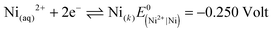The effects of problem-based learning (PBL) on the academic achievement of students studying ‘Electrochemistry’
Tuğçe
Günter
*a and
Sibel Kılınç
Alpat
b
aDepartment of Medical Services and Techniques, Ahmet Erdogan Vocational School of Health Services, Bulent Ecevit University, 67600 Esenköy, Kozlu, Zonguldak, Turkey. E-mail: tugcegunter85@gmail.com
bDepartment of Chemistry Education, Buca Education Faculty, Dokuz Eylul University, Izmir, Turkey. E-mail: skilincalpat@gmail.com
First published on 7th October 2016
Abstract
This study investigates the effects of problem-based learning (PBL) on students' academic achievements in studying ‘Electrochemistry’ within a course on Analytical Chemistry. The research was of a pretest–posttest control group quasi-experimental design and it was conducted with second year students in the Chemistry Teaching Program at the Faculty of Education at Dokuz Eylul University. The topic was taught with PBL in the experimental group and with expository teaching strategies in the control group. The ‘Chemistry achievement test (CAT)’ was applied as the pre-test and post-test in both groups. Post-test scores from the CAT demonstrated that there was a significant difference between the academic achievements of the two groups in favour of the experimental group (U = 8.500, p < 0.05). After using PBL for a period, eleven open-ended questions related to the topic were asked to both groups. The results of these questions were categorized and the scores obtained from the categories were evaluated using content analysis. The results of the scores obtained from the open-ended questions showed that the students in the experimental group were able to make statements containing fewer misconceptions and also had a better understanding of this topic when compared to the students in the control group (U = 2.500, p < 0.05). The correlation between the scores in the CAT post-test and the scores obtained from the open-ended questions of the experimental and control groups was also examined. The Spearman correlation coefficients were found to be 0.907 and 0.595 for the experimental and the control group, respectively. This finding indicated that there was a positive and very high level of statistically significant correlation between the scores in the CAT post-test and the scores obtained from the open-ended questions of the students in the experimental group. Therefore this finding also supports the conclusion that the students in the experimental group were better able to understand the topic and its structure compared to the students in the control group. At the end of the application, the results of semi-structured interviews carried out showed that students in the experimental group had positive opinions regarding PBL.
Introduction
Today, various active learning methods are used in the teaching process in order to produce individuals who are creative, can adapt themselves to team work and who are able to find solutions to the problems of daily life by using the knowledge and skills gained. Problem-Based Learning (PBL) is among these methods, with the intention that students gain critical thinking skills, analyse the problems of daily life, access and use appropriate learning sources, organize information into meaningful conceptual frameworks, work in cooperation in teams and small groups, and demonstrate effective communication skills by conveying information to others (Duch et al., 2001). The application of problem-based learning is based on three fundamental characteristics: ‘A quality problem’, ‘Self-learning’ and ‘Collaborative Learning’. In PBL, students gain problem-solving skills, work in collaborative groups, and increase their self-confidence and this method provides an impetus to students for self-learning and permanent learning (Hung et al., 2008). Working in cooperative groups during the teaching process also improves the achievement of students (Johnson et al., 1991). Problems related to everyday life and semi-structured problems are posed due to the fact that they help any knowledge gained to become permanent. In addition, students' problem-solving and higher-order cognitive skills, self-directed learning and cooperative working skills, self-perception and self-confidence increase (Hung et al., 2008). In recent years, PBL has been applied in the science education field in countries worldwide, including Turkey. Previous work in this field has shown that PBL is an effective teaching approach in learning chemistry involving difficult topics and requiring problem-solving and application, and it is also effective in improving academic achievement. In their study, Gallagher et al. (1992) showed that problem-based learning was an effective method in science education for improving students' problem-solving processes and skills. PBL applied in certain topics of biochemistry and bioanalytical chemistry was shown to be effective in improving students' higher-order cognitive learning and academic achievement (Dods, 1996; Grover, 2004; Osgood et al., 2005; Mabrouk, 2007). PBL applied in analytical chemistry and analytical chemistry laboratory courses was found to be an effective strategy for improving students' problem-solving, scientific research, self-learning and self-assessment skills and increasing students' learning and deep understanding of topics (Belt et al., 2002; Yoon et al., 2014; Jansson et al., 2015). Selco et al. (2003) found that students were able to prepare high-quality reports thanks to a laboratory application involved in designing a real-world problem about sea water analysis in the general chemistry course. In another study on ionization of water and acid and base strength in the general chemistry course, it was shown that PBL was effective in learning the concepts in chemistry education and increased students' positive opinions after the application (Tarhan and Şeşen, 2013). It was suggested in some studies that real-life cases had to be used to allow students to deal with problems and it was also stated that PBL improved students' communication, cooperative working, access to and use of information skills and their motivation (Marks and Eilks, 2010; Lopes et al., 2011; Festus and Ekpete, 2012). It was observed that problem-based laboratory education improved students' problem-solving and metacognitive skills and provided them with a more comprehensive experience compared to conventional education (Urena et al., 2012; Laredo, 2013).In the related literature, the topics of ‘Chemical Equilibrium’, ‘Mole Concept’ and ‘Oxidation-Reduction Reactions’ have been included among those chemistry topics most difficult to understand (Akram et al., 2014). ‘Electrochemistry’, based on oxidation–reduction (redox) reactions, is a topic found across courses in Analytical Chemistry, Physical Chemistry and General Chemistry in chemistry degrees at the undergraduate level and has a wide range of applications. The topic of ‘Electrochemistry and Electrochemical Analysis’, falling within the scope of Analytical Chemistry – the field of chemistry which produces concepts, principles and strategies and applies them to real examples in order to determine the properties of substances, has an important role because of its usage in other various fields of science (Serjant, 1984). An Analytical Chemistry course aims for students to become familiar with analytical techniques, to become self-learners, acquire self-assessment skills and critical thinking skills oriented towards analytical chemistry problems and also aims to encourage them to ‘think like scientists’ (Wright et al., 1998; Williamson and Rowe, 2002). Students must be able to follow and interpret new publications related to Analytical Chemistry and find answers to analytical problems or to open-ended problems which are encountered suddenly (Bransford et al., 2000). There are numerous studies in the literature which use problem-based learning to improve students' academic achievements in chemistry education (Ying, 2003; Schaber et al., 2011; Chamely-Wiik et al., 2012; Flynn and Biggs, 2012; Hicks and Bevsek, 2012; Li-Teh and Yakob, 2013). On the other hand, there are only a limited number of studies which use problem-based learning to improve students' academic achievements in electrochemistry specifically, and these studies generally deal with electrochemistry at the level of general chemistry (Ying, 2003; Tarhan and Acar, 2007; Sağır et al., 2009; Aydoğdu, 2012). When the studies in the relevant literature are taken into account, it can be considered that the implementation of PBL could be effective for Analytical Chemistry courses especially in teaching the topic of electrochemistry.
The aim of this study was to answer the question: “What is the effect of problem-based learning on students' academic achievements when it is implemented for teaching electrochemistry within a course on Analytical Chemistry in an undergraduate program of Chemistry Teaching?” Three sub-questions were as follows: “Is there any significant difference between the experimental and control groups relating to scores obtained from open-ended questions related to the topic?”; “Is there any significant difference between the experimental and control groups relating to the scores obtained from the Chemistry achievement post-test, and the scores obtained from open-ended questions?”, and “Is there any significant difference between the experimental and control groups relating to opinions obtained from semi-structured interviews?”
Method
Research design
The research had a pretest–posttest control group quasi-experimental design in order to determine the effect of PBL applied in the ‘Electrochemistry’ topic of the Analytical Chemistry course on students' academic achievement. Subjects were randomly assigned to the experimental and control groups (Büyüköztürk et al., 2008). Table 1 shows the Experimental Procedure process:| Groups | Pre-test | Process | Post-test | Students' opinions |
|---|---|---|---|---|
| Experimental | Chemistry achievement test (CAT) | Problem-based Learning (PBL) | Chemistry achievement test (CAT) | Semi-structured interview |
| Open-ended questions | ||||
| Control | Chemistry achievement test (CAT) | Expository teaching strategies | Chemistry achievement test (CAT) | Semi-structured interview |
| Open-ended questions |
The study group
The study group of this research consisted of second year students (N = 15) who were taught in the Department of Chemistry Education, Secondary Science and Mathematics Education, Faculty of Buca Education, Dokuz Eylul University. Problem-based learning was implemented in the spring semester of the 2013–2014 academic year. The permit for the implementation of the research was obtained from the Department of Chemistry Education, Buca Education Faculty, Dokuz Eylul University with the petition dated 19 March 2014 and numbered 53321635.B.8.6.YOK.2.DE.F.14.0.16.00/74. All the students were asked to participate in the study and gave their informed consent before participating in research and the researchers use individuals' existing data for research purposes. Students had a right to choose whether to contribute to the study or not. All the students were informed that their test results wouldn't count in their analytical chemistry assessment, they wouldn't be exposed to any chemical substances and there also wouldn't be any health risks in this study. All these ethical precautions were appropriate as the research activity was considered to be a useful learning opportunity for the students and that all those participating in this study were doing in full knowledge of their involvement (British Education Research Association; as cited in Taber, 2014). Second grade students attending the chemistry teaching program were randomly divided into two groups, the control group (N = 7) and the experimental group (N = 8). There is an additional potential ethical concern about educational research designs which divide a cohort of students to compare two treatments in this way. We return to this issue in our discussion, later in the paper. ‘Electrochemistry’ as a topic was taught with problem-based learning for a three week period in the experimental group by the researcher, while the same topic was taught according to expository teaching strategies for the same period of time in the control group by the same researcher. So the implementation of PBL and the expository teaching strategy was carried out by the same researcher and the researcher took into consideration that teaching in both groups was not deliberately restricted and in the educational process all students included discussion activities and could state their own ideas (Taber, 2014). It was specified as a criterion that students in the study group must be second-year students taking the Analytical Chemistry course for the first time. The low number of the study group constitutes a limitation for the study.Data collection tools
Data analysis
The analysis of data obtained from the CAT was made by the Mann–Whitney U Test and Wicoxon Sign Test, non-parametric tests, because the data did not show normal distribution and homogeneous variance and also due to the small number of individuals involved. On the basis of the Mann–Whitney U Test, CAT pre- and post-test scores of the experimental and control groups, it was determined whether there was a significant difference between them. Additionally, the Wilcoxon Sign Test was utilized to determine whether there was any change between the CAT pre-test and post-test scores within each group (Büyüköztürk, 2007; Salkind, 2007; Alpar, 2012).The students' responses to the open-ended questions were classified and scored in the content analysis by categorizing them as ‘Clear Understanding’ (CU), ‘Partial Understanding’ (PU), ‘Partial Understanding with Specific Misconception’ (PUSM), ‘Specific Misconception’ (SM) and ‘Incomprehension/Pointless’ (I) (Marek, 1986; Abraham et al., 1992; Ayas and Özmen, 1995). The frequencies and percentages of each response were determined within these categories. In addition, the Mann–Whitney U Test was used with the aim of determining whether there was a significant difference between the experimental and control groups according to the scores obtained from the open-ended questions. In this study, the categories, their descriptions and the scores used in the content analysis of the answers given by the students to these questions are given in Table 2. As seen in Table 2, the highest possible score that could be obtained from the eleven open-ended questions regarding the topic is 44. The Spearman correlation coefficient was calculated in order to determine whether there was a significant difference between the scores obtained from the CAT-post test of experimental and control groups and the scores obtained from the open-ended questions of these groups. Content analysis was also used to analyze semi-structured interview questions.
