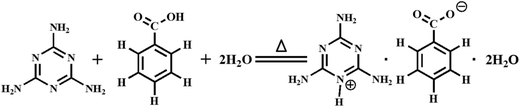A thermo-sensitive supramolecular hydrogel derived from an onium salt with solution–gel–crystal transition properties†
Xin-Jian Jia a,
Jinshu Wang*a,
Di-Chang Zhong
a,
Jinshu Wang*a,
Di-Chang Zhong b,
Junshu Wua,
Bingxin Zhaoa,
Daniel den Engelsen
b,
Junshu Wua,
Bingxin Zhaoa,
Daniel den Engelsen a and
Xu-Zhong Luo*b
a and
Xu-Zhong Luo*b
aKey Laboratory of Advanced Functional Materials for Ministry of Education, College of Materials Science and Engineering, Beijing University of Technology, Beijing 100124, China. E-mail: wangjsh@bjut.edu.cn
bKey Laboratory of Organo-Pharmaceutical Chemistry of Jiangxi Province, Key Laboratory of Jiangxi University for Functional Material Chemistry, Gannan Normal University, Ganzhou 341000, China. E-mail: luoxuzhong@hotmail.com
First published on 4th November 2016
Abstract
The gelation ability of melamine was evaluated under various acidic conditions, and the related gelator aggregates were investigated with scanning electron microscopy, transmission electron microscopy, single-crystal X-ray diffraction, thermo-gravimetric analysis, differential scanning calorimetric analysis, rheological experiments, 1H nuclear magnetic resonance spectroscopy, Fourier transform infrared spectroscopy and powder X-ray diffraction. It was found that melamine interacted strongly with a number of organic/inorganic acids in water, forming thermal-reversible supramolecular hydrogels with different critical gelator concentrations (CGCs). The CGCs of gelators successively made with salicylic acid, m-hydroxybenzoic acid and p-hydroxybenzoic acid decreased because of the steric hindrance, whereas those related to oxalic acid dihydrate and orthoboric acid presented higher values due to the lack of a phenyl ring in the molecular structures. More interestingly, a unique onium salt, 2,4,6-triamino-1,3,5-triazin-1-ium benzoate dihydrate (TTIBD), was formed via the Lewis acid–base reaction of benzoic acid and melamine. It crystallized in the monoclinic space group C2/c (Z = 8) with lattice parameters a = 21.477 (3) Å, b = 10.2253 (14) Å, c = 12.3312 (17) Å and β = 98.717 (3)°. The formed hydrogel not only exhibited thermo-sensitive characteristics and solid-like behavior, but also showed a solution–gel–crystal transition, being an amorphous-to-crystalline phase transition. By increasing the concentration from 0.04 to 0.12 mol L−1, the gel–solution transition temperature increased from 23.0 to 49.5 °C, and the gel–crystal transition time decreased from 430 to 253 min, but both leveled off upon increasing the concentration. The TTIBD crystal along with the corresponding hydrogel was self-assembled via hydrogen bonds and π–π stacking interactions.
1. Introduction
As one of the most versatile and efficient approaches for fabricating supramolecular nanoarchitectures, molecular self-assembly has long attracted widespread attention of scientists in various fields.1,2 It is characterized by weak non-covalent interactions, viz. hydrogen bonds, halogen bonds, π–π stacking interactions, metal–ligand coordination bonds, host–guest interactions, ionic bonds, hydrophobic interactions and van der Waals interactions, and has played a significant role in the design and development of intelligent functional materials.3–8Gels made from gelators are a type of adaptive and viscoelastic solid-like materials, which widely exist in nature with a variety of forms, such as pectins, inorganic clays, latexes, gelatins, etc. Supramolecular hydrogels, driven by molecular self-assembly and composed of low molecular weight gelators (LMWGs) and water, are well known for their potential applications in catalysis, photoactive devices, chemical sensors, drug delivery, pollutant capture and removal, genetic engineering, tissue engineering, etc.9–13 Over the past decade, inspired by the fascinating self-assembly processes, considerable attention has been paid to the supramolecular hydrogels owing to the biocompatibility and stimuli-responsiveness toward temperature, salt concentration, pH, magnetism, light, etc.14–16 However, the prevailing view for supramolecular hydrogel applications is that the long-term stability must be addressed; another suggestion is that the metastable state may be more suitable in some cases (such as in the crystallization of gelators and their application as smart switches).
