligands better suits “hard” and small metal ions such as Ga(III).
The stability constant for [Ga(Dpaa.dab)] complex (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaL = 22.08) is surprisingly much higher than those of the other two Ga(III) complexes. This is due to the different structure of the [GaL] species. The Ga(III) ion in [Ga(Dpaa)] and [Ga(Dpaa.ga)] complexes is coordinated by the fully deprotonated ligand. Ligand H3Dpaa.dab contains the highly basic terminal amino group. Consequently, the proton bound to the coordinated water molecule is more acidic than the proton bound to the amino group. The first protonation constant (log
KGaL = 22.08) is surprisingly much higher than those of the other two Ga(III) complexes. This is due to the different structure of the [GaL] species. The Ga(III) ion in [Ga(Dpaa)] and [Ga(Dpaa.ga)] complexes is coordinated by the fully deprotonated ligand. Ligand H3Dpaa.dab contains the highly basic terminal amino group. Consequently, the proton bound to the coordinated water molecule is more acidic than the proton bound to the amino group. The first protonation constant (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ka = 5.40, Table S2†) is similar to those describing formation of the monohydroxide species in both the Ga(III)-Dpaa and Ga(III)-Dpaa.ga systems and should be ascribed to the formation of the hydroxide species as well. Thus, the [Ga(Dpaa.dab)] complex is zwitterionic, with a hydroxide anion bound to the Ga(III) and with a protonated amine group. Dissociation constant of the amino group in the complex cannot be determined as it would dissociate at very high pH where the complex is fully decomposed to [Ga(OH)4]−. To compare the stability constants, it is more suitable to consider equilibrium between Ga(III) ion and monoprotonated ligand molecule (log
Ka = 5.40, Table S2†) is similar to those describing formation of the monohydroxide species in both the Ga(III)-Dpaa and Ga(III)-Dpaa.ga systems and should be ascribed to the formation of the hydroxide species as well. Thus, the [Ga(Dpaa.dab)] complex is zwitterionic, with a hydroxide anion bound to the Ga(III) and with a protonated amine group. Dissociation constant of the amino group in the complex cannot be determined as it would dissociate at very high pH where the complex is fully decomposed to [Ga(OH)4]−. To compare the stability constants, it is more suitable to consider equilibrium between Ga(III) ion and monoprotonated ligand molecule (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaHL 16.13, Table 4) where influence of the above processes is not considered. The value is lower and in line with those for the other systems if the presence of a positive charge, due to a protonated amino group in the ligand molecule, is taken into account. This is reflected in the pM values which are 8.91, 6.34 and 8.21 (pH = 7.4, [Ga] = 10−6 M, [L] = 10−5 M) for [Ga(Dpaa)], [Ga(Dpaa.dab)] and [Ga(Dpaa.ga)], respectively. When comparing the formation constant of [Ga(Dpaa)] of 18.53 with that of [Ga(DOTA)] and [Ga(NOTA)] (log
KGaHL 16.13, Table 4) where influence of the above processes is not considered. The value is lower and in line with those for the other systems if the presence of a positive charge, due to a protonated amino group in the ligand molecule, is taken into account. This is reflected in the pM values which are 8.91, 6.34 and 8.21 (pH = 7.4, [Ga] = 10−6 M, [L] = 10−5 M) for [Ga(Dpaa)], [Ga(Dpaa.dab)] and [Ga(Dpaa.ga)], respectively. When comparing the formation constant of [Ga(Dpaa)] of 18.53 with that of [Ga(DOTA)] and [Ga(NOTA)] (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KGaL = 26.05 and 29.60, respectively)45,46 the thermodynamic stability is lower, but still it may be sufficient for the application due to the short half-life of 68Ga.
KGaL = 26.05 and 29.60, respectively)45,46 the thermodynamic stability is lower, but still it may be sufficient for the application due to the short half-life of 68Ga.
The stability constants obtained for [Cu(Dpaa)] and [Zn(Dpaa)] complexes are significantly lower than that of the Ga(III) complex (Tables S2 and S3†). This may be rationalized due to the low flexibility of the ligand preventing the complex from fulfilling the ideal coordination geometry of these two ligands and due to the high charge density of Ga(III) compared to Cu(II) and Zn(II). The ligands are highly charged with hard oxygen donor atoms and interaction of the ligands with Ga(III) is highly electrostatic in its nature and, therefore, thermodynamic stability of the Ga(III) complexes is increased compared to Cu(II) and Zn(II) complexes. This preference for Ga(III) complexation is encouraging for biomedical imaging using [68Ga][Ga(Dpaa)] as Cu(II) and Zn(II) are two of the most abundant transition metal ions in vivo.47
Whilst the Cu(II) and Zn(II) complexes of H3Dpaa.dab and H4Dpaa.ga are less thermodynamically stable than the Ga(III) complexes, the resulting complexes are more stable than those seen with H3Dpaa. This suggests that the additional coordinating arms may be involved in the complexation of these two metals.