| Categories (Abbreviation) | Description of Categories | Score |
|---|---|---|
| Clear Understanding (CU) | The answer to the question is accurate. | 4 |
| Partial Understanding (PU) | The answer to the question does not include all aspects of the desired response, but the response contains some aspects. | 3 |
| Partial Understanding with Specific Misconception (PUSM) | This is an understanding in which the answer is correct but its explanation is wrong or in which the answer is wrong but its explanation is correct. | 2 |
| Specific Misconception (SM) | This is an answer which it is not true scientifically but is considered true by the student. | 1 |
| Incomprehension/Pointless (I) | The answer is not related to the question. | 0 |
Creating a problem-based learning scenario
The views of four experts and students were sought in order to prepare a scenario and the tutor's questions regarding this scenario were prepared by Tunç (2015). There were four sessions in total for this prepared scenario, which was entitled ‘Wow, Emre, Wow!’ (Appendix 1). The first three sessions consisted of four sections in total and the last session consisted of one section. The scenario is about a five-year-old boy called Emre who poisoned himself by eating matches and concealing this from his family. The scenario was inspired by a real-life case of a teenager who committed suicide by eating matches (Ünver et al., 2005). It also drew on the current news regarding this event (Güncel Haber, 2013, 23 January). The use of this scenario was intended to increase the interest in the clinical findings and the biochemical data provided in the scenario. By using the scenario as a teaching aid, two separate activities were prepared from it. These were intended for (i) the consolidation of an event-oriented calculation of electrode potential in an electrochemical cell and (ii) for finding electrode potential on the basis of solubility product constant (Appendices 2 and 3).The application of problem-based learning
Information about problem-based learning and its process was given to the students in the experimental group prior to application. PBL was implemented for 18 course hours in a three-week period (Table 3). At the beginning of each session, students were asked to identify a problem and make a hypothesis on the basis of a specific aspect of the scenario distributed and discussed. After each session, students revised their hypothesis, determined learning objectives and which new information they needed to learn, and tried to find a solution to the problem. In the relevant sections of each education session, the time required was granted to the students in order to hold group discussions and establish appropriate hypotheses. In this process, students were under the guidance of the researcher. Each of the sessions was recorded by video.| Implementation plan | Course hours |
|---|---|
| Giving information about PBL | 1 |
| PBL Education session I | 3 |
| PBL Education session II | 4 |
| PBL Education session III | 4 |
| PBL Education session IV | 4 |
| Completing the PBL Education sessions and General Review | 2 |
Learning objectives of the scenario in accordance with the sessions
The application of expository teaching strategies
In the control group, sub-topics included in the ‘Electrochemistry’ topic were presented using expository teaching strategies and a question–answer technique. The same questions posed by tutors and sub-topics in the ‘Electrochemistry’ topic in the PBL approach were also solved in the control group. The ‘Electrochemistry’ topic was taught over 18 course hours by the same researcher, as in the PBL teaching.Results and discussion
The results of the education sessions
In the experimental group, the participation of students in each education session and their hypotheses about the problem and whether they would be able to achieve the learning objectives for each session were recorded on video and also examined within the session. The students established hypotheses both individually and in groups, carried out the research, engaged in discussions and proposed solutions under the guidance of the tutor in order to solve the problem at hand.• Emre could have become sick because of the things he had eaten or drunk.
• He could have been affected by any chemicals in the toys he played with.
• He could have had any chronic or acute diseases.
• He could have developed a cold.
• He could have been poisoned.
• He could have swallowed something or been able to eat and drink something from the shopping bags he was playing with/without anyone knowing.
They also tried to determine which new information they would need in order to solve the related problem.
In the second section of Education session I, in light of the new information, students disproved most of their hypotheses developed in the first section, they developed new hypotheses and indicated Emre's blood and urine results needed to be looked at in order to evaluate the remaining hypotheses in the next section.
In the third section of Education session I, in line with the new information, students evaluated the hypotheses and investigated the normal values of respiratory rate, blood pressure, pulse and fever in children and adults. They discovered that Emre's respiratory rate and pulse were at a high level, that he appeared to have a fever and that his blood pressure was at the normal level. Some students also extrapolated that Emre had renal failure.
In the fourth section of Education session I, students researched the meaning of the results of the analyses given in the table and they discussed the reasons why specific values were high or low with their classmates. Some students concluded that Emre had methemoglobinemia and metabolic acidosis and that cyanosis had occurred as a result of methemoglobinemia on the basis of the low pH level, the ion of bicarbonate, pCO2 and the high level of pO2 and methaemoglobin in the analysis of blood gas. They found that the colour of the blood taken from Emre was ‘chocolate-brown.’ The result of renal failure, extrapolated by several students in the third section, was also confirmed in this section. The tutor asked the students about the reasons for the peaked T waves in the electrocardiogram results and for the high level of the ions of potassium in the biochemistry of blood. As a result of their investigations, students found that Emre had hyperkalemia (hyperpotassemia) due to the high level of the ions of potassium and that therefore the peaked T waves occurred in the electrocardiogram at the same time.
At the end of Education session I, students achieved the required aims through group discussions and investigations. Students made hypotheses about how Emre could have become poisoned and become ill as a result of eating or drinking something in the shopping bags.
In the second section of this session, the students investigated how gastric lavage, activated charcoal and hyperbaric oxygen therapy are carried out, for what purposes the medications concerning aforementioned diseases are used, and what active substance had caused Emre's poisoning. As a result of their investigations they learnt the purposes for which the medication for hyperkalemia and acidosis is used and also found out that hyperbaric oxygen therapy is administered in order to eliminate breathing difficulties (Ünver et al., 2005). But the students were not yet able to discover the active substance causing these diseases and poisoning.
In the third section of this session, students decided that Emre could have been poisoned by eating matches and they then investigated the content of matches. As a result of their investigations, they concluded that there was potassium chlorate (55%), potassium dichromate, carboxymethyl cellulose, gelatin, granulated glass, zinc oxide, iron oxide and starch in the head of match and that there was also monoammonium phosphate in the stem of the match (Türkmen, 2006).
In the fourth section of this session students investigated which substances in the matches caused Emre's poisoning and how this substance had caused the diseases of methemoglobinemia and hyperkalemia. Some of the students expressed the view that the concentration of potassium chlorate in the matches was higher than the other substances and this substance could cause poisoning and related diseases. They discovered what the active substance was and what kind of diseases it caused through group discussions.
In the second section of this session, the information that the ion of chlorate (ClO3−) can convert the ion of iron (+2) to the ion of iron (+3) was given to the students. In light of this information, students investigated what kind of reaction could occur and expressed the view that the ion of chlorate (ClO3−) was reduced and the ion of iron(II) was oxidized and a redox reaction occurred as below:
| ClO3(aq)− + 6Fe(aq)2+ + 6H(aq)+ ⇌ Cl(aq)− + 3H2O(s) + 6Fe(aq)3+ |
The researcher asked the students questions with the intention that students understand the redox reactions, the oxidation and reduction events and the oxidization and reduction of chemical species better. In this manner, students learnt about the redox reactions, the oxidation and reduction events, the oxidization and reduction of chemical species and how the redox reactions can be equalized in an acidic or a basic environment.
In the third section of this session students learned the calculation of cell potential, Gibbs free energy and equilibrium constant by using standard electrode potentials. They could categorize the electrochemical cell types and could explain the events occurred when the redox reaction reaches the equilibrium.
In this session two different activities were given to the students. In association with the first activity, students were able to write the net reaction about the formation of Fe2O3 (rust) by using related half-reactions, could make a decision about the electrochemical cell type by calculating the cell potential and could find the redox reaction of hydroxylamine with the ion of iron(III). In addition, the related issues were reinforced through the tutor's questions. In the second activity students tried to calculate the electrode potential of the redox reaction as below:
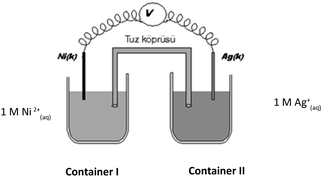
| PbI2(k) + 2e− ↔ Pb(k) + 2I(aq)− |
In the fourth section of Education session III students were able to explain the selection of the indicator in redox titrations, the requirement of titration of two analyte pairs with a single titrant and how the electrode potential of the equivalence point can be calculated. In this way, they were able to reach the expected learning objectives.
| Groups | N | Median | Min–Max | U | p |
|---|---|---|---|---|---|
| Experimental | 8 | 23 | 14–28 | 14.000 | 0.100 |
| Control | 7 | 16 | 10–26 | ||
| Total | 15 |
| Groups | N | Median | Min–Max | U | p |
|---|---|---|---|---|---|
| Experimental | 8 | 68 | 58–76 | 8.500 | 0.022 |
| Control | 7 | 58 | 40–68 | ||
| Total | 15 |
| Groups | Pre-test | Post-test | z | p |
|---|---|---|---|---|
| Experimental | 23 (14–28) | 68 (58–76) | −2.524 | 0.012 |
| Control | 16 (10–26) | 58 (40–68) | −2.371 | 0.018 |
| Groups | Median | Min–Max | z | p |
|---|---|---|---|---|
| Experimental | 1.87 | 1.07–3.86 | −0.694 | 0.536 |
| Control | 2.38 | 1.54–3.43 |
Considering the values in Tables 4 and 5, it can be seen that there was no significant difference between the experimental and control groups according to the scores of the CAT pre-test (U = 14.000, p > 0.05). On the other hand, in accordance with the scores of the CAT post-test, it can also be seen that there was a significant difference between both groups in favour of the experimental group (U = 8.500, p < 0.05). According to Table 6, changes between the CAT pre-test and post-test scores in the experimental and control groups were found to be statistically significant (z = −2.524, p = 0.012 and z = −2.371, p = 0.018, respectively). There was a significant increase in the scores of both groups. Furthermore, according to Table 7, it could be observed that the increase rates of pre- and post-test scores in both groups were statistically the same (z = −0.694, p = 0.536). This finding also showed that there was no statistically significant difference between the PBL and the expository teaching strategy. The PBL and the expository teaching strategy increased students' scores at the same rate. Observing a significant difference in favour of the experimental group in the CAT post-test scores suggested that problem-based learning was an effective tool to improve the academic achievement of students. Studies on electrochemistry and the other chemistry topics in the literature have also supported these findings, namely that PBL has an effective role in improving students' academic achievement (Şenocak et al., 2007; Tarhan et al., 2008; Oezden, 2009; Özeken and Yıldırım, 2011; Buyukdokumacı, 2012; İnce Aka, 2012; Li-Teh and Yakob, 2013; Jansson et al., 2015).
