Open Access Article
Open Access ArticleTeaching of chemical bonding: a study of Swedish and South African students' conceptions of bonding†
Anders
Nimmermark
*ab,
Lars
Öhrström
a,
Jerker
Mårtensson
a and
Bette
Davidowitz
c
aDepartment of Chemistry and Chemical Engineering, Physical Chemistry, Chalmers University of Technology, SE-412 96 Gothenburg, Sweden
bTingsholmsgymnasiet, SE-523 86 Ulricehamn, Sweden. E-mail: anders.nimmermark@ulricehamn.se
cDepartment of Chemistry, University of Cape Town, Rondebosch, 7701 South Africa
First published on 12th July 2016
Abstract
Almost 700 Swedish and South African students from the upper secondary school and first-term chemistry university level responded to our survey on concepts of chemical bonding. The national secondary school curricula and most common textbooks for both countries were also surveyed and compared for their content on chemical bonding. Notable differences between the countries were found in textbooks and in the curriculum regarding the topics of ionic bonding, bond energetics and use of the VSEPR model, the latter being absent in the Swedish curriculum and ionic bonding not explicitly mentioned in the South African curriculum. To some extent these differences are reflected in the students’ responses to the survey. It is also clear that university teachers in both countries must prepare effective counter-measures against deep rooted misunderstandings. For the upper secondary school level it is suggested that the bond energetics and exothermic and endothermic reactions be clearly and carefully presented and separated as the study indicates that mixing of these two concepts is a major cause of confusion.
Introduction
Background
In order to understand chemistry, it is essential to master chemical bonding. However, learning chemical bonding can be challenging as the level of abstraction is high and the acceptance of new, refined models can be hampered by students using heuristics (Talanquer, 2006), or by a limited understanding of previous models (Coll and Treagust, 2003a, 2003b). This may be a problem in particular for the transition from secondary to tertiary level as the typical university teacher may not be aware of the specific models used previously, nor the impact these have had on the students.The present study is the result of a project where an experienced secondary school teacher, author AN, was enrolled in a 2.5 year graduate program at Chalmers University of Technology‡ (Chalmers). The combined experiences of teaching at several levels, and in three educational frameworks (Swedish, South African, and International Baccalaureate§) made the team suitable to investigate how some key concepts of chemical bonding were understood by students in secondary school and those starting tertiary level chemistry. A pilot study was undertaken with selected student groups in Sweden but the full study included a questionnaire administered to students at a large number of Swedish high schools, and undergraduates starting their first term at Chalmers and the University of Cape Town (UCT).¶ We aimed to obtain some first insights about which problems and misconceptions are inherent difficulties in learning chemical bonding, and which are dependent on the foci in the two national curricula.
Literature review
All students create their own individual interpretations of the explanations they have been subjected to in different learning situations. These interpretations make up the students' framework theories, the ensemble constituting their conceptual ecology (Hewson, 1992). The framework theories may be very entrenched and take priority over the scientifically superior theories and thus actual conceptual change may be hard to achieve (Vosniadou, 1994).The high status of this alternative framework must be lowered or no meaningful learning will occur. To achieve this teachers can employ the four conditions for conceptual change proposed by Posner et al. (1982). First, the student has to become dissatisfied with the old framework; secondly, the new concept needs to be presented in a way that seems intelligent to the learner. The third point is that the new concept must be perceived as initially plausible and lastly it must be capable of providing fruitful solutions to the problems presented.
Aside from fulfilling these conditions, the teacher must create a learning environment of trust, situations that stimulate the asking of questions, and activities that challenge the students' beliefs and encourage them to step outside their regular thought patterns, all of which lie at the heart of being a teacher. Moreover, the teacher needs to take into account that students and instructors often enter into dialogue with different interpretive frameworks (Driver and Easley, 1978).
From a cognitive perspective, it can be argued that the understanding and subsequent creation of a conceptual framework of a topic can be influenced by what Talanquer calls “Commonsense chemistry” (Talanquer, 2006). The theory relies heavily on cognitive science and assigns the students' use of mainly involuntary empirical and heuristic thought processes as the origin of alternative conceptions. Two examples of heuristic reasoning and empirical assumptions leading to the wrong conclusions are described below.
“Copper is red, thus the Cu atom is also red”
“At equilibrium the forward reaction is completed before the reverse reaction commences” (Talanquer, 2006)
From the examples above it follows that without knowledge of the empirical assumptions and reasoning heuristics employed by the students, it is impossible to construct effective learning activities. The average school system, to some extent, fails to realize such conceptual change as several studies reveal that the framework adopted early on might be retained at higher levels in the educational system (Taber, 1998; Nicoll, 2001; Coll and Treagust, 2003).
Prior studies of bonding concepts
A number of studies of students' conceptual framework of chemical bonding have been published during the last two decades and a good starting point is the literature review by Özmen (2004). In this review he reported on a study in the late 1980s in which Peterson et al. (1989) developed and used a diagnostic instrument to investigate the misconceptions of Grade 12 students (17–18 years old). One of their findings was that molecular shape is a problematic concept, one where students are likely to create and retain misconceptions.A later study in Australia of lower secondary school students' mental models of atoms and molecules (Harrison and Treagust, 1996) discussed the risk of the teacher creating misconceptions and alternative frameworks when using analogies, metaphors and models without sufficient discussion or explanation. One very common misconception was that students used a covalent model for describing ionic compounds. This was also shown by Taber (1997) who described the formation of a “molecular” alternative framework and compares it to the curricular science framework of electrostatic interactions. Consequently, Taber recommends that bonding should be taught in the order; metallic, ionic, covalent, to minimize the risk of the student creating a molecular view of the ionic bond. In contrast, Dhindsa and Treagust (2014) using a cognitive approach, proposed that the best sequence for teaching bonding was to follow the order of: covalent, polar covalent and ionic bonding.
Boo (1998) investigated different aspects of the energetics of bonding and her research revealed that many students created a mental model where both bond formation and bond breaking require energy. She proposed that this model is formed through a blend of school science stating that bond breaking requires energy and the students' everyday perception that the breaking of a bond releases energy. The mental model starts with a bond requiring energy to break, but as the atoms move apart, energy is released. A related aspect of this framework is an assumption that something cannot be made without input of energy. In their report on a thermochemistry concept inventory, Wren and Barbera (2013), found the same misconception as reported by Boo. In addition, they noted that students often have difficulties interpreting endothermic and exothermic processes.
That such misconceptions are highly resistant to change, as discussed above (Vosniadou, 1994), was also shown by Nicoll (2001) in an cross age investigation of science and chemistry majors at a US university. Misconceptions at the microscopic and macroscopic level were shown to be common among the sample of students and, as might be assumed, the frequency of misconceptions is higher for the science majors than for the chemistry majors.
The difference between secondary school, undergraduate and graduate students mental models of ionic bonding was investigated by Coll and Treagust (2003). Students at all levels of education displayed alternative mental models and the main difference was in the explanatory details where the graduate students in general gave more details. Coll and Treagust recommend that teachers should emphasise the link between the macroscopic and microscopic level and to be careful when using visual clues, whether in diagrams or in real life models. In a study conducted in Israel, Levy Nahum et al. (2008) proposed an alternative approach to this subject that might decrease the formation of alternative frameworks. Their bottom-up framework starts with the fundamental aspects of bonding, namely electrostatic interactions, after which they suggested introducing a continuum of bond strengths and finally the structures and the properties that follows from the bond types.
Taber et al. (2012), undertook a cross-cultural investigation in England, Greece and Turkey, of the misconceptions of ionic bonding. They found that the misconceptions tend to be less related to the teaching tradition in each individual country than to the actual phenomenon. Thus they infer that it is not the social or cultural context that creates the alternative frameworks but the difficulties of the concepts themselves. A recent study in Croatia (Vladušić et al., 2016) using Taber's truth about bonding instrument (Taber, 2002) provides additional evidence for origins of this misconception. Bergqvist et al. (2013) recently reported on the way in which verbal, symbolic or visual modelling in Swedish textbooks helps or hinders the students understanding of chemical bonding. One finding from this study was that focusing on the separate atoms involved in bond forming was detrimental to the proper build-up of an adequate framework for chemical bonding.
The Swedish – South African chemical bonding study
We are not aware that any large-scale investigation into students' ideas of chemical bonding has been carried out with Swedish upper secondary school chemistry students. An in-depth study of a group of students at a specific Swedish upper secondary school understanding of matter and phase changes was published recently (Taber and Adbo, 2013). While some data has been collected about South African secondary students level of preparedness for tertiary studies (Potgieter and Davidowitz, 2011), there has been no in depth investigation of their ideas on chemical bonding. From these perspectives it would be especially fruitful for Swedish and South African chemistry teachers and researchers to obtain data regarding the frameworks of bonding held by their students. To gain cross-cultural and cross-contextual comparisons, students starting university level chemistry at Chalmers University of Technology (Chalmers) and at University of Cape Town (UCT) together with science and technology students in Swedish upper secondary schools were invited to participate in the study. We also performed a survey with the Swedish tertiary entry-level group, after they had studied the bonding section of their general chemistry course. With the findings from these different groups, we hope to gain some insight into strengths and weaknesses in the different educational systems in Sweden and in South Africa.Comparison of school systems
The Swedish upper secondary school chemistry is rather unregulated in terms of learning goals, activities, tests and exams syllabus (Swedish National Agency for Education, 2000, 2010) and the syllabus in chemistry contains only certain core concepts that are to be covered. The extent to which this should be done is not specified in detail. It is the individual teacher or team of teachers at the specific schools who determines the order in which the concepts are introduced, the types of learning activities, the level of complexity in the learning activities, the amount of time spent on any specific subject and the content and difficulty of the chemistry tests. The actual content taught and time spent on specific topics may thus vary to a large extent. In a system such as the Swedish secondary schools, textbook content can have a much larger influence on the teaching than the syllabus for the course.The current South African school system contains a more regulated and detailed curriculum and syllabus in the physical sciences (Dept. of Basic Education, RSA, 2012). The freedom of the individual teacher to decide activities, the order and time allocation of the different topics is much more limited in South Africa than in Sweden. This has not always been the case, since the previous curriculum, the NCS, initially was similar to the Swedish and left much freedom to the teachers and schools (Department of Education, 2003). After strong criticism from both teachers and the research community, a content description was added in which more details and guidance was given (Department of Education, 2006).