Radiolabelling
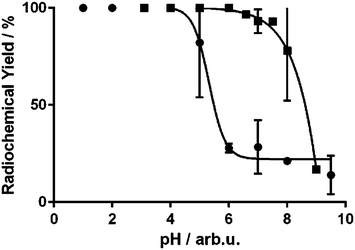
The radiochemical yield (RCY) of H3Dpaa complexing 68Ga was found to have a distinct pH dependence (Fig. 4); in acidic aqueous solution the radiochemical yield achieved by 100 μM H3Dpaa was very high up to pH 4, with a radiochemical yield >95% achieved in 15 minutes at ambient temperature. Above pH 5 the radiochemical yield at ambient temperature fell to approximately 30% after a reaction time of 15 minutes. This is likely due to the formation of gallium hydroxide species with slower complexation kinetics above pH 4.6,13 However, in the presence of 0.1 M phosphate buffer the high RCY was maintained up to pH 7.5, with a 92% yield being achieved in 15 minutes at ambient temperature. Under these conditions ([L] = 100 μM, pH = 7.5, T = 25 °C, I = 0.1 M phosphate buffer, t = 15 minutes), macrocyclic chelators DOTA (0% RCY) and NOTA (48% RCY) performed poorly, but acyclic chelates EDTA (95% RCY) and THP (95% RCY) performed comparably to H3Dpaa (Table S4†). Above pH 8 the RCY of H3Dpaa fell even in buffered solutions, likely due to the formation of [Ga(OH)4]− as indicated by the potentiometric results (Fig. 3). When heated to 37 °C the radiochemical yield achieved at pH 7.5 by 100 μM H3Dpaa in 15 minutes increased to 95%.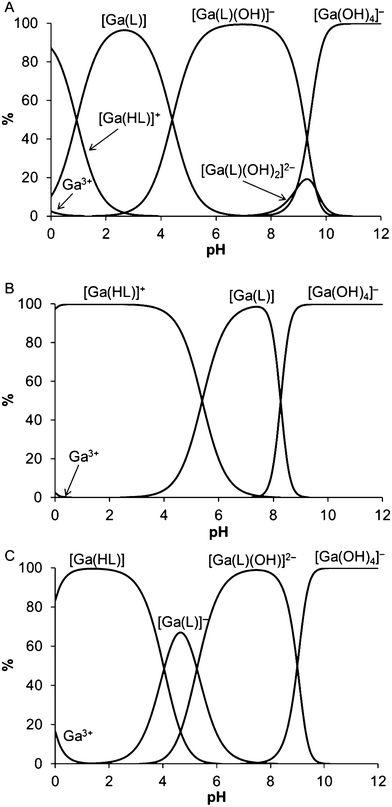 | ||
| Fig. 3 Speciation diagrams for Ga(III) – ligand systems. (A) H3Dpaa, (B) H3Dpaa.dab(NH2), (C) H4Dpaa.ga (T = 25 °C, I = 0.1 M (NMe4)Cl, [L] = [Ga(III)] = 0.004 M). | ||
This difference between buffered and aqueous solutions may be explained by a weak gallium–phosphate complex being formed. This may act as a “pre-coordination” complex, preventing the rapid formation of gallium hydroxide species that would result in slower complexation due to the kinetically inert gallium–hydroxide bonds.19
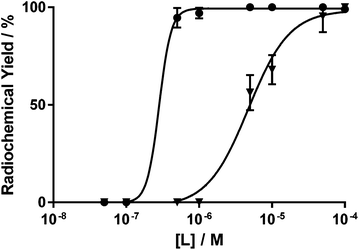
The pH of the radiolabelling reaction also has a significant effect on the concentration of ligand required for efficient radiolabelling (Fig. 5). When radiolabelling at pH 4, efficient complexation is achieved at ligand concentrations as low as 500 nM, with radiochemical yields >90% achieved in 15 minutes at ambient temperature. However, the radiochemical yield sharply decreases below this concentration with no radiolabelling seen when [H3Dpaa] = 100 nM. In contrast, at pH 7.4 the radiochemical yield after 15 minutes at ambient temperature is maintained above 90% at 50 μM, however drops below 90% at ligand concentrations of 10 μM.
The ability to rapidly complex 68Ga at neutral pH has the potential to simplify the production of 68Ga labelled radiopharmaceuticals by reducing the post-reaction conditioning required. To develop this further, the complexation of 68Ga by 100 μM H3Dpaa in saline and phosphate buffered saline (PBS) was assessed. High radio-chemical yields of 99% and 95% respectively were achieved with mild heating (37.7 °C) after 5 minutes. Furthermore, the pH of the PBS solution remained at pH 7.4 after complexation, although the pH of the saline solution was lower (pH 5.5) after addition of the ligand.
The bifunctional chelates H3Dpaa.dab and H4Dpaa.ga achieved 99% RCYs after 5 minutes at pH 4 and ambient temperature, and the RCY remained as high as 84% and 94% respectively at pH 7.4 in PBS after 5 minutes at 37 °C (Table 5 and Fig. S9†). Specific activities of 20.0 GBq μmol−1 (541 mCi μmol−1) and 28.9 GBq μmol−1 (781 mCi μmol−1) were achieved with H3Dpaa.dab and H3Dpaa.ga respectively (Fig. S12 and S13†) after radiolabelling at pH 4 (T = 25 °C, t = 5 minutes, I = 0.1 M acetate buffer), however H3Dpaa achieved a specific activity of only 3.9 GBq μmol−1 (105 mCi μmol−1)(Fig. S11†).
