Development of a novel smartphone-based application for accurate and sensitive on-field hemoglobin measurement
Niraj S. Ghatpandea,
Priti P. Aptea,
Bimba N. Joshia,
Sadanand S. Naikb,
Dhananjay Bodas*c,
Vishwajit Sanded,
Pravin Uttarward and
Prasad P. Kulkarni*a
aBioprospecting Group, Agharkar Research Institute, GG Agarkar Road, Pune-411004, India. E-mail: kulkarniari@gmail.com; Fax: +91 020 25651542; Tel: +91 020 25655101
bK. E. M Hospital and Research Center, Pune-411011, India
cNanobioscience Group, Agharkar Research Institute, GG Agarkar Road, Pune-411004, India. E-mail: dsbodas@aripune.org; Tel: +91 020 25655127
dMindbowser Infosolutions Pvt. Ltd., Gwalior, India 474012
First published on 21st October 2016
Abstract
Here, we report on the development of a smartphone-based application (app) for the accurate and sensitive measurement of hemoglobin (Hb). Hb values obtained using the app correlated significantly (R2 = 0.976) with those obtained by an automated hematology analyzer. Bland–Altman analysis showed that the standard deviation of the difference between Hb measured by the two methods was 0.35 g dL−1 and the limits of agreement between the two methods were −0.62 to 0.75. A limit of detection of 0.66 g dL−1 was achieved using the app along with a 2 g dL−1 limit of quantification. Furthermore, measurement using the app was highly sensitive (94%) and specific (90%). The app was developed for a smartphone and it will be particularly useful in resource-limited conditions for the rapid, accurate, highly sensitive and specific assessment of anemia.
The mobile phone is a device that has been projected to record human health in the future, since there are more than 7.1 billion users globally (as of 2014) and this value is expected to rise up to 9.2 billion by 2020.1 Therefore, mobile phone infrastructure can be utilized to develop low-cost diagnostics for monitoring existing and emerging diseases. In recent years, there has been an increase in the adoption of smartphone cameras and related applications by healthcare professionals for disease diagnosis and drug reference.2,3 The use of technology to deliver healthcare or telemedicine has proved effective, particularly for communities located in rural and remote areas.
Compared to standard laboratory testing, point-of-care (POC) diagnostics are fast, simple and inexpensive, and therefore have excellent applicability in resource-limited settings. Previously, Martinez et al.4 reported on paper-based microfluidic devices for performing multiple assays of glucose and protein simultaneously, and a camera phone was used. The digital information (an image of the assay) was sent to an off-site laboratory for intensity-based analysis. Nowadays, various external platforms/devices have been developed for the quantitative measurement of biomarkers with the help of a smartphone.5–7 Recently, researchers have also demonstrated that integrating the imaging and analysis capabilities is possible through the development of an application (app).8–10 Yetisen et al.8 demonstrated the use of such an app with inter-phone repeatability for the colorimetric analysis of the pH, protein and glucose levels in an artificial urine sample. Hence, the development of a fully integrated (capture and analysis) app is desirable to facilitate the rapid and cost effective assessment of various colorimetric tests.