The Swedish upper secondary school curriculum contains no mandatory final test in the natural sciences but there is a voluntary national test, which schools can choose to use if they wish to do so. The South African system is based on a mandatory National Senior Certificate (NSC) examination in each of the subjects taken, commonly called the matric examination. This examination, which is used not only for evaluation of the students results, but also of the individual schools performance, has a profound effect of guiding the learning activities related to areas covered in each subject. It has been argued that it can be problematic if the exam questions are not aligned with current scientific views and often focus on memorization, (Levy Nahum et al., 2007). If this is the case, then the examination is very likely to steer teaching towards rote learning.
The Swedish system might in that sense have more freedom to focus on the understanding and knowledge of chemistry but will lack the normative aspect of a mandatory examination. Another difference between the two systems is the fact that in Sweden chemistry is taught as a separate subject, whereas in South Africa chemistry is part of the larger subject, physical science, comprising both chemistry and physics. We note that the main difference between the Swedish and South African school systems seems to be the degree of freedom, thus it is impossible to deduce difference in subject teaching based only on the curriculum documents. Thus a textbook-survey was included in this study.
Objectives
The goal is to develop an understanding of the student's likely misconceptions and their origin in order for both secondary and tertiary-level teachers to facilitate the transition from high school studies to the more stringent scientific teaching at universities within the current curricula. The long-term goal is to suggest changes in the secondary school curriculum and teaching methods that could further improve this process.Aim and research questions
The aims of our study are to investigate differences and similarities in understanding some key concepts in chemical bonding of groups of students in upper secondary school and those starting tertiary level chemistry at Chalmers and UCT. We further wanted to see if the difference in the educational systems in Sweden and South Africa could indicate a reason for any differences we might observe. We also wanted to see how, and if, the first year chemistry course changed this understanding. To guide the study the following research questions were investigated:(1) Are there differences between the groups regarding the understanding of chemical bonding concepts, and if so, can such a difference be traced back to specific concepts?
(2) Are there differences between Sweden and South Africa at the curriculum level, and if so are these related to the findings for question 1?
(3) Is there a quantifiable difference between Sweden and South Africa when it comes to teaching in the classroom and the contents of the textbooks used?
(4) To what extent is there a quantifiable evolution in the understanding before and after a first year university chemistry course?
Methodology
Data collection
Data were collected using surveys, interviews and a survey of curricula and textbooks. The majority of the data used in this study stems from the surveys in terms of answers to multiple choice and open-ended questions, these data are mostly numerical or text generated by the respondent. A survey of the textbooks was carried out to link the results from the student survey to the use of different models and teaching methods. Interviews were conducted to give further insights in the reasoning of the students and comments from the interviews are used to give examples that highlight the findings in the surveys or textbooks. The surveys were performed between 2012 (preliminary) and 2014, and the interviews were performed in September 2013.Ethical considerations
In the case of the students at Chalmers and University of Cape Town the study was approved by the department heads. Permission to include the Swedish upper secondary school students was given by their individual teacher. Prior to being asked to volunteer to participate, students were informed of the aim of the study and were informed that all participation was voluntary and anonymous.Surveys
The surveys were presented to the Swedish students in Swedish and to the South African students in an English translation.The language of instruction at UCT is English and all students admitted would have completed their Matriculation examination in English or had English as a subject at high school. Thus, all respondents are expected to have sufficient understanding of English to not be disadvantaged while answering the survey.
The manner of data collection varied as described for the different sample groups.
General comments on the student population
Different cohorts were chosen to represent the students:(1) Swedish upper secondary school science students with no special interest or aptitude in chemistry.
(2) Swedish entry-level tertiary chemistry students with interest and aptitude for chemistry (inferred from their choice of university studies, and generally higher achievements) in a longitudinal study through their first year at university.
(3) A South African reference group outside the Swedish school system.
A student at the Swedish Natural Science Programme (Swedish National Agency for Education, 2012) takes one mandatory and one optional course in chemistry during which a total of 170–220 teaching hours are delivered in chemistry over two years. A student of the Technology Programme (Swedish National Agency for Education, 2012) takes one mandatory course of 85–110 study hours. In South Africa 50% of the course time in Physical Science is spent on chemistry, which over three years equals 210 hours of chemistry.
CH13 (N = 66)
This sample group comprised 191 Swedish tertiary level students in the first term of the programmes: chemical engineering, chemical engineering with physics, and biotechnology at Chalmers.|| The students were admitted based on their grades; the lowest grade accepted was an average (all courses in the upper secondary school exam) of 18.2 out of 22.5 grade points. Most of the students were drawn from the Natural Science programme, some from the Technology programme and a few students had different backgrounds. The students in this group could be expected to be interested in chemistry and were in general high achievers in the upper secondary school. This is evident from the number of students who achieved the highest grade MVG in Chemistry (66%).A short introduction to the survey was given during a lecture and the students were asked to use a web link to complete it in their own time. Two reminders were sent to request students to complete the survey. In total 35% (N = 66) of the students completed the survey. The survey was completed before students started their study of bonding in their first general chemistry course.
CH14 (N = 98)
This group is the same cohort as in the CH13, but after completion of the topic of bonding in the first university level chemistry course, i.e. the survey was completed in the beginning of their second term. The survey included mostly the same questions as in their first term but this time the questionnaire was presented as a paper-and-pencil test and time was allotted for them to complete the survey in class. This lead to different number of respondents compared to CH13 cohort.SWE (N = 346)
This group is drawn from students in the science programmes of the Swedish upper secondary school, the Natural science, Technology and to a lesser degree the IB-Diploma programmes.** A short e-mail invitation with an introduction, a web link and a request to forward the mail to the chemistry teachers, were sent to 285 upper secondary schools listed at the Swedish National Board of Education's website giving a mix of 80% government schools and 20% private schools. The teachers were asked to include only students who had completed the topic of bonding in the first mandatory chemistry course. The group contained students (N = 346) from schools randomly spread around Sweden and represented a cross section of students of these programmes having an average aptitude and interest in chemistry. Average grade data for all subjects were not collected for this group. The average grade in the first upper secondary chemistry course, Chemistry A/1, was 77% or 15.3 out of 20 pts. for the group. The number of students having received the highest grade, MVG, in Upper secondary school was much lower for this group (38%) compared to the CH13 group (66%).UCT (N = 188)
Admission to UCT is limited to students achieving an average of at least 71% across 6 subjects on the upper secondary school leaving exam. The lowest score of students accepted in this cohort was 71.5% while the average score for the UCT sample was 83.9%. The majority of the students admitted (88%) attended schools located in urban areas. The students have a mixed background with the largest language groups being English (63%), IsiXhosa (11%) and IsiZulu (6%), the remainder having a wide variety of first languages.They were introduced to the survey during a lecture a few weeks into their first term at UCT before any teaching about chemical bonding had taken place. Students were asked to complete the survey in their own time through a web link sent to their e-mail address. They were reminded three times to complete the survey. 188 students (42%) completed the survey. Of these students, 81% had graduated according to the old South African curriculum (NCS 2006). The remaining 19% were students of different nationalities, mainly from other southern African countries.
Curricula, textbooks and teaching situations
It is also relevant to investigate how and to what extent the different national textbooks describe aspects of bonding, bond energetics and molecular geometry. The use and choices of representational models, the teaching order and the starting point for the explanations are also important. These aspects as well as the three general foci of our study were used to create a matrix to give structure to the survey of the texts. The elements of the matrix shown in Table 1 were chosen based on the following factors:| Elements | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Textbook | Number of space filling models | Number of ball and stick models | Total number of representational models (BS + SF) | Shell/Bohr model main explanatory tool | Quantum models/atomic orbitals main explanatory tool | Describes and uses molecular orbitals to some extent when describing bonding | Order of introducing bond types | Uses the potential bond curve as a fundament for chemical bonding | Describes and uses the VSEPR model | Main descriptive model for ionic bonding | Energy level diagrams describing bond breaking and formation |
• recent findings on representations of chemical bonding models in school textbooks by Bergqvist et al. (2013) (1–3),
• known border-line case of quantum mechanics that may or may not be introduced in upper secondary school but where 1st year university chemistry starts (4–6),
• differing recommendations about order of introducing bond types by Taber (1997) and Dhindsa and Treagust (2014) (7) and
• specific detected differences in the curriculum documents of the two countries (8–11)
An excerpt of the matrix is shown in Table 1.
The individual teacher in the Swedish system decides which textbook to use, while only textbooks approved by the Department of Education may be used in South Africa. The four Swedish textbooks chosen were based on AN's experience and the five South African books (each consisting of two volumes) were selected after input from a South African researcher in chemical education who also has experience as a chemistry teacher.††
The following Swedish textbooks were selected for this study namely, Gymnasiekemi A (Andersson et al., 2000), Gymnasiekemi 1 (Andersson, et al., 2012), Syntes kemi 1 (Henriksson, 2011), Modell och Verklighet (Pilström, et al., 2011). The choice of the South African books included the following textbooks: Study and Master Physical Sciences Grades 10 and 11 (Kelder, 2005, 2006), Spot On – Physical Sciences Grades 10 and 11 (Elferink et al., 2012a, 2012b), Physical Sciences explained, Grades 10 and 11 (Jones and Berens, 2005, 2006), Everything Science Grades 10 and 11 (FHSST, 2010; Siyavula, 2012) and Oxford Successful Physical Sciences Grades 10 and 11 (Broster et al., 2005; Broster et al., 2006).
The survey instrument
The Swedish authors discussed certain common traits in student learning about chemical bonding at both the upper secondary and tertiary levels. This discussion and subsequent analysis of the literature lead to the development of the survey consisting of three main topics: (1) basic bonding, (2) structure and shape of molecules, and (3) properties and bonding. The first version of the questionnaire consisted of questions developed by the Swedish authors and questions from literature.The questionnaire was evaluated and adjusted after a pilot study in autumn 2012 and was validated by an upper secondary school chemistry teacher. At first an additional choice regarding properties of the chemical bond was included in the survey for the CH13 group. This additional choice made the interpretation of the answers more difficult, due to the fact that the students were presented with two opposing statements, which would have given them the information that only one of the two was correct. Therefore, to provide a better check of the students' knowledge of properties and bonding, the second choice was omitted from the following surveys. In the follow-up study with the CH14 group, the question, “What is true about chemical bonding”, was split into two, one dealing with energetics and one with properties, furthermore in the energetics question a fourth alternative (D CH14) was included to gain more information on this issue as the initial data analysis revealed that many students choose mutually exclusive responses. The modifications described above led to the final version of the survey which was distributed to students between 2013 and 2014 (Appendix 1). In this final version the basic bonding section (Q1 & Q2) contained two multiple-choice questions. The structure, shape and bonding section (Q3, Q4 & Q5) consisted of one multiple-choice and two open ended questions. The last part (Q6 & Q7) concerning properties and bonding had two multiple-choice questions.