The gelation and crystallization of LMWGs are two closely related types of non-equilibrium self-assembly processes; both take place in over-saturated solutions and undergo nucleation and growth processes.17 A good way to understand how molecules self-assemble in gel state is investigating the properties of supramolecular gels and focusing on the transformation of gel to crystal, notably the corresponding LMWG crystal structure. But to the best of our knowledge only very few reports have been published on this subject. For instance, Dastidar and co-workers described a pyridyl-urea-based hydrogelator for the first time, which could be crystallized from its gelling solvent.18 The spontaneous transition of a hydrogel formed from a meta-hydroxy pyridinium salt into macroscopic crystals was explored by Tang's group.19 Lloyd and Steed have obtained crystals of a low molecular weight salt hydrogelator induced by the addition of salt to the gel formed in acidified water.20 The phase transition from gel to crystal of an organogel, based on a glycosylated amino acid derivative, in dichloromethane, chloroform and ethanol has been reported by Li and co-workers.21 Sureshan's team has studied the role of water in selection between gelation and crystallization of cyclohexane-1,3-diols based organogelators.22 Based on the scarcity of supramolecular hydrogels with the transformation of gel to crystal, we decided to explore new gelators that show both gelation and crystallization. The underlying objective is to guide us to design more complex supramolecular gels with a vaster variety of gelators. Nitrogen-rich melamine can easily interact with a carboxylic acid derivative, forming hydrogen bonds.23 Nandi and co-workers have made an outstanding contribution in two-component hydrogels with melamine and another component.24–29 Inspired by their excellent achievements, we tried to further enrich the types of onium salts formed from melamine and carboxylic acids and investigate their gelation behavior.
Herein, we present the gelation ability of melamine within a number of acidic solutions, and then describe in detail the synthesis and solution–gel–crystal transition behavior of an onium salt, 2,4,6-triamino-1,3,5-triazin-1-ium benzoate dihydrate (C3H7N6+·C6H5COO−·2H2O, TTIBD), formed via a Lewis acid–base reaction of benzoic acid and melamine in aqueous solution. The molecular arrangement of TTIBD in crystals was determined, and its gelation ability in water was investigated. The morphology and microstructure of corresponding xerogels were then characterized. The rheological behaviors were analyzed. In addition, the stability was also studied taking into account the influences of temperature and ageing time. The formation mechanism of self-assembly was further explored.
2. Experimental section
2.1. Chemicals
Analytical grade benzoic acid and melamine were purchased from Beijing Chemical Works (Beijing, China) and utilized to prepare TTIBD without further treatment. Analytical grade caffeic acid, tartaric acid, citric acid monohydrate, salicylic acid, p-hydroxybenzoic acid, m-hydroxybenzoic acid, orthoboric acid and oxalic acid dihydrate were supplied by Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Deuterium oxide (99.9 atom% D) was obtained from J&K Scientific Ltd. (Shanghai, China). All solutions were prepared from deionized water with a resistivity of 18 MΩ cm unless otherwise specified.2.2. Preparation of hydrogels
The gelation ability of melamine in various acidic solutions was determined by the typical ‘inversion test’.11 For this test, a certain amount of melamine and an equivalent amount of acid were dissolved in water with continuous stirring at 90 °C, respectively. Subsequently, the solutions were immediately mixed in a scintillation vial and cooled to room temperature. Finally, the scintillation vials were turned upside down, in which the viscoelastic mixture that did not fall down was defined as a hydrogel.2.3. Synthesis of TTIBD
In a typical experiment, melamine (12.6 mg, 0.1 mmol) and benzoic acid (12.2 mg, 0.1 mmol) were separately dissolved into water (1.0 mL) by ultrasonic cavitation at 90 °C. These solutions were then immediately mixed in a scintillation vial and cooled at 20 °C in a shaking water bath until a hydrogel was generated. After standing for about 6 h, colorless granular crystals, which were suitable for single-crystal X-ray diffraction analysis, gradually formed (23.4 mg, 82.4%) (Scheme 1 and Fig. S1 (ESI†)). Anal. calcd for TTIBD: C, 42.25; H, 5.67; N, 29.56. Found: C, 43.11; H, 5.44; N, 29.73.2.4. Characterization