The stability of the radiolabelled complexes formed were assessed against biological competitors, apo-transferrin and foetal bovine serum (FBS). Some stability to the iron transport protein apo-transferrin was seen, 92% of 68Ga activity was associated with the [68Ga][Ga(Dpaa)] complex after 2 hours of incubation (Fig. S14†). In FBS complete decomplexation of the 68Ga was seen within 30 minutes for all chelate derivatives (Fig. S15†). This suggests that having the vacant coordination site filled by H2O (Fig. 2), allows for 68Ga to be more readily taken up by competitor proteins found in the serum and therefore H3Dpaa is not the ideal system for 68Ga application in vivo.
Conclusions
We describe a ligand system based on amino acids allowing for synthesis of bifunctional chelators. This family of ligands are able to complex 68Ga under physiological conditions (5 minutes, pH 7.4 and T = 37.7 °C) at 100 μM. The H3Dpaa family of ligands are also able to complex 68Ga efficiently across a wide pH range. The ligands presented here are more suited to the complexation of small, hard cations like Ga(III) than they are to softer Zn(II) and Cu(II) cations, as indicated by their higher association constants. Studies with biological competitors, FBS and apo-transferrin suggest that these chelates are not suited for in vivo application with 68Ga. However the chelator's preference for Ga(III) and the high radiochemical yield achievable under physiological pH and temperature provides the groundwork to support future studies into acyclic chelators for 68Ga.Experimental section
NMR spectra were recorded on a JEOL ECP 400 MHz/JEOL Lambda 400 MHz spectrometer using the residual protic solvent signal as an internal reference. ESI Mass spectra were recorded on Advion MS SOP electrospray ionisation spectrometer. pH measurements were carried out using a Jenway model 3520 pH/mV/temperature meter with a three point calibration.48 All commercially available starting materials were used without further purification. Ethyl 6-(chloromethyl)picolinate was synthesised from 2,6-dipicolinic acid according to literature methods.49,50 H3Dpaa and H3Dpaa.dab were synthesised according to literature methods.32,33Synthesis of Et4Dpaa.ga
To a suspension of L-glutamic acid diethyl ester hydrochloride (1.82 g, 7.6 mmol), potassium carbonate (4.00 g, 28.9 mmol) and potassium iodide (2.64 g, 15.9 mmol) in anhydrous acetonitrile (10 mL) was added ethyl 6-(chloromethyl)picolinate hydrochloride (3.75 g, 15.9 mmol). The mixture was heated to 60 °C for 12 hours. The reaction was quenched with water (50 mL) and extracted with dichloromethane (3 × 100 mL). The combined organic layers were dried with magnesium sulfate and concentrated under reduced pressure. The dark orange oil was purified by column chromatography (silica, 50 mm × 150 mm, hexane/ethyl acetate 20–50%) to yield an orange oil (2.91 g, 5.5 mmol, 72%).1H NMR (400 MHz, CDCl3, 298 K): 7.95 (dd, 2H, J = 6, 2.5 Hz), 7.76 (m, 4H, py), 4.46 (q, 4H, 3JHH = 7.1 Hz), 4.22 (qd, 2H, 3JHH = 7.1 Hz, J = 1.8 Hz), 4.16 (d, 2H, 2JHH = 15.5 Hz), 4.10 (d, 2H, 2JHH = 15.5 Hz), 4.01 (qt, 2H, 3JHH = 7.1 Hz, J = 3.7 Hz), 3.45 (dd, 1H, J = 9.2, 6.1 Hz), 2.50 (m, 2H), 2.13 (tq, 1H, 2JHH = 14.1 Hz, 3JHH = 7.5 Hz), 2.02 (dqt, 1H, 2JHH = 14 Hz, 3JHH = 7.5 Hz, J = 1.6 Hz), 1.44 (t, 6H, 3JHH = 7.1 Hz), 1.33 (t, 3H, 3JHH = 7.1 Hz), 1.18 (t, 3H, 3JHH = 7.1 Hz) 13C NMR (100 MHz, CDCl3, 298 K): 173.04, 172.36, 165.26, 160.23, 147.76, 137.21, 125.93, 123.35, 62.33, 61.73, 60.65, 60.25, 57.08, 30.66, 24.79, 14.38, 14.25, 14.08.
Synthesis of H4Dpaa.ga
To Et4Dpaa.ga (887 mg, 1.72 mmol) was added 6 M HCl (14 mL). The solution was heated to reflux for 16 hours and then allowed to cool to room temperature. The solvent was removed to yield a yellow oil. Acetone (10 mL) was added to yield an off-white precipitate (617 mg, 1.26 mmol, 73%).1H NMR (400 MHz, D2O (pD = 7.1), 298 K), δ: 7.72–7.55 (m, 4H), 7.42–7.32 (m, 2H), 4.47–4.33 (m, 4H), 3.77–3.63 (m, 1H), 2.43–2.27 (m, 2H), 2.27–2.06 (m, 2H). 13C NMR (100 MHz, D2O (pD = 7.1), 298 K), δ: 181.70, 176.22, 171.57, 153.47, 151.81, 138.88, 126.17, 123.11, 69.52, 57.14, 34.51, 25.67 MS (ESI), m/z: 418.04 [M + H]+. Elemental analysis (C/H/N), %: expected for H4Dpaa.ga·2HCl (C19H21Cl2N3O8): 46.55/4.32/8.57, found: 46.22/4.40/7.99.