Interviews
To gain further insights into the students' ideas of chemical bonding they were invited to participate in one-on-one interviews. The 6 semi-structured interviews took place between September and October 2013 and focused on bonding and atomic build up. The 7 main and the 12 subsequent situational questions were prepared by AN and discussed and validated by the Swedish co-authors (see Appendix 2). Each of the interviews lasted 25–40 minutes.Basic bonding
Two multiple-choice questions were designed for which the students were asked to mark all statements that they found correct. The first question, Q1, “What is true about waves?”, aimed to investigate whether the students have the required knowledge of basic wave functions crucial for understanding the quantum models of bonding, i.e. interference of waves and summation of trigonometric functions. The next question, Q2, focused on energetics as learners have been shown to exhibit misconceptions regarding the energetics of bond breaking and bond formation (Boo, 1998; Wren and Barbera, 2013). Included in this question is also one item regarding the connection between the properties and the bond type, since other studies (Lemke, 1990; Taber, 2001; Othman et al., 2008) have shown that students are inclined to believe that the inherent (macroscopic) properties of the substance determine the bond type and not vice versa. A sample question is shown below.What is true about bonding? (Correct answers are marked with (C))
(A) It takes energy to create a chemical bond from single atoms!
(B) When a chemical bond is formed, energy is liberated! (C)
(C) It always takes energy/work to break a chemical bond! (C)
(D) The properties (hardness, conductivity etc.) of the substance determines its bond type (ionic, covalent etc.)
(E) Sometimes energy is liberated and sometimes energy is needed when a chemical bond is formed. (This statement was only presented to CH14.)
(F) The bond type (ionic, covalent etc.) of the substance determines its properties (hardness, conductivity etc.) (C) (This statement were only presented to CH13.)
Structure, shape and bonding
Models play an important role in the learning and understanding of chemistry, and textbooks and lectures are rich in presenting different types of representational modes. (Coll and Treagust, 2003; Bergqvist et al., 2013) Based on empirical observations and the studies above we found it beneficial to investigate which models chemistry students' use when asked to describe the geometry of a methane molecule. For question 3, we choose four different shapes (Fig. 1), a wheel spanner, a caltrop, a tetrahedron and a ceramic tile, that in one way or another could be seen as a representation of a methane molecule. Students could only give one answer for this item. The caltrop makes sense for students used to ball and stick models. Tetrahedral representations indicate that the students understand that atoms can be seen as large spheres in close connection to each other, i.e. space filling models. Students choosing the wheel spanner and the ceramic tile may have problems relating atoms and molecules to three-dimensional structures. | ||
| Fig. 1 The representations that the student could choose from as being the “truest” depiction of a methane molecule. | ||
The latter part of this theme consisted of two open-ended questions, Q4 & 5, aimed at uncovering the conceptual frameworks the students employ when asked to describe the bonding and distribution of electrons in methane. The students were instructed to give an as comprehensive an answer as possible. When asked to explain a complex concept in a precise way there will be cues in the answers indicating to which extent a student have consolidated the fundamental and specific concepts regarding the subject.
Properties and bonding
AN's experience is in agreement with several studies (Butts and Smith, 1987; Taber, 1997; Taber et al., 2012; Vladušić et al., 2016) that an issue that confuses secondary level students is the difference between covalent and ionic bonding as well as molecular compounds and salts. We used a question, Q6, formulated in the chemical bonding instrument (Tan and Treagust, 1999) where we substituted NaCl for KCl. It is a two-tiered question, with an initial statement to be answered with true or false, in the second part the students are requested to choose one of four statements as being their rationale for the previous answer of true and false. In the last question, Q7, the students were asked to first identify the bonding in water and hydrogen sulfide as either inter- or intramolecular, and then choose a statement that explained their reasoning. This question was taken in its entirety from Tan and Treagust (1999).Data analysis
| Category | Description and an example of a typical answer |
|---|---|
| (1) Simple model | Lack of or with very weak conceptual explanations or descriptions. Ex “Each carbon atom is covalently bonded to a single hydrogen.” |
| (2) Conceptual covalent model | Use of a contextual explanation in which the student makes some reference to electron sharing, orbital overlap or bond types (covalent, sigma or π). Ex “By sharing of electrons via covalent bonding” |
| (3) Advanced model | Use of the accepted concepts of covalent bonding and incorporates, electron sharing, the octet rule and/or orbital reasoning in the explanation. Ex “through the sharing of electrons the four hydrogen's are able to fill up their orbitals with the carbon's valence electrons” |
| (4) Intermolecular. (Indicates a faulty model of covalent bonding) | Indication of the use of concepts and with evidence of that the view of the interaction is intermolecular. Ex “Carbon and Hydrogen are covalently bonded and are kept together by Van der Waals intermolecular forces.” |
| (5) Ionic interaction. (Indicates a faulty model of covalent bonding) | Indication of the use of concepts with evidence of the view of the interaction is of an ionic nature and/or due to charge attraction. Ex “Hydrogen atoms have a small positive charge and carbon atoms have a small negative charge. As like charges attract these atoms come together to form a bond. These intermolecular forces hold the atoms together.” |
| (6) General explanations or descriptions. | The respondent only gives a word/phrase without any elaboration or uses a very limited or general explanation. Ex “Single bonds from carbon to the 4 hydrogen atoms in a tetrahedral shape.” |
The analysis of Question 1 about waves, relationship between properties and bonds and the second open ended question, “Explain where you find the electrons in methane”, which relates to the localisation of electrons was found to be beyond the scope of this article and were therefore omitted from this analysis. The response rates are, however, reported and discussed at the end of this section.
Quantitative data from the surveys and textbooks
| Question/group | SWE (346) (%) | CH13 (66) (%) | CH14 (98) (%) | UCT (188) (%) |
|---|---|---|---|---|
| Bond energetics | 81 | 85 | 100 | 88 |
| Geometry of methane | 70 | 59 | 99 | 72 |
| Bonding in methane | 49 | 45 | 89 | 60 |
| Molecularity of KCl | 66 | 52 | 95 | 68 |
| Average response | 62 | 57 | 93 | 67 |
Findings
In the following sections we present data from the investigation of the first research question: “Are there differences between the groups regarding the understanding of chemical bonding concepts, and if so, can such a difference be traced back to specific concepts?”Students' understanding of bond energetics
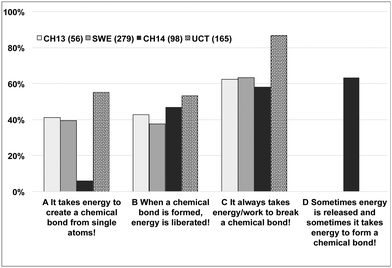
In the question on bond energetics, we asked the students to choose all the answers they considered to be correct from the four statements below:What is true about bond energetics?
(A) It takes energy to create a chemical bond from single atoms!
(B) When a chemical bond is formed, energy is liberated!
(C) It always takes energy/work to break a chemical bond!?
(D) Sometimes energy is released and sometimes it takes energy to form a chemical bond! (CH14 only)
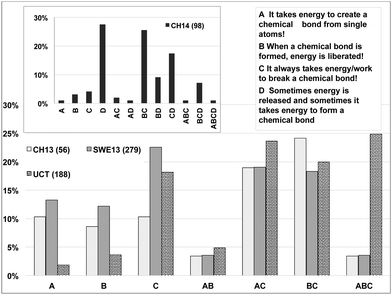
The students' responses are shown in Fig. 2 in the order: students entering Chalmers (CH13), Swedish high school students (SWE); Chalmers students after one term (CH14), students entering University of Cape Town (UCT) We note that the preferences of the CH13 group are similar to the SWE group, which is surprising given the different level of chemical aptitude of the two groups, as manifested in the difference in number of highest chemistry grades and choice of tertiary chemistry education. The more chemically inclined students were expected to have a higher retention and understanding of basic bonding concepts. However, when combinations of selected statements are considered, the expected difference between the two groups can be seen with the CH13 performing better on average.
 | ||
| Fig. 2 Question investigating the students understanding of bond energetics. Correct alternatives are B and C. Alternative D was only tested for the CH14 group. | ||
In the case of the UCT group, Fig. 2, we note that they have a higher preference for the incorrect alternative, A, with over half the UCT cohort choosing this alternative. The choice of response A for the SWE and CH13 Swedish groups is ∼15 percentage-points lower. The combinations of statements shown in Fig. 3 reveal that alternative A was almost always chosen in combinations AC and ABC, very few UCT students choose only A. The UCT students also have a high preference for choosing alternative C. The South African group is also dissimilar to any of the Swedish groups in the sense that the UCT group are more than five times as inclined to mark all three statements as being true.
In the data derived from combining choices of statements, Fig. 3, we find that the numbers of students choosing the correct combination of B + C for any of the groups is between 18–26%, indicating that few students have a clear grasp of bond energetics. The finding that approximately two thirds of the CH14 students included the incorrect alternative D in their combination of selected alternatives in the follow-up survey (Fig. 3, inset), however shows that bond energetics remains a problem even at higher levels in the educational system.
Textbooks and teaching aspects of bond energetics
We used the survey of textbooks to answer research question 2: “Are there differences between Sweden and South Africa at the curriculum level, and if so can these be related to the answers to question 1 and 3” “Is there a quantifiable difference between Sweden and South Africa when it comes to teaching in the classroom and the contents of the textbooks used?”A comprehensive survey of the textbooks described above reveals that there is a marked difference in the approach of how basic bond types and the explanations of bonding are presented in the Swedish textbooks compared to those from South Africa. This difference was quantified in Appendix 3.