| Questions | Experimental group | Control group | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| % Distributions according to categories | % Distributions according to categories | |||||||||
| CU | PU | PUSM | SM | I | CU | PU | PUSM | SM | I | |
| (1) What do you understand by ‘redox reaction’? | 100 | — | — | — | — | 100 | — | — | — | — |
| (2) What are oxidation and reduction? | 100 | — | — | — | — | 100 | — | — | — | — |
| (3) Consider an electrochemical cell. What comes into your mind when “Anode” and “Cathode” are mentioned? | 83.4 | — | — | 8.3 | 8.3 | 100 | — | — | — | — |
| (4) What do you know about the relationship between Gibbs free energy and electrode potential? | 100 | — | — | — | — | 57.1 | 14.3 | — | 14.3 | 14.3 |
| (5) What do you know about the difference between cell potential and standard electrode potential? | 12.5 | 62.5 | — | — | 25.0 | 14.3 | 14.3 | — | 14.3 | 57.1 |
| (6) What do you know about the SHE of reference electrodes? | 75.0 | — | — | — | 25.0 | 28.6 | 28.6 | — | — | 42.8 |
| (7) What do you know about the relationship between equilibrium constant (Kd) and electrode potential? | 87.5 | — | — | — | 12.5 | 28.6 | 57.1 | — | — | 14.3 |
| (8) What do you know about the relationship between solubility product constant (Ksp) and electrode potential? | 80.0 | — | 20.0 | — | — | — | 14.2 | 42.9 | — | 42.9 |
| (9) What do you understand about how you find the equivalence point electrode potential in redox titrations? | 62.5 | 37.5 | — | — | — | — | 14.3 | — | — | 85.7 |
| (10) What do you understand about whether an environment containing two analyte pairs can be titrated by one titrant simultaneously? | 87.5 | — | — | — | 12.5 | 57.1 | — | — | — | 42.9 |
| (11) What do you know about how the indicator in redox titrations is determined? | 100 | — | — | — | — | 42.9 | — | — | — | 57.1 |
| Total | 81.44 | 8.25 | 2.06 | 1.03 | 7.22 | 48.05 | 12.98 | 3.90 | 2.60 | 32.47 |
| CU | PU | PUSM | SM | I | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| f | % | f | % | F | % | f | % | f | % | |
| Experimental group | 79 | 81.44 | 8 | 8.25 | 2 | 2.06 | 1 | 1.03 | 7 | 7.22 |
| Control group | 37 | 48.05 | 10 | 12.98 | 3 | 3.90 | 2 | 2.60 | 25 | 32.47 |
| Groups | N | Median | Min–Max | U | p |
|---|---|---|---|---|---|
| Experimental | 8 | 93.50 | 71–100 | 2.500 | 0.003 |
| Control | 7 | 61.00 | 36–84 | ||
| Total | 15 |
| The explanation of the open-ended questions | Students' responses |
|---|---|
| After examining the students' responses for both groups, it was observed that all students in both groups gave answers which fell in the category of ‘Clear Understanding’ to the first and second questions. These were about redox reactions and the concepts of oxidation/reduction respectively: |
“Redox reactions are the oxidation and reduction reactions. For example; the oxidation of Fe2+ion to Fe3+ion or the reduction of Fe3+ion to Fe2+ion.” (EG-S1) (CU)
“Redox reactions are the oxidation and reduction reactions. These reactions are based on electron exchange reactions.” (CG-S7) (CU) “The oxidation reaction is an oxidation of Fe2+ion to Fe3+ion. It loses an electron. In this case, losing the electron is called oxidation; gaining the electron is called reduction. For example, chlorate ions are reduced to chloride ions in the scenario.” (EG-S1) (CU) “Giving up an electron is called oxidation; gaining an electron is called reduction.” (CG-S5)(CU) |
| Examining the responses of students in the experimental group to the third question, it was determined that most students defined the concepts of ‘anode’ and ‘cathode’ correctly. However, one student referred to ‘the salt bridge’ and made a mistake about the flow of electrons in the salt bridge when he/she defined the concepts of anode and cathode and one student did not understand the question. In the literature, it can be seen that there are also misconceptions about the electron flows in the salt bridge (Allsop and George, 1982; Sanger and Greenbowe, 1997; Rogers et al., 2000; Yılmaz et al., 2002; Cullen and Pentecost, 2011; Tasdelen, 2011; Akram et al., 2014; Brandiet and Bretz, 2014). It was seen that all students in the control group gave statements within the category of ‘Clear Understanding’ for the same question. |
“The anode is the site of oxidation, while the cathode is the site of reduction.” (EG-S3) (CU)
“When you say anode and cathode, the salt bridge comes to my mind. Electrons flow from the anode to the cathode.” (EG-S8) (SM) “When you say that, the electrochemical cell, the salt of bridge, Ecell, Ksp, Kd, the schematic representation of cells, galvanic and electrolytic cells come to my mind.” (EG-S1) (I) “Oxidation occurs at the anode, while reduction occurs at the cathode.” (CG-S4) (CU) |
| Examining the responses of the students in the experimental and control groups to the fourth question, it was seen that all students in the experimental group and four students in the control group gave answers with regard to determining electrode potential according to whether Gibbs free energy is greater or less than zero. However, for this fourth question, one student in the control group gave an answer only on whether the reaction occurring spontaneously in accordance with Gibbs free energy was greater or less than zero, without mentioning electrode potential. This therefore belonged to the category of ‘Partial Understanding’. It was also observed that there was a misconception concerning this issue in the response of one student who suggested that if the electrode potential was lower than zero, the reaction occurred spontaneously. One other student didn't answer the question. This finding is also supported by several studies in the literature. Akram et al. (2014) concluded that there were misconceptions by students in interviews concerning the relationship between ‘occurring spontaneously’ and the electrode potential. |
“ΔG is Gibbs free energy.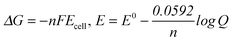 ; In the event of ΔG < 0 Ecell> 0; the type of electrochemical cell is a galvanic cell, while in the event of ΔG > 0, Ecell< 0; the type of electrochemical cell is an electrolytic cell. In a galvanic cell, the reactions take place spontaneously, while the reactions do not take place spontaneously in an electrolytic cell.” (EG-S8) (CU) ; In the event of ΔG < 0 Ecell> 0; the type of electrochemical cell is a galvanic cell, while in the event of ΔG > 0, Ecell< 0; the type of electrochemical cell is an electrolytic cell. In a galvanic cell, the reactions take place spontaneously, while the reactions do not take place spontaneously in an electrolytic cell.” (EG-S8) (CU)
“In the event of ΔG < 0, Ecell> 0; the type of electrochemical cell is a galvanic cell and the reaction takes place spontaneously. In the event of ΔG > 0, Ecell< 0; the type of electrochemical cell is an electrolytic cell and the reaction doesn't take place spontaneously.” (CG-S4) (CU) “In the event of ΔG < 0; the reaction occurs spontaneously. In the event of ΔG > 0; the reaction does not occur spontaneously. The spontaneous occurrence of the reaction shows that the type of electrochemical cell is a galvanic cell. On the contrary, when the reaction does not occur spontaneously, this shows that the type of the electrochemical cell is an electrolytic cell. In this situation, it needs to get outside power so the reaction can take place.” (CG-S3) (PU) “When the electrode potential is negative, the reaction occurs spontaneously.” (CG-S1) (SM) “Very good question. I do not remember at the moment.” (CG-S2) (I) |
| For the fifth question, it was seen that the percentage of responses of the students in the control group in the ‘Incomprehension’ category was higher than the percentage of responses of the students in the experimental group. There were also responses in the category of ‘Specific Misconceptions’ in the control group. In the control group, four students didn't give an answer to the question and one student also made statement including a misconception because he/she expressed wrongly that the difference between cell potential and standard electrode potential was to do with galvanic and electrolytic cells. |
“Standard electrode potential occurs in the standard conditions (25 °C); while electrode potential occurs in normal, non-standard conditions and in one of them it is processed by taking the reference of H+; and in the other one, it is processed without taking the reference of H+.” (EG-S4) (CU)
“Standard electrode potential occurs under standard conditions; while electrode potential is also the collection of oxidation and reduction potentials of two half reactions.” (CG-S2) (CU) “E0of anode half-cell and E0of cathode half-cell are written into the half reactions and gathered side by side. Therefore we have achieved a standard electrode potential. From there, we can find the electrode potential with Nernst equation. When we get the equation with Ecell= Ecathode− Eanodewe find the electrode potential. But we write as reduction potentials in the anode and cathode. Therefore we find electrode potential. “Standard electrode potential is the electrode potential of hydrogen. In fact, they measured hydrogen and agreed it to be zero. They measured the electrode potentials of the others by taking hydrogen as a reference.” (CG-S7) (PU) “The difference between standard electrode potential and electrode potential is galvanic and electrolytic. I remember it as being occurrence spontaneously and non-occurrence spontaneously.” (CG-S5) (SM) “I don't know. Let's pass on this one.” (EG-S3) (I) |
| For the sixth question, it was observed that the percentage of the ‘Incomprehension’ category for students in the control group was higher than for the students in the experimental group and also that there were no answers in the ‘Specific Misconception’ category in either group. The majority of students in the experimental group could define the SHE reference electrode exactly and state that its value of standard electrode potential is 0.00 volts. For the same question, it was observed that the percentage of responses in the category of ‘Clear Understanding’ was also lower for the students in the control group and that they could not define the SHE reference electrode exactly: |
“It is the standard hydrogen reference electrode. Its electrode potential is 0.00 volts. For this reason, we can find electrode potentials of the other substances. It is used as the reference.” (EG-S5) (CU)
“In SHE, there was HCl solution under it. We made it with platinum. This was due to release hydrogen gas. Due to the fact that electrode potential was 0.00 volts, it was taken as the reference.” (CG-S6) (CU) “I do not know. Nothing comes to mind.” (CG-S4) (I) “There was H2gas. I remember it. There was a figure similar to the salt bridge, we performed our processes over it. There was a reaction of H2gas. I do not remember the electrode potential. Let's pass on this question.” (CG-S5) (PU) |
| For the seventh question, the majority of the students in the experimental group gave responses in the ‘Clear Understanding’ category, and the majority of the students in the control group gave responses in the ‘Partial Understanding’ category. Although the proportion was low, there were responses for students in both groups in the ‘Incomprehension’ category as regards the relationship between equilibrium constant and electrode potential. Only one student in the experimental group was not able to describe the relationship between equilibrium constant (Kd) and electrode potential correctly, getting it confused with finding the electrode potential of the equivalence point in the redox titrations. Again, one student in the control group also gave only the formula of the equilibrium constant, showing that the student had not understood the question: |
“In the Nernst equation of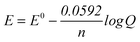 ; we write Kdinstead of Q. We first write cathode and then the anode separately in the Nernst equation. In balance, Ecell= Ecathode= Eanodeequalizes to zero. In this case, we could find Kdby equalizing together the Nernst equations of cathode and anode.” (EG-S1) (CU) ; we write Kdinstead of Q. We first write cathode and then the anode separately in the Nernst equation. In balance, Ecell= Ecathode= Eanodeequalizes to zero. In this case, we could find Kdby equalizing together the Nernst equations of cathode and anode.” (EG-S1) (CU)
“In the Nernst equation, we can find it from the part of logarithm.” (CG-S2) (PU) “We find Eanodeand Ecathodeseparately. And we accumulate both of them and find the electrode potential of equivalence point.” (EG-S6) (I) |
| For the eighth question, the majority of students in the experimental group gave answers in the category of ‘Clear Understanding’. In the control group the percentages of responses given in the categories of ‘Partial Understanding with Specific Misconception’ and ‘Incomprehension’ were high and, again, in the control group only one student gave an answer in the category of ‘Partial Understanding’. For the same question, it was observed that the percentage of ‘Incomprehension’ in the control group was higher than the related percentage in the experimental group. It was seen that the majority of students in the experimental group could explain how the solubility product constant would be found on the basis of electrode potential by giving examples and only two students gave an answer suggesting the solubility product constant could be found by writing Ksp instead of Kd in the Nernst equation, thus putting their answers in the category of ‘Partial Understanding with Specific Misconception’. It was seen that three students in the control group also gave a similar answer in the category of ‘Partial Understanding with Specific Misconception’ about being able to find the solubility product constant by writing Ksp instead of Kd in the Nernst Equation. Three students were not able to answer the question: |
“For example; AgCl ⇌ Ag++ Cl−Ksp= [Ag+][Cl−] Hence; we find that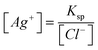 . Of course; the question must give the reduction potential of silver. And then, in the Nernst equation of . Of course; the question must give the reduction potential of silver. And then, in the Nernst equation of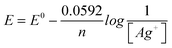 ; we write ; we write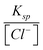 instead of [Ag+]. In the event of [Cl−] 1.00 M; that is E = E0. Therefore we find Ksp.” (EG-S5) (CU) instead of [Ag+]. In the event of [Cl−] 1.00 M; that is E = E0. Therefore we find Ksp.” (EG-S5) (CU)
“We write the Nernst equation again. From part of the logarithm, we write the concentration of product or reactant by taking 1.00 Molar. And then, we replace it in the Nernst equation.” (CG-S2) (PU) “In order to find the solubility product constant from the Nernst equation, when we write “The solubility product constant is found by the Nernst equation. We write Kspthis time.” (EG-S8) (PUSM) “In the Nernst equation, when we accumulate half reactions from part of the logarithm, we also find Ksplike the equilibrium constant.” (CG-S3) (PUSM) “I do not remember right now.” (CG-S7) (I) |