H3Dpaa
Et3Dpaa (38 mg, 0.089 mmol) in 6 M HCl (6 mL) was heated to reflux for 16 h. The sample was concentrated, ethanol added, and the precipitate collected and washed with diethyl ether to yield a white solid (36.5 mg, 0.089 mmol, 100%).1H NMR (400 MHz, D2O (pD = 8.8), 298 K), δ: 7.72–7.62 (m, 4H), 7.38 (br d, 2H, 3JHH = 7.8 Hz), 3.92(br s, 4H), 3.27 (br s, 2H). 13C NMR (100 MHz, D2O (pD = 8.8), 298 K), δ: 172.96, 152.71, 138.14, 125.87, 122.35, 60.07, 59.22 MS(ESI), m/z = 346.4 [M + H]+. Elemental analysis (C/H/N), %: expected for H3Dpaa·1.3(HCl)0.25(diethyl ether) (C17H19.05Cl1.3N3O6.25): 49.62/4.67/10.21, found: 49.63/4.40/9.94.
H3Dpaa.dab
Et2MeDpaa.dab(Boc) (676.4 mg, 1.21 mmol) was dissolved in 6 M HCl (20 mL) and heated to reflux overnight. The solvent was removed under reduced pressure to yield a yellow oil. Addition of acetone resulted in precipitation of a solid. Isolation of this precipitate yielded a yellow solid (446.3 mg, 0.84 mmol, 70%).1H NMR (400 MHz, D2O (pD = 1.6), 298 K), δ: 8.09 (t, 2H, 3JHH = 7.8 Hz), 7.96 (d, 2H, 3JHH = 7.8 Hz), 7.71 (d, 2H, 3JHH = 7.8 Hz), 4.41 (s, 4H), 4.00 (t, 1H, 3JHH = 7.3 Hz), 3.37–3.18 (m, 2H), 2.40–2.23 (m, 2H). 13C NMR (100 MHz, D2O (pD = 1.6), 298 K), δ: 174.50, 164.62, 154.61, 145.26, 143.74, 128.24, 124.92, 64.99, 55.53, 37.66, 26.10. MS (ESI) m/z: 389.4 [M + H]+. Elemental analysis (C/H/N), %: expected for H3Dpaa.dab(HCl)3(Acetone)0.55 (C19.65H28.3Cl3N4O7.55): 43.09/5.21/10.23, found 42.85/5.15/9.93.
Synthesis of [Ga(Dpaa.ga)]
To H4Dpaa.ga (25 mg, 0.062 mmol) in a solution of methanol (1 mL) and water (1 mL) was added GaCl3 (10.9 mg, 0.062 mmol) in water (1.2 mL). The solution was heated to 70 °C for 16 hours and allowed to cool. The precipitate was collected by centrifugation (4000 rpm, 3 minutes) to give a white solid (14.6 mg, 0.026 mmol, 42%).1H NMR (400 MHz, D2O (pD = 6.0), 298 K) δ: 8.30–8.11 (m, 4H), 7.81–7.73 (m, 2H), 4.66 (br d, 2H, 2JHH = 17 Hz), 4.33 (br d, 2H, 2JHH = 17 Hz), 3.05–2.97 (m, 1H), 2.44–2.33 (m, 1H), 2.26–2.15 (m, 1H), 2.06–1.88 (m, 2H). 13C NMR (100 MHz, D2O (pD = 6.0), 298 K) δ: 180.94, 178.13, 168.10, 152.32, 151.03, 144.69, 144.45, 142.83, 142.76, 127.57, 126.38, 123.11, 123.06, 62.08, 58.28, 53.07, 34.80, 22.33. MS (ESI), m/z: 483.94 [69GaM + H]+, 485.97 [71GaM + H]+. Elemental analysis (C/H/N), %: expected for GaDpaa.ga(COOH)·3.5H2O·0.5MeOH (C19.5H25GaN3O12): 41.59/4.47/7.46, found: 41.56/4.68/7.42.
Synthesis of [Ga(Dpaa)]
To H3Dpaa (76 mg, 0.22 mmol) in water (5 mL) was added GaCl3 (38.8 mg, 0.22 mmol) in water (3.8 mL). The pH was adjusted to 4.5 with sodium hydroxide. The solution was heated to reflux for 3 days. After cooling, the solution was filtered to give a white solid (76 mg, 0.14 mmol, 64%).1H NMR (400 MHz, D2O (pD = 8.8), 298 K) δ: 8.21 (t, 2H, 3JHH = 7.3 Hz), 8.14 (d, 2H, 3JHH = 7.3 Hz), 7.75 (d, 2H, 3JHH = 7.3 Hz), 4.62 (dd, 2H, 2JHH = 16.0 Hz, 4JHH = 2.75 Hz), 4.48 (br d, 2H, 2JHH = 16.0 Hz), 3.39 (d, 2H, 4JHH = 2.3 Hz). 13C NMR (100 MHz, D2O (pD = 8.8), 298 K) δ: 176.86, 168.40, 151.26, 144.57, 142.35, 126.76, 122.99, 60.60, 59.89. MS (ESI), m/z: 411.95 [69GaM + H]+, 413.89 [71GaM + H]+. Elemental Analysis (C/H/N), %: expected for GaDpaa·H2O·3HCl (C16H17Cl3GaN3O7): 35.63/3.18/7.79, found: 35.85/3.09/7.59.
