A high-temperature flexible supercapacitor based on pseudocapacitive behavior of FeOOH in an ionic liquid electrolyte†
Abstract
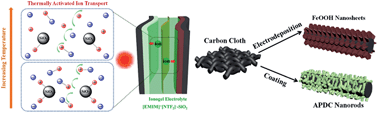
Although flexible all-solid-state supercapacitors (f-SSCs) have been receiving much attention as promising flexible energy storage devices, most of them cannot operate at high temperatures due to the volatility or flammability of currently used aqueous and organic electrolytes. Here, we report an ionic liquid (IL) gel-based asymmetric supercapacitor having excellent heat-resistant performance and flexibility. To this end, low-cost γ-FeOOH is firstly electrodeposited on carbon cloth, and its pseudocapacitive behavior in a typical IL is investigated through an electrochemical quartz crystal microbalance (EQCM) for the first time. The results show that the pseudocapacitance mainly originates from a diffusion-controlled insertion process of the cations. By taking advantage of the prominent pseudocapacitance of γ-FeOOH, as well as excellent characteristics of IL gel electrolytes (thermostability, non-flammability, chemical inertness and wide potential), an advanced high-temperature f-SSC is fabricated by using γ-FeOOH as the anode and porous N-doped activated carbon as the cathode. The f-SSC exhibits outstanding electrochemical performance at elevated temperatures, and can achieve a maximum volumetric energy density of 1.44 mW h cm−3 (based on the whole device volume) at 200 °C. Moreover, it is able to maintain a stable energy-storage ability during the bending process even at 180 °C, providing the highest reported temperature for flexibility tests in f-SSCs to date.

 Please wait while we load your content...
Please wait while we load your content...