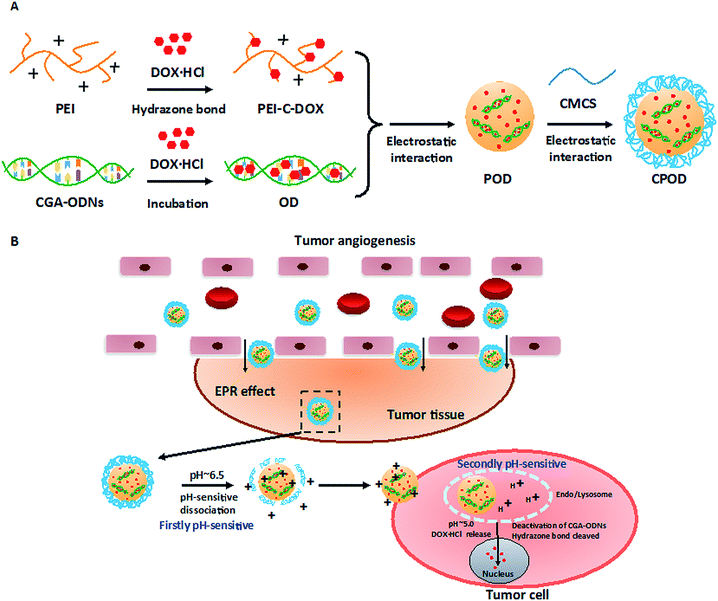
Two-stage pH-sensitive doxorubicin hydrochloride loaded core–shell nanoparticles with dual drug-loading strategies for the potential anti-tumor treatment†
Xiaoyue Yua,
Bo Zhanga,
Tianqi Wanga,
Jing Zhanga,
Shengjun Mua,
Chunxi Liu*b and
Na Zhang*a
aDepartment of Pharmaceutics, School of Pharmaceutical Science, Shandong University, 44 Wenhuaxi Road, Jinan 250012, Shandong Province, China. E-mail: zhangnancy9@sdu.edu.cn; Fax: +86 0531 88382548; Tel: +86 0531 88382015
bPharmaceutical Department, Qilu Hospital of Shandong University, 107 Wenhuaxi Road, Jinan 250012, Shandong Province, China. E-mail: liuchunxi1985@163.com
First published on 26th October 2016
Abstract
A novel two-stage pH-sensitive doxorubicin hydrochloride (DOX·HCl) delivery system, programmed to respond to tumor extracellular circumstrances (about pH 6.5) and intercellular endo/lysosome (pH 5.0), was designed with dual drug-loading strategies. Hydrazone linked cationic conjugates poly(ethyleneimine)-C6-succinimidyl 6-hydrazinonicotinate acetone hydrazone-DOX·HCl (PEI-C6-SANH-DOX·HCl, PEI-C-DOX) was synthesized, then condensed the DOX·HCl loaded 21-base (CGA)7 oligodeoxynucleotides (CGA-ODNs/DOX·HCl, OD) to get the dual methods DOX·HCl loaded nanoparticles PEI-C-DOX/OD (POD) as inner core. Subsequently, O-carboxymethyl-chitosan (CMCS) was coated on POD to construct the core–shell nanoparticles CMCS/POD (CPOD). The average size and zeta potential of CPOD were (165.0 ± 3.3) nm and −(15.6 ± 0.82) mV, respectively. In vitro evaluation showed that CMCS could dissociate from POD at tumor extracellular pH values, and the cellular uptake of CPOD was much higher than that at pH 7.4 (p < 0.005 in B16 cells, p < 0.01 in HepG2 cells). Besides, in vitro release study revealed that the drug release amount from CPOD at endo/lysosome pH was significantly more than that at pH 7.4 (p < 0.005), which was further indicated by nuclear localization test. In vivo NIRF imaging illustrated the accumulation in tumor of CPOD for 24 h. Furthermore, in vivo anti-tumor test indicated that CPOD exhibited more superior anti-tumor efficacy even than double dosage DOX·HCl solution. No visible tissue lesions of main organs in CPOD group were observed, indicating preliminary in vivo safety of CPOD. These results suggest that the two-stage pH-sensitive CPOD would be a promising delivery platform for improving anti-tumor efficacy of DOX·HCl with a fair degree of safety.
1. Introduction
Cancer has become the most serious life-threatening disease in the world, with more than 10 million new cases every year.1,2 As a common treatment (chemotherapy, radiotherapy and surgery), chemotherapy is widely employed in for many cancers. However, its success has been limited by several drawbacks, such as low efficacy and toxic side effects, which are mainly attributed to the non-selectivity biodistribution of drug in the whole body.3,4 For improving chemotherapy efficacy, achieving tumor target and site-specific drug release in tumor, appropriate drug delivery systems are greatly required.5 In recent years, nano-drug delivery systems (NDDSs) have been widely developed with notable advantages including biocompatibility, controlled size and shape, and stimuli-sensitive properties for precise spatiotemporal control.6 Furthermore, the suitable size (50–200 nm) of NDDSs enables them to passively reach tumor target through the enhanced permeability and retention (EPR) effect, which make NDDSs preferentially accumulate in tumor tissues following better anti-tumor efficacy than low molecular weight anti-tumor agents.7–11Doxorubicin hydrochloride (DOX·HCl), a type of anthracycline broad-spectrum antineoplastic drug, has been used clinically to treat several kinds of tumors, such as hepatocellular carcinoma,12 lung cancer13 and breast cancer.14 It's clinical application is often coupled with severe side effects, such as cardiotoxicity and renal damage.15,16 It has been reported that the anti-tumor efficacy of DOX·HCl depends on both the drug-loading concentrations in NDDSs and the composition of tumor matrix.17 Therefore, in order to enhance anti-tumor efficacy and overcome side effects of DOX·HCl, NDDSs with higher drug-loading and better drug release capability triggered by tumor microenvironment need to be developed.18–21
In order to obtain high drug-loading, several researchers have made various efforts, namely. Li et al. have built a (3-aminopropyl)triethoxysilane (APTES)-modified mesoporous silica nanoparticles (MSNs) with high drug-loading, DOX has been efficient encapsulated inside MSNs because of the large surface area and pore volume of MSNs.22 However, MSNs are involved in a complicated process and slow biodegradation rate.23 In addition, Tan et al. have synthesized a D-α-tocopherol polyethylene glycol 1000 succinate (TPGS)-based prodrug conjugated with DOX, which was based on the high drug-loading efficiency of TPGS.24 DOX was loaded by chemical conjugation. In our work, dual drug-loading strategies, in which deoxyribonucleic acid (DNA) loading method combined with chemical modification were used to achieve a higher drug-loading.
Since Trouet et al. reported the first use of DNA as a carrier for anthracycline drugs in 1972,25 DOX·HCl has been known to intercalate within the DNA strand, especially in 5′-GC-3′ or 5′-CG-3′ sequences due to the presence of flat aromatic rings in its molecule structure.26 Jon et al. has established a hybridized aptamer with 21-base (CGA)7 oligodeoxynucleotides (CGA-ODNs) extended at 3′ end and verified its high drug loading capacity for DOX·HCl, which improved the loaded DOX·HCl molecules from 0.6 to 7.5 per aptamer.26,27 Subsequently, many researchers have been reported CGA-ODNs as a DOX·HCl carriers with a high and easily controlled drug loading through modifying the length of -CG- repeat sequence.28–31
Moreover, chemical modification is one of common drug loading methods of NDDSs by conjugation of drugs to hydrophilic or amphiphilic polymers through chemical bonds.32,33 The benefits of chemical conjugates, include high drug loading and accurate loading efficiency.34–37 To enable triggered responsiveness inside tumors, drugs are usually conjugated via special linkages which are designed according to the tumor microenvironments, such as pH, redox, enzymes and temperature field.38,39 Among the stimuli-responsive bonds, pH-sensitive bonds including hydrazone,40 cis-acotinyl,41 and acetal21 are the most commonly used due to the distinct pH gradients among normal tissues (pH 7.4), tumor extracellular environments (about pH 6.5)42,43 as well as endosome (pH 5.0–6.0) and lysosome (pH 4.0–5.0) compartments.44–46 By taking advantages of the gradual pH decrease in tumor tissues and endo/lysosome, designing a two-stage pH-sensitive nanoparticles, which can respond to the pH changes step by step is necessary for accurate intracellular drug release control.