Synthesis of [Ga(Dpaa.dab)]
To H3Dpaa.dab (25 mg, 0.064 mmol) in methanol (5 mL) was added GaCl3 (11.3 mg, 0.064 mmol) in water (1.2 mL). This solution was heated to 70 °C for 1 hour and allowed to cool. The precipitate was collected by centrifugation (4000 rpm, 3 minutes) to give a white solid (17 mg, 0.039 mmol, 60%).1H NMR (400 MHz, D2O (pD = 1.1), 298 K) δ: 8.32–8.24 (m, 2H), 8.24–8.12 (m, 2H), 7.88–7.75 (m, 2H), 4.56–4.33 (m, 3H), 4.07 (br d, 1H, 2JHH = 17.4 Hz), 3.18–3.12 (m, 1H), 3.11–3.02 (m, 2H), 2.28–2.07 (m, 3H). 13C NMR (100 MHz, D2O (pD = 1.1), 298 K) δ: 177.21, 167.18, 167.05, 153.37, 152.71, 145.06, 144.69, 143.97, 128.49, 127.67, 123.77, 123.63, 61.19, 65.00, 51.95, 38.11, 30.30, 24.07. MS (ESI), m/z: 454.43 [69GaM + H]+, 456.39 [71GaM + H]+. Elemental Analysis (C/H/N), %: expected for GaDpaa.dab(NH2)·H2O·0.9HCl (C18H19.9Cl0.9GaN4O7): 42.73/3.97/11.07, found: 43.02/3.63/10.87.
Potentiometry
Potentiometry was carried out according to previously published procedures.51,52 Protonation and stability constants were determined in 0.1 M (NMe4)Cl at 25.0 °C with pKw = 13.81. Protonation constants ([L] = 0.004 M) were determined from data obtained in pH range 1.6–12.1 (∼40 points per titration and three parallel titrations) with electrode calibrated by acid–base titration in extended pH ranges (1.7–12.2 for each titration set). Complex stability constants ([L] = [M] = 0.004 M) were determined from data obtained in pH range 1.5–12.1, 50 data points per titration, three parallel titrations.UV-VIS spectra were recorded on spectrophotometer Specord 50 Plus (Analytik Jena AG). Temperature was maintained by Peltier block. UV-VIS titration of the Ga(III)-H3Dpaa system ([L] = [M] = 0.0001 M) was performed at pH range 2.4–6.9 in 0.1 M (NMe4)Cl, pH was adjusted with ∼0.2 M (NMe4)OH using a glass electrode. UV-VIS titrations of the Cu(II) and Zn(II) systems with H4Dpaa.ga and H3Dpaa.dab ([L] = [M] = 0.00001 M) were performed at pH range 0.0–2.0 without ionic strength control, pH was calculated from added amount of HCl.
The titration and UV-VIS data were treated simultaneously with OPIUM program package.53,54 Calculated constants are concentration constants defined as βhl = [HhLl]/[H]h·[L]l or βhlm = [HhLlMm]/[H]h·[L]l·[M]m and standard deviations are given directly by the program. pM values were also calculated by OPIUM from determined protonation and stability constants.
The full version of the OPIUM program is available (free of charge) on http://www.natur.cuni.cz/_kyvala/opium.html.
Radiolabelling
The IGG100 generator was eluted with 0.6 M aq. HCl (3 mL). This eluate (300–200 MBq) was diluted with H2O (15 mL) and passed through a Strata-X-C 33 μM Cation Mixed-mode polymeric support. The activity was liberated from the column using 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 acetone
2 acetone![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 M aq. HCl (1 mL). Aliquots (∼30 MBq) of this solution were dried under a stream of inert gas at 90 °C and allowed to cool before use. 1 mL of ligand solution was added to the dried 68Ga and shaken. 5 μL aliquots were taken for analysis by TLC and 20 μL aliquots for analysis by HPLC.
0.1 M aq. HCl (1 mL). Aliquots (∼30 MBq) of this solution were dried under a stream of inert gas at 90 °C and allowed to cool before use. 1 mL of ligand solution was added to the dried 68Ga and shaken. 5 μL aliquots were taken for analysis by TLC and 20 μL aliquots for analysis by HPLC.
TLC analysis was performed on Kieselgel 60 F254 plates (Merck) with an eluate of 0.1 M citric acid in water. HPLC analysis was carried out using an Agilent Zorbax Eclipse XDB-C18 column and a solvent system of water + 0.1% TFA and methanol.
Assessment of stability to apo-transferrin
100 μL of radiolabelling solution containing 100 μM H3Dpaa in 0.1 M acetate buffer (pH 4.5) after incubation with gallium-68 for 5 minutes was added to 600 μL of 1 mg mL−1 transferrin in 0.1 M sodium hydrogen carbonate solution to give a final pH of 7.2. This solution was incubated at 37 °C with aliquots taken at 60 and 120 minutes for HPLC analysis (Fig. S13†).Assessment of stability in foetal bovine serum
100 μL of radiolabelling solution containing 100 μM ligand in PBS (pH 7.4) was added to 1.5 mL of foetal bovine serum and incubated at 37 °C. Aliquots were taken at 30 minutes for TLC analysis (Fig. S14†).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B). [0–3](95
B). [0–3](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). [3–18](95
5). [3–18](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5–5
5–5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95). [18–20](5
95). [18–20](5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95). [20–25](5
95). [20–25](5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95–95
95–95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). [25–30](95
5). [25–30](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5).