A Morse potential energy diagram, showing the change in potential energy during bond formation, is used as a foundation for describing bonding in several places in the South African curriculum and in the South African textbooks, whereas it is neither mentioned nor used in the Swedish curriculum or textbooks. Moreover, the South African curriculum specifically states that teachers should emphasize that the main reason for bonding is the increased stability due to a potential energy minimum at a given inter-nuclear distance. No such guidance regarding stability and potential energy is given in the Swedish syllabus or textbooks and an informal survey of Swedish chemistry teachers reveal that they usually do not use this type of diagram when they introduce chemical bonding.
Instead, in the Swedish teaching tradition, as experienced by this group of teachers, chemical bonds are connected to energetics through the use of Born–Haber type diagrams depicting atomisation of reactants and the subsequent energy release as the new bonds form. Most of the Swedish textbooks use these diagrams to illustrate bond energetics but such illustrations are generally missing from the South African books. The South African curriculum and textbooks instead use text-based descriptions to emphasise that energy is released when a single bond forms and that energy is absorbed when a single bond is broken. These written definitions are usually less visible in the Swedish textbooks, and the students are often left to infer this for themselves from the diagrams supplied.
Students' choices of geometrical representation for the shape of a molecule
The following question (Q3) was posed to the students:If you were to photograph a methane molecule, which of the representations above would be the closest to the actual shape? (See Fig. 1 for shapes presented to the students).
The radar diagrams of students responses shown in Fig. 4 reveal that most students have grasped the 3-dimensionality of molecules as they choose either the caltrop or the tetrahedron. Almost none of the students choose the ceramic tile as their favoured representation.
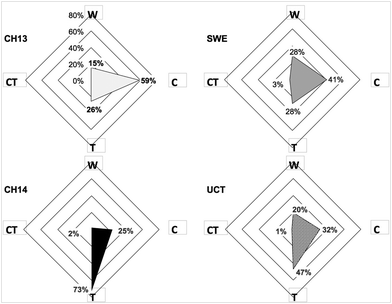 | ||
| Fig. 4 Radar diagram of student responses to the multiple-choice question: “If you were to photograph a methane molecule, which of the representations above (see Fig. 1) would be the closest to the actual shape?” The sharpest point in the figure indicates the most preferred representation; a blunter shape indicates a lower preference for any given representation. W = wheel spanner, C = caltrop. T = tetrahedron, CT = ceramic tile, see Fig. 1. | ||
We note that some groups have a clear preference which is visible in the sharp points in the diagrams in Fig. 4. The choice of the CH13 group is the caltrop while a semester later the CH14 cohort favours the tetrahedron. The students in the less chemically inclined SWE group display no specifically favoured visualisation. The UCT group is similar in their choice of representations to the CH14 group.
Textbook and teaching aspects of geometrical representations and models
There is a large difference in the use of visual representational models, i.e. ball and stick or space filling, between the Swedish and the South African books. The latter are much more limited with respect to the use of illustrations, using less than half the number of visualisations compared to those used in Sweden. In the Swedish books we also note a shift from a majority (59%) of ball and stick models in the old curriculum Chemistry A (Andersson et al., 2000) towards more space filling models (56%) in the books for the new curriculum Chemistry 1 (Pilström, et al., 2011; Andersson, et al., 2012). In the South African books, apart from Everything Science by Siyavula, the dominant model of representation for methane is the ball and stick model (56–67%).If we analyse the overall use of illustrations the difference is even more profound in particular for molecular representations where the Swedish books use between 39 and 59 illustrations whereas the South African books use 6 to 25 such illustrations (Appendix 3). The Swedish books have at least one illustration on each page, these illustrations are very varied and use anything from pictures, to visual models, to drawings of molecules and atoms in anthropomorphic or animistic settings (Bergqvist, et al., 2013, p. 594). The books from South Africa have, in general, much fewer illustrations. In particular, anthropomorphic or animistic representations were not found in any of the South African books. An exception is the Oxford Successful series which is similar to the Swedish books in terms of number of illustrations, but is more restrictive in the use of anthropomorphic or animistic representations.
To some extent all books introduce and try to visualize the three-dimensionality of chemical compounds. This is mainly done by using ball-and-stick and space-filling models, sometimes inscribed in geometrical shapes, e.g. a tetrahedron for methane. The foundation for these representations is VSEPR theory which is both introduced and explained in the South African books but this is not the case in the Swedish books where it is only implied.
While space-filling models are fairly well represented in the textbooks, there is a large predominance of the use of ball and stick models in the actual teaching situations in Sweden since most chemistry kits provided are of this type. One of the respondents in the interviews describes the learning situation: “We mostly worked with ball and sticks, when we had things to test by ourselves” The use of models at schools in South Africa is more limited than in Sweden due to the cost of prefabricated model kits. According to an experienced South African teacher and researcher, only about 500 schools out of approximately 6500 have any kind of model kits. To compensate for this lack of model kits it is recommended in the curriculum to make use of play dough and tooth picks to construct 3D models of molecules and compounds.
Student’s descriptions of the bonding in methane
In this open-ended question, Q4, the students were asked to answer the following question:Describe in detail how the carbon atom and the hydrogen atoms are held together in the methane molecule!
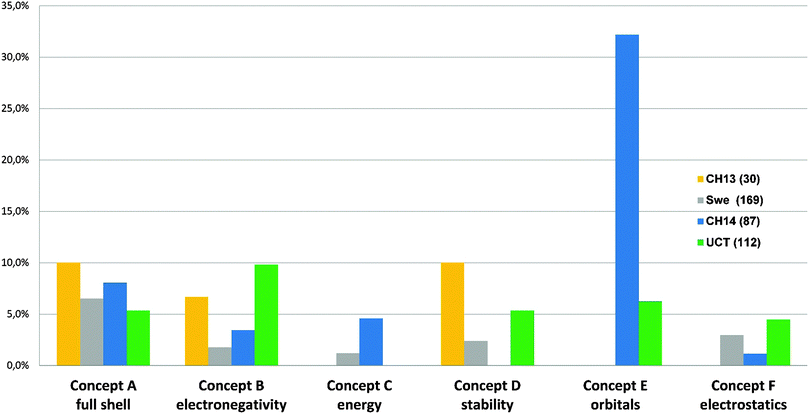
Students' responses were classified according to the framework in Table 2. In Fig. 5 we note that most students used an explanation that adheres to a covalent framework for chemical bonding. It is also clear that few of the students see bonding as being intermolecular or as a result of charge attraction. In terms of the depth of the answers (categories 1–3) we find that the less chemically inclined SWE group has the highest incident of simple answers, while the CH14 group gave more advanced explanations. The UCT students' explanations are on average of a higher conceptual level (category 2 and 3) than the Swedish CH13 or SWE students.
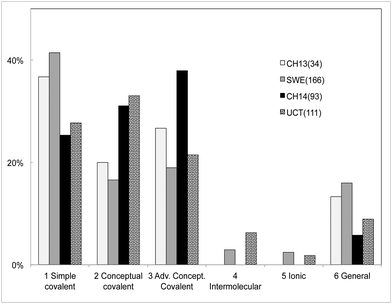 | ||
| Fig. 5 Classification of open-ended answers explaining bonding in methane. See Table 2. | ||
On a more detailed level it was found that the South African students have a higher inclination, compared to the CH13 group, to include orbital and quantum explanations in their descriptions of the bonding in methane, see Appendix 4. In the initial survey of the CH13 cohort, no students used orbital or quantum concepts, however, in the CH14 group these explanations have increased to almost 35%. We interpret this change in the pattern of responses as conceptual advancement during the first term of higher chemistry education.
Students' views of ionic bonding and lattices
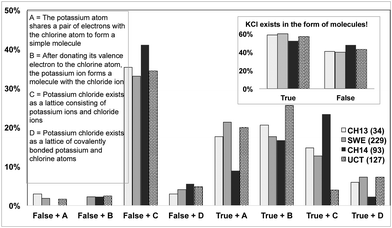
In this two-tiered question, Q6, the students were asked to choose whether the statement about KCl was true or false and then to give the reason for their answer.Potassium chloride exists in the form of molecules! True or False!
(A) The potassium atom shares a pair of electrons with the chlorine atom to form a simple molecule!
(B) After donating its valence electron to the chlorine atom, the potassium ion forms a molecule with the chloride ion!
(C) Potassium chloride exists as a lattice consisting of potassium ions and chloride ions!
(D) Potassium chloride exists as a lattice of covalently bonded potassium and chlorine atoms!
The inset in Fig. 6 shows that in most of the groups the majority of the students have a molecular view of potassium chloride. In addition, it shows which explanatory choices the students made to rationalize their initial true or false statement. There seems to be a very sharp dividing line between the students knowing that KCl does not exist in the form of molecules and those who believe that KCl exists as a molecular entity in the solid phase. The former student group almost always chose the correct alternative C, while among the students who have the molecular view, the choice of explanation indicates no strong preference. Even though many students state that KCl is a molecular compound, few of these students indicate that they see the lattice as being of a pure covalent type (D).
The UCT and CH13 students have a slight preference for alternative B. The SWE group on the other hand prefers alternative A which is more consistent with the molecular view. Another interesting fact in this figure is that even after completing entry-level tertiary chemistry many students in the CH14 group retain the view of KCl being a molecule which is in line with the findings of Nicoll (2001).
Textbooks and teaching aspects of chemical bonding
The content coverage of bonding in the two countries differs mainly in the case of the foundation for bonding, where the focus in South Africa is on the electrostatic nature of all bonds and an emphasis on the octet rule, (depicted in Lewis dot formulas), while the Swedish curriculum and books only discusses the electrostatic framework very briefly.In the Swedish curriculum for Chemistry A the following paragraph is the complete description of the learning outcomes of the chemical bonding section in chemistry, “Be able to describe how models of different types of chemical bonding are based on the electronic structure of the atom and be able to relate the properties of elements to type of bonding and its strength, as well as to the structure of the element” (Swedish National Agency for Education, 2000, p. 56).
The current Swedish curriculum uses the same brief descriptions for the bonding content of the course, “Models and theories of the structure and classification of matter. Chemical bonding and its impact on e.g. the occurrence, properties and application areas of organic and inorganic substances.” (Swedish National Agency for Education, 2010) The South African NCS curriculum is much more detailed in describing the learning outcomes of the chemical bond, but omits the metallic and ionic bonds. The following quote from the NCS content document (Department of Education, 2006) illustrates the level of detail in the South African curricula, “-Describe a chemical bond as a shared pair of electrons. -Describe and apply simple rules to deduce bond formation, viz.: -Different atoms, each with an unpaired valence electron can share these electrons or form a chemical bond, -different atoms, with unpaired valence electrons can share these electrons and form a chemical bond for each electron pair shared (multiple bond formation).