| For the ninth question, it was observed that the percentage of responses in the category of ‘Clear Understanding’ was high in the experimental group, while the percentage of responses in the category of ‘Incomprehension’ was quite high in the control group. Five students in the experimental group gave answers in the category of ‘Clear Understanding’ and three students also gave answers in the category of ‘Partial Understanding’. For the same question, the majority of students in the control group said that they could not remember how the electrode potential of the equivalence point could be found in the redox titrations. Only one student could not express exactly how the electrode potential of equivalence point could be found and gave an answer in the category of ‘Partial Understanding’: |
“Eanodeand Ecathodeare written by Eeqin the Nernst equation separately. Two of Eeqare accumulated and divided by the number of electrons. If the number of electrons is not the same, it is multiplied including Eeq. Therefore, Eeq's are accumulated and divided.” (EG-S6)(CU)
“For example; in the reaction of Fe2++ Ce4+ “When finding the electrode potential of equivalence point, mL's were given in the question. And we can find and calculate mmoles. We can find it from the formula of “We write Eanodeand Ecathode. We calculate both of them separately. The addition of these is equivalence point.” (CG-S2) (PU) “We find it by calculation. We calculate with numbers.” (CG-S1) (I) “I have forgotten at the moment.” (CG-S3) (I) “I guess it is the potential when it reaches equilibrium.” (CG-S7) (I) |
| For the tenth question, it was observed that the percentage of responses in the ‘Clear Understanding’ category was higher for the students in the experimental group than for the students in the control group, while the percentage of responses in the ‘Incomprehension’ category for the students in the experimental group was lower than for the students in the control group. It was observed that only one student in the experimental and three students in the control group could not remember that the difference between the electrode potentials of pairs should be over 0.2 volts in order to be able to titrate by one titrant simultaneously in a medium containing two analyte pairs in the redox titrations: |
“For example, if the medium containing the ions of Fe2+and Fe3+titrates by the ion of Ce4+, the difference between standard electrode potentials of iron ions would be over 0.2 volts.” (EG-S1)(CU)
“The difference between electrode potentials should be 0.2 volts or over this value.” (CG-S6) (CU) |
| For the eleventh question, it was observed that all students in the experimental group gave answers in the category of ‘Clear Understanding’, three students in the control group gave answers in the category of ‘Partial Understanding’, and four students in the control group gave answers in the category of ‘Incomprehension’. While students in the control group made scientific statements suggesting that the solution's electrode potential of the equivalence point should be equal to the indicator's electrode potential of the turning point, some students in the control group made statements unrelated to the question, suggesting that the indicator should change colour in the solution: |
“If the standard electrode potential is 1.75 volts, the electrode potential of the indicator should be close to that potential.” (EG-S2) (CU)
“If Eeqis 1.25 volts in the analyte, the electrode potential of the indicator should also be within this range.” (CG-S2) (CU) “When we drop a certain amount of titrant into a certain amount of solution, we see that the portion of solutions varies. It could change colour. This situation happens with the indicator.” (CG-S3) (I) |
Correlation between scores from the CAT post-test and the scores from open-ended questions
The results concerning the correlation between the scores from the CAT post-test and the scores obtained from open-ended questions of students in the experimental and control groups are presented in Table 12.| Interview | Experimental group | Control group | |
|---|---|---|---|
| CAT | r | 0.907* | 0.595* |
| p | 0.002 | 0.159 | |
| N | 8 | 7 |
Examining the values in Table 12, it was determined that there was a high level, positive and significant relationship between the scores from the CAT post-test and the scores obtained from the open-ended questions of students in the experimental group [r(8) = 0.907 and p < 0.01]. Considering answers given to open-ended questions by experimental group students, it was observed that student EG-S8 gave an answer in the Specific Misconception category to the third question regarding the anode and the cathode. It was found that EG-S8 incorrectly answered two questions about determining the anode and the cathode according to electrochemical cell types in the CAT test (Appendix 4). This finding shows that the student incorrectly determined electrolytic and galvanic cell types, thus the anode and cathode. Similarly, other experimental group students answered either one of the questions incorrectly or answered all of the questions correctly. EG-S6 and EG-S8 were observed to give answers in the Partial Understanding with Specific Misconception category to the eighth open-ended question regarding the relationship between electrode potential and solubility constant. A total of six questions was asked in the CAT test regarding the sub-topic of standard potentials of systems related to precipitates. EG-S8 answered only two of these questions correctly, whereas other experimental group students answered one or two of these questions incorrectly. This finding shows that EG-S8 partially misunderstood the relationship between electrode potential and Ksp (Appendix 5).
Examining the values in Table 12, it was found that there was also an insignificant relationship between the scores from the CAT post-test and the scores obtained from the open-ended questions of students in the control group [r(7) = 0.595 and p > 0.01]. To the fourth open-ended question regarding the relationship between the electrode potential and Gibbs free energy, CG-S1 gave an answer in the Specific Misconception category, CG-S2 gave an answer in the Incomprehension/Pointless category, CG-S3 gave an answer in the Partial Understanding category and other control group students gave answers in the Clear Understanding category. Three questions were asked in the CAT test regarding this topic. CG-S1 answered two of these questions incorrectly and CG-S3 answered one of these questions incorrectly, whereas CG-S2 answered all of the questions correctly. The fact that CG-S2 who gave an answer in the Incomprehension/Pointless category to the open-ended question correctly answered all of the related questions in the CAT test, whereas answers given by CG-S1 and CG-S3 to CAT test questions were partial consistent with their answers to open-ended questions supports the idea that the correlation is partial. Similarly, it was observed that CG-S7 gave an answer in the Incomprehension/Pointless category to the eighth open ended question regarding the relationship between Ksp and electrode potential. The fact that the same student correctly answered four out of six questions in the CAT test regarding the sub-topic of standard potentials of systems related to precipitates support the idea that the correlation is partial. A sample question in the CAT test regarding the topic can be seen in Appendix 6.
This finding shows that there is a high level, positive and significant relationship between the scores from the CAT post-test and the scores obtained from open-ended questions of students in the experimental group, but there is also a partial significant correlation between the scores for the related test and the scores obtained from open-ended questions for the students in the control group.
The results of content analysis of questions in semi-structured interviews
Answers given by experimental and control group students to semi-structured interview questions were analyzed with content analysis. Table 13 shows positive and negative opinions of experimental group students regarding PBL and group work. As can be seen from Table 13, experimental group students had positive opinions regarding the PBL method and stated that it led them to research, it was understandable, informative, and relevant to everyday life and allowed for permanent learning and increased their knowledge and interpretation ability. Two students, on the other hand, stated before the application that they felt as if they were passive and did not believe that they were going to learn anything at all, whereas they expressed how wrong they were after the application. This finding is supported by some studies in the literature (Lopes et al., 2011; Flynn and Biggs, 2012; Ashraf, 2013). Experimental group students had negative opinions as well, such as the view that PBL was unusual, time-consuming and exhausting. In a study conducted by Tatar (2007) on this subject, it was stated that there was a time limitation in problem-based learning and method's unusualness led to certain limitations. Some of the statements of experimental group students regarding PBL can be found below:| Positive opinions | f | % | Negative opinions | F | % | Positive opinions | f | % | Negative opinions | f | % |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sub-themes: about PBL | 33 | 80.49 | Sub-themes: about PBL | 8 | 19.51 | Sub-themes: group work | 25 | 83.33 | Sub-themes: group work | 5 | 16.67 |
| Understandable/informative | 8 | 24.24 | Unusual | 4 | 50.00 | Information exchange | 8 | 32.00 | Discussion | 2 | 40.00 |
| Completely positive | 6 | 18.18 | Time-consuming | 2 | 25.00 | Completely positive | 8 | 32.00 | Incorrect information transfer | 1 | 20.00 |
| Leading to research | 5 | 15.15 | Hesitating due to camera | 1 | 12.50 | Communicating | 3 | 12.00 | Difficulty of changing incorrect information | 1 | 20.00 |
| Application and interpretation | 5 | 15.15 | Exhausting | 1 | 12.50 | Discussion environment | 2 | 8.00 | Communication gap | 1 | 20.00 |
| Relevant to everyday life | 4 | 12.12 | Fun | 2 | 8.00 | ||||||
| Information exchange | 2 | 6.06 | Enriched hypotheses | 2 | 8.00 | ||||||
| Change in feeling of passivity | 2 | 6.06 | |||||||||
| Necessity to apply in other courses as well | 1 | 3.03 |
“I think it is good. It is easier in terms of learning. For example, we learned how to relate our knowledge to our everyday lives after the application. I think it was positive. It increases the incentive for research and also makes it easier and more understandable, examples from everyday life make it easier and more understandable. There were no negative aspects. However, it takes a lot of time.” (EG-S2)
“At first, I considered myself as a victim. But as we learnt more, we started to practice more. We did not have a foundation. We started to learn by research. I did not know anything about PBL. The more I learned, the more knowledge I had. As a matter of fact, it has both pros and cons. In terms of its negative aspects, we are used to being taught something from the beginning. Since we are not used to learning on our own, this method seemed a little too complicated. That is why I could not answer some questions. As for its positive aspects, the method is very good. It felt very good to learn something through research and practice what we learned.” (EG-S5)
Students stated that they found group work to be positive because it allowed them to exchange information, find the correct answer by providing an environment for discussion, communicate with each other and enrich their hypotheses. This finding is supported by some studies in the literature (Senocak et al., 2007; Tarhan et al., 2008; Urena et al., 2012; Tosun and Taşkesenligil, 2013). However, students stated that group work provided a disadvantage due to discussions that started among friends as a result of opposing ideas, the difficulty of changing incorrect information and possible communication gaps. It was reported in some studies in the literature that certain disadvantages were encountered during the implementation of problem-based learning due to heterogeneous groups, problems among students in the same group, inadequate knowledge acquisition, competition among students and limited time (Tosun, 2010; Tosun and Taşkesenligil, 2013). Some of the statements of experimental group students regarding group work can be found below:
“Group work has its advantage, because we exchange ideas with everyone. Everyone's ideas go through a filter. You get to hear about ideas different from yours. For example, a lot of hypotheses were suggested other than mine. I had not thought about those. It offers diversity. I have not seen any disadvantages.” (EG-S3)
“Group work is very advantageous. We exchange ideas, we laugh and have fun. These were nice things. I mean, I believe these are advantages. I do not believe these are disadvantages.” (EG-S7)
Positive and negative opinions of control group students regarding the expository teaching strategy are given in Table 14. As can be seen from Table 14, control group students had positive opinions since they were familiar with the expository teaching strategy, they were able to better understand concepts with the method presented by the teacher, teachers had positive approaches and the students did not like research. The novelty and unfamiliarity of the problem-based learning for students are among its limitations (Tatar, 2007; Tosun, 2010). For this reason, it can be said that it is only natural for control group students studying ‘Electrochemistry’ with the expository teaching strategy to have such opinions. Control group students also had negative opinions regarding the expository teaching strategy, because they found it to be inadequate, uninteresting and leading to rote learning. They also believed that it allowed for an insufficient information transfer, stated that they did not have a chance to stand out, did not understand the topic and that teachers sometimes had negative approaches. This finding is supported by some studies in the literature as well. In a study conducted by Serin (2009), it was noted that students were more enthusiastic about research, designing and performing experiments in problem-based learning, whereas students usually listened to the teacher, noted down definitions, formulae and graphics given by the teacher and solved problems requiring mathematical operations in the expository teaching strategy. In this context, it was concluded that students were more enthusiastic and curious in the problem-based learning. In addition, students have a higher level of motivation in problem-based learning (Marks and Eilks, 2010; Madhuri and Goteti, 2011; Zhang et al., 2011; Moralar, 2012; Figueira and Rocha, 2014). Content analysis results of control and experimental group students are given in Table 15.