5).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B). [0–10](95
B). [0–10](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). [10–11](95
5). [10–11](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5–5
5–5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95). [11–14](5
95). [11–14](5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95). [14–15](5
95). [14–15](5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95–95
95–95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) [15–20](95
5) [15–20](95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5).
5).
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors would like to thank the University of Hull for the PhD studentship and The Royal Society for the research grant RG160156, for a 68Ga generator, which funded this project. The authors would also like to thank Sergei Palmer for his input. We thank the EPSRC UK National Crystallography Service at the University of Southampton for the collection of the crystallographic data.55References
- S. S. Gambhir, Nat. Rev. Cancer, 2002, 2, 683–693 CrossRef CAS PubMed.
- S. M. Ametamey, M. Honer and P. A. Schubiger, Chem. Rev., 2008, 108, 1501–1516 CrossRef CAS PubMed.
- P. W. Miller, N. J. Long, R. Vilar and A. D. Gee, Angew. Chem., Int. Ed., 2008, 47, 8998–9033 CrossRef CAS PubMed.
- T. W. Price, J. Greenman and G. J. Stasiuk, Dalton Trans., 2016, 45, 15702–15724 RSC.
- D. Brasse and A. Nonat, Dalton Trans., 2015, 44, 4845–4858 RSC.
- M. Fani, J. P. André and H. R. Maecke, Contrast Media Mol. Imaging, 2008, 3, 53–63 CrossRef CAS PubMed.
- E. W. Price and C. Orvig, Chem. Soc. Rev., 2014, 43, 260–290 RSC.
- F. Rösch, Appl. Radiat. Isot., 2013, 76, 24–30 CrossRef PubMed.
- I. Velikyan, J. Labelled Compd. Radiopharm., 2015, 58, 99–121 CrossRef CAS PubMed.
- M. D. Bartholomä, A. S. Louie, J. F. Valliant and J. Zubieta, Chem. Rev., 2010, 110, 2903–2920 CrossRef PubMed.
- X. Sun, Y. Li, T. Liu, Z. Li, X. Zhang and X. Chen, Adv. Drug Delivery Rev., 2017, 110–111, 38–51 CrossRef CAS PubMed.
- B. P. Burke, G. S. Clemente and S. J. Archibald, J. Labelled Compd. Radiopharm., 2014, 57, 239–243 CrossRef CAS PubMed.
- M. D. Bartholomä, Inorg. Chim. Acta, 2012, 389, 36–51 CrossRef.
- P. Spang, C. Herrmann and F. Roesch, Semin. Nucl. Med., 2016, 46, 373–394 CrossRef PubMed.
- L. Wei, Y. Miao, F. Gallazzi, T. P. Quinn, M. J. Welch, A. L. Vāvere and J. S. Lewis, Nucl. Med. Biol., 2007, 34, 945–953 CrossRef CAS PubMed.
- I. Velikyan, H. Maecke and B. Langstrom, Bioconjugate Chem., 2008, 19, 569–573 CrossRef CAS PubMed.
- G. J. Stasiuk and N. J. Long, Chem. Commun., 2013, 49, 2732–2746 RSC.
- G. Kramer-Marek, N. Shenoy, J. Seidel, G. L. Griffiths, P. Choyke and J. Capala, Eur. J. Nucl. Med. Mol. Imaging, 2011, 38, 1967–1976 CrossRef CAS PubMed.
- I. Velikyan, Theranostics, 2014, 4, 47–80 CrossRef CAS PubMed.
- B. M. Zeglis, J. L. Houghton, M. J. Evans, N. Viola-Villegas and J. S. Lewis, Inorg. Chem., 2014, 53, 1880–1899 CrossRef CAS PubMed.
- D. J. Berry, Y. M. Ma, J. R. Ballinger, R. Tavare, A. Koers, K. Sunassee, T. Zhou, S. Nawaz, G. E. D. Mullen, R. C. Hider and P. J. Blower, Chem. Commun., 2011, 47, 7068–7070 RSC.
- J. Seemann, B. P. Waldron, F. Roesch and D. Parker, ChemMedChem, 2015, 10, 1019–1026 CrossRef CAS PubMed.
- J. Seemann, B. Waldron, D. Parker and F. Roesch, EJNMMI Radiopharm. Chem., 2016, 1, 1–12 Search PubMed.
- M. T. Ma, C. Cullinane, C. Imberti, J. Baguña Torres, S. Y. A. Terry, P. Roselt, R. J. Hicks and P. J. Blower, Bioconjugate Chem., 2016, 27, 309–318 CrossRef CAS PubMed.
- M. T. Ma, C. Cullinane, K. Waldeck, P. Roselt, R. J. Hicks and P. J. Blower, EJNMMI Res., 2015, 5, 1–11 CrossRef CAS PubMed.