Anemia is a major worldwide public health problem with more than 1.6 billion people estimated to be anemic.11 In particular, anemia during pregnancy leads to low birth weight and increased risk of maternal and perinatal mortality.12,13 Hemoglobin (Hb) is the most commonly used indicator for assessing anemia.14 Hb is a tetrameric protein containing ‘heme’ and ‘globin’ and is present in red blood cells. In clinical laboratories, Hb is analyzed using a spectrophotometer (mainly via the cyanmethemoglobin method), an automated hematology analyzer, blood gas analyzers, and CO-oximeters.15 An automated hematology analyzer combines absorption spectrometry, impedance and conductivity measurement and flow cytometry for cell counting and Hb estimation. Although these analyzers showed greater sensitivity, accuracy and precision for Hb estimation, the higher cost of the analysis, the requirement of skilled labour and bulky instrumentation limits their usage in resource-limited conditions. The World Health Organization (WHO) Hb color scale (HbCS) test and the Hemocue Hb 201+ system are widely used as low-cost POC devices for Hb estimation in resource-limited conditions. However, the HbCS depends on a comparison between the color of a drop of blood absorbed onto chromatography paper and the standard colors on a laminated card displayed in increments of 2 g dL−1.16 These tests have several drawbacks including low accuracy and sensitivity.17 In the Hemocue system, blood is drawn into a small microcuvette through capillary action, and the Hb concentration is read by a portable device. Although this method is relatively simple, accurate, and rapid, the use of costly disposable cuvettes (approximately $2) and an analyzer (approximately $800) make it expensive for developing and third world countries. Recently, the development of POC devices for the estimation of various biomarkers including Hb has been carried out extensively.18,19 Both Xang et al.18 and Bond et al.19 reported on paper-based devices for Hb estimation. In these studies, they used a portable scanner and a low-cost reader to digitize and analyze the Hb levels. However, these paper-based devices are not yet used in clinical practices and moreover, the app cannot provide the analysis.20 Therefore, the applications need to be developed not only to digitize/capture the assay, but also to analyze the digital information. Only then could these apps provide a better alternative to currently available methods for accurate and sensitive on-field Hb estimation. These methods will be particularly useful in resource-limited settings, such as remote areas in developing countries where screening of a large number of samples is involved. In this study, we have concentrated on the development of a smartphone-based app for on-field colorimetric-based Hb estimation. The analytical performance of the app in terms of sensitivity, specificity, limit of detection (LOD) and limit of quantification (LOQ) is evaluated and compared with the standard procedures. For the development and assessment of correctness of the app we have used the colorimetric cyanmethemoglobin method.21 Briefly, in a clean and dry 24 well-plate (Nest Biotech, USA), 1000 μL of Hemocor-D (containing potassium ferricyanide and potassium cyanide, Crest Biosystems, India) solution was added.
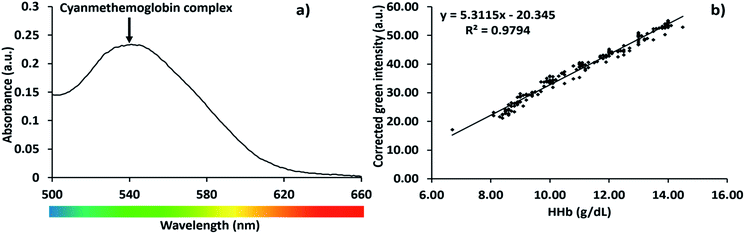
5 μL of whole blood was mixed with the Hemocor-D solution and incubated at 25 °C for 3 min. Following incubation, we recorded the absorption spectrum of the solution between 300 to 700 nm using a plate reader (Biotek SYNERGY HT, microplate reader, USA). In this method, Hb is first converted into methaemoglobin using potassium ferricyanide, followed by reaction with potassium cyanide to form a stable cyanmethemoglobin complex.21 The cyanmethemoglobin complex showed maximum absorbance at 540 nm as shown in Fig. 1a, corresponding to the green region of the spectrum (Fig. 1a). Therefore, we selected green color intensity for the development of the app for the colorimetric estimation of Hb levels.
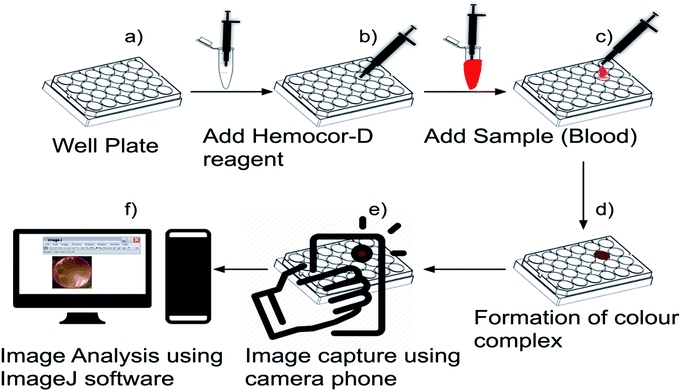
A linear relationship between the Hb concentration and the green color intensity was established using 168 blood samples and the protocol shown in Fig. 2. All of the experiments were performed in compliance with the “Biomedical research ethics review method involving people” (India) guidelines, and approved by the medical ethics committee at King Edward Memorial Hospital Research Center, Pune, India. Informed consent was obtained from human participants in this study. The blood samples were collected in 5 mL anticoagulant tubes (K2 EDTA Labtech Disposables) and stored at 4 °C. The samples were used for the estimation of Hb using a Lab life Nobel III automated hematology analyzer (Diagnova India) in KEM Hospital laboratory. Briefly, 1000 μL of Hemocor-D solution was added to a clean and dry 24 well-plate (Fig. 2a and b). Next, 5 μL of whole blood was mixed with the Hemocor-D solution and incubated at 25 °C for 3 min (Fig. 2c and d), followed by image capture using a Lenovo S660 (Lenovo Group Ltd. Beijing, China, Android version 4.1.2) smartphone equipped with an 8 megapixel camera. The images of the individual wells were captured one by one at a fixed distance of 20 cm from the well-plate without using the internal flash of the smartphone. Finally, ImageJ software (NIH public software) was used to measure the intensity of the green channel from the captured images.22 We have plotted the graph of the corrected [blank ¬ sample] green values obtained using ImageJ against the Hb concentration obtained by the automated hematology analyser (see Fig. 1b). The calibration curve was recorded by capturing the blank reading and subtracting the test sample from it. The method developed by us is designed to negate the effects of different lighting conditions. For this purpose, a blank reading is recorded before every test sample. The blank reading provides all of the details regarding the ambient light conditions which are then subtracted from the sample to achieve the absolute color intensity. Hence the sample tests can be compared with the calibration curve. An equation was derived to correlate the green intensity with the Hb values by considering all of the factors related to the image processing. Furthermore, this equation was used in the development of the app to correctly estimate the Hb concentration in whole blood.
We then developed the application called “Hb calculator app” (Fig. 3a–f) which operates on an Android operating system for the colorimetric estimation of Hb from whole blood. To test a given blood sample, the user needs to follow the four steps shown in Fig. 2a–d followed by image capture and Hb estimation using the app.
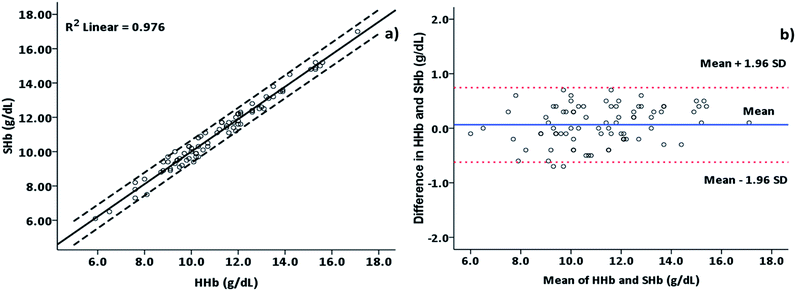
This user-friendly smartphone app was designed with the necessary instructions to guide the user for the Hb estimation. Fig. 3a and b provides a screenshot of the icon and splash screen of the “Hb calculator” app. Before sample testing begins, the user needs to calibrate the app by capturing an image of the ‘calibration’ well (i.e. the well containing only the reagent). A guide (see red circle in Fig. 3c and e) is provided, which ensures a fixed camera to well-plate distance as well as the area to be captured. The calibration image must be captured under laboratory light conditions. It is advisable to calibrate every time before analysing the test sample to ensure similar light conditions for the analysis. After selecting the calibration image, the user needs to capture an image of the test well (Fig. 3e). The app processes the image information (Fig. 3g) and transforms it into Hb concentrations by using the derived equation and reading the measured value from the calibration curve (Fig. 1b). The Hb value (g dL−1) is then displayed on the screen (Fig. 3g). Briefly, the app processes the image by splitting red, green and blue channels and selecting the green channel for further analysis. It further corrects for the green intensity by fitting the derived equation and then reading the data from the calibration curve (Fig. 1b) to provide accurate Hb values in g dL−1. To evaluate the accuracy of the measurement by the developed smartphone app, we further measured the Hb concentrations of 82 blood samples using the same protocol as described in Fig. 2 and 3. The Hb values measured using the app were compared to those obtained by the automated hematology analyzer at KEM hospital (see Fig. 4). The linear least-squares regression analysis (Fig. 4a) showed a significant correlation (R2 = 0.976) between the values obtained from the automated hematology analyzer and those measured using the developed app. No significant difference (P > 0.05) was observed in the mean values of Hb (g dL−1) estimated using both of the techniques (hematology analyser 11.17 ± 2.22 vs app 11.11 ± 2.13) over the range of 6 to 18 g dL−1 of Hb. Recently, Zhu et al.6 used a cell phone-based blood analyzer for white blood cells (WBC), red blood cells (RBC) and Hb measurements. They reported a good correlation coefficient of 0.92 with the automated hematology analyzer. However, our technique is significantly better in terms of a faster turn-around time, and higher sensitivity and accuracy of the Hb measurement using an inexpensive tool.