Herein, intracellular drug release is expected because it can reduce drug loss and increase intracellular drug concentration.47 Moreover, DOX·HCl, as a kind of topoisomerase inhibitor,48 intracellular release is beneficial to nuclear localization for its anti-tumor efficacy. For effective intracellular drug release, nanoparticles must first traverse the cell membranes. Since cell membranes are negatively charged, positively charged nanoparticles show higher affinity for cell membrane and help bypass the cellular barriers.49 However, cationic nanoparticles have strong non-specific cellular uptake in the bloodstream and interact strongly with serum components, which cause severe aggregation and rapid clearance from circulation.50,51 Therefore, it will be useful to create NDDSs that are negatively charged during blood circulation, but positively charged after accumulated in tumor tissues.52,53 Core–shell nanoparticles have been considered to be a promising strategy.
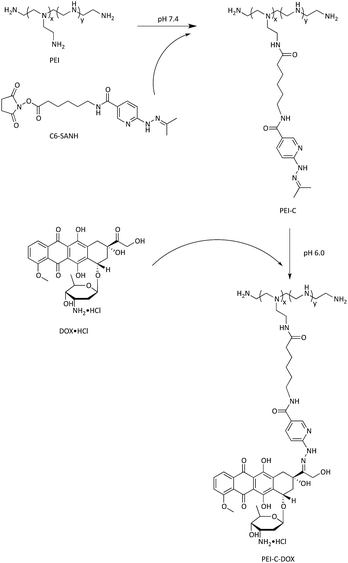
Branched poly(ethylenimine) (PEI) with high density of dissociative amine groups has extensive use in biomedical applications, particularly for oligodeoxynucleotides and DNA delivery based on the highly condensing ability of PEI to them.54,55 Meanwhile, the dissociative amino groups are suitable for chemical modification. Furthermore, compared with some other cationic polymers, such as chitosan and poly-D-lysine, PEI is more efficient in promoting cell attachment and adsorptive endocytosis through proteoglycans in the cell membrane.56,57 In the present study, DOX·HCl was conjugated to PEI by hydrazone bonds through a heterobifunctional crosslinking agent C6-S-HyNic (C6-succinimidyl 6-hydrazinonicotinate acetone hydrazone, C6-SANH) to form an intracellular pH-sensitive cationic conjugates PEI-C6-SANH-DOX·HCl (PEI-C-DOX). Hydrazone bond appears to be more stable at pH 7.4 than other pH-sensitive bond while showing excellent pH-sensitivity.58 On the other hand, DOX·HCl loaded CGA-ODNs (CGA-ODNs/DOX·HCl, OD) was obtained by easily incubation at room temperature. There were no organic reagents incorporated in the preparation process, which was of safety and non-immunogenicity. PEI-C-DOX could further condense negatively charged OD to obtain PEI-C-DOX/OD (POD). Herein, the DOX·HCl loading in POD could be increased through the dual drug-loading strategies, i.e. chemical modification combining with CGA-ODNs loading method. Afterwards, O-carboxymethyl-chitosan (CMCS) is a suitable choice of shell material for coating on POD, which is a biodegradable pH-sensitive amphiprotic polyelectrolyte with good biocompatibility.59,60 Scheme 1A and B show preparation and functionalization of CPOD, respectively. In blood circulation and normal tissues (pH 7.4), CMCS was expected to retain negative charge and coated on POD, keep CPOD stable and prolong the circulation time, thus relieve side effects of DOX·HCl caused by drug leakage. After CPOD accumulated in tumor tissues via EPR effect, the outer CMCS shell dissociated at the subacid microenvironment (about pH 6.5) and POD was exposed to facilitate the tumor cellular uptake. Upon internalization, DOX·HCl was pH-sensitively released due to both the hydrolysis of hydrazone bonds and the deactivation of CGA-ODNs under acid endo/lysosome environment (about pH 5.0), allowing nuclear accumulation of released DOX·HCl and which resulted in DOX·HCl toxicity in target cells. The effective drug delivery and controlled intracellular drug release made CPOD a potential NDDS to achieve greater anti-tumor efficacy with increased safety.
In this study, cationic intracellular pH-sensitive PEI-C-DOX was synthesized, and 1H nuclear magnetic resonance (1H NMR) spectroscopy was used to characterize the successful synthesis. The appropriate molar ratio of CGA-ODNs to DOX·HCl was tested using a fluorescence spectrofluorometer. The proper weight ratios of PEI-C-DOX to OD were evaluated by agarose gel electrophoresis assay, and the optimal formulations of POD and CPOD were chosen after further physical and chemical properties evaluation. The two-stage pH-sensitive of CPOD was confirmed. Firstly, the pH-triggered dissociating of CMCS from CPOD at subacid pH was confirmed in vitro, and pH-sensitive CPOD internalization were tested under different pH conditions using B16 and HepG2 cell lines, respectively. Secondly, the intracellular pH-sensitive DOX·HCl release was proved in vitro, and nuclear localization of released DOX·HCl was tested in B16 and HepG2 cell lines, respectively. The cytotoxicities of CPOD, DOX-free blank nanoparticles and PEI were evaluated in both the two cell lines. Furthermore, tumor accumulation of CPOD was confirmed using a real-time near infrared fluorophore (NIRF) imaging system. The in vivo anti-tumor efficacy of CPOD was evaluated in murine hepatoma cell line H22 tumor-bearing Kunming (KM) mice model. The plasma stability, hemolytic tests and histological analysis were used to investigate the preliminary safety of CPOD.
2. Materials and methods
2.1. Materials, cells and animals
Branched poly(ethyleneimine) (PEI, Mw 25 kDa) was purchased from Sigma-Aldrich (USA). C6-S-HyNic (C6-succinimidyl 6-hydrazinonicotinate acetone hydrazone, C6-SANH) was purchased from Solulink, Inc. (San Diego, CA, USA). Doxorubicin hydrochloride (DOX·HCl) was purchased from Dalian Meilun Biology Technology Co., Ltd (Dalian, China). O-Carboxymethyl-chitosan (CMCS) (average Mw 50 kDa; degree of carboxymethyl substitution = 60%; degree of deacetylation = 85%) was obtained from Jinan Haidebei Biological Engineering Co. (Jinan, China). 21-base (CGA)7 oligodeoxynucleotides (CGA-ODNs) (sequence: 5′-CGACGACGACGACGACGACGA-3′; complementary sequence: 5′-TCGTCGTCGTCGTCGTCGTCG-3′) were purchased from BGI tech (Shenzhen, China). GoldView was purchased from Beijing Saibaisheng Biological Engineering Co., Ltd (Beijing, China). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was purchased from Sigma-Aldrich (USA). Hoechst 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 342 was obtained from Invitrogen (USA). Sulfo-Cyanine5 carboxylic acid (Cy5) was obtained from LITTLE-PA Sciences Co., Ltd. (Wuhan, China). Cy5 labeled CGA-ODNs (Cy5-CGA-ODNs) (sequence: 5′-Cy5-CGACGACGACGACGACGACGA-3′; complementary sequence: 5′-TCGTCGTCGTCGTCGTCGTCG-3′) were purchased from BGI tech (Shenzhen, China).