| Positive opinions | f | % | Negative opinions | f | % |
|---|---|---|---|---|---|
| Sub-themes | 17 | 50.00 | Sub-themes | 17 | 50.00 |
| Teacher | 6 | 35.29 | Inadequate | 7 | 41.18 |
| Understandable/informative | 4 | 23.53 | Not encouraging for the course | 3 | 17.65 |
| Completely positive | 2 | 11.77 | Rote learning | 3 | 17.65 |
| Non-necessity of research | 2 | 11.77 | Not Understandable/informative | 1 | 5.88 |
| Not preferring teamwork | 1 | 5.88 | Not interesting | 1 | 5.88 |
| Catchy | 1 | 5.88 | Not leading to research | 1 | 5.88 |
| Being familiar with the method | 1 | 5.88 | Not being able to stand out | 1 | 5.88 |
| Groups | Positive opinions | Negative opinions | ||
|---|---|---|---|---|
| f | % | F | % | |
| Experimental | 58 | 81.69 | 13 | 18.31 |
| Control | 17 | 50.00 | 17 | 50.00 |
As can be seen from Table 15, the percentage of positive opinions in the experimental group (81.69%) was higher compared to the percentage of positive opinions in the control group (50.00%), whereas the percentage of negative opinions in the experimental group (18.31%) was lower compared to the percentage of negative opinions in the control group (50.00%). This finding shows that experimental group students had positive opinions regarding PBL and group work.
In this study we divided a group of students to compare two treatments. Such comparisons are potentially valuable for comparing different pedagogical approaches. However, they also potentially raise ethical concerns. It is in the nature of research of this kind that it cannot be known in advance which treatment (i.e. teaching approach etc.) will be found to be most effective or otherwise advantageous in a study. However, such studies are motivated by theories, and often the existing studies from other contexts, which suggest that an innovation may be more advantageous than the default treatment. If it were considered certain that an innovation would be more effective (or indeed less effective) in some teaching context, then there would be no justification for carrying out research to test this. If there were good reasons to expect that an innovation would prove detrimental in some teaching context, it would be perverse to seek to test that, as this would require acting in a way expected to be to the detriment of some learners. More usually researchers are in a position where there are good grounds to expect an innovation to be advantageous for students and/or teachers, but are not certain of this. This means, however, that research is conducted that assigns some students in a cohort to a control condition that researchers expect to disadvantage them compared to those peers selected for the intervention (‘experimental’) condition. There is clearly an ethical issue here, as researchers are deliberately putting some students in a condition they expect to be relatively detrimental to those students' best interests. There is clearly a balance to be considered here between the likely value of the research findings in informing future practice and protecting the interests of research participants. Making judgements about the acceptability of such designs will in part depend on how strong the grounds are considered to be for expecting the innovation to be advantageous in the research context, given that studies without suitable comparison conditions are always open to the question of what the outcomes would actually have been without the innovation. This is an issue that researchers, and indeed the wider research community, need to consider.
Conclusion
This study examined the effect of PBL on the academic achievement of students when applied to the topic of electrochemistry. The effect of applying PBL on students' academic achievement was evaluated by the Mann–Whitney U Test. According to the results of the Mann–Whitney U Test, although there is no significant difference between the results of the pre-test in the experimental and control groups, there is a significant difference in the results of the post-test in favour of the experimental group (U = 8.500, p < 0.05). Furthermore, the result of the Wilcoxon Sign Test showed that the changes in the CAT pre-test and post-test scores of students from the experimental and control groups were found to be statistically significant (z = −2.524, p = 0.012 and z = −2.371, p = 0.018, respectively); and accordingly, it could be seen that the increase rates of pre- and post-test scores of students from both groups were statistically the same (z = −0.694, p = 0.536). These findings also suggested that the PBL and the expository teaching strategy increased students' scores at the same rate. The reason why there was no significant difference between the experimental and control groups according to the CAT pre-test scores was a result of inadequate sample size. If the sample size increases, a statistically significant difference could be determined between the CAT pre-test scores of students from the experimental and control groups. Additionally, it was considered that the significant difference between the CAT post-test scores of students from the experimental and control groups was a result of greater variation of the median values of the CAT post-test scores in both groups. Since the purpose of the present study was to determine the difference between the PBL and the expository teaching strategy in terms of their effect on students' academic achievements, the comparison of percentage changes of the CAT pre- and post-test scores in both groups would be a more accurate approach than the comparison of the CAT pre- and post-test scores between these groups. Observing a significant difference in favour of the experimental group in the CAT post-test scores suggested that academic achievement of students taught through the problem-based learning was higher than that of the students taught through the expository teaching strategy. This finding suggests that problem-based learning, applied to the topic of electrochemistry, is more effective in increasing the achievement of students in contrast to an expository teaching approach applied to the same topic. The positive effect of PBL on students' academic achievement also shows similarities to earlier studies carried out for the topic of electrochemistry and other topics in chemistry. There are studies in the literature which showed that problem-based learning applied in chemistry and chemistry laboratory courses was effective in improving students' academic achievement and conceptual understanding, scientific research and social skills and giving them positive attitudes toward chemistry (Groh, 2001; Kelly and Finlayson, 2007; Senocak et al., 2007; Tarhan et al., 2008; Oezden, 2009; Hicks and Bevsek, 2012). Schaber et al. (2011) designed a scenario entitled “The Case of Nut Poisoning” for the “Upper-level Analytical Chemistry Course” in their study. The study showed that PBL improved students' interest in the subject, that they felt like researchers and scientists and believed that the course was more fun and taught them science in a better way. In a study conducted by Chamely-Wiik et al. (2012), it was shown that problem-based learning applied in the General Chemistry course enhanced students' academic writing skills and improved their knowledge regarding chemistry. In their study, Flynn and Biggs (2012) showed that problem-based learning improved students' independent learning, research and critical thinking skills. In the study conducted by Ashraf (2013), a biotechnology experiment developed in accordance with problem-based learning allowed students to effectively learn how enzymes work and what factors affect enzymes (pH, temperature, substrate/enzyme concentration and de-naturalizers). In their study, Figueira and Rocha (2014) showed that problem-based learning applied in an “Introduction to Biochemistry” course university students to understand the subject more easily and improved students' motivation. Li-Teh and Yakob (2013) examined the effect of problem-based learning in teaching the topic of ‘Cell Potential’. Li-Teh and Yakob's findings showed that using problem-based learning to teach the topic of cell potential had a positive effect on the academic performance of students in the experimental group compared to students in the control group. For this reason, it can be suggested that problem-based learning is an effective technique in the teaching of difficult chemical concepts. Aydoğdu (2012) examined the effect of problem-based learning on the academic achievements and attitudes of undergraduate students for the topic of ‘Electrolysis and Battery’ in chemistry courses. The research showed that the academic achievements of students in the experimental group were higher than those of students in the control group, and the students in the experimental group also had better attitudes toward chemistry courses than students in the control group. Sağır et al. (2009) examined the effect of problem-based learning on undergraduate students' learning for the topic ‘The Activity of Metals’ and the results showed that problem-based learning improved the academic achievements of students. At the same time, it has also been proposed elsewhere in the research that problem-based learning is an effective method for teaching chemistry and should be used in the teaching of other scientific topics. Tarhan and Acar (2007) examined the effect of problem-based learning on secondary school students' understanding of ‘The Effect of Temperature, Concentration and Pressure on Cell Potential’ and found that PBL eliminates misconceptions and had a positive effect on the achievements of students. This study showed that PBL students are more motivated, have higher self-esteem, are more willing to engage in problem-solving and sharing information and are more active in cooperative group activities. Ying (2003) applied problem-based learning using a prepared scenario concerning air pollution to teach electrochemistry within a course on ‘Physical Chemistry’ in which laboratory work was also included. The results showed that students had a better understanding of the areas of the usage of electrochemistry and could apply chemical analysis methods.The integration and application of information learned in real life situations are becoming more important every day in terms of chemistry. In this context, teaching Analytical Chemistry, which requires knowledge of mathematics, interpretative and critical skills, on the basis of scenarios and problems found in daily life problem is important. In the open-ended questions, it can be seen that the percentage of responses (81.44%) in the category of ‘Clear Understanding’ for students in the experimental group was higher than the percentage of responses (48.05%) in the same category for students in the control group. It can also be observed that the percentages of responses in the other categories (Partial Understanding, Partial Understanding with Specific Misconception, Specific Misconception and Incomprehension/Pointless) were lower than the percentage of responses in these categories for students in the control group. In general it was identified that students had specific misconceptions concerning the flow of electrons in the salt bridge and not being able to connect Gibbs free energy with electrode potential, the solubility equilibrium constant with electrode potential, and cell potential with standard electrode potential (Sanger and Greenbowe, 1997; Rogers et al., 2000; Cullen and Pentecost, 2011; Akram et al., 2014; Brandiet and Bretz, 2014).
However, in this study, it was also observed that students in the experimental and control groups made statements in the category of ‘Incomprehension’ concerning the description of the standard hydrogen reference electrode, the relationship between equilibrium constant and electrode potential, and the explanation of how the medium including two analyte pairs could be titrated by one titrant simultaneously in the redox titrations. In particular, the students in the control group made erroneous statements in the same category concerning finding the electrode potential of the equivalence point and determining the indicator in the redox titrations. In evaluating this study in general, the results of the Mann–Whitney U Test of the scores of the CAT post-test, as well as the scores obtained from open-ended questions, have shown that there was a significant difference in favour of the experimental group (U = 2.500, p < 0.05). This supports the conclusion that the students in the experimental group had a better understanding of the topic than the students in the control group.