The morphology of the freeze-dried hydrogels (xerogels) was studied in a scanning electron microscope (SEM, Hitachi S-4800). All samples used for the SEM experiments were coated with a layer of Au in order to minimize the effect of surface charging.3 The crystal-phase transition was studied with a transmission electron microscope (TEM, FEI Tecnai G2 F30) combined with selected area electron diffraction (SAED) at an acceleration voltage of 300 kV. The molecular arrangement of TTIBD in crystals was confirmed by a single-crystal X-ray diffractometer (SCXRD, Bruker Smart APEX-CCD) with Mo-Kα radiation (λ = 0.71073 Å) at 298 K. All empirical absorption corrections were applied by using the SADABS program.30 The non-hydrogen atoms were refined with anisotropic thermal parameters, whereas the positions of the hydrogen atoms were found by difference Fourier synthesis and refined isotropically. The structure was determined by the direct method and refined with a full-matrix least-squares technique against F2 by using the SHELXL software package.31 The structural phase transition of the TTIBD crystal was also investigated with a powder X-ray diffractometer (PXRD, Bruker D8 Advance) with Cu-Kα radiation (λ = 1.54056 Å) at different temperatures (298, 308, 318, 328, 338, 348, 358 and 368 K), and the data were collected in the range of 5° < 2θ < 60° in steps of 0.02°. The accelerating voltage was set at 40 kV with an emission current of 40 mA. The thermo-responsiveness of the hydrogel was investigated by a 1H Nuclear Magnetic Resonance (1H NMR, Bruker AV400) spectrometer at 400 MHz, and the chemical shifts (δ) were recorded in parts per million (ppm). The thermo-stabilities of the TTIBD crystal and xerogel were characterized by a thermo-gravimetric analyzer (TGA, Netzsch STA 449 C) at a heating rate of 10 °C min−1 under an Ar atmosphere. The mechanical properties of hydrogels were performed using a stress-controlled rheometer (Haake RheoStress 6000) with the parallel plate geometry of 3.5 cm diameter and 1 mm gap. A solvent trap equipped with the rheometer was used to minimize water evaporation. The temperature dependences of storage shear moduli (G′) and loss shear moduli (G′′) were conducted by heating the sample from 25 to 95 °C at a rate of 0.1 °C s−1. The frequency sweep at 25 °C was carried out in the linear viscoelastic region, which was determined by the strain sweep measurement with the stress varying at a constant frequency of 1.0 Hz. The functional groups were analyzed with a Fourier transform infrared (FT-IR, Nicolet AVATAR 360) spectrometer in the range from 4000 to 400 cm−1 at a resolution of 4 cm−1. KBr pellets and CaF2 substrates were used for the transmission spectrum measurements of the crystal and dried hydrogel, respectively. The gel–solution transition temperatures (TGS) of hydrogels at different concentrations of TTIBD were evaluated by the ‘falling drop’ method32 and confirmed by a differential scanning calorimeter (DSC, Setaram μDSC7-Evo) at a heating rate of 1.0 °C min−1 under a N2 atmosphere.3. Results and discussion
3.1. Gelation behavior
The gelation ability of melamine within various acidic solutions is summarized in Table 1. It can be seen that melamine has the capability to interact with benzoic acid, salicylic acid, p-hydroxybenzoic acid, m-hydroxybenzoic acid, orthoboric acid and oxalic acid dihydrate in water, forming supramolecular hydrogels at different critical gelator concentration (CGC), which is defined as the minimum amount of melamine and acid required for gelatinizing. Caffeic acid, tartaric acid and citric acid monohydrate could not form a hydrogel upon mixing with the melamine solution. All formed hydrogels are thermally reversible. Moreover, the CGCs of gelators successively correlated with salicylic acid, m-hydroxybenzoic acid and p-hydroxybenzoic acid showed a decreasing trend, which may be attributed to the steric hindrance effect.33,34 In contrast, the CGCs of gelators related to oxalic acid dihydrate and orthoboric acid yielded higher values due to the lack of a phenyl ring in molecular structures. To our surprise, the supramolecular hydrogel formed from melamine and benzoic acid gradually transformed into colorless granular crystals upon extension of the aging time (Fig. 1). The phenomenon is ascribed to the changes in crystallization kinetics and geometrical constraints of the hydrophobic segments in this supramolecular hydrogel. Furthermore, these crystals readily dissolved in water upon heating, and the opaque hydrogel was obtained again upon cooling down to room temperature. By prolonging the aging time, the colorless granular crystals were formed again. This solution–gel–crystal transition process was reversible.| Reagents | Molar ratio (melamine![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) acid) acid) |
CGCs (g L−1) | Appearance |
|---|---|---|---|
| a OG = opaque gel; TS = turbid solution. | |||
| Benzoic acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
7.09 | OG |
| Caffeic acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | TS |
| Tartaric acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | TS |
| Citric acid monohydrate | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | TS |
| Salicylic acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13.20 | OG |
| p-Hydroxybenzoic acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.64 | OG |
| m-Hydroxybenzoic acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
10.56 | OG |
| Orthoboric acid | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
55.80 | OG |
| Oxalic acid dihydrate | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
25.20 | OG |
3.2. Morphology and microstructure
Generally speaking, supramolecular hydrogels are composed of a three-dimensional network and entrapped water because of surface tension and capillary forces.35,36 To investigate the aggregation morphology and microstructure of gelators, the xerogels were characterized visually by SEM and TEM. All xerogels were prepared via freeze-drying to prevent the damage from high vacuum condition or drying of the samples. As illustrated in Fig. S2 (ESI†), different aggregates of gelators exhibit a variety of forms. The gelators fabricated with melamine and hydroxybenzoic acids self-assembled into strip-shaped (p-hydroxybenzoic acid), sheet-like (m-hydroxybenzoic acid) and block-shaped (salicylic acid) aggregates, respectively. The columnar and crushed granular aggregates were separately obtained from the xerogels correlated with oxalic acid dihydrate and orthoboric acid. These significant distinctions also explain why the different hydrogels show different CGCs. Furthermore, a new phenomenon was observed when the aging time of the supramolecular hydrogel formed from melamine and benzoic acid was prolonged. As indicated in Fig. 2a, the xerogel aged at room temperature for 5 min mainly consists of flocculent structure which is approximately 200 nm wide, and the corresponding SAED image presents a diffuse pattern indicating that it is an amorphous state (Fig. 2d). By extending the aging time, the flocculent structure gradually becomes shorter and wider, and then transforms into a fibrous structure. The SAED pattern of this structure exhibits a distinct diffraction ring illustrating an increase in crystallinity (Fig. 2b and e). When the as-prepared hydrogel was aged for 300 min, granular crystals were eventually generated (Fig. 2c and f). We also tried to observe high-resolution TEM (HR-TEM) images to further analyze the crystal-phase transition of gelators. Unfortunately, the HR-TEM micrographs could not show any meaningful information due to the knock-on damage induced by electron beam in the irradiated samples.37The phase purity of the granular crystals was confirmed by PXRD measurements at ambient temperature (Fig. S3, ESI†). All positions of the diffraction peaks closely corresponded to those in the simulated pattern generated from the SCXRD data, indicating that a single-crystalline phase of TTIBD was obtained.
3.3. Effects of temperature and ageing time
Taking into account the relationship between the thermal stability of a supramolecular hydrogel and the concentration of the gelator, we measured the TGS of hydrogels for TTIBD at different concentrations. As indicated in Fig. 3, a remarkable increase in TGS from 23.0 to 49.5 °C was found when the concentration was gradually increased from 0.04 to 0.12 mol L−1. Subsequently, a leveling off was reached at higher gelator concentrations. This result indicates that a more rigid three-dimensional network structure intertwisted and interlocked with fibers was formed upon increasing the concentration of TTIBD, giving rise to higher values of TGS. However, the TGS will become constant when the concentration is increased further. Similar results were also observed in the DSC analyses (Fig. S4, ESI†). | ||
| Fig. 3 Scatter plots of the TGS (green line) and gel–crystal transition time (blue line) versus the concentration of TTIBD in aqueous solution. | ||
As the molecules aggregate, precipitation or crystallization may occur instead of gelatinization, because the self-assembly of gelators is a non-thermodynamically stable and kinetically trapped process.38 Therefore, another important factor affecting the stability, and thus lifetime, of supramolecular gels is the gel–crystal transition time. As depicted in Fig. 3, the gel–crystal transition time decreased considerably from 430 to 253 min when the concentration of TTIBD was gradually increased from 0.04 to 0.12 mol L−1. Subsequently, a leveling off was reached at higher concentrations of the gelator. This result implies that the gelatinization and crystallization processes are well-separated in time: i.e. gelatinization occurs over a period of minutes followed by a crystallization process of many hours, which is consistent with the results published before.17 Initially, the gel–crystal transition time decreases quickly upon increasing the concentration of the gelator due to supersaturation. But, it will reach a constant value at high concentrations.
3.4. Mechanical property
The mechanical property of a hydrogel is very important in their practical applications, so rheological measurements were carried out to investigate the viscoelastic property of TTIBD hydrogel. As depicted in Fig. 4a, the TTIBD hydrogel begins to lose the solid-like behavior at a shear stress of about 4 Pa. When the shear stress is less than the critical shear stress, G′ and G′′ are independent and the deformation is close to 0, which indicates that the hydrogel structure is completely intact. Subsequently, the structure collapse occurs, as proved by a dramatic decrease in the values of both modulus and the reversal of viscoelastic signal. The frequency dependences of modulus were tested to confirm the elastic behavior of TTIBD hydrogel. As expected, the values of G′ are greater than those of G′′, and both level off during the sweeps from 0.01 to 10 Hz, exhibiting the typical viscoelastic feature of a hydrogel (Fig. 4b). The thermo-responsive of TTIBD hydrogel was also characterized by the variation of G′ and G′′ with increasing temperature. As described in Fig. 4c, both G′ and G′′ of the TTIBD hydrogel are almost constant from room temperature to 40 °C. When the temperature is above 40 °C, the values of modulus decrease significantly, implying the occurrence of gel–solution transition. However, both will level off after the hydrogel is heated over 50 °C. These results are in accordance with those obtained from the TGS and DSC experiments. Additionally, the time sweep rheological measurement of the TTIBD hydrogel was conducted. As shown in Fig. 4d, a decline in G′ and G′′ is observed with increasing time, which can be attributed to the breakup of TTIBD hydrogel structure. When the time is increased to a certain value, the trend becomes slow. This phenomenon is similar with that of previous report.39 | ||
| Fig. 4 Rheological measurements of the TTIBD hydrogel (0.1 mol L−1 in H2O) with the modes of (a) strain sweep, (b) frequency sweep, (c) temperature sweep and (d) time sweep for G′ and G′′. | ||
3.5. SCXRD structure
X-ray crystallography is one of the most commonly-used techniques for analyzing the arrangement of atoms and molecules in crystals.40 However, PXRD recordings of the crystalline materials could only provide less structural information at a lower accuracy than SCXRD due to the serious overlapping of diffraction peaks.41 To get an idea about how molecules self-assemble in crystals and the relationship between gelation and crystallization, we decided to investigate the TTIBD structure with SCXRD. The crystal data are listed in Table S1 (ESI†), and the selected bond lengths and angles are summarized in Table S2 (ESI†). Further details about the structure determination have been deposited at the Cambridge Crystallographic Data Centre (CCDC) as supplementary publication CCDC 1504096. As shown in Table S1 (ESI†), TTIBD crystallizes in the monoclinic space group C2/c (Z = 8) with lattice parameters a = 21.477 (3) Å, b = 10.2253 (14) Å, c = 12.3312 (17) Å and β = 98.717 (3)°. The asymmetric unit is composed of one deprotonated benzoic acid molecule, one protonated melaminium moiety and two water molecules. The O atoms of the carboxylate anion form two intramolecular hydrogen bonds (N1⋯O2, 2.667 Å, N6⋯O1, 2.920 Å) with the protonated N atom and amino group in melaminium moiety. Two water molecules establish a hydrogen-bonded dimeric structure (O2W⋯O1W, 2.860 Å). Meanwhile, an N–H⋯O hydrogen bond (N4⋯O2W, 2.876 Å) links the melaminium moiety and water molecule. In addition, adjacent TTIBD molecules are connected to each other via N–H⋯N (N4⋯N2, 3.046 Å), N–H⋯O (N5⋯O1W, 2.916 Å, N5⋯O2W, 2.991 Å) and O–H⋯O (O1W⋯O1, 2.731 Å, O1W⋯O2, 2.836 Å, O2W⋯O1, 2.903 Å) hydrogen bonds, generating a two-dimensional (2D) bilayer structure (Fig. 5a) in which the triazine rings are stabilized by π–π stacking interaction. These 2D-layers are further stacked along the b-axis direction in a staggered manner, creating a three-dimensional (3D) structure (Fig. 5b). | ||
| Fig. 5 (a) Top view of the 2D bilayer in a ball-and-stick model and (b) side-view of the stacked 2D sheets in a space-filling model. | ||
3.6. Thermal stability
Temperature is a very important factor for the stability of material structure, so TGA was carried out in order to get insight into the thermal stability of TTIBD crystal and xerogel. As illustrated in Fig. 6, three weight loss regions can be distinguished in the TGA curve of TTIBD crystals. The first weight loss of 12.58% in the temperature range between room temperature and 105 °C is attributed to the loss of two lattice water molecules per TTIBD unit (calculated: 12.67%). The second weight loss of 43.33% occurring from 105 to 240 °C is assigned to the loss of one benzoic acid molecule per TTIBD unit (calculated: 42.95%). The third weight loss of 44.09% in the temperature range from 240 to 400 °C corresponds to the loss of one melamine molecule per TTIBD unit (calculated: 44.37%). This result indicates that one TTIBD unit can be divided into three parts: two lattice water molecules, one benzoic acid molecule and one melamine molecule, which agrees favorably with the structure obtained from the SCXRD analysis. In contrast with the TTIBD crystal, the xerogel did not show an obvious weight loss from 25 to 105 °C after the removal of a large amount of free water in freeze-drying process. It reveals that there was a lack of lattice water for crystallization when the supramolecular hydrogel formed.22To obtain a better understanding of the thermal stability of the TTIBD crystal, the variable-temperature PXRD diagrams were measured from ambient temperature to 95 °C (Fig. 7). From ambient temperature to 55 °C, no significant change was observed in the PXRD patterns. When TTIBD crystals were successively heated over 65 °C, the intensities of the diffraction peaks at 2θ = 9.7°, 11.7°, 12.5°, 14.6°, 19.3°, 23.5°, 26.5°, 27.4°, 28.3°, 30.8°, 33.8° and 35.2° were much weaker or even disappeared. Moreover, new diffraction peaks gradually appeared at 2θ = 9.2°, 13.4°, 20.1°, 21.1°, 21.8°, 25.8° and 36.6° upon increasing temperature, indicating that the framework of the TTIBD crystal dramatically changed. This low stability of the TTIBD crystal may be attributed to breaking of the asymmetric hydrogen bonds.42
3.7. Study of 1H NMR spectra
The variable-temperature 1H NMR spectra of TTIBD hydrogel formed in D2O were measured to investigate the thermo-responsiveness and self-assembly process in the gel state. As depicted in Fig. 8, only the resonances of benzoic acid were observed in the downfield region owing to the proton exchange with bulk-solvent molecules.43 The aromatic signals at about 7.80 (d, H2,6-ph), 7.46 (d, H4-ph) and 7.38 (t, H3,5-ph) ppm had low sensitivity, and were too broad to be observed at 20 °C, which might be ascribed to the long correlation time and slow tumbling rate in the gel state,44–46 indicating a strong intermolecular aggregation. By increasing the temperature from 30 to 70 °C, the unresolved peaks became well-dispersed and also gradually shifted downfield to 8.35 (m, H2,6-ph), 8.01 (d, H4-ph) and 7.91 (m, H3,5-ph) ppm, respectively. It can be inferred that the π–π stacking interaction promotes the self-assembly of hydrogelators. This result indicates a remarkable temperature-dependent behavior, and suggests that the gelatinous structure collapses at a higher temperature, which is in accordance with the reports of previous workers.47,483.8. Study of the FT-IR spectrum
FT-IR spectroscopy is a rapid and simple detection method, which can present some unique information from the absorption peaks.49 It has been widely utilized in the characterization of non-covalent interactions.50–52 As displayed in Fig. 9, all characteristic vibration bands of benzoic acid and melamine agree with the data in previous reports.53,54 Compared with the spectra of benzoic acid and melamine, the peaks at 3468 and 3419 cm−1 for the N–H stretching vibration of primary amine,55 in the region of 3300–2500 cm−1 for the O–H stretching vibration of carboxylic acid dimer and at 1687 cm−1 for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of benzoic acid are strongly attenuated or almost invisible in the TTIBD crystal. A new peak appears at 3360 cm−1, and the peaks for the quadrant (1560 cm−1) and semicircle (1466 cm−1) stretching of triazine ring56 shift to lower wavenumbers (1508 and 1394 cm−1), respectively. For the corresponding xerogel, however, the O–H stretching band (3360 cm−1) and N–H deformation band (1655 cm−1) shift to lower wavenumbers (3335 and 1647 cm−1), respectively, as compared with the spectrum of the TTIBD crystal. Furthermore, an additional peak is observed at 1439 cm−1. These observations indicate that the TTIBD crystals have been synthesized successfully, and the assembly structure of TTIBD hydrogel is likely to have a little difference compared with that of TTIBD crystal.
O stretching vibration of benzoic acid are strongly attenuated or almost invisible in the TTIBD crystal. A new peak appears at 3360 cm−1, and the peaks for the quadrant (1560 cm−1) and semicircle (1466 cm−1) stretching of triazine ring56 shift to lower wavenumbers (1508 and 1394 cm−1), respectively. For the corresponding xerogel, however, the O–H stretching band (3360 cm−1) and N–H deformation band (1655 cm−1) shift to lower wavenumbers (3335 and 1647 cm−1), respectively, as compared with the spectrum of the TTIBD crystal. Furthermore, an additional peak is observed at 1439 cm−1. These observations indicate that the TTIBD crystals have been synthesized successfully, and the assembly structure of TTIBD hydrogel is likely to have a little difference compared with that of TTIBD crystal.
 | ||
| Fig. 9 FT-IR spectra of benzoic acid (black line), melamine (red line), TTIBD crystal (green line) and TTIBD xerogel (blue line). | ||
3.9. Study of the PXRD pattern
To provide further evidence for the findings in the FT-IR spectra, the PXRD patterns of benzoic acid, melamine, TTIBD crystal and xerogel were also collected (Fig. 10). As expected, the positions of diffraction peaks for the TTIBD xerogel are evidently not coinciding with those for benzoic acid and melamine. Compared with the TTIBD crystal, the diffraction peaks at 2θ = 8.3°, 9.7°, 12.5°, 14.6°, 19.3°, 22.6°, 26.5° and 27.4° are appeared in the same position for the TTIBD xerogel, and there are also new broad peaks observed at around 2θ = 20.3–21.3° and 29.2–29.9°. In some previous reports, this phenomenon was ascribed to the escape of lattice water from gel fibers during the xerogel-forming process.18,57,58 Therefore, it can be concluded that both the crystal and hydrogel have similar assembly structures, though there is a little difference.4. Conclusions
Melamine shows the remarkable gelation ability towards many organic/inorganic acids in water, forming thermal-reversible supramolecular hydrogels with varying CGCs. Most of the formed supramolecular hydrogels display the conventional characteristics. Via the Lewis acid–base reaction of benzoic acid and melamine in aqueous solution, an onium salt TTIBD was synthesized. Interestingly, the hydrogel formed from TTIBD not only exhibited the thermo-sensitive characteristic and the solid-like behavior, but also showed a solution–gel–crystal transition. Furthermore, a step-wise gel-to-crystal transformation, also being an amorphous-to-crystalline phase transition process, was observed upon extension of the aging time. Both the crystal and hydrogel of TTIBD are self-assembled via hydrogen bonds and π–π stacking interactions. They have similar assembly structures even though there is a little difference. Although further research is still required, this supramolecular hydrogel with selective molecular recognition ability is expected to be used as a new phase-change-driven switching device.Acknowledgements
The authors are grateful to the National Natural Science Foundation of China (No. 51471006, No. 51225402, No. 51268002 and No. 51402008), Beijing Natural Science Foundation (2151001, 2142008, and 2154043), Guangxi Natural Science Foundation (2014GXNSFBA118039 and 2014GXNSFBB118001), Importation and Development of High-Caliber Talents Project of Beijing Municipal Institutions (CIT&TCD201404038) for financial support of this work.References
- M. Liu, L. Zhang and T. Wang, Chem. Rev., 2015, 115, 7304–7397 CrossRef CAS PubMed.