342 was obtained from Invitrogen (USA). Sulfo-Cyanine5 carboxylic acid (Cy5) was obtained from LITTLE-PA Sciences Co., Ltd. (Wuhan, China). Cy5 labeled CGA-ODNs (Cy5-CGA-ODNs) (sequence: 5′-Cy5-CGACGACGACGACGACGACGA-3′; complementary sequence: 5′-TCGTCGTCGTCGTCGTCGTCG-3′) were purchased from BGI tech (Shenzhen, China).
Human hepatoma cell line HepG2, murine melanoma cell line B16 and murine hepatoma cell line H22 were kindly provided by Institute of Immunopharmacology and Immunotherapy of Shandong University (Jinan, China). HepG2 cell line was cultured in Dulbecco's Modified Eagle's Medium (DMEM) medium, and B16 cell line was cultured in Roswell Park Memorial Institute (RPMI) 1640 medium supplemented with 10% FBS at 37 °C and 5% CO2.
Female Kunming mice weighing 18–22 g were supplied by Medical Animal Test Center of the New Drugs Evaluation Center, Shandong University. All experiments were carried out in compliance with the Animal Management Rules of the Ministry of Health of the People's Republic of China (document number 55, 2001) and the Animal Experiment Ethics Review of Shandong University. All procedures were approved by the Animal Care and Use Committee.
2.2. Synthesis and characterization of PEI-C-DOX
As illustrated in Scheme 2, synthesis of PEI-C-DOX was divided into two steps. The synthesis route was shown in Scheme 2.The solutions were mixed in a balloon flask (50 mL) under stirring, and pH 6.0 of PBS was added into the flask up to 20 mL. The reaction solution was stirred for 24 h at room temperature in dark and environment of nitrogen. The obtained product was purified by dialysis against distilled water (MWCO 3500), lyophilized, the chemical structure was confirmed by 1H NMR spectroscopy in D2O and infrared spectroscopy (IR) (6700 FT-IR NXR FT-RAMAN, Nicolet, USA).
 | (1) |
2.3. Preparation and characterization of OD
DOX·HCl was dissolved in double distilled water (ddH2O), then DOX·HCl and CGA-ODNs were mixed and incubated at room temperature to obtain OD. To confirm the intercalating molar ratio of DOX·HCl to CGA-ODNs, CGA-ODNs (50 μM) was added stepwise to a fixed concentration of DOX·HCl (2.5 μM) in ddH2O, and the fluorescence intensity of OD was monitored at an excitation wavelength (excitation) of 493 nm and emission wavelength (emission) from 520 nm to 700 nm using a fluorescence spectrofluorometer (F-7000, HITACHI, Japan). The drug loading capacity of OD was calculated using the following equation:
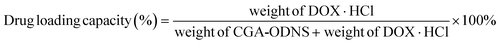 | (2) |
The drug loading capacities of POD and CPOD were calculated using the following equation, respectively, and the results were expressed as the mean ± SD (n = 3):
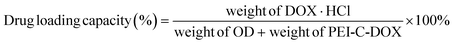 | (3) |
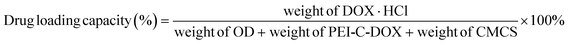 | (4) |
To observe a possible fluorescence recovery of DOX·HCl, the OD was placed at 4 °C for 24 h. The fluorescence intensity of OD was measured at the same condition.
2.4. Preparation and characterization of CPOD
An agarose gel electrophoresis assay was used to evaluate the weight ratios between PEI-C-DOX and OD. POD prepared at various weight ratios of PEI in PEI-C-DOX to CGA-ODNs (339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4096, 339
4096, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2048, 339
2048, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1024, 339
1024, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 512, 339
512, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 256, 339
256, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128, 339
128, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64, 339
64, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32, 339
32, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16) was mixed with an appropriate amount of 6× loading buffer and electrophoresed on a 1% (w/v) agarose gel containing an appropriate amount of GoldView (TAE buffer, 90 V, 10 min). The electrophoretic mobility of POD was visualized using a UV transilluminator and digital imaging system (IS-2200, Alpha Innotech, USA). The particle size of POD was determined using dynamic light scattering (DLS), (Zetasizer Nano ZS-90, Malvern, UK) at 25 °C.
16) was mixed with an appropriate amount of 6× loading buffer and electrophoresed on a 1% (w/v) agarose gel containing an appropriate amount of GoldView (TAE buffer, 90 V, 10 min). The electrophoretic mobility of POD was visualized using a UV transilluminator and digital imaging system (IS-2200, Alpha Innotech, USA). The particle size of POD was determined using dynamic light scattering (DLS), (Zetasizer Nano ZS-90, Malvern, UK) at 25 °C.
Similarly, the mixture binding conditions for CPOD with different weight ratios of CMCS to CGA-ODNs (75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128, 75
128, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64, 75
64, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32, 75
32, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16, 75
16, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, 75
8, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, 75
4, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 75
2, 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were evaluated by agarose gel electrophoresis assay with the same method. The study was conducted to examine whether the addition of CMCS led to the decomposition of the complex. The zeta potentials of CPOD were determined using DLS at 25 °C.
1) were evaluated by agarose gel electrophoresis assay with the same method. The study was conducted to examine whether the addition of CMCS led to the decomposition of the complex. The zeta potentials of CPOD were determined using DLS at 25 °C.
2.5. In vitro evaluation of firstly/extracellular pH-sensitive
To quantify the cellular uptake, the cells were treated as above. After washing three times with cold PBS, all the cells were trypsinized and washed with cold PBS, then the cells were resuspended in 100 μL of PBS after centrifugated. The quantified cellular uptake was carried out in triplicate and detected using a flow cytometer (FACSCaliber, Becton Dickinson, USA). The results were expressed as the mean ± SD (n = 3).
2.6. In vitro evaluation of secondly/intracellular pH-sensitive
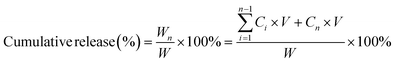 | (5) |
2.7. Cytotoxicity assay
In vitro antitumor efficiency of CPOD was evaluated by MTT assay using B16 and HepG2 cells, respectively. B16 or HepG2 cells were seeded into 96-well plates at a density of 5 × 103 and cultured for 24 h at 37 °C before the assay, respectively. Different concentrations of free DOX·HCl, CPOD, PEI and blank nanoparticles (CMCS/PEI/CGA-ODNs) prepared as the same method of CPOD were added to five wells each concentration and incubated for 48 h, respectively. Following that, 20 μL MTT (5 mg mL−1) was added to each well and the cells were incubated for another 4 h at 37 °C. The media were removed and then 150 μL DMSO was added to each well to dissolve the formazan crystals formed by living cells. Cells without treatment were used as control. Cell culture media (RPMI 1640 or DMEM) without cells was used as blank. The absorbance at a test wavelength of 570 nm and a reference wavelength of 630 nm in each well was recorded using a Microplate Reader (Elx800, BioTek, USA). The MTT assay was carried out in triplicate. Cell viability was calculated according to the following equation, and the results were expressed as the mean ± SD (n = 3):
 | (6) |
2.8. Plasma stability and hemolytic tests
In order to investigate the stability and safety of CPOD in blood circulation, and prepared for the following in vivo experiments. Plasma stability and hemolytic tests were conducted.