Furthermore, this study also examined whether there was any relationship between intra-group scores received from the CAT post-test and the scores received from open-ended questions for the experimental and control groups. The findings obtained showed that there was a positive, high level, significant relationship between the scores from the test and the scores from the open-ended questions for the experimental group, but that there was an insignificant relationship between the scores from the test and the scores from the open-ended questions for the control group. The findings in favour of students in the experimental group obtained from the CAT post-test scores and the open-ended questions support the conclusion that PBL improves academic achievement and that students in the experimental group had a better understanding of information they had learned. The semi-structured interview results showed that students had positive opinions regarding PBL and found it to be interesting, relevant to everyday life, to allow for permanent learning, to lead them to carry out research and improve their communication. As for negative opinions, they thought it was a time-consuming and exhausting method.
In addition, since the scope of this study remained relatively limited within a single class from a single institution, the effect of the PBL could be measured more extensively in the future if the study group size could be increased; and/or students from the experimental and control groups could be selected from various grades.
Implications
The research suggests that the implementation of problem-based learning for the specific topic of ‘Electrochemistry’ in Analytical Chemistry is an effective method to enhance academic achievement. In order to overcome some misconceptions emerging after applying it, the period of implementation could be extended and in this period students could further discuss the topic by being given additional scenarios to eliminate misconceptions. Within the topic of ‘Electrochemistry’ itself, the use of PBL to teach the sub-topics of ‘auxiliary oxidizing and reducing agents in oxidation/reduction titration applications’ and ‘the applications of standard reducing and oxidizing agents’ could be improved.Appendix 1. The problem-based learning sessions (“Wow, Emre, Wow!”)
Emre, who was five years old, was playing with his toys next to his mother. After a while his mother went into the kitchen. While he was playing with his toys, there was a sudden knock on the door. His father, returning from shopping, left some the bags in the doorway. He kissed Emre and went to see Emre's mother in the kitchen. Emre took the shopping bags and started to poke around in them without anyone knowing. When his mother and father came back from the kitchen, they took Emre to go for his after-dinner nap. After about three hours, he woke up, crying and with abdominal pain. Emre began to vomit, have diarrhea and experience breathing difficulties.
(1) What do you think is the problem with Emre?
(2) From what things do you think this problem has resulted? Discuss them by establishing hypotheses.
(3) What new information do you need to solve this problem?
His mother told his father that Emre had been playing with the toys that he always played with and she hadn't seen anything different happening. His mother and father were worried about Emre who was usually quite healthy and didn't have any difficulties in breathing. They took him to the hospital nearest to their home.
(1) Summarize the new knowledge gained.
(2) In the light of this new knowledge, review your hypotheses.
(3) What do you think is the solution to this problem? Discuss this and justify it.
Dr Haluk, the emergency doctor in charge, asked Emre's mother how long Emre's abdominal pain, vomiting, diarrhea, breathing difficulties and bruising had lasted, whether he had previously had an allergic condition or any chronic diseases, and if he took any medication regularly. Meanwhile, he also asked her what Emre had recently eaten and drunk, what he had been doing and where he had been. His mother said that Emre had not previously had any chronic diseases or an allergic condition, he hadn't previously taken any medications, he hadn't wanted to have any breakfast, but she had made meatballs – because Emre liked them best – for dinner. She also said that Emre hadn't drunk anything, that he had recently been playing with his toys in the living room, that they had taken him to go to sleep at afternoon and that these symptoms were very new and had lasted for approximately three hours. In the first examination, Dr Haluk found that the general situation was bad. Emre's respiratory rate was 34 breaths per minute; BP was 120/70 mm Hg; pulse was 114 minute−1; fever was 37 °C (arm-pit). In neurological examination Emre had good muscle relaxation, his pupils were abnormal and his tendon reflexes were hypoactive. Dr Haluk wanted complete blood analysis, biochemistry of blood, blood gas and urinalysis, and electrocardiography and ultrasonography in order to be able to establish a final diagnosis. He monitored the patient. He assigned Mr Erol, the laboratory technician, to expedite the analyses.
Tutor's question: Are there any terms you don't know?
Tendon reflexes are hypoactive: There are no motor activities on either side of the body and no reaction occurs.
BP: Blood pressure.
Monitored: A device is connected to the patient with the purpose of keeping the patient's vital signs under observation and understanding how they change over time.
(1) Summarize the new knowledge gained.
(2) In the light of the new knowledge, review your hypotheses.
(3) What do you think is the solution to this problem? Discuss and justify this.
In accordance with the analyses of blood biochemistry, blood gas and urinalysis, electrocardiography and ultrasonography, the laboratory technician Mr Erol found the following results. The blood taken from Emre was ‘chocolate-brown’ colour:
| Data | Quantity | Normal values (5 years old) | |
|---|---|---|---|
| Hemogram | Hgb | 11.1 g dL−1 | 11.5–15.5 g dL−1 |
| HCT | 33% | 35–45% | |
| Plt | 264 × 103 mm−3 | 150–400 × 103 mm−3 | |
| Biochemistry of blood | Ure | 59 mg dL−1 | 7–26 mg dL−1 |
| Creatinine | 3.4 mg dL−1 | 0.30–1.40 mg dL−1 | |
| K | 7.5 mEq L−1 | 3.4–4.4 mEq L−1 | |
| Urinalysis | Density of urine | 1012 | 1015–1025 |
| Proteinuria | 200 mg L−1 | <150 | |
| Blood gas analysis | pH | 7.10 | 7.35–7.45 |
| HCO3− | 18 mEq L−1 | 22–26 mEq L−1 (blood plasma) | |
| pCO2 | 25 mm-Hg | 35–45 mm-Hg | |
| pO2 | 120 mm-Hg | 80–100 mm-Hg | |
| Methaemoglobin | 30% | <1% | |
| Electrocardiography | Sharp T waves | Normal | |
| Ultrasonography | The sizes of both kidneys are normal, there is renal failure in the third degree | The size of kidney is normal | |
References
http://www.sanal-hastane.com/tam-kan-sayimi-hemogram-cbc
http://www.beyazhastane.com/13/Hemogram-Normal_Değerleri.aspx
http://www.saglikekibi.com/can/pratikbilgi/tum-testlerin-normal-değer-araliklari.html
Tutor's question: Are there any terms you don't know?
Hemogram: Blood Count
Renal failure: Kidney failure
(1) Summarize the new knowledge gained.
(2) In the light of the new knowledge, review your hypotheses.
(3) What do you think is the solution to this problem? Discuss and justify this.
According to the laboratory analyses and the results of ultrasonography and electrocardiography, Dr Haluk found out that methemoglobinemia, cyanosis, hyperkalemia and acidosis were taking place in Emre and considered that this was a sign of poisoning. Then he again urgently asked Emre's mother what things Emre had eaten during the day and whether he took any medication. His mother again said that Emre didn't want to have breakfast and also didn't take any medication. His parents were interviewed by the police.
(1) Summarize the new knowledge gained.
(2) In the light of this new knowledge, review your hypotheses.
(3) How could Dr Haluk determine that methemoglobinemia, cyanosis, hyperkalemia and acidosis were taking place in Emre?
(4) What suggestions could Dr Haluk make for the treatment of Emre in this situation?
Emre fell into a coma. Dr Haluk carried out a gastric lavage and gave activated charcoal because he didn't know the cause of the poisoning. He gave him 1–2 mg kg−1 (0.1–0.2 mL kg−1 of 1% solution) of methylene blue solution intended for methemoglobinemia and cyanosis. He applied 10% 0.5 mL kg−1 of calcium gluconate, 1 gram kg−1 of glucose, 0.1 U kg−1 of insülin in 10% of dextrose, 1 mEq kg−1 of sodium bicarbonate I.V., 5 mg albuterol intended for acidosis and hyperkalemia. In accordance with the suggestion of the neurological consultant, he also gave him between 20 and 40 mg of furosemide I.V. as anti-edema treatment. At the same time he applied hyperbaric oxygen therapy.
Tutor's question: Are there any terms you don't know?
I.V.: Intravenously.
Gastric lavage: This means washing out the stomach with a gastric tube.
Activated charcoal: Medical charcoal can be given for the acceleration of detoxification.
(1) Summarize the new knowledge gained.
(2) In the light of this new knowledge, review your hypotheses.
(3) What do you think the main active substance poisoning Emre could be?
(4) What do you think the reasons are for Dr Haluk using the medications he does for Emre's treatment?
Dr Haluk sent Emre's father home to check all the medications and chemicals there one by one and see whether any drug was missing, whether all chemicals used at home were necessary and whether their lids or tops were open, because he didn't know the cause of the poisoning. Emre's father looked at all the drug containers and all chemicals used at their home. Nothing was open. But in the living room he found the shopping bags. He realised that he hadn't been aware of this situation while he was talking to his wife and leaving the house in a hurry. It was obvious that Emre had taken the shopping bags secretly and looked inside them. Emre's father leaned down close to the floor and an empty matchbox under the coffee table caught his attention.
(1) Summarize the new knowledge gained.
(2) In the light of this new knowledge, review your hypotheses.
(3) Do you think that Emre could get poisoning from the matches he secretly took from a shopping bag after he looked inside it?
His father picked up the empty matchbox and took the road back to the hospital. When he arrived at the hospital, he said to his wife that he had found an empty matchbox under the coffee table. He asked his wife whether he had brought the matches in his shopping. His wife said that there weren't any matches at home and that he had got these matches. Both of them suspected that Emre could have eaten the matches and checked this situation with Dr Haluk. Then Dr Haluk dialled 114 for the Drug and Poison Information Center and got information about how to treat a five-year old child who had eaten matches.
(1) Summarize the new knowledge gained.
(2) Which active substance or substances in the content of the matches do you think caused Emre to get poisoned?
(3) Which active substance in the content of the matches do you think resulted in Emre developing hyperkalemia and methemoglobinemia?
The laboratory technician Mr Erol felt bad about Emre's situation. He was confused that Emre had been poisoned by eating matches. He started to investigate how the matches had led to the poisoning. He explained the situation to his friend Emel who was working in a private factory as an analytical chemist and asked her to investigate the content of the matches. On the following day, as a result of the analyses she had carried out, Emel reported to Mr Erol that the matches contained a high concentration of potassium chlorate.
(1) Summarize the new knowledge gained.
(2) How do you think the potassium chlorate caused methemoglobinemia and hyperkalemia?
Mr Erol, the laboratory technician, started to investigate how potassium chlorate destroys the structure of haemoglobin. He researched this both from books and online. Finally, he found that the ion of chlorate (ClO3−) destroys the structure of the atom of iron involved in the structure of haemoglobin and destroys the iron atom's property of being an oxygen carrier. He found out that it converted the (+2)-loaded iron atom in the structure of haemoglobin to the (+3)-loaded iron atom.
(1) Summarize the new knowledge gained.
(2) According to the information found, is there a reaction of the ion of chlorate (ClO3−) with the ion of iron(II) (Fe2+)? If there is, how could this occur?
While Mr Erol was doing his research, he found a reaction as follows:
| ClO3(aq)− + 6Fe(aq)2+ + 6H(aq)+ ⇌ Cl(aq)− + 3H2O(s) + 6Fe(aq)3+ |
He determined that this reaction occurs spontaneously in the body, because he found out that the Gibbs free energy of this reaction was smaller than zero (ΔG0 < 0).
(1) Summarize the new knowledge gained.
(2) How do you think the Gibbs free energy of this reaction is found? Discuss.
(3) How do you think the equilibrium constant of this reaction can be found? Discuss.
In light of this information, the laboratory technician Mr Erol wondered about the quantity of the ion of chlorate (ClO3−) in the match eaten by Emre. He asked his friend Emel how this could be found by tests in the laboratory environment. Emel told him that this situation could have occurred by back titration of extra iron(II) (Fe2+) ions in the reaction with cerium(IV) (Ce4+) ions. She also indicated that the turning point of this reaction could be found in the way that the pale blue colour of the indicator of the 5-nitro-1, 10-phenanthroline iron(II) complex turns into red-violet colour in the medium of 1 M of H2SO4 solution.