- B. Baur, E. Andreolli, E. Al-Momani, N. Malik, H.-J. Machulla, S. N. Reske and C. Solbach, J. Radioanal. Nucl. Chem., 2014, 299, 1715–1721 CrossRef CAS.
- B. Baur, C. Solbach, E. Andreolli, G. Winter, H.-J. Machulla and S. Reske, Pharmaceuticals, 2014, 7, 517–529 CrossRef PubMed.
- C. Zhai, D. Summer, C. Rangger, H. Haas, R. Haubner and C. Decristoforo, J. Labelled Compd. Radiopharm., 2015, 58, 209–214 CrossRef CAS PubMed.
- S. Ray Banerjee, Z. Chen, M. Pullambhatla, A. Lisok, J. Chen, R. C. Mease and M. G. Pomper, Bioconjugate Chem., 2016, 27, 1447–1455 CrossRef CAS PubMed.
- E. Boros, C. L. Ferreira, J. F. Cawthray, E. W. Price, B. O. Patrick, D. W. Wester, M. J. Adam and C. Orvig, J. Am. Chem. Soc., 2010, 132, 15726–15733 CrossRef CAS PubMed.
- A. M. Nonat, C. Gateau, P. H. Fries, L. Helm and M. Mazzanti, Eur. J. Inorg. Chem., 2012, 2012, 2049–2061 CrossRef CAS.
- A. Nonat, P. H. Fries, J. Pécaut and M. Mazzanti, Chem. – Eur. J., 2007, 13, 8489–8506 CrossRef CAS PubMed.
- G. J. Stasiuk, S. Tamang, D. Imbert, C. Gateau, P. Reiss, P. Fries and M. Mazzanti, Dalton Trans., 2013, 42, 8197–8200 RSC.
- A. Forgacs, R. Pujales-Paradela, M. Regueiro-Figueroa, L. Valencia, D. Esteban-Gomez, M. Botta and C. Platas-Iglesias, Dalton Trans., 2017, 46, 1546–1558 RSC.
- D. M. Weekes, C. F. Ramogida, M. d. G. Jaraquemada-Peláez, B. O. Patrick, C. Apte, T. I. Kostelnik, J. F. Cawthray, L. Murphy and C. Orvig, Inorg. Chem., 2016, 55, 12544–12558 CrossRef CAS PubMed.
- K. G. Kadiyala, T. Tyagi, D. Kakkar, N. Chadha, K. Chuttani, B. G. Roy, M. Thirumal, A. K. Mishra and A. Datta, RSC Adv., 2015, 5, 33963–33973 RSC.
- E. Boros, Y.-H. S. Lin, C. L. Ferreira, B. O. Patrick, U. O. Hafeli, M. J. Adam and C. Orvig, Dalton Trans., 2011, 40, 6253–6259 RSC.
- A. Forgács, M. Regueiro-Figueroa, J. L. Barriada, D. Esteban-Gómez, A. de Blas, T. Rodríguez-Blas, M. Botta and C. Platas-Iglesias, Inorg. Chem., 2015, 54, 9576–9587 CrossRef PubMed.
- B. Gerey, M. Gennari, E. Goure, J. Pecaut, A. Blackman, D. A. Pantazis, F. Neese, F. Molton, J. Fortage, C. Duboc and M.-N. Collomb, Dalton Trans., 2015, 44, 12757–12770 RSC.
- Y. Bretonnière, M. Mazzanti, J. Pécaut, F. A. Dunand and A. E. Merbach, Inorg. Chem., 2001, 40, 6737–6745 CrossRef.
- N. Chatterton, Y. Bretonnière, J. Pécaut and M. Mazzanti, Angew. Chem., Int. Ed., 2005, 44, 7595–7598 CrossRef CAS PubMed.
- A. Pellissier, Y. Bretonnière, N. Chatterton, J. Pécaut, P. Delangle and M. Mazzanti, Inorg. Chem., 2007, 46, 3714–3725 CrossRef CAS PubMed.
- S. L. Madsen, M. J. Welch, R. J. Motekaitis and A. E. Martell, Int. J. Rad. Appl. Instrum. B, 1992, 19, 431–444 CrossRef CAS PubMed.
- A. Schmidtke, T. Läppchen, C. Weinmann, L. Bier-Schorr, M. Keller, Y. Kiefer, J. P. Holland and M. D. Bartholomä, Inorg. Chem., 2017, 56, 9097–9110 CrossRef CAS PubMed.
- V. Kubíček, J. Havlíčková, J. Kotek, G. Tircsó, P. Hermann, É. Tóth and I. Lukeš, Inorg. Chem., 2010, 49, 10960–10969 CrossRef PubMed.
- J. Simecek, M. Schulz, J. Notni, J. Plutnar, V. Kubicek, J. Havlickova and P. Hermann, Inorg. Chem., 2012, 51, 577–590 CrossRef CAS PubMed.
- P. J. Blower, J. S. Lewis and J. Zweit, Nucl. Med. Biol., 1996, 23, 957–980 CrossRef CAS PubMed.
- P. K. Glasoe and F. A. Long, J. Phys. Chem., 1960, 64, 188–190 CrossRef CAS.
- R. Fornasier, D. Milani, P. Scrimin and U. Tonellato, J. Chem. Soc., Perkin Trans. 2, 1986, 233–237 RSC.