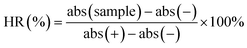 | (7) |
| Number | |||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 (−) | 7 (+) | |
| CPOD (mL) | 0.1 | 0.2 | 0.3 | 0.4 | 0.5 | — | — |
| Normal saline (mL) | 2.4 | 2.3 | 2.2 | 2.1 | 2.0 | 2.5 | — |
| Distilled water (mL) | — | — | — | — | — | — | 2.5 |
| 2% RBCs solution (mL) | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| Concentration of DOX·HCl (μg mL−1) | 2 | 4 | 6 | 8 | 10 | ||
2.9. In vivo biodistribution of CPOD
The healthy female Kunming (KM) mice (age of six weeks; body weight of 18–22 g) were obtained from Medical Animal Test Center of the New Drugs Evaluation Center, Shandong University.The tumor-bearing mice model was established by inoculating subcutaneously of H22 hepatic cancer cells (2 × 107) in the right flank of each mouse. Water-soluble dye free Cy5 was used to mimic the biodistribution of free DOX·HCl as a positive control, CPOD prepared by Cy5-CGA-ODNs was used as experimental group. When tumor volume reached about 100 mm3, the tumor-bearing mice were administrated with free Cy5 and CPOD prepared with Cy5-CGA-ODNs through tail vein at a dosage of 62.5 nmol kg−1, respectively. At desired time intervals (1 h, 2 h, 4 h, 8 h, 24 h), the mice were imaged using a real-time near infrared fluorophore (NIRF) imaging system (IVIS Kinetic, Cailper Life Science, USA) at appropriate wavelength (Ex 640 nm, Em 694 nm) after narcosis. After 24 h, the mice were sacrificed, tumors and main organs including heart, liver, spleen, lung, and kidney were excised and imaged using a NIRF imaging system. The fluorescence intensities of tumors and main organs were then quantitated and analyzed by Living Image 3.1 software (Cailper Life Science, USA).
2.10. In vivo anti-tumor efficacy
The tumor-bearing KM mice model was established as the same method in Section 2.9. Then the tumor-bearing mice (n = 24) were randomly and equally divided into four groups: normal saline group, free DOX·HCl group (1 mg kg−1 and double dosage 2 mg kg−1) and CPOD group (1 mg kg−1). When the tumor volumes reached about 100 mm3, mice in the four groups were administrated with the formulations through tail vein everyday (total for six times), respectively. The tumor dimensions were measured with a vernier caliper on day 1, 3, 5, 7, 9, 11, and the tumor volume was calculated using the formula:
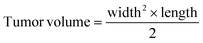 | (8) |
In addition, body weights of each mice were recorded everyday. On day 11, each group of mice were sacrificed, and the tumors were excised, following weighting.
2.11. Histological analysis
The healthy female KM mice were administrated with the formulations through tail vein at the same dosage and frequency as mentioned in Section 2.10. On day 11, each group of mice was sacrificed, the organs including heart, liver, spleen, lung, and kidney were excised and fixed with formalin, embedded in paraffin, and cut into 5 μm thick sections for hematoxylin and eosin (H&E) staining. Images were observed using a light microscope.3. Results and discussion
3.1. Synthesis and characterization of PEI-C-DOX
In order to obtain the feature of dual drug-loading, branched PEI was selected as the material both for chemical conjugation and OD condensing. A heterobifunctional crosslinking agent, C6-SANH, was used to prepare the PEI-C-DOX with intracellular pH-sensitive hydrazone bonds. C6-SANH helped to avoid the complex synthesis steps, harsh reaction conditions and high amounts of organic solvents in hydrazone bond synthesis process. Thus, the synthesis of PEI-C-DOX took advantages of simple operation and mild reaction conditions, which was divided into two steps, and synthesis route was as shown in Scheme 2.PEI-C-DOX was further verified by IR spectroscopy. As shown in Fig. 1G and H, the carbonyl peak of DOX·HCl at 1730 cm−1 was disappeared after linked to PEI-C. In addition, although the peaks of several functional groups appeared around 1600 cm−1, such as amide, carbon–carbon double bond, benzene and hydrazone bond, the peak at 1652 cm−1 (Fig. 1H) was most likely attributed to the absorption of hydrazone bond. Because as shown in Fig. 1F and H, the intensity of absorption peak at around 1600 cm−1 (1633 cm−1 in Fig. 1F and 1652 cm−1 in Fig. 1H) was obviously increased after conjugation of DOX·HCl (Fig. 1H). It indicated that PEI-C conjugated to the carbonyl of DOX·HCl via hydrazone bond, and confirmed the successful synthesis of intracellular pH-sensitive PEI-C-DOX.
3.2. Preparation and characterization of OD
The preparation of OD was easily operated by incubation at room temperature, and did not induce any other modifications of the drug or CGA-ODNs in the process. It has been reported that the fluorescence of DOX·HCl will quench after intercalated into CGA-ODNs.61,62 As shown in Fig. 2A, the fluorescence intensity of DOX·HCl decreased progressively along with the increase of CGA-ODNs after incubation with CGA-ODNs, suggesting the successful intercalation. When the molar ratio of CGA-ODNs to DOX·HCl was higher than 0.12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, there was no significant change of fluorescence intensity. It demonstrated that DOX·HCl was totally intercalated into CGA-ODNs with a high loading capacity of 8.33 DOX·HCl molecules per CGA-ODNs. Therefore, in the present work, the molar ratio was fixed at 0.12
1, there was no significant change of fluorescence intensity. It demonstrated that DOX·HCl was totally intercalated into CGA-ODNs with a high loading capacity of 8.33 DOX·HCl molecules per CGA-ODNs. Therefore, in the present work, the molar ratio was fixed at 0.12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for the following experiment. The drug loading capacity of OD was 27.3%, and the drug loading capacities of POD and CPOD were 16.2 ± 0.977% and 4.72 ± 0.09%, respectively. To investigate the store stability of OD, OD at molar ratio of 0.12
1 for the following experiment. The drug loading capacity of OD was 27.3%, and the drug loading capacities of POD and CPOD were 16.2 ± 0.977% and 4.72 ± 0.09%, respectively. To investigate the store stability of OD, OD at molar ratio of 0.12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was placed at 4 °C for 24 h and the emission spectrum was as shown in Fig. 2B, and fluorescence of DOX·HCl was hardly recovered indicating the stability of the intercalation.
1 was placed at 4 °C for 24 h and the emission spectrum was as shown in Fig. 2B, and fluorescence of DOX·HCl was hardly recovered indicating the stability of the intercalation.