(1) Summarize the new knowledge gained.
(2) How do you think this process happens by the method of back titration? Discuss this.
(3) What do you think about how the usage of the indicator is determined in accordance with the turning point? Explain this.
(4) What do you think about how the medium containing the analyte pairs of Fe(aq)2+ and Fe(aq)3+ can be titrated by the titrant of Ce(aq)4+? Discuss this.
Laboratory technician Mr Erol shared all the information he had investigated with Dr Haluk. According to the information obtained from the Drug and Poison Information Center, Dr Haluk learnt that he had applied the right treatment but that conventional hemodialyses also had to be administered at the same time. Dr Haluk then applied to Emre four conventional hemodialyses and ten clinical treatments of hyperbaric oxygen. Emre was discharged from the hospital on the nineteenth day of his hospitalization with normal findings.
Reminder:
Conventional hemodialyses: This is a means of providing intracellular, extracellular and solute equilibrium. Acute renal failure is a case frequently encountered in intensive care patients. In general, the hemodynamics of the patients being treated in intensive care and with acute renal failure are disordered, with multiple organ failure, and shocks are often accompanied by acute renal failure. Today, the conventional treatment for this type of patient is intermittent hemodialysis therapy. This can provide equilibrium to the hemodynamics, acid–bases and electrolytes and can treat acute renal failure (Fresenius Medical Care (2012, 4 February). Sürekli Renal ReplasmanTedavileri (SRRT) Accessed Date: 10.02.2013 nternet address: http://www.fresenius.com.tr/pg16c.html).
(1) Summarize the new knowledge gained.
(2) For what purpose do you think that conventional hemodialysis is applied? Discuss this.
(3) Summarize the scenario with a flow chart.
Appendix 2: activity 1
The maintenance of hydraulic fluid and coating are performed in an automotive plant in Aydın. Anti-corrosion additives (inhibitors) are used for the coating in particular. A high concentration of hydroxylamine is found among these substances. Hydroxylamine is a substance which is an ammonia derivative like semi-hydrazine and semi-hydrogen peroxide. It is both a reducing and oxidizing agent. As a result of an explosion occurring in this plant, a high concentration of hydroxylamine is mixed into the city's water supply. As a result, a high concentration of iron(II) ion (Fe2+) is formed and N2O gas is released. However, the city's water pipes are not made of stainless steel. You are an analytical chemist and your specialty is electroanalytical methods. You are assigned to find out which reactions occurred, how iron(II) ion (Fe2+) was formed and to measure the electrode potential of iron(II) ion by making an electrochemical cell. You are asked to write a report about the situation. What do you do?Appendix 3: activity 2
In 1998 a high concentration of lead was found in the samples of mussels taken from Sinop and from the point where the waste materials from the Hopa Copper Enterprises in the Eastern Black Sea reached the sea. This was measured in a project named “Heavy Metal Pollution in the Black Sea” carried out by the Institute of Marine Sciences (Erdemli, Icel), General Directorate of Agricultural Research, Ministry of Agriculture and Rural Affairs, and the Fishery Research Institute (Yomra, Trabzon) (Ünsal et al., 1998). As we know, iodine is present as a dissolved iodide, found in the environment mainly in seawater. In this region, the high concentration of lead has precipitated with the iodide as lead iodide. You are an analytical chemist and you are carrying out the electrochemical analyses. You are asked to measure the electrode potential of the solid of lead iodide (PbI2) that has occurred. What do you do?| PbI2(k) ↔ Pb(aq)2+ + 2I(aq)−Ksp = 7.1 × 10−9(25 °C) |
| PbI2(k) + 2e− ↔ Pb(k) + 2I(aq)−E = ? |
Appendix 4: a sample question in CAT test regarding electrochemical cell types
19th question:Which of the following statements about the electrolytic cell system in the figure are incorrect?
(A) The Ag(k) metal is more active than the Ni(k) metal.
(B) Anions in the salt bridge head towards container II.
(C) The mass of the Ag electrode decreases over time.
(D) The concentration of Ni(aq)2+ in container I decreases over time.
(E) Electrons move towards the Ni(k) electrode from the Ag(k) electrode-through the conductive wire.
Appendix 5: a sample question in CAT test regarding the relationship between electrode potential and Ksp
39th question: the solubility constant of AgCl(k) is (Ksp) 1.82 × 10−10. Accordingly;How many volts is the electrode potential for the
| AgCl(k) + e− ⇌ Ag(k) + Cl(aq)− reaction? |
(A) −0.350
(B) −0.244
(C) −0.222
(D) +0.222
(E) +0.244
Appendix 6: a sample question in the CAT test regarding the relationship between electrode potential and Gibbs free energy
29th question: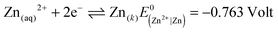
According to half cell potentials given;
(I) Mg(k) + Zn(aq)2+ ⇌ Mg(aq)2+ + Zn(k) reaction occurs spontaneously
(II) ΔG > 0
(III) In Mg∥Zn cell, if water at the same temperature is added to the container containing Zn(k), the standard electrode potential increases.
Which of the above statements are correct?
(A) Only I (B) Only II (C) Only III (D) I and II (E) I and III
Acknowledgements
The authors wish to thank Prof. Dr. Canan Nakiboğlu, Assoc. Prof. Dr Gülten Şendur, Assoc. Prof. Dr Suat Türkoğuz, Assist. Prof. Dr A. Murat Ellez and Lecturer Dr M. Çağatay Büyükuysal for supporting this research. We thank all undergraduate students who participated in this study. In addition, we wish to thank the referees for suggestions and having time for the study. We also especially thank the editor for advice on discussing the ethics of the research design adopted in this study, suggestions and having time for the study.References
- Abraham M. R., Grzybowski E. B., Renner J. W. and Marek E. A., (1992), Understandings and misunderstandings of eight graders of five chemistry concepts found in textbook, J. Res. Sci. Teach., 29(2), 105–120.
- Akram M., Surif J. B. and Ali M., (2014), Conceptual Difficulties of Secondary School Students In Electrochemistry, Asian Soc. Sci., 10(19), 276–281.
- Allsop R. T. and George N. H., (1982), Redox In Nuffield Advanced Chemistry, Educ. Chem., (19), 57–59.
- Alpar R., (2012), Spor, Sağlık ve Eğitim Bilimlerinden Örneklerle Uygulamalı statistik ve Geçerlilik-Güvenirlik, DetayYayıncılık, 2nd edn, Ankara, p. 271.
- Ashraf S. S., (2013), Raising Environmental Awareness through Applied Biochemistry Laboratory Experiments, Biochem. Mol. Biol. Educ., 41(5), 341–347.
- Ayas A. and Özmen H., (1995), Lise 1 Kimya Öğrencilerinin Maddenin Tanecikli Yapısı Kavramını Anlama Seviyeleri Üzerine Bir Çalışma, II, Ankara: Fen Bilimleri Eğitim Kongresi, ODTÜ.
- Aydoğdu C., (2012), The Effect of Problem Based Learning Strategy In Electrolysis And Battery Subject Teaching, Hacettepe Üniversitesi Eğitim Fakültesi Dergisi, 42, 48–59.
- Belt S. T., Evans E. H., McCreedy T., Overton T. L. and Summerfield S., (2002), A Problem Based Learning Approach to Analytical and Applied Chemistry, Univ. Chem. Educ., 6(2), 29–36.
- Blane P. D., (2012), Methemoglobinemi, in Savcı V. (ed.), Zehirlenmeler &laçAşırıDozu, stanbul: Nobel, pp. 292–294.
- Brandiet A. R. and Bretz S. L., (2014), Measuring Meta-Ignorance through the Lens of Confidence: Examining Students' Redox Misconceptions about Oxidation Numbers, Charge and Electron Transfer, Chem. Educ. Res. Pract., 15, 729–746.
- Bransford J. D., Brown A. L. and Cocking R. R., (2000), How People Learn: Brain, Mind, Experience, and School, Washington, DC: National Academy Press.
- Büyükdokumacı H., (2012), lköğretimSekizinciSınıf Fen ve Teknoloji Dersinde Probleme Dayalı Öğrenmenin (PDÖ) Öğrenme Ürünlerine Etkisi, Yüksek Lisans Tezi, Pamukkale Üniversitesi Fen Bilimleri Enstitüsü.
- Büyüköztürk Ş., (2007), Sosyal Bilimler çin Veri Analizi El Kitabı, Ankara: PegemAYayıncılık.
- Büyüköztürk Ş., Çakmak K. E., Akgün E. Ö., Karadeniz Ş. and Demirel F., (2008), Bilimsel Araştırma Yöntemleri, kinci Baskı Ankara: Pegem Akademi.
- Chamely-Wiik D. M., Haky J. E. and Galin J. R., (2012), From Bhopal To Cold Fusion: A Case-Study Approach To Writing Assignments In Honors General Chemistry, J. Chem. Educ., 89, 502–508.
- Cullen D. M. and Pentecost T. C., (2011), A Model Approach To The Electrochemical Cell: An Inquiry Activity, J. Chem. Educ., 88(11), 1562–1564.
- Dimitrov D., (2007), Reliability, in Erford B. T. (ed.), Assessment for Counselors, Boston: Houghton-Mifflin/Lahaska Press, pp. 99–122.
- Dods R. F., (1996), A Problem-Based Learning Design For Teaching Biochemistry, J. Chem. Educ., 73(3), 225–228.
- Duch B. J., Groh S. E. and Allen D. E., (2001), Why Problem Based Learning?: A Case of Institutional Change in Undergraduate Education, The Power of Problem Based Learning: A Practical “How To” For Teaching Undergraduate Courses in Any Discipline, 1st edn, Stylus Publishing, LLC.
- Festus C. and Ekpete O. A., (2012), Improving Students' Performance and Attitude towards Chemistry through Problem-Based-Solving Techniques (PBST), Int. J. Acad. Res. Prog. Educ. Dev., 1(1), ISSN: 2226–6348.
- Figueira A. C. M. and Rocha J. B. T., (2014), A Proposal For Teaching Undergraduate Chemistry Students Carbohydrate Biochemistry By Problem-Based Learning Activities, Int. Union Biochem. Mol. Biol., 42(1), 81–87.
- Flynn A. B. and Biggs R., (2012), The Development and Implementation Of A Problem-Based Learning Format In A Fourth-Year Undergraduate Synthetic Organic And Medicinal Chemistry Laboratory Course, J. Chem. Educ., 89, 52–57.
- Gallagher S. A., Stepien W. J. and Rosenthal H., (1992), The Effects Of Problem-Based Learning On Problem Solving, Gifted Child Q., 36(4), 195–200.
- Groh S. E., (2001), Using Problem-Based Learning In General Chemistry, in Duch B. J., Groh S. E. and Allen D. E. (ed.), A Practical “How To” For Teaching Undergraduate Courses In Any Discipline: The Power Of Problem-Based Learning, Stylus Publishing.
- Grover N., (2004), Introductory Course Based on a Single Problem: Learning Nucleic Acid Biochemistry from AIDS Research, Biochemistry and Molecular Biology Education, 32(6), 367–372.
- Güncel Haber, (2013), ErişimTarihi: 24/01/2013, nternet Adresi: http://www.genelhaber.org/kutahyada-kibrit-copu-yalayan-cocuk-zehirlendi/.