- M. Mato-Iglesias, A. Roca-Sabio, Z. Pálinkás, D. Esteban-Gómez, C. Platas-Iglesias, É. Tóth, A. de Blas and T. Rodríguez-Blas, Inorg. Chem., 2008, 47, 7840–7851 CrossRef CAS PubMed.
- P. Táborský, P. Lubal, J. Havel, J. Kotek, P. Hermann and I. Lukeš, Collect. Czech. Chem. Commun., 2005, 70, 1909–1942 CrossRef.
- M. Försterová, I. Svobodová, P. Lubal, P. Táborský, J. Kotek, P. Hermann and I. Lukeš, Dalton Trans., 2007, 535–549 RSC.
- M. Kývala and I. Lukeš, Presented in part at the International Conference Chemometrics ‘95, Pardubice, Czech Republic, 1995.
- M. Kývala, P. Lubal and I. Lukeš, Presented in part at the Spanish-Italian and Mediterranean Congress on Thermodynamics of Metal Complexes (SIMEC 98), Girona, Spain, 1998.
- S. J. Coles and P. A. Gale, Chem. Sci., 2012, 3, 683–689 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available. CCDC 1549314 ([Ga(Dpaa)]), 1530703 ([Ga(Dpaa.ga)]) and 1530704 (H4Dpaa.ga). For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c7dt03398b |
| ‡ H4Dpaa.ga crystal structure data: refined formula C19H19N3O8; Mr = 417.37; crystal dimensions 0.275 × 0.130 × 0.056 mm3; monoclinic; P21/n; a = 7.0852(6) Å, b = 32.290(4) Å, c = 7.6786(6) Å, β = 94.108(7)°, V = 1752.2(3) Å3; Z = 4; ρcalcd = 1.582 g cm−3; μ = 0.125 mm−1; Mo Kα radiation λ = 0.7173 Å; T = 150 K; 2θmax = 51.10°; no. of reflections measured (independent) = 9845 (3229); Rint = 0.0571; R = 0.0401; wR2 = 0.0612; ρmax/min = 0.172/−0.197 e Å−3; data collected using a Stoe IPDS2 diffractometer; structure solved by routine dual space methods and refined against all observed F2 values. |
§ GaDpaa crystal structure data: refined formula C16H20Ga1N3O10; Mr = 484.07; crystal dimensions 0.040 × 0.005 × 0.005 mm3; triclinic; P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ; a = 7.06810(10) Å, b = 8.21920(10) Å, c = 15.9952(2) Å, α = 93.6180(10)°, β = 93.7920(10)°, γ = 91.9130(10)°, V = 924.69(2) Å3; Z = 1; ρcalcd = 1.739 g cm−3; μ = 1.425 mm−1; synchrotron radiation λ = 0.6889 Å; T = 100 K; 2θmax = 72.358°; no. of reflections measured (independent) = 20 ; a = 7.06810(10) Å, b = 8.21920(10) Å, c = 15.9952(2) Å, α = 93.6180(10)°, β = 93.7920(10)°, γ = 91.9130(10)°, V = 924.69(2) Å3; Z = 1; ρcalcd = 1.739 g cm−3; μ = 1.425 mm−1; synchrotron radiation λ = 0.6889 Å; T = 100 K; 2θmax = 72.358°; no. of reflections measured (independent) = 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 701 (8614); Rint = 0.057; R = 0.0425; wR2 = 0.1062; ρmax/min = 1.606/−0.543 e Å−3; data collected at Diamond synchrotron UK, station I19; structure solved by routine dual space methods and refined against all observed F2 values. 701 (8614); Rint = 0.057; R = 0.0425; wR2 = 0.1062; ρmax/min = 1.606/−0.543 e Å−3; data collected at Diamond synchrotron UK, station I19; structure solved by routine dual space methods and refined against all observed F2 values. |
¶ GaDpaa.ga crystal structure data: refined formula C19H24Ga1N3O12.5; Mr = 564.12; crystal dimensions 0.060 × 0.005 × 0.005 mm3; orthorhombic; Pccn; a = 20.7850(12) Å, b = 30.2224(18) Å, c = 7.2085(6) Å, V = 4528.2(5) Å3; Z = 8; ρcalcd = 1.646 g cm−3; μ = 1.186 mm−1; synchrotron radiation λ = 0.6889 Å; T = 100 K; 2θmax = 49.67°; no. of reflections measured (independent) = 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 211 (4251); Rint = 0.2109; R = 0.0999; wR = 0.2359; ρmax/min = 2.429/−1.265 e Å−3; disordered water was modelled using the SQUEEZE routine; data collected at Diamond synchrotron UK, station I19; the crystal suffers from radiation damage but structure solution and refinement were routine; structure solved by routine dual space methods and refined against all observed F2 values. 211 (4251); Rint = 0.2109; R = 0.0999; wR = 0.2359; ρmax/min = 2.429/−1.265 e Å−3; disordered water was modelled using the SQUEEZE routine; data collected at Diamond synchrotron UK, station I19; the crystal suffers from radiation damage but structure solution and refinement were routine; structure solved by routine dual space methods and refined against all observed F2 values. |
| This journal is © The Royal Society of Chemistry 2017 |