 | ||
| Fig. 2 Fluorescence emission spectra of OD with different molar ratios of CGA-ODNs to DOX·HCl (A). Fluorescence emission spectra of OD stored at 4 °C for 24 h (B). | ||
3.3. Preparation and characterization of CPOD
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4096 to 339
4096 to 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 and agarose gel electrophoresis assay was carried out to determine the OD binding ability of PEI-C-DOX. As shown in Fig. S1A,† at low weight ratios of PEI to CGA-ODNs, a fraction of migration-free OD could still be visualized. When the weight ratio of PEI to CGA-ODNs was up to 339
16 and agarose gel electrophoresis assay was carried out to determine the OD binding ability of PEI-C-DOX. As shown in Fig. S1A,† at low weight ratios of PEI to CGA-ODNs, a fraction of migration-free OD could still be visualized. When the weight ratio of PEI to CGA-ODNs was up to 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 256, free OD could not be observed, indicating that OD was completely condensed by PEI-C-DOX. Subsequently, the size distributions of POD with weight ratios between 339
256, free OD could not be observed, indicating that OD was completely condensed by PEI-C-DOX. Subsequently, the size distributions of POD with weight ratios between 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 256 to 339
256 to 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 were measured as shown in Fig. S1B.† For mass ratios ≤339
16 were measured as shown in Fig. S1B.† For mass ratios ≤339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 512, POD could not be accurately analyzed due to incomplete complex formation and too broad complex size ranges. When the weight ratio of PEI to CGA-ODNs increased from 339
512, POD could not be accurately analyzed due to incomplete complex formation and too broad complex size ranges. When the weight ratio of PEI to CGA-ODNs increased from 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 256 to 339
256 to 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64, the average sizes of POD were 140–150 nm. In this phase, the mean size of POD was hardly changed, the binding ability between PEI-C-DOX to already formed POD may be described as an exponential function.63 Then, an increase size of POD was observed in the weight ratio from 339
64, the average sizes of POD were 140–150 nm. In this phase, the mean size of POD was hardly changed, the binding ability between PEI-C-DOX to already formed POD may be described as an exponential function.63 Then, an increase size of POD was observed in the weight ratio from 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 to 339
32 to 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16, indicating that the POD suspension became unstable. At the weight ratio of 339
16, indicating that the POD suspension became unstable. At the weight ratio of 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64, a more symmetrical size distribution with a small polydispersity index (PDI) was observed, and some excessive positive charges provided by PEI-C-DOX was necessary for electrostatic crosslinking with CMCS. In summary, 339
64, a more symmetrical size distribution with a small polydispersity index (PDI) was observed, and some excessive positive charges provided by PEI-C-DOX was necessary for electrostatic crosslinking with CMCS. In summary, 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64 was chosen as the optimal weight ratio for the following experiments.
64 was chosen as the optimal weight ratio for the following experiments.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128 to 75
128 to 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were shown in Fig. S2A.† At the test weight ratios of CMCS to CGA-ODNs, no free OD was observed, indicating that CMCS could not affect the stability of POD. Additionally, as shown in Fig. S2B,† the addition of negatively charged CMCS could result in surface charge (zeta potential) variations. Along with the weight ratios increasing, the zeta potentials of CPOD decreased from (20.4 ± 1.9) mV and finally reached a plateau of about −15 mV. The charges overturn from positive to negative indicating that anionic CMCS was coated on cationic POD at pH 7.4. The plateau reflection point of 75
1) were shown in Fig. S2A.† At the test weight ratios of CMCS to CGA-ODNs, no free OD was observed, indicating that CMCS could not affect the stability of POD. Additionally, as shown in Fig. S2B,† the addition of negatively charged CMCS could result in surface charge (zeta potential) variations. Along with the weight ratios increasing, the zeta potentials of CPOD decreased from (20.4 ± 1.9) mV and finally reached a plateau of about −15 mV. The charges overturn from positive to negative indicating that anionic CMCS was coated on cationic POD at pH 7.4. The plateau reflection point of 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (CMCS to CGA-ODNs) was selected as the ideal weight ratio for the following experiments.
4 (CMCS to CGA-ODNs) was selected as the ideal weight ratio for the following experiments.| Average size (nm) | PDI | Zeta potential (mV) | |
|---|---|---|---|
POD (w/w = 339![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 64) 64) |
140.2 ± 5.2 | 0.144 ± 0.0182 | 20.4 ± 1.9 |
CPOD (w/w = 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
165.0 ± 3.3 | 0.191 ± 0.0097 | −15.6 ± 0.82 |
The TEM picture and size distributions were as shown in Fig. 3A and B. Both POD and CPOD displayed a spherical morphology with a narrow distribution, and the results of PDI (<0.2 both for POD and CPOD) showed in Table 2 was further indication of the characteristic colloidal suspension reliable disparity.
3.4. In vitro evaluation of firstly/extracellular pH-sensitivity
At pH 7.4, in both B16 and HepG2 cell lines, POD displayed similar internalization to DOX·HCl solution, which were all close to 100%, indicating the strong cellular uptake ability of POD. However, less uptake of CPOD than POD at pH 7.4 was observed, while, at pH 6.5 and pH 6.0 the cellular uptake of CPOD was no significant differences to that of POD and DOX·HCl solution at pH 7.4. It may be because in normal physiological environment (pH 7.4), the negatively charged CMCS coating made CPOD difficult to approach the same electronegative cytomembranes and caused little internalization of CPOD.66–68 Meanwhile, in tumor subacid extracellular pH (pH 6.5 and pH 6.0), the outermost anionic layer, CMCS, may completely fall off and then exposed to the cationic POD. The exposed positive POD enhanced the cellular uptake of the payloads.
3.5. In vitro evaluation of secondly/intracellular pH-sensitivity
 | ||
| Fig. 6 In vitro DOX·HCl release from DOX·HCl solution, CPO and COD (A); DOX·HCl solution and CPOD (B) at pH 5.0 and pH 7.4 of PBS buffers (0.01 M), respectively, *p < 0.05, ***p < 0.005. | ||
As shown in Fig. 6B, the amount of DOX·HCl released from CPOD was both time- and pH-dependent. After 48 h, 73.89 ± 2.91% DOX·HCl was released from CPOD at pH 5.0, which was significantly more than that at pH 7.4 (p < 0.005, about 40.44 ± 1.27% of DOX·HCl was released from CPOD at pH 7.4 under the same conditions). These results revealed that intracellular pH-sensitive CPOD would rapidly release DOX·HCl in response to endo/lysosomal pH. It confirmed that CPOD was an ideal platform for constructing intracellular pH-responsive system.
Although, most of red fluorescence accumulated in cytoplasm of both cell lines treated with POD (at pH 7.4) and CPOD (at pH 6.5 and pH 6.0) and much weaker purple fluorescence was showed in the nuclei. It may be attributed to that internalization of free DOX·HCl was due to efficient passive diffusion,70 while the cellular uptake of POD (at pH 7.4) and CPOD (at pH 6.5 and 6.0) as well as intracellular pH-sensitive DOX·HCl release needed longer time. On the other hand, B16 and HepG2 cell lines were treated with POD and CPOD for a longer incubation time (4 h), respectively. After 4 h, there was stronger purple fluorescence in nuclei of both cell lines treated with POD (at pH 7.4) and CPOD (at pH 6.5 and 6.0) than that at 3 h, indicating much more DOX·HCl was intracellular released and located in nuclei. For CPOD, no purple fluorescence was observed at pH 7.4 of both cell lines, while at pH 6.5 (of both cell lines) and pH 6.0 (of B16 cell line) the intensity of purple fluorescence was similar to that of POD. At pH 6.0 of HepG2 cell line, the intensity of purple fluorescence was much stronger than others, further indicating the extracellular pH-sensitive function. The effective nuclear localization of CPOD at tumor extracellular pH (pH 6.5 and 6.0) after 4 h demonstrated two-stage pH-sensitive function, which had first high cellular uptake at tumor extracellular pH and second intracellular pH-sensitive DOX·HCl release.
3.6. Cytotoxicity assay
The in vitro therapeutic efficacy of CPOD was tested using B16 and HepG2 cell lines and compared with DOX·HCl solution as a positive control. Both DOX·HCl and CPOD exhibited a dosage-dependent cytotoxic effect in B16 and HepG2 cells (Fig. 8A and B, respectively). In B16 cell line, the (half maximal inhibitory concentration) IC50 of DOX·HCl solution was 0.277 ± 0.031 μg mL−1 while IC50 of CPOD was 1.11 ± 0.085 μg mL−1. DOX·HCl solution showed higher cytotoxicity than CPOD (p < 0.05). Meanwhile, in HepG2 cell lines, the cytotoxicity of CPOD was similar to DOX·HCl solution (IC50 of DOX·HCl solution and CPOD was 3.82 ± 0.51 μg mL−1 and 3.66 ± 0.14 μg mL−1, respectively). The results can be explain by different sensibility of various cell lines to DOX·HCl. In addition, the internalization of CPOD and DOX·HCl release from CPOD needed a longer time than passive diffusion of DOX·HCl solution, thus the in vitro cytotoxicity of CPOD was reduced at the test concentration range.The cytotoxicity of nanocarriers is an important factor that should be taken into consideration. As shown in Fig. 8C and D, after incubation with blank nanoparticles for 48 h, the cell viabilities were higher than 90% at all experimental dosage for both B16 and HepG2 cells, indicating the low toxicities of blank nanoparticles. Meanwhile as shown in Fig. 8E and F, high cytotoxicity of PEI was observed in both the two cells (the cell viabilities were lower than 40% at all experiment dosages). This may be attributed to the fact that the positive charges of PEI was shielded by the anionic shell, CMCS, which was able to improve the viability of the cells, thus preliminary proving the in vitro safety of the bland nanoparticles.
3.7. Plasma stability and hemolytic tests
 | ||
| Fig. 9 The average size of POD and CPOD after incubation with 10% plasma (A). The image of hemolysis test and hemolysis percentage of CPOD (B), *p < 0.05. | ||
However, the size of POD had abrupt increase (p < 0.05), and after 2 h, macroscopic sediments were observed. It may be caused by the existence of substances in plasma, such as proteins. The plasma proteins were easy to absorb on the surface of positive POD, which caused the change of particle size and formation of coagulations.71,72 CMCS coating could relieve the adsorption and make CPOD stable in blood circulation.
3.8. In vivo biodistribution of CPOD
The tumor target and tissue biodistribution of CPOD was evaluated by the in vivo biodistribution study using H22 tumor-bearing KM mice model. As shown in Fig. 10A, after 1 h of administration through tail vein, fluorescence signal was observed in both free Cy5 and CPOD groups. The distribution of free Cy5 was non-selective, at 24 h, no fluorescence signal was observed indicating the elimination of free Cy5. However, CPOD accumulated in tumor within 1 h and were distinctly observed up to 24 h post-injection at the tumor site. At the same time, the live accumulation of CPOD was observed from 1 h to 8 h, and CPOD was also shown in spleen at 8 h. The liver or spleen accumulation pattern of CPOD was highly representative of charged nanoparticles, widely known to undergo a sequestration in the major organs of the reticuloendothelial system (RES), mainly in the liver and spleen.74 The effective tumor target of CPOD was based on EPR effect, which was due to the suitable particle size of CPOD [(165.0 ± 3.3) nm]. This caused an efficient translocation through the tumor angiogenesis and accumulation in tumor tissues. The fluorescence signal of CPOD in bladder indicated the renal excretion of CPOD. At 24 h, free Cy5 was already eliminated, but CPOD still accumulated in tumor and no fluorescence signal in other tissues was observed. Subsequently, the tumor and major organs including heart, liver, spleen, lung, and kidney were collected and imaged after the mice were sacrificed at 24 h post-injection. The image is shown in Fig. 10B. After quantitation, the fluorescence intensity of tumor in CPOD group was 2.261 × 107, which was 3.46 times stronger than that of free Cy5 group (6.533 × 106). It further demonstrated the effective tumor accumulation of CPOD. Simultaneously, CPOD showed a high liver accumulation. The fluorescence intensity of liver (3.676 × 108) was 7.95 times stronger than that of free Cy5 group (4.624 × 107), which was consistent with the in vivo imaging results. The fluorescence intensity of kidney in CPOD group (3.211 × 107) was 4.03 times stronger than that of free Cy5 group (7.964 × 106), further indicating the renal excretion of CPOD.In profile, the retention of CPOD in tumor tissues observed in both the in vivo and ex vivo imaging was beneficial for anti-tumor efficacy. It was also helpful in elimination in other tissues, which improved medicinal safety of CPOD.
3.9. In vivo anti-tumor efficacy
Anti-tumor efficacy of CPOD was assessed on H22 tumor-bearing KM mice. The changes of tumor volumes, tumor weights and body weights of the mice were as shown in Fig. 11. After 11 days of treatment, the average tumor volume of mice treated with normal saline reached about 600 mm3 (Fig. 11A). For CPOD group, the average tumor volume was about 100 mm3, which was significantly smaller than that of 1 mg kg−1 DOX·HCl group (p < 0.005) and double dosage 2 mg kg−1 DOX·HCl group (p < 0.005). Thereafter, the mice were sacrificed, and then the solid tumors were excised. As seen in Fig. 11B, the tumors of CPOD group were smaller than tumors of other groups. Upon weighing of the tumors (Fig. 11C), the average tumor weight of mice treated with CPOD was 146.8 ± 76.8 mg, which was significantly lighter than that of 1 mg kg−1 DOX·HCl group (p < 0.005) and 2 mg kg−1 DOX·HCl group (p < 0.05). Overall, in contrast to the lower cytotoxicity in vitro, CPOD showed preliminarily greater in vivo anti-tumor efficacy than free DOX·HCl (even better than double dosage of DOX·HCl solution) in H22 tumor-bearing KM mice model. The result could be explained as follows. Firstly, the in vivo biodistribution of DOX·HCl was changed after loaded into CPOD, CPOD was demonstrated to target tumor tissue and accumulate in it for a longer time in Section 3.8. Secondly, in the Sections 3.4 and 3.5, the internalization in tumor subacid microenvironment and controlled intracellular pH-sensitive drug release of CPOD was confirmed, and it was beneficial to improve the anti-tumor efficacy of CPOD.The body weight changes were monitored for reflecting the adverse effects of various DOX·HCl formulations. As shown in Fig. 11D, all groups exhibited similar smooth fluctuations of body weight (p > 0.05), suggesting weight loss caused by systemic toxicities wasn't revealed in the test.
3.10. Histological analysis
To further evaluate the safety of CPOD on main organs, histological evaluation effect was conducted, and administration was the same with in vivo anti-tumor efficacy test. The H&E staining histological sections of main organs including heart, liver, spleen, lung and kidney were shown in Fig. 12. The heart, liver, spleen, lung and kidney of CPOD group showed normal histological morphology, and no visible difference was observed in comparison with the negative control (normal saline) group. However, for 1 mg kg−1 and 2 mg kg−1 DOX·HCl solution groups, there was vacuolation degeneration in the cells of hearts, livers, and inflammatory cells infiltration in the lungs. Moreover, the heart, liver and lung tissues damage and inflammation of 2 mg kg−1 DOX·HCl solution group was more serious than that of 1 mg kg−1 DOX·HCl solution group, indicating that DOX·HCl solution may have a dosage-dependent toxicity on main organs caused by non-specific in vivo biodistribution. In the in vivo biodistribution test, CPOD showed liver accumulation, however lesions were not observed in the liver section. It may be that CPOD could keep stable in normal tissues and seldom DOX·HCl was released. It preliminarily indicated that CPOD was a potential platform for DOX·HCl loading to improve medicinal safety.4. Conclusion
In conclusion, a two-stage pH-sensitive DOX·HCl loaded core–shell nanoparticles with dual drug-loading strategies aiming at improving antitumor efficacy and medicinal safety were simultaneously designed and investigated. DOX·HCl was loaded in inner core POD by dual drug-loading strategies of chemical modification combining with CGA-ODNs loading method, and thereafter extracellular pH-sensitive CMCS was coated on POD to construct the core–shell nanoparticles CPOD. Both POD and CPOD showed spherical and uniform size distribution. CMCS could shelter the positively charged POD, stailize and prolong blood circulation time of CPOD. CPOD was observed to accumulate in tumor tissues, which lasted for 24 h. Meanwhile, at 24 h the ex vivo fluorescence intensity of tumor in CPOD group was 3.46 times stronger than that of free drug group. After enriched in tumor tissues, CPOD played the two-stage pH-sensitive functions that were firstly pH-sensitive internalization and secondly intracellular pH-sensitive DOX·HCl release, which finally achieved the cytotoxicity of nuclear localizated DOX·HCl. More importantly, CPOD displayed a good curative efficacy on the inhibition of tumor growth with a fair degree of safety. This approach provides a new platform with effective DOX·HCl delivery and controlled drug release to improve anti-tumor efficacy and improve medicinal safety.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81402867). A special thanks to Dr Livesey Olerile for his support of English grammar corrections.Notes and references
- J. Chen, W. Zhang, M. Zhang, Z. Guo, H. Wang, M. He, P. Xu, J. Zhou, Z. Liu and Q. Chen, Nanoscale, 2015, 7, 12542–12551 RSC.
- Q. Cheng, L. Du, L. Meng, S. Han, T. Wei, X. Wang, Y. Wu, X. Song, J. Zhou, S. Zheng, Y. Huang, X. J. Liang, H. Cao, A. Dong and Z. Liang, ACS Appl. Mater. Interfaces, 2016, 8, 4347–4356 CAS.
- H. Mcilwain, Nature, 1943, 151, 270–273 CrossRef CAS.
- Z. Khatun, M. Nurunnabi, M. Nafiujjaman, G. R. Reeck, H. A. Khan, K. J. Cho and Y. K. Lee, Nanoscale, 2015, 7, 10680–10689 RSC.
- J. Liu, B. Zhang, Z. Luo, X. Ding, J. Li, L. Dai, J. Zhou, X. Zhao, J. Ye and K. Cai, Nanoscale, 2015, 7, 3614–3626 RSC.
- D. B. Pacardo, F. S. Ligler and Z. Gu, Nanoscale, 2015, 7, 3381–3391 RSC.
- V. Torchilin, Adv. Drug Delivery Rev., 2011, 63, 131–135 CrossRef CAS PubMed.
- D. Chao, Z. Zhong, Y. Jiang, C. Ru and F. Meng, Nano Today, 2012, 7, 467–480 CrossRef.
- F. Danhier, O. Feron and V. Preat, J. Controlled Release, 2010, 148, 135–146 CrossRef CAS PubMed.
- N. Bertrand, J. Wu, X. Xu, N. Kamaly and O. C. Farokhzad, Adv. Drug Delivery Rev., 2014, 66, 2–25 CrossRef CAS PubMed.
- Y. J. Son, J. S. Jang, Y. W. Cho, H. Chung, R. W. Park, I. C. Kwon, I. S. Kim, J. Y. Park, S. B. Seo, C. R. Park and S. Y. Jeong, J. Controlled Release, 2003, 91, 135–145 CrossRef CAS PubMed.
- H. Li, Y. Cui, J. Sui, S. Bian, Y. Sun, J. Liang, Y. Fan and X. Zhang, ACS Appl. Mater. Interfaces, 2015, 7, 15855–15865 CAS.
- S. Lv, Z. Tang, M. Li, J. Lin, W. Song, H. Liu, Y. Huang, Y. Zhang and X. Chen, Biomaterials, 2014, 35, 6118–6129 CrossRef CAS PubMed.
- T. Wei, C. Chen, J. Liu, C. Liu, P. Posocco, X. Liu, Q. Cheng, S. Huo, Z. Liang, M. Fermeglia, S. Pricl, X. J. Liang, P. Rocchi and L. Peng, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 2978–2983 CrossRef CAS PubMed.
- J. Lu, W. Zhao, Y. Huang, H. Liu, R. Marquez, R. B. Gibbs, J. Li, R. Venkataramanan, L. Xu, S. Li and S. Li, Mol. Pharm., 2014, 11, 4164–4178 CrossRef CAS PubMed.
- Y. F. Song, D. Z. Liu, Y. Cheng, M. Liu, W. L. Ye, B. L. Zhang, X. Y. Liu and S. Y. Zhou, Sci. Rep., 2015, 5, 16125 CrossRef CAS PubMed.
- C. Fang, F. M. Kievit, O. Veiseh, Z. R. Stephen, T. Wang, D. Lee, R. G. Ellenbogen and M. Zhang, J. Controlled Release, 2012, 162, 233–241 CrossRef CAS PubMed.
- J. Yu, Y. Zhang, W. Sun, C. Wang, D. Ranson, Y. Ye, Y. Weng and Z. Gu, Nanoscale, 2016, 8, 9178–9184 RSC.
- J. Xu, Q. Zhao, Y. Jin and L. Qiu, Nanomedicine, 2014, 10, 349–358 CAS.
- J. Guo, H. Hong, G. Chen, S. Shi, Q. Zheng, Y. Zhang, C. P. Theuer, T. E. Barnhart, W. Cai and S. Gong, Biomaterials, 2013, 34, 8323–8332 CrossRef CAS PubMed.
- C. Liu, F. Liu, L. Feng, M. Li, J. Zhang and N. Zhang, Biomaterials, 2013, 34, 2547–2564 CrossRef CAS PubMed.
- Y. Wang, Y. Sun, J. Wang, Y. Yang, Y. Li, Y. Yuan and C. Liu, ACS Appl. Mater. Interfaces, 2016, 8, 17166–17175 CAS.
- Y. Chen, H. Chen and J. Shi, Adv. Mater., 2013, 25, 3144–3176 CrossRef CAS PubMed.
- Y. Bao, M. Yin, X. Hu, X. Zhuang, Y. Sun, Y. Guo, S. Tan and Z. Zhang, J. Controlled Release, 2016, 235, 182–194 CrossRef CAS PubMed.
- A. Trouet, D. D. Campeneere and C. De Duve, Nature, 1972, 239, 110–112 CrossRef CAS.
- V. Bagalkot, O. C. Farokhzad, R. Langer and S. Jon, Angew. Chem., Int. Ed. Engl., 2006, 45, 8149–8152 CrossRef CAS PubMed.
- D. Kim, Y. Y. Jeong and S. Jon, ACS Nano, 2010, 4, 3689–3696 CrossRef CAS PubMed.
- M. Chang, C. S. Yang and D. M. Huang, ACS Nano, 2011, 5, 6156–6163 CrossRef CAS PubMed.
- I. H. Lee, M. K. Yu, I. H. Kim, J. H. Lee, T. G. Park and S. Jon, J. Controlled Release, 2011, 155, 88–95 CrossRef CAS PubMed.
- L. Meng, L. Yang, X. Zhao, L. Zhang, H. Zhu, C. Liu and W. Tan, PLoS One, 2012, 7, e33434 CAS.
- T. Liu, M. Wang, T. Wang, Y. Yao and N. Zhang, Colloids Surf., B, 2015, 126, 531–540 CrossRef CAS PubMed.
- J. Kopecek, Adv. Drug Delivery Rev., 2013, 65, 49–59 CrossRef CAS PubMed.
- M. Li, Y. Liu, L. Feng, F. Liu, L. Zhang and N. Zhang, Colloids Surf., B, 2015, 131, 191–201 CrossRef CAS PubMed.
- L. Zhang and N. Zhang, Int. J. Nanomed., 2013, 8, 2927–2941 CrossRef PubMed.
- A. J. Ferdous, N. Y. Stembridge and M. Singh, J. Controlled Release, 1998, 50, 71–78 CrossRef CAS PubMed.
- M. Elsabahy and K. L. Wooley, Chem. Soc. Rev., 2012, 41, 2545–2561 RSC.
- Z. Su, Y. Liang, Y. Yao, T. Wang and N. Zhang, J. Mater. Chem. B, 2016, 4, 1122–1133 RSC.
- Y. Bae, S. Fukushima, A. Harada and K. Kataoka, Angew. Chem., Int. Ed., 2003, 42, 4640–4643 CrossRef CAS PubMed.
- S. Yoon, W. J. Kim and H. S. Yoo, Small, 2013, 9, 284–293 CrossRef CAS PubMed.
- R. Tang, W. Ji, D. Panus, R. N. Palumbo and C. Wang, J. Controlled Release, 2010, 151, 18–27 CrossRef PubMed.
- Z. Y. Qiao, F. S. Du, R. Zhang, D. H. Liang and Z. C. Li, Macromolecules, 2010, 43, 6485–6494 CrossRef CAS.
- D. M. Prescott, H. C. Charles, J. M. Poulson, R. L. Page, D. E. Thrall, Z. Vujaskovic and M. W. Dewhirst, Clin. Cancer Res., 2000, 6, 2501–2505 CAS.
- C. J. Gulledge and M. W. Dewhirst, Anticancer Res., 1996, 16, 741–749 CAS.
- E. S. Lee, Z. Gao and Y. H. Bae, J. Controlled Release, 2008, 132, 164–170 CrossRef CAS PubMed.
- M. Murakami, H. Cabral, Y. Matsumoto, S. Wu, M. R. Kano, T. Yamori, N. Nishiyama and K. Kataoka, Sci. Transl. Med., 2011, 3, 599–612 Search PubMed.
- S. Ganta, H. Devalapally, A. Shahiwala and M. Amiji, J. Controlled Release, 2008, 126, 187–204 CrossRef CAS PubMed.
- H. Maeda, T. Sawa and T. Konno, J. Controlled Release, 2001, 74, 47–61 CrossRef CAS PubMed.
- T. Li, M. Zhang, J. Wang, T. Wang, Y. Yao, X. Zhang, C. Zhang and N. Zhang, AAPS J., 2016, 18, 146–155 CrossRef CAS PubMed.
- S. E. Gratton, P. A. Ropp, P. D. Pohlhaus, J. C. Luft, V. J. Madden, M. E. Napier and J. M. DeSimone, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 11613–11618 CrossRef CAS PubMed.
- J. Z. Du, T. M. Sun, W. J. Song, J. Wu and J. Wang, Angew. Chem., Int. Ed., 2010, 49, 3703–3708 CrossRef.
- Y. Huang, Z. Tang, X. Zhang, H. Yu, H. Sun, X. Pang and X. Chen, Biomacromolecules, 2013, 14, 2023–2032 CrossRef CAS PubMed.
- H. Deng, X. Zhao, J. Liu, J. Zhang, L. Deng, J. Liu and A. Dong, Nanoscale, 2016, 8, 1437–1450 RSC.
- Y. Dong, J. Yang, H. Liu, T. Wang, S. Tang, J. Zhang and X. Zhang, Theranostics, 2015, 5, 890–904 CrossRef CAS PubMed.
- W. T. Godbey, K. K. Wu and A. G. Mikos, J. Controlled Release, 1999, 60, 149–160 CrossRef CAS PubMed.
- S. Kumar, S. Raj, K. Sarkar and K. Chatterjee, Nanoscale, 2016, 8, 6820–6836 RSC.
- A. R. Vancha, S. Govindaraju, K. V. Parsa, M. Jasti, M. González-García and R. P. Ballestero, BMC Biotechnol., 2004, 4, 663–664 CrossRef PubMed.
- F. Chen, J. Zhang, L. Wang, Y. Wang and M. Chen, Nanoscale, 2015, 7, 15763–15779 RSC.
- Y. Bae and K. Kataoka, Adv. Drug Delivery Rev., 2009, 61, 768–784 CrossRef CAS PubMed.
- L. Upadhyaya, J. Singh, V. Agarwal and R. P. Tewari, J. Controlled Release, 2014, 186, 54–87 CrossRef CAS PubMed.
- N. N. Inamdar and V. K. Mourya, Adv. Mater. Lett., 2010, 1, 11–33 CrossRef.
- H. T. Haj, M. Salerno, W. Priebe, H. Kozlowski and A. Garnier-Suillerot, Chem.-Biol. Interact., 2003, 145, 349–358 CrossRef CAS PubMed.
- L. Valentini, V. Nicolella, E. Vannini, M. Menozzi, S. Penco and F. Arcamone, Farmaco, Ed. Sci., 1985, 40, 377–390 CAS.
- I. Y. Perevyazko, M. Bauer, G. M. Pavlov, S. Hoeppener, S. Schubert, D. Fischer and U. S. Schubert, Langmuir, 2012, 28, 16167–16176 CrossRef CAS PubMed.
- Y. Yao, Z. Su, Y. Liang and N. Zhang, Int. J. Nanomed., 2015, 10, 6185–6198 CAS.
- F. Liu, M. Li, C. Liu, Y. Liu and N. Zhang, J. Biomed. Nanotechnol., 2014, 10, 3397–3406 CrossRef CAS PubMed.
- P. L. Rodriguez, T. Harada, D. A. Christian, D. A. Pantano, R. K. Tsai and D. E. Discher, Science, 2013, 339, 971–975 CrossRef CAS PubMed.
- T. Stylianopoulos, K. Soteriou, F. Dai and R. K. Jain, Ann. Biomed. Eng., 2013, 41, 68–77 CrossRef PubMed.
- T. Stylianopoulos, M. Z. Poh, N. Insin, M. G. Bawendi, F. Dai, L. Munn and R. K. Jain, Biophys. J., 2010, 99, 1342–1349 CrossRef CAS PubMed.
- E. Kim, Y. Jung, H. Choi, J. Yang, J. S. Suh, Y. M. Huh, K. Kim and S. Haam, Biomaterials, 2010, 31, 4592–4599 CrossRef CAS PubMed.
- C. Fang, F. M. Kievit, Y. C. Cho, H. Mok, O. W. Press and M. Zhang, Nanoscale, 2012, 4, 7012–7020 RSC.
- K. Fukukawa, R. Rossin, A. Hagooly, E. D. Pressly, J. N. Hunt, B. W. Messmore, K. L. Wooley, M. J. Welch and C. J. Hawker, Biomacromolecules, 2008, 9, 1329–1339 CrossRef CAS PubMed.
- E. D. Pressly, R. Rossin, A. Hagooly, K. Fukukawa, B. W. Messmore, M. J. Welch, K. L. Wooley, M. S. Lamm, R. A. Hule, D. J. Pochan and C. J. Hawker, Biomacromolecules, 2007, 8, 3126–3134 CrossRef CAS PubMed.
- T. Ramasamy, Z. S. Haidar, T. H. Tran, J. Y. Choi, J. H. Jeong, B. S. Shin, H. G. Choi, C. S. Yong and J. O. Kim, Acta Biomater., 2014, 10, 5116–5127 CrossRef CAS PubMed.
- T. Maldiney, B. Ballet, M. Bessodes, D. Scherman and C. Richard, Nanoscale, 2014, 6, 13970–13976 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra19242d |
| This journal is © The Royal Society of Chemistry 2016 |