- Hicks, R. W. and Bevsek H. M., (2012). Utilizing Problem-Based Learning in Qualitative Analysis Lab Experiments, J. Chem. Educ., 89, (2), 254-257.
- Hung W., Jonassen D. H. and Liu R., (2008), Problem Based Learning, in Spector M., Merrill D., Van Merrienböer J. and Driscoll M. (ed.), Handbook of Research on Educational Communications and Technology, New York, NY: Erlbaum, 3rd edn, pp. 485–506.
- İnce Aka, E., (2012), Asit Bazlar Konusunun Öğretiminde Kullanılan Probleme Dayalı Öğrenme Yönteminin Farklı Değişkenler Üzerine Etkisi Ve Yönteme lişkin Öğrenci Görüşleri, DoktoraTezi, Gazi Üniversitesi, Eğitim BilimleriEnstitüsü.
- Jansson S., Soderstrom H., Andersson P. L. and Nording M. L., (2015), Implementation of Problem-based Learning in Environmental Chemistry, J. Chem. Educ., 92, 2080–2086.
- Johnson D. W., Johnson R. T. and Smith K. A., (1991), Cooperative Learning: Increasing College Faculty Instructional Productivity, (ASHE-ERIC Higher Education Report No. 4), Washington, DC: George Washington University.
- Kelly O. C. and Finlayson O. E., (2007), Providing Solutions through Problem-Based Learning for The Undergraduate 1(st) Year Chemistry Laboratory, Chem. Educ. Res. Pract., 8(3), 347–361.
- Laredo T., (2013), Changingthe First-Year Chemistry Laboratory Manual to Implement A Problem-Based Approach That Improves Student Engagement, J. Chem. Educ., 90(9), 1151–1154.
- Li-Teh K. and Yakob N., (2013), Problem-Based Learning As An Approach To Teach Cell Potential In Matriculation College, Malaysia, Chemistry Education and Sustainability In The Global Age, 121–130. DOI: http://10.1007/978-94-007-4860-6_11.
- Lopes R. M., Silva Filho M. V. and Marsden M., (2011), Problem Based Learning: A Teaching Toxicology Chemistry Experience, Quimica Nova, 34(7), 1275–1280.
- Mabrouk P. A., (2007), Bioanalytical Chemistry: Model For A Fully Integrated Problem-Based Learning Approach. Active Learning: Models From The Analytical Sciences, ACS Symp. Ser., 970, 69–86.
- Madhuri G. V. and Goteti P. (2011), Imparting Transferable Skills and Creating Awareness Among Students on Non-Conventional Energy Sources Using Problem Based Learning, in Torres I. C., Chova L. G., Martinez A. L. (ed.), 4th International Conference of Education, Research and Innovation (ICERI), Madrid, Spaın, pp. 6925–6929, Date: Nov 14–16, 2011.
- Marek E. A., (1986), They Misunderstand, But They'll Pass, Sci. Teach., 53(9), 32–35.
- Marks R. and Eilks I., (2010), Research-Based Development of a Lesson Plan on Shower Gels And Musk Fragrances Following a Socio-Critical and Problem-Oriented Approach to Chemistry Teaching, Chem. Educ. Res. Pract., 11, 129–141, DOI: 10.1039/C005357K.
- Moralar A., (2012), Fen Eğitiminde Probleme Dayalı Öğrenme Yaklaşımının Akademik Başarı, Tutum ve Motivasyona Etkisi, Yüksek Lisans Tezi, TrakyaÜniversitesi Fen Bilimleri Enstitüsü.
- Oezden M., (2009), Enhancing Prospective Teachers' Development through Problem-Based Learning in Chemistry Education, Asian J. Chem., 21(5), 3671–3682.
- Osgood M. P., Mitchell S. M. and Anderson W. L., (2005), Teachers as Learners in a Cooperative Learning Biochemistry Class, Biochem. Mol. Biol. Educ., 33(6), 394–398.
- Özeken Ö. F. and Yıldırım A., (2011), The Effect of Problem-Based Learning Method In Teaching Acid–Base Subject On Science Teachers' Academic Success, Pegem Eğitimve Öğretim Dergisi, 1(1), 33–38.
- Rogers F., Huddle P. A. and White M. D., (2000), Using A Teaching Model To Correct Known Misconceptions In Electrochemistry, J. Chem. Educ., 77(1), 104.
- Sağır S. U., Çelik A. Y. and Armağan F.O., (2009), The Effect Of Problem Based Learning Strategy In Metalic Activity Subject Teaching, Hacettepe Üniversitesi Eğitim Fakültesi Dergisi, 36, 283–293.
- Salkind N. J., (2007), Encyclopedia of Measurement and Statistics, SAGE Publications, vol. 1, ISBN: 1-4129-1611-9.
- Sands T. R., (2012a), Hiperkalemi Ve Hipokalemi, in Savcı V. (ed.), Zehirlenmeler & laçAşırıDozu, stanbul: Nobel, pp. 36–39.
- Sands T. R., (2012b), Kloratlar, in Savcı V. (ed.) Zehirlenmeler & laçAşırıDozu, stanbul: Nobel, pp. 252–253.
- Sanger M. J. and Greenbowe T. J., (1997), Students' Misconceptions in Electrochemistry: Current Flow in Electrolyte Solutions and The Salt Bridge, J. Chem. Educ., 74(7), 819–823.
- Schaber P. M., Pines H. A., Larkin J. E., Shepherd L. A. and Wierchowski E. E., (2011), The Case Of Nut Poisoning (Or Too Much Of A Good Thing?): Implementation And Assessment, J. Chem. Educ., 88, 1012–1013.
- Selco J. I., Roberts Jr. J. L. and Wacks D., (2003), The Analysis of Seawater: A Laboratory-centered Learning Project in General Chemistry, J. Chem. Educ., 80(1), 54–57.
- Şencan H., (2005), Sosyal ve Davranışsal Ölçümlerde Güvenilirlik ve Geçerlilik, Ankara: Seçkin Yayıncılık.
- Şenocak E., Taşkesenligil Y. and Sözbilir M., (2007), A Study on Teaching Gases to Prospective Primary Science Teachers Through Problem-Based Learning, Res. Sci. Educ., 37, 279–290.
- Serin G., (2009), The Effect Of Problem Based Learning Instruction On 7th Grade Students' Science Achievement, Attitude Toward Science And Scientific Process Skills, The Degree of Doctor of Philosophy, Middle East Technical University, The Graduate School of Natural And Applied Sciences.
- Serjant E. P., (1984), Potentiometry and Potentiometric Titrations, New York: John Wiley and Sons.
- Taber K. S., (2014), Ethical considerations of chemistry education research involving ‘human subjects’, Chem. Educ. Res. Pract., 15, 109–113.
- Tarhan L. and Acar B., (2007), Problem-Based Learning In An Eleventh Grade Chemistry Class: ‘Factors Affecting Cell Potential’, Res. Sci. Technol. Educ., 25(3), 351–369.
- Tarhan L. and Şeşen B. A., (2013), Problem Based Learning In Acids And Bases: Learning Achievements And Students' Beliefs, J. Balt. Sci. Educ., 12(5), 565–578, ISSN 1648–3898.
- Tarhan L., Kayalı A. H., Ürek Ö. R. and Acar B., (2008), Problem-Based Learning in 9th Grade Chemistry Class: Intermolecular Forces, Res. Sci. Educ., 38, 285–300.
- Taşdelen U., (2011), The Effects Of Computer Based Interactive Conceptual Change Texts On 11th Grade Students' Understanding Of Electrochemistry Concepts And Attitude Toward Chemistry, The Degree of Doctor of Philosophy, Middle East Technical University, The Graduate School of Natural And Applied Sciences.
- Tatar E., (2007), Effect of Problem Based Learning Approach on Understanding of First Law of Thermodynamics, DoktoraTezi, Atatürk Üniversitesi Fen BilimleriEnstitüsü.
- Tosun C., (2010), Probleme Dayalı Öğrenme Yönteminin Çözeltiler Ve Fiziksel Özellikleri Konusunun Anlaşılmasına Etkisi, DoktoraTezi, Atatürk Üniversitesi Fen BilimleriEnstitüsü.
- Tosun C. and Taşkesenligil Y., (2013), The Effect of Problem-Based Learning on Undergraduate Students' Learning about Solutions and Their Physical Properties and Scientific Processing Skills, Chem. Educ. Res. Pract., 14, 36–50.
- Tunç T., (2015), Analitik Kimya Dersi “Elektrokimya” Konusunda Probleme Dayalı Öğrenmenin Öğrencilerin Akademik Başarılarına Etkisi, Doktora Tezi, Dokuz Eylül Üniversitesi Eğitim Bilimleri Enstitüsü.
- Tunç T. and Alpat S. K., (2015), Developing a Valid and Reliable Chemistry achievement test in Electrochemistry, J. High. Educ. Sci., 5(3), 347–360.
- Türkmen E. Y., (2006), (ErişimTarihi: 10/02/2013), PratiKimya: TeknolojikUygulamalar,nternetAdresi: http://www.kimyasanal.com/konugoster.php?yazi=dzxtgwqlik.
- Ünsal M., Çağatay N., Bekiroğlu Y., Kıratlı N., Alemdağ N., Aktaş M. and Sarı E., (1998), Karadeniz'de Ağır Metal Kirliliği, Proje No: YDEBÇAG-456/G-457/G.
- Ünver S., Atasoyu E. M., Evrenkaya T. R., Solmazgül E., Ay H. and Saraçoğlu M., (2005), Potasyum KloratToksisitesine Bağlı Nörolojik Bulguların Eşlik Ettiği Ender Bir Akut Tübüler Nekroz Olgusu, Türk Nefroloji Diyaliz ve Transplantasyon Dergisi, 14(3), 143–145.
- Urena S. S., Cooper M. and Stevens R., (2012), Effect of Cooperative Problem Based Lab Instruction on Metacognition and Problem-Solving Skills, J. Chem. Educ., 89(6), 700–706.
- Williamson V. M. and Rowe M. W., (2002), Group Problem-Solving Versus Lecture in College-Level Quantitative Analysis: The Good, The Bad And The Ugly, J. Chem. Educ., 79, 1131–1134.
- Wright J. C., Millar S. B. and Kosciuk S. A., (1998), A Novel Strategy For Assessing The Effects Of Curriculum Reform On Student Competence, J. Chem. Educ., 75, 986–992.
- Yılmaz A., Erdem E. and Morgil İ., (2002), Öğrencilerin Elektrokimya Konusundaki Kavram Yanılgıları, Hacettepe Üniversitesi Eğitim Fakültesi Dergisi, 23, 234–242.
- Ying Y., (2003), Using Problem-Based Teaching and Problem-Based Learning to Improve the Teaching of Electrochemistry, The China Papers, July, 42–47.
- Yoon H., Woo A. J., Treagust D. and Chandrasegaran A. L., (2014), The Efficacy of Problem-based Learning in an Analytical Laboratory Course for Pre-service Chemistry Teachers, Int. J. Sci. Educ., 36(1), 79–102.
- Zhang H., Zhang Q. and Jing H., (2011), Application For The Method Of PBL In Pharmaceutical Chemistry Teaching, in Xiong J. Q. (ed.), Advanced Materials and Information Technology Processing, Advanced Materials Research, part: 1–3, vol. 271–273, pp. 1670–1673.
| This journal is © The Royal Society of Chemistry 2017 |

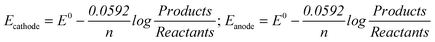
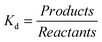
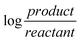
![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif)