Online remote monitoring of explosives by optical fibres†
Shuai Ruana,
Yizhao Chenb,
Peng Zhangac,
Xuanzhao Pana,
Cheng Fangd,
Anjun Qinb,
Heike Ebendorff-Heidepriema,
Ben Zhong Tange,
Youhong Tangf and
Yinlan Ruan*a
aARC Centre of Excellence for Nanoscale BioPhotonics, Institute for Photonics and Advanced Sensing, The University of Adelaide, Adelaide, SA 5005, Australia. E-mail: Yinlan.ruan@adelaide.edu.au
bGuangdong Innovative Research Team, State Key Laboratory of Luminescent Materials & Devices, South China University of Technology, Guangzhou, P. R. China
cWuhan National Laboratory for Optoelectronics, Huazhong University of Science and Technology, Wuhan, 430074, China
dGlobal Centre for Environmental Remediation, The University of Newcastle, Callaghan, New South Wales, Australia
eDepartment of Chemistry, Hong Kong Branch of Chinese National Engineering Research Centre for Tissue Restoration and Reconstruction, The Hong Kong University of Science & Technology, Clear Water Bay, Kowloon, Hong Kong, P. R. China
fCentre for NanoScale Science and Technology, Centre for Maritime Engineering, Control and Imaging, School of Computer Science, Engineering and Mathematics, Flinders University, South Australia, Australia
First published on 25th October 2016
Abstract
An AIE-active polytriazole coated fibre tip sensor was developed to detect picric acid with its detection limit down to 100 ppb. The fibre sensor requires an analysis time of only a few minutes and shows strong photostability and high reusability, and is promising for remote sensing of explosives in dangerous environments.
Rapid detection of explosives is essential because it has important applications in criminal investigations and homeland security.1 Picric acid (2,4,6-trinitrophenol) (PA) is one of the most common components in explosives. It is also an important organic acid and can react with certain drugs such as cinchonine and quinine to form insoluble precipitates. Attention has therefore been paid to the direct determination of PA in the area of biological chemical analysis and to indirect determination of these drugs.2
Various spectroscopic methods of PA detection are currently available. For example, ion mobility spectrometry (IMS) is one of the most widely used detection techniques in routine use.3 Mass spectrometry (MS) has also been used to detect explosives, due to its specificity in identifying substances.4 Terahertz (THz) spectroscopy has been demonstrated as another technique for the detection of explosive vapour signatures.5 Infrared spectroscopic methods can detect finger-print absorption of certain groups of chemicals at specific wavelengths, allowing detection of many materials.6 These methods, though highly sensitive, require tedious sample pre-treatment and lack of portability, and they were not designed for remote sensing. It is essential, therefore, to develop a rapid and sensitive method to detect the presence of these compounds on site.
Much effort has been devoted to the development of relatively inexpensive and simple sensing methods for industrial processes and environmental monitoring. For example, Chu et al. demonstrated a suspended core optical fibre-based sensor for explosive detection based on the fluorescence quenching of a surface-attached conjugated polymer.8 These fibres permit evanescent field interactions with the surrounding media due to their small core size. However, the explosive detection limit achieved was merely 6.3 ppm, which may not be sensitive enough for practical use, and its response time was limited to several minutes or longer due to time cost for sample liquid filling into the micro-structured fibre holes through capillary force. Long et al. reported an electro-spun nano-fibrous film doped with a fluorescent conjugated polymer for the detection of another explosive, dinitrotoluene (DNT), in chloroform. The conjugated polymer has greater fluorescent quantum yields and amplified sensory responses in comparison with small fluorescent molecules. However, it undergoes self-aggregation and thus self-fluorescent quenching.7
There are many PA-sensitive fluorophores for explosive detection.9 In most of the reported fluorophores, emission is often quenched when they have high concentration or are in the aggregate state, which is known as aggregation-caused quenching (ACQ).10 The ACQ effect often limits the applications of fluorophores to be diluted to a large degree, which requires a higher excitation laser power. Instead, a fluorophore called P3a reported by Qin et al. with the aggregation-induced emission (AIE) effect overcomes the ACQ problem and is able to detect explosives.11 The P3a was reported as a group of new tetraphenylethene (TPE)-containing polytriazoles synthesized from the click polymerization of diyne with diazides.11 The aggregation of P3a was found to result in strong fluorescence at 520 nm due to the restriction of intra-molecular rotations, which leads to a lower excitation power required.11 In its aggregated state, the fluorescence of P3a is quenched by PA molecules due to energy transfer.12 Compared to the short-range process of charge transfer, energy transfer is a long-range process and hence quenching of P3a fluorescence occurs already when PA molecules surround the fluorophores and direct contact is not required.
In this paper, we report a simple fibre tip explosive sensor by immobilizing the P3a molecule onto the tip of a multimode (MM) fibre through silica sol–gel (see ESI†).13 Sol–gel derived glass is an ideal matrix material for sensing applications since it is highly permeable and has favourable mechanical properties, excellent chemical stability and superior optical clarity.14 The sol–gel acted as the medium to host the P3a molecules and its porous structure allowed PA molecules to permeate from the surroundings to P3a, resulting in fluorescence quenching of P3a.16 The detection limitation of our reported sensor is demonstrated to be down to 100 ppb (mass concentration), about 60 times enhanced detection limit partly due to highly aggregated P3a molecules allowed on the fibre tip because of its AIE property.
The experimental setup used for characterisation of the P3a fluorescence quenching by PA is shown in Fig. 1. Continuous wave (CW) light from a 407 nm Ar+ laser was reflected off a long-pass filter and coupled into multimode fibre with 200 μm core diameter using a 40× microscope objective. An optical chopper was used to reduce optical bleaching of the fluorophores. The fluorescence of the fluorophores could be detected when the fibre probe end was dipped into the sample solution. The fluorescence signal was collected in backscattering mode of the fibre, then passed the long pass filter and recorded by a fibre-coupled cooled-CCD spectrometer.
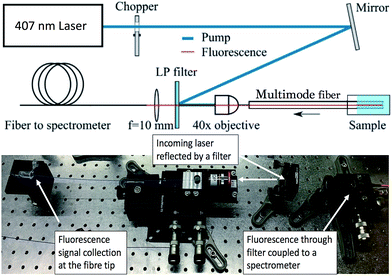 | ||
| Fig. 1 Experimental setup for detection of PA in Milli-Q water using multimode optical fibres.11 LP filter: long pass filter. | ||
We commenced with investigation of fluorescence quenching of P3a in aqueous solution by inserting the fibre into the P3a/PA mixture simply for signal excitation and collection. AIE-active P3a was reported to be non-luminescent when dissolved in an organic solvent such as THF or DCM, but strong fluorescence was observed with the addition of water into the well-dissolved P3a solution.11 This effect was successfully confirmed in our work by detecting the fluorescent intensity from P3a solution with various THF/water ratios (Fig. S1a†), and P3a showed the strongest fluorescent intensity in the THF/water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 v/v) (Fig. S1b†) which is in agreement with literature.11 Therefore, the quenching tests of PA to the fluorescence of P3a were conducted by detecting the fluorescence of the P3a in the THF/water mixture (1
9 v/v) (Fig. S1b†) which is in agreement with literature.11 Therefore, the quenching tests of PA to the fluorescence of P3a were conducted by detecting the fluorescence of the P3a in the THF/water mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 v/v) with a series of PA concentrations.
9 v/v) with a series of PA concentrations.
In order to investigate how the concentration of P3a affects the sensitivity of detection, three batches of tests with P3a concentrations of 0.2, 0.5 and 1.0 mg L−1 in THF/water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 v/v) were chosen for comparison. Upon adding and increasing the PA concentration from 0.1 to 200 ppm, the fluorescence intensity of the mixtures gradually decreased. The detection limit was found to be as low 0.1 ppm for all tests (Fig. 2, S2 and S3 in ESI†).13 This limit of detection is very similar to that reported by Qin et al.,11 who measured the P3a fluorescence in a cuvette by a spectrophotometer with an unspecified P3a concentration.
9 v/v) were chosen for comparison. Upon adding and increasing the PA concentration from 0.1 to 200 ppm, the fluorescence intensity of the mixtures gradually decreased. The detection limit was found to be as low 0.1 ppm for all tests (Fig. 2, S2 and S3 in ESI†).13 This limit of detection is very similar to that reported by Qin et al.,11 who measured the P3a fluorescence in a cuvette by a spectrophotometer with an unspecified P3a concentration.
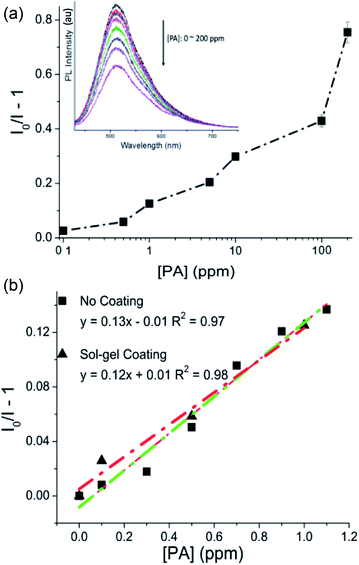
Fig. 2a shows the Stern–Volmer plots of 0.2 mg L−1 P3a, which were linear with quenching constants of 1.3 × 105 (R2 = 0.98), 4.6 × 103 (R2 = 0.98) and 2.3 × 103 (R2 = 0.97) M−1 when the PA concentrations were in the ranges of 0–1 ppm, 6–45 ppm and 45–200 ppm, respectively.13 These results suggest that P3a solution with concentration of 0.2 mg L−1 was more sensitive to low concentrations of PA than to the relatively high concentration ranges. Fig. 2b compares the Stern–Volmer plots of these three P3a concentrations in the low PA concentration ranges (0–4 ppm). It can be seen that lower P3a concentration (0.2 mg L−1) was the most sensitive (quenching constant 1.3 × 105 M−1) to the change of PA concentration, but at the cost of a narrow detection range (0.1 to 1.0 ppm). In contrast, high P3a concentrations (1.0 mg L−1), even though less sensitive (quenching constant 0.3 × 105 M−1), had a relatively broader detection range (0.1 to 3.5 ppm). These results were not beyond our expectations, as the quenching mechanism was proposed to be an energy transfer as the absorption spectrum of PA overlaps with the PL spectra of the P3a aggregates.11 When the energy donor P3a and energy acceptor PA are in a close proximity, the electronically excited state P3a may transfer energy to PA through non-radiative dipole–dipole coupling, resulting in the quenching of fluorescence.15 Consequently, compared to high concentrated p3a, lower concentrations of P3a are more easily quenched upon the addition of the same concentrated PA, thus show higher sensitivity.
In all the tests described above, the optical fibre was merely used as a tool for optical signal excitation and collection. Next, we immobilised P3a on fibre tips by using dip coating method to enable remote detection of PA molecules in water. For the dip coating procedure, the cleaved fibre tip was firstly treated with piranha solution for 30 min to remove any organic residues before coating. Then it was immersed into a mixed solution of silica sol–gel and P3a molecules. We chose the P3a solution with concentration of 1.0 mg L−1 for the fibre coating to enable a large detection range shown by our previous experiments. This solution was mixed with the same volume of freshly prepared 45 wt% silica sol–gel, and then dip-coated to the clean fibre end face (the distal end). A wet layer was then formed by withdrawing the fibre. Finally, the solvent evaporation resulted in the gelation of P3a/sol–gel mixture layer on the fibre tip. The fibre was then dried in the open air for two hours before any PA detection test.
In order to investigate the sensing performance of the sol–gel/P3a coated fibre, the same setup as shown in Fig. 1 was used for the quenching measurements. The power from the fibre output end was 250 μW. When the distal end of the fibre with P3a coating was inserted into PA solutions, the fluorescent intensity from the fibre tip was observed to gradually decrease with an increase of PA concentration from 0.1 to 200 ppm with the detection limit 0.1 ppm (Fig. 3a). Fig. 3b compares the Stern–Volmer plots of P3a attached to the fibre tip and P3a molecules that are aggregated in solution with a concentration 1.0 mg L−1. The curve for sol–gel coated fibre (fitted red line) shows good linearity in the range of 0–1 ppm with a quenching constant 0.27 × 105 M−1. This result is similar to the PA detection by P3a aggregated in solution with the optical fibre merely used for signal transmission (fitted green line in Fig. 3b).
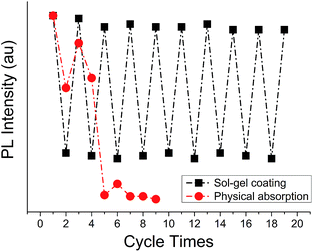
To verify reversibility of the fibre dip sensor with the P3a attached, recovery tests were conducted. For comparison, the same tests were also performed for the fibre sensor with the P3a attached to the fibre tip by simple physical adsorption using the same concentration of P3a solution (1.0 mg L−1 P3a dispersed in THF). 100 ppm PA was used for the tests and after each measurement the fibre tips were carefully rinsed with Milli-Q water and dried with N2. This procedure ensured no PA residuals remained on fibre tips before each test and meanwhile verified that sol–gel embedded P3a can stand the repeated rinsing force from Milli-Q water. As shown by the red dots in Fig. 4, for the fibre sensor with the P3a attached by physical absorption, no obvious fluorescence was detected after testing 5 times of the fibre tip, which indicated escape of P3a from fibre substrate to the solution. In contrast, the recovery rate was 94% for the sol–gel coated fibre, even after repeatedly testing 20 times. This result demonstrates that P3a molecules are firmly embedded in the network structure of the silica sol–gel so that they resist water rinsing and enable reusability of the fibre sensor, compared to their physical absorption to the fibre surface, which carried out by the same dip coating procedure but not mixing the P3a with sol–gel solution.
In summary, we have successfully demonstrated detection of trace PA explosive using AIE-active P3a. The detection limit of 0.1 ppm was achieved for the P3a/sol–gel mixture coated fibre tip sensor. Although the detection area (0.03 mm2) of 200 μm diameter of the MM fibre is much smaller than that of suspended core fibre with the detection area being the side surface along the whole length of the fibre, the demonstrated MM fibre sensor still achieved 65 times smaller detection limit compared to Chu's work by using the suspended core fibre.8 This is benefited from the highly aggregated P3a molecules functionalized into the fibre tip without causing any quenching. This suggests that the detection limit and sensitivity can be further improved by coating the P3a molecules on a larger surface of suspended or enclosed core fibre due to the outstanding performance of P3a molecules. The strongly aggregated p3a molecules also enabled an excitation laser power of only 250 μW required, which can be met by a cheap LED for practical applications. By immobilizing the P3a on a MM fibre end face using silica sol–gel, a simple and reliable optical tip sensor was demonstrated for remote detection/monitoring of the explosive, which is very useful for applications in dangerous environments.
Acknowledgements
Considerable assistance in research discussions provided by Dr Sabrina Heng and Dr Victoria Peddie during the work is gratefully acknowledged. This work was performed in part at the OptoFab node of the Australian National Fabrication Facility utilizing Commonwealth and SA State Government funding.Notes and references
- (a) B. R. McCord, K. D. Smith, K. Mount, W. F. Rowe and W. A. MacCrehan, J. Forensic Sci., 1999, 44, 789 Search PubMed; (b) S.-A. Barshick, J. Forensic Sci., 1998, 43, 284 CAS.
- J. Lu and Z. Zhang, Anal. Chim. Acta, 1996, 318, 175 CrossRef CAS.
- R. G. Ewing, D. A. Atkinson, G. Eiceman and G. Ewing, Talanta, 2001, 54, 515 CrossRef CAS PubMed.
- J. Yinon, Counterterrorist Detection Techniques of Explosives, 2007, p. 41 Search PubMed.
- M. R. Leahy-Hoppa, M. J. Fitch and R. Osiander, Anal. Bioanal. Chem., 2009, 395, 247 CrossRef CAS PubMed.
- O. M. Primera-Pedrozo, Y. M. Soto-Feliciano, L. C. Pacheco-Londoño and S. P. Hernández-Rivera, Sens. Imag. Int. J., 2009, 10, 1 CrossRef.
- Y. Long, H. Chen, Y. Yang, H. Wang, Y. Yang, N. Li, K. Li, J. Pei and F. Liu, Macromolecules, 2009, 42, 6501 CrossRef CAS.
- F. Chu, G. Tsiminis, N. A. Spooner and T. M. Monro, Sens. Actuators, B, 2014, 199, 22 CrossRef CAS.
- (a) Y. Engel, R. Elnathan, A. Pevzner, G. Davidi, E. Flaxer and F. Patolsky, Angew. Chem., Int. Ed., 2010, 49, 6830 CrossRef CAS PubMed; (b) D. Gao, Z. Wang, B. Liu, L. Ni, M. Wu and Z. Zhang, Anal. Chem., 2008, 80, 8545 CrossRef CAS PubMed; (c) S. Guo, D. Wen, Y. Zhai, S. Dong and E. Wang, Biosens. Bioelectron., 2011, 26, 3475 CrossRef CAS PubMed; (d) A. Díaz Aguilar, E. S. Forzani, M. Leright, F. Tsow, A. Cagan, R. A. Iglesias, L. A. Nagahara, I. Amlani, R. Tsui and N. Tao, Nano Lett., 2009, 10, 380 CrossRef PubMed.
- D. Ding, K. Li, B. Liu and B. Z. Tang, Acc. Chem. Res., 2013, 46, 2441 CrossRef CAS PubMed.
- A. Qin, J. W. Lam, L. Tang, C. K. Jim, H. Zhao, J. Sun and B. Z. Tang, Macromolecules, 2009, 42, 1421 CrossRef CAS.
- J. Wang, J. Mei, W. Yuan, P. Lu, A. Qin, J. Sun, Y. Ma and B. Z. Tang, J. Mater. Chem., 2011, 21, 4056 RSC.
- ESI.†.
- (a) P. Lehner, C. Staudinger, S. M. Borisov and I. Klimant, Nat. Commun., 2014, 5, 4460 CAS; (b) S. M. Borisov, A. S. Vasylevska, C. Krause and O. S. Wolfbeis, Adv. Funct. Mater., 2006, 16, 1536 CrossRef CAS; (c) C.-S. Chu and Y.-L. Lo, Sens. Actuators, B, 2008, 134, 711 CrossRef CAS.
- V. Helms, Principles of computational cell biology, John Wiley & Sons, 2008 Search PubMed.
- C.-S. Chu and C.-Y. Chuang, Sens. Actuators, B, 2015, 209, 94 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Sol–gel synthesis procedures, fluorescent responses of concentrated P3a solutions towards serial PA solution, high photo-stability of dip-coated P3a. See DOI: 10.1039/c6ra24080a |
| This journal is © The Royal Society of Chemistry 2016 |