The way in which chemical bonds are introduced and modelled varies between the books. The South African NCS curricula emphasizes that bonding should be explained in the following way, “A chemical bond as the net electrostatic force two atoms sharing electrons exert on each other” (Department of Education, 2006). The exclusion of metallic and ionic bonds from the NCS curriculum is also observed in some of the textbooks, where the book, Physical Sciences Explained, omits metallic bonding and the Spot On series excludes both ionic and metallic bonding. All other South African books examined include all basic bond types. Swedish chemistry books commonly start with the ionic bond followed by the covalent and lastly the metallic bond, the South African books starts with either the covalent or metallic bond. All the South African books also introduce the dative bond, which is omitted from the Swedish books.
Shared valence electrons and the octet rule constitute the main explanation of covalent bonding in all textbooks but the South African books use the Lewis dot formulas more frequently. Three of the Swedish books mention that the electrons reside in a shared electron cloud surrounding the molecule. The South African book, Everything Science, (2012) takes the explanation further and uses the concept of overlapping orbitals instead of shared electron clouds. In the Study and Master series even more detail is given as it uses hybridisation of atomic orbitals to explain bonding and geometry. Some Swedish textbooks have a short extra-curricular section where atomic orbitals are introduced, but the level of detail is much less than what is presented in the South African curriculum. Most of the South African books use an orbital description, and employ Hund's rule and Pauli's exclusion principle to explain how electrons fill the orbitals and why some electronic structures are stable and others not.
The South African books mainly rely on the octet rule when describing the ionic bonding and the fact that the atoms obtain full octets upon formation of ions. Thus Lewis dot diagrams, electron transfer and the subsequent charge attraction are used as the foundation for explaining the ionic bond. Two of the Swedish books (Henriksson, 2011; Pilström et al., 2011) clearly describes an electrostatic model as the explanation for ionic bonding, these two books also clearly separate the ionic bonding from the formation of ions. The two books by Andersson et al. (2000, 2012) indicate that ionic bonding is connected to the transfer of electrons to complete the octet rule.
All books discussing the ionic bond start with single atoms or ions. These books also introduce and explain the lattice concept. The common representational choice is to depict the crystal lattice with ball and sticks. None of the textbooks analysed try to explain how to interpret the depicted lattice model in terms of bonding interactions and only one book emphasises that the bonding is non-directional. The different ways of describing and explaining ionic bonding might yield very different results as has been observed by AN in teaching situations. Students taught ionic bonding through the octet framework and electron transfer struggle to understand why silver chloride precipitates when aqueous solutions of sodium chloride and silver nitrate are mixed together. This confusion is visible in the following discussion about precipitation, observed by AN during a practical activity.
Student A: “What makes them stick together?… I mean they both already have full octets!…
Student B: No idea!… No electrons are transferred! Is it a salt?… Silver is +1 and chloride is −1, I know opposites attract but why these two and not the others?…”
Bewilderment about schematic representations in figures of a lattice, as reported previously by Coll and Treagust (2003), are also common in AN's experience, i.e. when asked: “What do the lines in the picture mean?” Student C reveals her uncertainty: “-aren't those the bonds? In between the atoms!” Many also struggle with the concept of formula units and molecules as revealed in this statement from Student D: “we didn't differentiate between molecules and ionic compounds (From the interviews with CH14 students)”.
Discussion
Our research indicates that the Swedish and South African upper secondary school chemistry education fails for the most part to help students to generate a correct understanding of some of the basic aspects of chemical bonding; bond energetics, molecular geometry and ionic bonding. For the students in Sweden this general picture agrees with the more in-depth study of a limited number of Swedish students by Taber and Adbo (2013).Student conceptions of bond energetics
All groups display misconceptions about energetics related to the breaking and formation of bonds, Q2, however, the majority of students correctly understands that bond breaking is associated with an uptake of energy. Fewer students make the connection that bond formation releases energy (statement B) and only about 20% chose both correct statements (B + C), see Fig. 2. Comparing the UCT group to the Swedish groups we note that the South African students are more inclined to choose the combinations A + B over B + C than the other groups, that is they are more inclined to see both breaking and formation of a bond as requiring energy. 63 percent of the CH14 cohort choose combinations with D “Sometimes energy is liberated and sometimes energy is needed when a chemical bond is formed” (Fig. 3, inset). These result shows that bond energetics remains a problem even at higher levels in the educational system.In contrast to previous studies (Boo, 1998; Wren and Barbera, 2013) we investigated how the students rationalize about individual bonds and not the net overall reaction or more specific thermochemical terms, thus it is interesting to see that our data is consistent with those found in the previous study. Like Boo we found that only few students are capable of reliably describing the energetics of bond breaking and bond formation. We also note that in the CH14 group many students choose the alternative D which is interpreted as an indication of a blending of the concepts of individual bond forming and the concept of reactions being exo- or endothermic. It is our opinion based on empirical observations over many years of teaching, that the use of exothermic and endothermic examples when discussing bonding are one of the largest contributions to this confusion of energetics of bond breaking and formation. Examples used in teaching include formation of e.g. solid sodium chloride or hydrogen iodide:
(1) 2Na(s) + Cl2(g) → 2NaCl(s) + heat
(2) H2(g) + I2(g) + heat → 2HI(g).
When examples such as these are presented they are often accompanied by a schematic energy level, Born–Haber type, diagram of the reaction. This is especially problematic since these diagrams are quite similar to enthalpy diagrams used to visualise overall reaction energetics, as being either as exo- or endothermic processes depicted in Fig. 7. A student not versed in how to interpret enthalpy or energy level diagrams and what they signify, may easily deduce that in the case of an endothermic reaction, bond formation is associated with an uptake of energy.
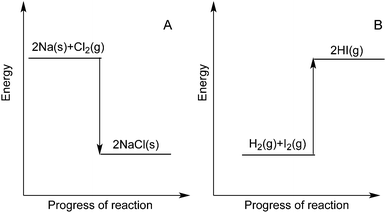 | ||
| Fig. 7 Traditional enthalpy reaction diagram depicting A, an exothermic reaction and B, an endothermic reaction. | ||
Aspects of molecular geometry and the bonding in methane
Most Swedish chemistry students in grade 10–12 as well as those starting tertiary level prefer the caltrop as their representation of methane, Q3, whereas 47% of the South African students choose the tetrahedron, see Fig. 4. From the data it can be argued that the curriculum for Chemistry A (Swedish National Agency for Education, 2000, 2010) taught in the Swedish upper secondary school, seems to foster a view of atoms and molecules as being close in shape to the ball and stick models (see CH13 in Fig. 4 as an example). The South African students have a slight preference, compared to the Swedish groups (CH14 excluded), for the tetrahedral shape. The slight preference for the tetrahedron within the group of South African students might be due to a scarcity of models in textbooks and a lack of ball-and-stick building kits in the classrooms. The lack of model kits, though problematic in many aspects, might have a slight positive influence; since it allows the teacher to place more emphasis on the space filling aspect of atoms and molecules in the learning activities. The fact that the students have to construct their own models of atoms and molecules from play dough etc. may lead them to a better understanding of the role of models in chemistry.Another difference between the South African and the Swedish curricula is the emphasis on the VSEPR theory in the former and the lack thereof in the latter. The UCT students, who have been exposed to the VSEPR theory during their upper secondary school education, show a much higher preference for the tetrahedron than the Swedish students whose secondary chemistry education was less rich in examples of the VSEPR theory. This finding is supported by the preference of the tetrahedron for the CH14 group, who are given a rich introduction to Lewis dot formulas, VSEPR theory and the geometrical shapes that follow from this during their first semester at the University.
In the results from the open-ended question about bonding in methane, Q4, there is a clear trend for the South African students to use a higher explanatory level than the Swedish groups of similar background. It can be argued that the richer foundation given to the South African students in terms of knowledge of the potential energy curve for bonds, atomic orbitals, Hund's rule, Pauli's exclusion principle and to some extent molecular orbitals or hybridisation equip them with a larger explanatory toolbox. In this sense our data are similar to the more in-depth study of conceptualisation by students performed by Adbo and Taber (2014). The UCT group, to a greater extent, also use concepts related to stability, electrostatics or quantum mechanics concepts to explain the bonding in methane, compared to the SWE or CH13 groups.
The lack of even the most basic quantum mechanical models in the Swedish textbooks and teaching situations clearly disadvantages the students when trying to understand the concept of bonding and especially the covalent bond. This conclusion is supported by inspection of the answers provided by the CH14 group, who studied chemical bonding at the tertiary level and thus have been exposed to quantum models etc. The students in this group show a higher adaptation (35%) to advanced conceptual explanations containing quantum mechanical or molecular orbital concepts compared to the other Swedish groups. Data from classroom observations and the answers in the survey, Fig. 5, agree with Taber and Adbo (2013) who found that many students misconceptions were caused by an “explanatory vacuum” i.e. in the absence of adequate tools creative students construct their own, faulty models. Our findings reveal that many students struggle to come to terms with the conflicting models presented to them.
Aspects of ionic bonding and lattices
A majority of students (55–60%) from all educational levels or curricula have a molecular view of the sub microscopic structure of a salt i.e. they answer true to the statement: “Potassium chloride exists in the form of molecules!”, Q6. This result is slightly better than that reported in the original study by Tan and Treagust (1999), which showed that 80% of the upper secondary school students in Singapore had a molecular view of NaCl.‡‡It can be seen in the textbook survey (Appendix 3) that the concepts of bonding in general are not introduced in a way which would minimize the formation of a molecular framework, as proposed by Taber (1997)i.e. first metal then ionic and lastly covalent bonding. It is very common to start the teaching of ionic bonds with a single metal atom that reacts with a single non-metal atom forming a formula unit of, for instance, NaCl or some similar compound. A formula unit drawn on the board or presented in a book, without proper explanation, is identical to that for molecular compounds such as hydrogen chloride, thus there is a risk of creating a picture of a salt being a molecular compound. In lectures, classes and discussions AN has overheard many lecturers, teachers and students use statements such as: “This sodium chloride molecule then reacts…” and other similar examples which might be more an indication of sloppy vocabulary than a faulty understanding, it still points to the fact that care must be exercised when discussing these issues.
Together these approaches promote the view of ionic compounds as consisting of molecules. Many students also have a confused picture of the actual bonding inside the lattice. Even among the groups (UCT and CH13) expected to have a higher aptitude and interest in chemistry, most students view KCl as being molecular and have trouble explaining how the lattice is constructed. Many of these students employ what could only be described as a hybrid bond model, visible in the choice of alternative B “After donating its valence electron to the chlorine atom, the potassium ion forms a molecule with the chloride ion!”. They know that KCl contains ions and that the ions are paired but not how they are held together. The mental model of these students then leads to the idea that the sets of ion pairs each constitute a molecule. It is probably inherent in this model that the students fail to see ionic bonding as being non-directional; instead it is strictly seen as a bond between the two ions. From this, the molecular build-up of the salt follows, since this is the only way for them to make sense of all the contrasting facts.
In the textbooks surveyed we have found that ionic bonding is not always presented clearly and with little emphasis on the charge attraction and its non-directionality (see Appendix 3). Many textbooks instead mix their explanations with the formation of ions and pairing of ions in the formula unit. These aspects together with the less than stringent use of phrasing and illustrations, shown to be present in the learning situation, probably leads to the formation of the hybrid framework described above.
For the students knowing that the initial statement is false there is a strong correlation of also choosing the correct explanatory statement C, “Potassium chloride exists as a lattice consisting of potassium ions and chloride ions!” From the answers it is clear that if the students through learning activities come to know how the ionic compounds are held together and learn to differentiate between a formula unit and a molecule, it may help them to visualize the ionic lattice in the correct way.
The results of the UCT group are similar to the Swedish groups, which is intriguing since the ionic bond was largely excluded from the NCS curricula. It is clear from the results that most South African chemistry teachers employed their pedagogical content knowledge and introduced and talked about ionic bonding in their classes, even if it was absent from the curricula. On this topic it can be noted that the lack of ionic and metallic bonding in the NCS curriculum has been remedied and these topics are included in the new CAPS syllabus (Department of Basic Education, 2012).
It is also important to note that this molecular framework persists even after tertiary level education on bonding (see CH14, Fig. 6). This indicates that this idea is probably set early on and is given a high status. The framework is further enhanced through the improper or less stringent use of schematic models of reactions, oral or written explanations and visualisations of ionic and molecular compounds in different learning situations. These aspects together make the alternate framework theory highly resistant to change, which is visible when comparing the results from the CH13 and CH14 groups. (It should be noted that the response rate to all questions is higher for CH14, see Table 3 this is probably an effect of the data collection method using a paper-and-pencil format for the survey.)
The first chemistry course at university level at Chalmers§§ clearly fails to decrease the prevalence of aspects of the molecular framework, as there is no significant difference in the response to the initial statement between the CH13 and CH14 groups. In one sense the tertiary-level education seems to be at least partly successful in fostering a proper view of the build-up of a lattice as shown by their choice of the correct alternative C where the preference for this alternative increased by 20% during the semester at Chalmers. One explanation for this result might be that the secondary level chemistry textbooks do not always properly introduce and explain the concept of the ionic lattice. During the higher level chemistry, more emphasis is laid on the aspects of lattices. This change of focus in tertiary level chemistry is also visible in Coll and Treagust's (2003) cross age study of ionic bonding, where the undergraduate and graduate students to a much higher degree used the ionic lattice as a starting point for explaining ionic bonding.
Limitations
In terms of the responses we do not know what considerations governed the teachers who received the request for participation, in the decision to involve their students. It might affect the result if it were a majority of students from progressive teachers. A further limitation is that the response frequencies for some questions are low.All questions in the survey must be interpreted with the knowledge that respondents might underachieve due to time constraints, disinterest etc. as shown by Trusted (1987) who found that a strong social imperative or the possibility of individual reward greatly influences respondents to make a much larger effort in answering. This is an aspect all questionnaires suffer from and especially open-ended questions are strongly affected by this. The ongoing discussion in Sweden regarding PISA etc. is precisely that students might underachieve in surveys etc., since they do not feel that it is included in their grade assessment. Our study included only one item testing energetics and this was of the multiple-choice type. What was missing from the energetics item was a statement checking how many students actually associate bond breaking with a release of energy, since this misconception is very common in classroom observations during the first author's ten years of teaching the subject. The different curricula strive to a varying degree to influence the teaching practice in the classroom, but there is no direct link between the curricula and the actual teaching practice.
In the follow up study with the CH14 group we added a fourth statement which might have affected the results for the three other statements. Another limitation is that when sorting choices in combinations of statements as in Fig. 2 some combinations will contain only a handful of students. The main results though are sound and do not suffer from very small numbers of respondents.
In the case of the bonding in methane question, the number of students giving elaborate and advanced answers containing specific concepts was small and therefore only general trends can be identified. The use of second order concepts given in the explanations, such as orbital theory, stability and energetics etc. are especially affected by lack of sufficient answers and cannot be used as anything else than trend indicators. The use of these concepts would need further investigation to give the complete picture.
Implications for teaching
The use of diagrams of energetics without thorough elaboration of the fate of the individual bonds can clearly foster a synthetic mental model congruent with the data obtained in the study. The term individual bond in this sense includes the breaking and formation of separate bonds, as for the processes shown in Fig. 7 above. Teachers need to think about how they present the topic of bond energetics so that they can avoid pitfalls such as the confusion between enthalpy and energy level diagrams. When using these types of diagrams care must be exercised to explain how they are to be interpreted. Another aspect to be considered is that teachers need to be more thorough in describing and modelling the energetics of individual bonds and firmly separate them from the already often misunderstood concepts of endo- and exothermic reactions.We observed that students who had been exposed to more information about the VSEPR theory have a higher tendency to view the geometry of the methane molecule as being a tetrahedron. We therefore suggest that the VSEPR theory should be given more time in the Swedish curriculum in the same way as it is in the South African curriculum. We can also see that students in South Africa, who are more aware of the restrictions of models in science due to specific activities stated in the syllabus, have a higher frequency of choosing the tetrahedron. The suggestion is therefore to let all students build and work with different kinds of models and preferably not only the prefabricated ball and sticks so common in Sweden and elsewhere.
Based on data in the study we suggest that it is beneficial to the students understanding of bonding if they meet and become familiar with at least a simplified quantum model for the build-up of the atom at the secondary level. Our recommendation is to include these concepts in the Swedish school curriculum.
When teaching about ionic compounds and lattices of salts care should be taken to avoid creating images of ionic compounds based on single ions or pairs thereof. This can be avoided if both lattices and single ions are used as the explanatory model for the ionic bond. The teacher should be careful not to depict and discuss ionic bonding based only on the formula unit, due to its resemblance to a molecule. It is also essential that the teacher is careful not to use expressions that may suggest similarities to molecules. To help the student correctly understand the bonding inside the lattice, the electrostatic aspect of the bonds and the fact that these bonds are non-directional must be stressed more strongly. Teachers and textbook authors should be careful to separate the formation of ions from the explanations of ionic bonding, since lack of separation can lead to a strong attachment of bond formation to electron transfer and the octet rule. Common to this mental model is that if there is no electron transfer there can be no ionic bonding. There is a risk of fostering this view in the South African students since in the CAPS document (Department of Basic Education, 2012) the following guidance is given to the teachers: “Ionic bonding: transfer of electrons in the formation of ionic bonding, cations and anions, electron diagrams of simple ionic compounds, ionic structure as illustrated by sodium chloride”.
Another issue that our studies and others (Harrison and Treagust, 1996) indicate is that models and diagrams showing bonding in terms of ball and stick representations should be complemented with space filling representations. This could minimize the risk of fostering the mental model of atomic shape, reflected in the choice of the caltrop in Fig. 1, or in the molecular mental model of a salt (Fig. 6). Care should be taken to always discuss models in terms of their strengths and weaknesses.
Recommended activities further include the use of electronic diagrams and physical models for describing formation of ions and the ionic bond. As in other studies, our findings indicate that teachers at university manage to influence the students into appreciating the importance of the crystal lattice of a salt. For example, it is possible to construct a model of an ionic solid using polystyrene spheres of different colours as has been done by BD in her first year course at UCT. The molecular model of the lattice shown to persist even at tertiary and graduate level is harder to remedy and teachers and lecturers need to be more thorough in describing the internal bonding and non-directional ionic build-up of a lattice.
Conclusions
Based on our findings we can assume that only about 20% of the first year students that university lecturers and instructors meet have a clear grasp of the concept of bond energetics. Most students confuse individual bond formation/breaking with exo- and endothermic processes, probably due to the use of examples such as the exothermic formation of solid sodium chloride from its elements as an example for teaching bond formation and breaking.Models used in teaching are taken at face value and seen as true representations of molecular shapes where an over-use of ball and stick models can lead to students having difficulties in the proper visualisation of the shape of molecules. We find that students versed in the VSEPR theory from secondary school have a better grasp of molecular shapes.
Teachers should be aware of that in a class of, students who have studied chemical bonding, only about half may have a correct model of ionic bonding. It is also important to keep in mind that the modelling of an ionic lattice on molecular building blocks has been shown to be retained by students even after the first tertiary level chemistry course.
Finally, we would like to point out that the somewhat divergent material covered in schools in the two countries gave rise to a difference in response patterns also to questions not directly related to these specific differences.
Appendix 1: survey instrument used in 2013 and 2014 (Groups CH13, SWE, UCT and CH14)
Section 1: basic bonding
Q1 What is true about waves?Mark all the answers you consider to be correct!
□ The sine function describes a wave motion!
□ Two waves that meet on a surface always enhance each other!
□ Two waves that meet on a surface extinguish each other in certain positions!
□ The square of a sine function is always positive!
Q2 What is true about chemical bonding?
Mark all the alternatives which you find to be true!
□ It takes energy to create a chemical bond from single atoms!
□ When a chemical bond is formed, energy is liberated!
□ It always takes energy/work to break a chemical bond!
□ The properties (hardness, conductivity etc.) of the substance determines it's bond type (ionic, covalent etc.)
Section 2: structure, shape and bonding
Different types of representations of a methane molecule!Q3 If you were to photograph a methane molecule, which of the representations above would be the closest to the actual shape?