The LOD of an analytical method is merely the lowest amount of analyte in a sample, which can be detected but not necessarily quantitated as an absolute value. Whereas, the LOQ is the lowest amount of analyte in a sample, which can be determined quantitatively with suitable precision and accuracy. The LOD and LOQ were calculated for the data obtained from the app using the intercept and standard error of the regression line (Fig. 4).23 An LOD of 0.66 g dL−1 was obtained using the app along with an LOQ of 2 g dL−1. Furthermore, the sensitivity and specificity were determined to differentiate anemic and non-anemic individuals by following the criteria laid down by WHO.24 We found that our technique showed a sensitivity of 94% and a specificity of 90% for the Hb estimation. Cohen’s Kappa test was carried out to determine the correlation between the data (82 individuals) obtained from the automated hematology analyser and the app on the correct differentiation between anemic and non-anemic individuals. There was good agreement between the two methods, κ = 0.802, P < 0.0005. Greater sensitivity, specificity and a higher Cohen’s Kappa for the app suggested that it had the ability to accurately differentiate between anemic or non-anemic subjects.
Previously, Darshana et al.16 reported an HbCS method that is commonly used in resource-limited settings, and showed an Hb estimation with an error of ± 1 g dL−1 for only 53% of the measurements when compared with laboratory Hb values. However, our app showed 100% agreement with the reference of Hb ± 1 g dL−1. Specifically, the difference between the Hb values obtained by the analyser and the app was within ± 1 g dL−1 suggesting the accuracy of the app in detecting anemia was higher.
A Bland–Altman analysis25 (see Fig. 4b) showed that the standard deviation of the difference between the Hb measured by the two methods was 0.35 g dL−1 and the limits of agreement between the two methods were −0.62 to 0.75. The higher sensitivity, specificity, accuracy and reliability of the smartphone app for the Hb measurements makes it an attractive alternative for Hb estimation in resource-limiting conditions.
Another advantage of using a smartphone is its reach in the developing world. The smartphone has taken its place as a personalized garment and hence is projected as a versatile healthcare device. Our technique captures an image and provides output using an app rather than using a remote location for the analysis. This makes the smartphone-based app extremely user-friendly and easy to operate in remote rural areas within developing countries, as in our technique we have used reagents and plates which are available off the shelf. This makes the Hb measurement technique highly cost effective (less than  10 ($0.15), excluding the cost of a smartphone). Overall, our results suggest taking Hb measurements using the smartphone-based app could be an attractive alternative to existing methods. The advantages of our method are that a low sample volume is required, the analysis is rapid and low cost, and the method has a higher specificity, sensitivity and reliability for Hb measurements.
10 ($0.15), excluding the cost of a smartphone). Overall, our results suggest taking Hb measurements using the smartphone-based app could be an attractive alternative to existing methods. The advantages of our method are that a low sample volume is required, the analysis is rapid and low cost, and the method has a higher specificity, sensitivity and reliability for Hb measurements.
Conclusions
Measurement techniques based on smartphone apps have been emerging in the field of the POC system as they provides a portable, inexpensive, and user-friendly solution. The smartphones allow for the measurement of analytes by converting the variation in color intensity to digitized values for a more accurate and versatile estimation. This work involves the use of a smartphone app for Hb estimation. Our results demonstrate a close agreement of Hb concentration estimated with the app and that of the reference method. Indeed, Hb detected using the app shows better sensitivity, specificity and reliability when compared to the automated hematology analyzer. This method enables storage, archiving, and retrieval of data when required. We believe that this technique will be helpful in improving patient care and reducing healthcare expenses. This method will be particularly useful in resource-limited conditions and will help in getting rapid on-the-spot Hb estimation. This will be advantageous in the rapid assessment of anemia in epidemiological studies where large numbers of subjects are involved.Acknowledgements
This study was supported by financial assistance from Agharkar Research Institute project (BIO-22).Notes and references
- Ericsson Mobility report, Stockholm, Sweden, June 2015 Search PubMed.
- A. S. Mosa, I. Yoo and L. Sheets, BMC Med. Inf. Decis. Making, 2012, 12, 67–98 CrossRef PubMed
.