○ Wheel spanner
○ Caltrop (foot trap)
○ Tetrahedron
○ Ceramic tile
Q4 Describe in detail how the carbon atom and the hydrogen atoms are held together in the methane molecule.
Use as much detail as possible
Q5 Describe in detail in which region you would find the different electrons in the methane molecule.
Section 3: properties and bonds
Q6 Potassium chloride, KCl, exists in the form of molecules!○ True
○ False
Choose among the statements below the reason for your answer to the question above
○ The potassium atom shares a pair of electrons with the chlorine atom to form a simple molecule!
○ After donating its valence electron to the chlorine atom, the potassium ion forms a molecule with the chloride ion!
○ Potassium chloride exists as a lattice consisting of potassium ions and chloride ions!
○ Potassium chloride exists as a lattice of covalently bonded potassium and chlorine atoms!
Q7 Water (H2O) and hydrogen sulfide (H2S) have similar chemical formulae and structures. At room temperature, water is a liquid and hydrogen sulfide is a gas. This difference in state is due to:
○ The forces between the molecules
○ The forces within the molecules
Choose among the statements below the reason for your answer to the question above
○ The difference in the forces attracting water molecules to each other and those attracting hydrogen sulfide molecules to each other is due to the difference in strength of the O–H and the S–H covalent bonds
○ The bonds in hydrogen sulfide are easily broken whereas those in water are not!
○ The hydrogen sulfide molecules are closer to each other, leading to greater attraction between molecules!
○ The forces between water molecules are stronger than those between hydrogen sulfide molecules!
Appendix 2: interview questions
A total of six semi structured interviews were conducted in Swedish.(1) Draw and describe the buildup of a calcium atom
Situational questions (to be asked if appropriate)
What do the rings symbolise?
What is the difference between two shells/orbitals?
Why do you start filling the N-shell before the M shell is completely full?
Do you know of any other way to represent the location of the electrons?
(2) What types of chemical bonding is there? Explain how they differ
Situational questions (to be asked if appropriate)
Are there big differences between them?
Give one example each of the different types of bonding?
(3) Which atoms form covalent bonds?
(4) Explain how the bond arises between the atoms
Situational questions (to be asked if appropriate)
What happens if the shell is full?
What is the influence of the electronegativity?
Where are the electrons?
(5) Are there any 100% covalent or ionic compounds?
(6) Draw a picture of a salt (NaCl) and explain how it is held together and built up!
Situational questions (to be asked if appropriate)
What constituents are there in the crystal?
Do the bonds you've drawn differ? (alternatively the bonds that you see in the presented model)
(7) The elements in the middle part of the periodic table, what are they called and what is special about them?
Situational questions (to be asked if appropriate)
How is it possible for these elements to have differently charged ions?
Appendix 3
Account of the survey of textbooks from Sweden and South Africa. The Swedish curriculum is not included since it is text based and do not particularly specify any of the terms or models below| Book | Number of space filling models | % of all model representations | Number of ball and stick models | % of all model representations | Total number of representational models | Shell/Bohr model as main explanatory tool | Qauntum models /atomic orbitals as main explanatory tool |
|---|---|---|---|---|---|---|---|
| Swedish curriculum | |||||||
| Syntes kemi 1 | 16 | 36% | 29 | 64% | 45 | Yes | 1/2 page extra curricular |
| Gymnasiekemi 1 | 27 | 56% | 21 | 44% | 48 | Yes | No |
| Modell och verklighet | 33 | 56% | 26 | 44% | 59 | Yes | 1 page extra curricular |
| Gymnasie kemi A | 16 | 41% | 23 | 59% | 39 | Yes | No |
| South African curriculum | |||||||
| NCS Syllabi | Yes | No | |||||
| CAPS syllabi | Yes | Yes | |||||
| Siyavula gr 10 gr11 CAPS | 16 | 64% | 9 | 36% | 25 | No | 6, 5 pages |
| Siyavula gr 10 11 NCS | 11 | 73% | 4 | 27% | 15 | No | Yes 5 pages aufbau, pauli |
| Physical sciences explained gr10, gr11 | 4 | 44% | 5 | 56% | 9 | Yes | No |
| Spot on-Physical Sciences | 2 | 33% | 4 | 67% | 6 | No | 3 pages |
| Study and master Physical Sciences | 4 | 44% | 5 | 56% | 9 | No | 7, 5 pages +2, 5 pages on hybridisation |
| Oxford successful Physical Sciences grade 10 and 11 | 4 | 36% | 7 | 64% | 11 | Yes short | No only in terms of quantisation of energy |
| Book | Describes and uses molecular orbitals to some extent when describing bonding | Uses the potential bond curve as a fundament for chemical bonding | Order of introducing bondtype s; I = ionic, C = covalent, PC = polar covalent, M = metallic bond | Describes and uses the VSEPR model | Main descriptive model of ionic bonding | Energy level diagrams describing bond breaking and formation |
|---|---|---|---|---|---|---|
| Swedish curriculum | ||||||
| Syntes kemi 1 | e-clouds mentioned Metal bonding explained as common e-cloud | No | ICPCM | Not mentioned but the idea is present, mainly in terms of lone pair repulsion | Charge attraction as main model. Both formula unit and lattice depicted and explained. | The energetics part not mentioned together with bonding |
| Gymnasiekemi 1 | e-clouds mentioned Metal bonding explained as common e-cloud | No | MICPC | Not mentioned but the idea is present |
Electron transfer due to the octet rule as main model.
Both formula unit and lattice depicted and explained |
2 step energy level diagram |
| Mod ell och verklighet | e-clouds | No, but explained in a way that makes it possible to construct the curve | ICPCM | Not mentioned but the idea is present | Charge attraction, points out that bonding is directionless, lattice and formula unit depicted. | 2 step energy level diagram |
| Gymnasie kemi A | Shared e clouds constitutes the bond | No | ICPCM | Not mentioned but the idea is present |
Electron transfer due to the octet rule as main model.
Both formula unit and lattice depicted and explained |
No connection made |
| South African curriculum | ||||||
| NCS Syllabi | No | Yes | Only covalent and polar covalent bonding included | Yes | Ionic bonding not introduced | No |
| CAPS syllabi | No | Yes | CIM gr10 C PC grade 11 | Yes | Transfer of electrons | Yes |
| Siyavula gr 10 gr11 CAPS | Yes | Yes | CIPCM | Yes | Transfer of electrons | Yes |
| Siyavula gr 10 11 NCS | AO overlap | Yes | CPCIM | Yes | Electron transfer due to high electronegativity then charge attraction, lattice and formula unit | Yes in text no diagram |
| Physical sciences explained gr10, gr11 | No | No, but explained in a way that makes it possible to construct the curve | CICPC, metal bonding missing in matter module | Yes | Mainly caused by electron transfer due to high electronegativity, which then causes charge attraction, lattice and formula unit | Yes |
| Spot on-Physical Sciences | No | Yes | Only covalent and polar covalent bonding included, but introduces dative bonds. | Yes | Ionic bonding not introduced | Yes |
| Study and master Physical Sciences | Yes hybrid orbitals and s and p bonding | Yes | MCI | Yes | Electron transfer. Both formula unit and lattice depicted and explained | Yes as Born Haber cycles |
| Oxford successful Physical Sciences grade 10 and 11 | No | No | MCPI | Yes | Very weak explanation, octet rule based, electrostatic attraction | No |
Appendix 4: detailed data Q4, subject groups are in order of CH13, Swe, CH14, UCT
Acknowledgements
AN, LÖ and JM thank the Swedish Science Council for support through Chalmers' Forskarskola i ingenjörsvetenskap, AN and LÖ also thank Swedish Science Council for support through the Swedish Research Links program with South Africa. The authors thank Ms Rene Toerien for sharing her expertise of the South African secondary curriculum and Dr Maja Elmgren, Uppsala University, for helpful discussions.References
- Adbo K. and Taber K. S., (2014), Developing an understanding of chemistry: a case study of one swedish student's rich conceptualisation for making sense of upper secondary school chemistry, Int. J. Sci. Educ., 36, 1107–1136.
- Andersson S., Sonesson A., Stålhandske B. and Tullberg A., (2000), Gymnasiekemi a, 2nd edn, Stockholm: Liber AB.
- Andersson S., Sonesson A., Svahn O. and Tullberg A., (2012), Gymnasiekemi 1, 4th edn, Stockholm: Liber.
- Bergqvist A., Drechsler M., De Jong O. and Rundgren S.-N.C., (2013), Representations of chemical bonding models in school textbooks – help or hindrance for understanding? Chem. Educ. Res. Pract., 14, 589–606.
- Boo H. K., (1998), Students' understandings of chemical bonds and the energetics of chemical reactions, J. Res. Sci. Teach., 35, 569–581.
- Broster P., James H. and Matshona M., (2005), Oxford successful physical sciences grade 10 learner's book, 1st edn, Cape Town: Oxford University Press.
- Broster P., Carter P., James H. and Matshona M., (2006), Oxford successful physical sciences grade 11 learner's book, 1st edn, Cape Town: Oxford University Press.
- Butts B. and Smith R., (1987), Hsc chemistry students' understanding of the structure and properties of molecular and ionic compounds, Res. Sci. Educ., 17, 192–201.
- Coll R. K. and Taylor N., (2002), Mental models in chemistry: senior chemistry students' mental models of chemical bonding, Chem. Educ. Res. Pract., 3, 175–184.
- Coll R. K. and Treagust D. F., (2003a), Investigation of secondary school, undergraduate, and graduate learners' mental models of ionic bonding, J. Res. Sci. Teach., 40, 464–486.
- Coll R. K. and Treagust D. F., (2003b), Learners' mental models of metallic bonding: a cross-age study, Sci. Educ., 87, 685–707.
- Department of Basic Education, (2012), National curriculum statement grades 10–12 (general) physical sciences curriculum and assessment policy statement, 2014-05-18, Department of Basic Education, Pretoria, South Africa, http://www.education.gov.za. Retrieved from http://www.education.gov.za/LinkClick.aspx?fileticket=GJVoSM8ie6E%3D&tabid=420&mid=1216.
- Department of Education, (2003), National curriculum statement grades 10–12 (general) physical sciences, 2014-05-18, Department of Education, Pretoria, South Africa, http://www.education.gov.za. Retrieved from http://www.education.gov.za/LinkClick.aspx?fileticket=VoSn0yzfkcE=.