- C. D. Chin, Y. K. Cheung, T. Laksanasopin, M. M. Modena, S. Y. Chin, A. A. Sridhara, D. Steinmiller, V. Linder, J. Mushingantahe, G. Umviligihozo, E. Karita, L. Mwambarangwe, S. L. Braunstein, J. van deWijgert, R. Sahabo, J. E. Justman, W. El-Sadr and S. K. Sia, Clin. Chem., 2013, 59, 629–640 CAS
.
- A. W. Martinez, S. T. Phillips, E. Carrilho, S. W. Thomas III, H. Sindi and G. M. Whitesides, Anal. Chem., 2008, 80, 3699–3707 CrossRef CAS PubMed
.
- V. Oncescu, D. O’Dell and D. Erickson, Lab Chip, 2013, 13, 3233–3238 RSC
.
- H. Zhu, I. Sencan, J. Wong, S. Dimitrov, D. Tseng, K. Nagashima and A. Ozcan, Lab Chip, 2013, 13, 1282–1288 RSC
.
- T. Guo, R. Patnaik, K. Kuhlmann, A. J. Rai and S. K. Sia, Lab Chip, 2015, 15, 3514–3520 RSC
.
- A. K. Yetisen, J. L. Martinez-Hurtado, A. Garcia-Melendrez, F. da Cruz Vasconcello and R. L. Christopher, Sens. Actuators, B, 2014, 196, 156–160 CrossRef CAS
.
- S. Wang, X. Zhao, I. Khimji, R. Akbas, W. Qiu, D. Edwards, D. W. Cramer, B. Ye and U. Demirci, Lab Chip, 2011, 11, 3411–3418 RSC
.
- J. L. Delaney, E. H. Doeven, A. J. Harsant and C. F. Hogan, Anal. Chim. Acta, 2013, 790, 56–60 CrossRef CAS PubMed
.
- S. R. Pasricha, H. Drakesmith, J. Black, D. Hipgrave and B. Biggs, Blood, 2013, 121, 2607–2617 CrossRef CAS PubMed
.
- P. J. Steer, Am. J. Clin. Nutr., 2000, 71, 1285S–1287S CAS
.
- Q. Zhang, C. V. Ananth, G. G. Rhoads and Z. Li, Ann. Epidemiol., 2000, 19, 793–799 CrossRef PubMed
.
- I. Adam, S. Ahmed, M. H. Mahmoud and M. I. Yassin, Diagn. Pathol., 2012, 7, 30–36 CrossRef CAS PubMed
.
- G. J. Myers and J. Browne, Perfusion, 2007, 22, 179–183 CrossRef PubMed
.
- L. G. Darshana and D. I. Uluwaduge, Anemia, 2014, 531670, 1–4 CrossRef PubMed
.
- B. Nkrumah, S. B. Nguah, N. Sarpong, D. Dekker, A. Idriss, J. May and Y. Adu-Sarkodie, BMC Clin. Pathol., 2011, 11, 5–11 CrossRef CAS PubMed
.
- X. Yang, N. Z. Piety, S. M. Vignes, M. S. Benton, J. Kanter and S. S. Shevkoplyas, Clin. Chem., 2013, 59, 1506–1513 CAS
.
- M. Bond, C. Elguea, J. S. Yan, M. Pawlowski, J. Williams, A. Wahed, M. Oden, T. S. Tkaczyk and R. Richards-Kortum, Lab Chip, 2013, 13, 2381–2388 RSC
.
- X. Li, D. R. Ballerini and W. Shen, Biomicrofluidics, 2012, 6, 11301–1130113 CrossRef PubMed
.
- E. J. vanKampen and W. G. Zijlstra, Clin. Chim. Acta, 1961, 6, 538–544 CrossRef CAS
.
- C. A. Schneider, W. S. Rasband and K. W. Eliceiri, Nat. Methods, 2012, 9, 671–675 CrossRef CAS PubMed
.
- Analytical Procedures and Methods Validation: Chemistry, Manufacturing, and Controls, Federal Register (Notices), 2000, 65, pp. 776–777 Search PubMed.
- WHO, Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. Vitamin and Mineral Nutrition Information System, World Health Organization, Geneva, 2011, WHO/NMH/NHD/MNM/11.1, http://www.who.int/vmnis/indicators/haemoglobin.pdf, accessed on May 2015 Search PubMed
.
- J. M. Bland and D. G. Altman, Lancet, 1986, 8, 307–310 CrossRef
.
| This journal is © The Royal Society of Chemistry 2016 |