- Department of Education, (2006), National curriculum statement grade 10–12 (general) physical sciences content document 2014-05-18, Department of Basic Education, Pretoria, South Africa, http://www.education.gov.za. Retrieved from http://wced.pgwc.gov.za/circulars/minutes07/NCS-PhysicalScience-Content-June06.pdf.
- Dhindsa H. S. and Treagust D. F., (2014), Prospective pedagogy for teaching chemical bonding for smart and sustainable learning, Chem. Educ. Res. Pract., 15, 435–446.
- Driver R. and Easley J., (1978), Pupils and paradigms: a review of literature related to concept development in adolescent science students, Stud. Sci. Educ., 5, 61–84.
- Elferink M., Kirstein W., Maclachlan L., Pillay N., Rens L. and Roos J., (2012a), Spot on-physical sciences grade 11 learners book, Johannesburg, South Africa: Heinemann Publishers Ltd.
- Elferink M., Kirstein W., Maclachlan L., Pillay N., Rens L., Roos J. and White M., (2012b), Spot on-physical sciences grade 10 learners book, Johannesburg, South Africa: Heinemann Publishers Ltd.
- FHSST V. A., (2010), Everything science -grade 10 physical sciences-version 0.5 – ncs (sep 2010), 2014-08-11, Siyavula, http://www.fhsst.org, retrieved from http://everythingscience.co.za/grade-11.
- Harrison A. G. and Treagust D. F., (1996), Secondary students' mental models of atoms and molecules: implications for teaching chemistry, Sci. Educ., 80, 509–534.
- Henriksson A., (2011), Syntes kemi 1, 2nd edn, Malmö: Gleerups utbildning.
- Hewson P. W., (1992), Conceptual change in science teaching and teacher education, Paper presented at a meeting by the National Center for Educational Research, Documentation and Assessment, Ministry of Education and Science, Madrid, Spain.
- Jones R. and Berens N., (2005), Physical sciences explained – learners book-grade 10 (ncs), 1st edn, Lansdowne, South Africa: Juta Gariep Ltd.
- Jones R. and Berens N., (2006), Physical sciences explained – learners book-grade 11 (ncs), 1st edn, Cape Town, South Africa: Juta Gariep Ltd.
- Kelder K. H., (2005), Study & master physical sciences grade 10 learner's book, Cape Town: Cambridge University Press.
- Kelder K. H., (2006), Study & master physical sciences grade 11 learner's book, Cape Town: Cambridge University Press.
- Kwak N. and Radler B., (2002), A comparison between mail and web surveys: response pattern, respondent profile, and data quality, J. Offic. Stat., 18, 257–273.
- Lemke J. L., (1990), Talking science: language, learning, and values, 355 Chestnut Street, Norwood, NJ 07648: Ablex Publishing Corporation.
- Levy Nahum T., Mamlok-Naaman R., Hofstein A. and Krajcik J., (2007), Developing a new teaching approach for the chemical bonding concept aligned with current scientific and pedagogical knowledge, Sci. Educ., 91, 579–603.
- Levy Nahum T., Mamlok-Naaman R., Hofstein A. and Kronik L., (2008), A new “bottom-up” framework for teaching chemical bonding, J. Chem. Educ., 85, 1680–1685.
- National Agency for Education, (2000), Chemistry a syllabus (sfs 2000:65), 2014-04-02, Department of Education, Stockholm, Sweden, http://www.skolverket.se, retrieved from http://www.skolverket.se/regelverk/skolfs/skolfs?_xurl_=http%3A%2F%2Fwww5.skolverket.se%2Fwtpub%2Fws%2Fskolfs%2Fwpubext%2Ffs%2FRecord%3Fk%3D759.
- National Agency for Education, (2010), Chemistry syllabus gy11 (skolfs 2010:108), 2014-04-02, Swedish Department of Education, retrieved from http://www.skolverket.se/polopoly_fs/1.194837!/Menu/article/attachment/Chemistry.pdf.
- National Agency for Education, (2012), Upper secondary school 2011, 2014-08-11, Department of Education, Stockholm, Sweden, Skolverket.se, retrieved from http://www.skolverket.se/publikationer?id=2801.
- Nicoll G., (2001), A report of undergraduates' bonding misconceptions, Int. J. Sci. Educ., 23, 707–730.
- Othman J., Treagust D. F. and Chandrasegaran A. L., (2008), An investigation into the relationship between students' conceptions of the particulate nature of matter and their understanding of chemical bonding, Int. J. Sci. Educ., 30, 1531–1550.
- Özmen H., (2004), Some student misconceptions in chemistry: a literature review of chemical bonding, J. Sci. Educ. Technol., 13, 147–159.
- Peterson R. F., Treagust D. F. and Garnett P., (1989), Development and application of a diagnostic instrument to evaluate grade-11 and -12 students concepts of covalent bonding and structure following a course of instruction, J. Res. Sci. Teach., 26, 301–314.
- Pilström H., Wahlström E., Lüning B., Viklund G., Aastrup L. and Peterson A., (2011), Modell och verklighet kemi 1/a, 2nd edn, Stockholm: Natur och Kultur.
- Posner G. J., Strike K. A., Hewson P. W. and Gertzog W. A., (1982), Accommodation of a scientific conception: toward a theory of conceptual change, Sci. Educ., 66, 211–227.
- Potgieter M. and Davidowitz B., (2011), Preparedness for tertiary chemistry: multiple applications of the chemistry competence test for diagnostic and prediction purposes, Chem. Educ. Res. Pract., 12, 193–204.
- Siyavula V. A., (2012), Everything science -grade 11 physical sciences-version 0.9 – ncs(2012), 2014-05-22, Siyavula, retrieved from http://everythingscience.co.za/grade-11.
- Taber K., (2002), Chemical misconceptions: prevention, diagnosis and cure, Royal Society of Chemistry.
- Taber K. and Adbo K., (2013), Developing chemical understanding in the explanatory vacuum: Swedish high school students' use of an anthropomorphic conceptual framework to make sense of chemical phenomena, in Tsaparlis G. and Sevian H. (ed.) Concepts of matter in science education, Dordrecht: Springer Netherlands, pp. 347–370.
- Taber K. S., (1997), Student understanding of ionic bonding: molecular versus electrostatic framework, Sch. Sci. Rev., 78, 85–95.
- Taber K. S., (1998), An alternative conceptual framework from chemistry education, Int. J. Sci. Educ., 20, 597–608.
- Taber K. S., (2001), Building the structural concepts of chemistry: some considerations from educational research, Chem. Educ. Res. Pract., 2, 123–158.
- Taber K. S., Tsaparlis G. and Nakiboglu C., (2012), Student conceptions of ionic bonding: patterns of thinking across three european contexts, Int. J. Sci. Educ., 34, 2843–2873.
- Talanquer V., (2006), Commonsense chemistry: a model for understanding students' alternative conceptions, J. Chem. Educ., 83, 811–816.
- Tan K.-C. D. and Treagust D. F., (1999), Evaluating students' understanding of chemical bonding, Sch. Sci. Rev., 81, 75–83.
- Trusted J., (1987), Inquiry and understanding: an introduction to explanation in the physical and human sciences.
- Vladušić R., Bucat R. B. and Ožić M., (2016), Understanding ionic bonding – a scan across the croatian education system, Chem. Educ. Res. Pract., 10.1039/C6RP00040A.
- Vosniadou S., (1994), Capturing and modeling the process of conceptual change, Learn. Instruct., 4, 45–69.
- Wren D. and Barbera J., (2013), Gathering evidence for validity during the design, development, and qualitative evaluation of thermochemistry concept inventory items, J. Chem. Educ., 90, 1590–1601.
Footnotes |
| † All diagrams in the manuscript were either created by the authors or in the case of the caltrop in Fig. 1 taken from Wikipedia commons. For the Caltrop: http://https://upload.wikimedia.org/wikipedia/commons/8/8d/CaltropTireSpikeCIA.jpg. The following attribution applies: by Central Intelligence Agency (Caltrop (Tire Spike) Uploaded by liftarn) [Public domain], via Wikimedia Commons. |
| ‡ Chalmers Tekniska Högskola or Chalmers University of Technology, located in Gothenburg, is one of Sweden's two ‘old’ engineering schools. It has over 8500 MSc and BSc students (25% women), more than 1000 PhD students (26% women) and 2450 employees. Currently the Chemical Engineering program is the most popular chemistry program in Sweden, recruiting top students from all over the country according to the Swedish Higher Education Authority, (uka.se). |
| § See http://www.ibo.org/diploma. |
¶ The University of Cape Town enrolls around 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 students and has about 5000 employees. It is Africa's oldest university (in the modern sense) and the only one to be placed in the top 150 on the Times Higher Education World University Rankings (http://https://www.timeshighereducation.co.uk/world-university-rankings/). 500 students and has about 5000 employees. It is Africa's oldest university (in the modern sense) and the only one to be placed in the top 150 on the Times Higher Education World University Rankings (http://https://www.timeshighereducation.co.uk/world-university-rankings/). |
| || The vast majority of these will have Swedish as their first language, which is also true for the teachers. While courses are taught in Swedish the textbooks used are exclusively in English. |
| ** This group is mixed in terms of curriculum followed at school, with the younger students, about 90% of the sample, following the Gy11 curriculum and the older ones following the old curriculum from 2000. The differences are not judged to have any significant bearing on this study. |
| †† We choose, due to language restrictions, to focus on the South African textbooks written in English (the other main language of science instruction in South Africa being Afrikaans). According to an experienced South African teacher, some instruction in physical science is probably in other languages, but all students are required to write the exam either in English or Afrikaans. To a much smaller degree this situation is mirrored in Sweden. It should be noted that the South African school system has a richer variety of textbooks than the Swedish. |
| ‡‡ It should perhaps be noted that less than 100 years ago the non-molecular nature of NaCl(s) was shocking news to chemists even of high academic standing, see for example: H. E. Armstrong, FRS, Poor Common Salt, letter to Nature, 1 October, 1929, p. 478. |
| §§ The first ¾ of year one chemical engineering at Chalmers comprise equal amounts of chemistry, including biochemistry, and mathematics. See http://https://student.portal.chalmers.se/en/ for information. |
| This journal is © The Royal Society of Chemistry 2016 |