- S. Zhang, Nat. Biotechnol., 2003, 21, 1171–1178 CrossRef CAS PubMed.
- Z. Y. Li, Y. Zhang, C. W. Zhang, L. J. Chen, C. Wang, H. Tan, Y. Yu, X. Li and H. B. Yang, J. Am. Chem. Soc., 2014, 136, 8577–8589 CrossRef CAS PubMed.
- R. Daly, O. Kotova, M. Boese, T. Gunnlaugsson and J. J. Boland, ACS Nano, 2013, 7, 4838–4845 CrossRef CAS PubMed.
- E. A. Appel, J. del Barrio, X. J. Loh and O. A. Scherman, Chem. Soc. Rev., 2012, 41, 6195–6214 RSC.
- A. Priimagi, G. Cavallo, P. Metrangolo and G. Resnati, Acc. Chem. Res., 2013, 46, 2686–2695 CrossRef CAS PubMed.
- N. M. Sangeetha and U. Maitra, Chem. Soc. Rev., 2005, 34, 821–836 RSC.
- M. R. Caplan, P. N. Moore, S. Zhang, R. D. Kamm and D. A. Lauffenburger, Biomacromolecules, 2000, 1, 627–631 CrossRef CAS PubMed.
- S. Shabbir, Y. Lee and H. Rhee, J. Catal., 2015, 322, 104–108 CrossRef CAS.
- S. Das, P. Chakraborty, A. Shit, S. Mondal and A. K. Nandi, J. Mater. Chem. A, 2016, 4, 4194–4210 CAS.
- M. O. M. Piepenbrock, G. O. Lloyd, N. Clarke and J. W. Steed, Chem. Rev., 2010, 110, 1960–2004 CrossRef CAS PubMed.
- S. Das, P. Chakraborty, S. Mondal, A. Shit and A. K. Nandi, ACS Appl. Mater. Interfaces, 2016, 8, 28055–28067 CAS.
- L. A. Estroff and A. D. Hamilton, Chem. Rev., 2004, 104, 1201–1218 CrossRef CAS PubMed.
- J. Zhuang, M. R. Gordon, J. Ventura, L. Li and S. Thayumanavan, Chem. Soc. Rev., 2013, 42, 7421–7435 RSC.
- K. Yamamoto, T. Serizawa, Y. Muraoka and M. Akashi, Macromolecules, 2001, 34, 8014–8020 CrossRef CAS.
- J. L. Zhong, X. J. Jia, H. J. Liu, X. Z. Luo, S. G. Hong, N. Zhang and J. B. Huang, Soft Matter, 2016, 12, 191–199 RSC.
- D. K. Kumar and J. W. Steed, Chem. Soc. Rev., 2014, 43, 2080–2088 RSC.
- D. K. Kumar, D. A. Jose, A. Das and P. Dastidar, Chem. Commun., 2005, 4059–4061 RSC.
- Y. Wang, L. Tang and J. Yu, Cryst. Growth Des., 2008, 8, 884–889 CAS.
- G. O. Lloyd and J. W. Steed, Soft Matter, 2011, 7, 75–84 RSC.
- J. Liu, F. Xu, Z. Sun, Y. Pan, J. Tian, H. C. Lin and X. Li, Soft Matter, 2016, 12, 141–148 RSC.
- A. Vidyasagar and K. M. Sureshan, Angew. Chem., Int. Ed., 2015, 54, 12078–12082 CrossRef CAS PubMed.
- P. Yadav and A. Ballabh, Colloids Surf., A, 2012, 414, 333–338 CrossRef CAS.
- A. Saha, B. Roy, A. Garai and A. K. Nandi, Langmuir, 2009, 25, 8457–8461 CrossRef CAS PubMed.
- B. Roy, A. Saha, A. Esterrani and A. K. Nandi, Soft Matter, 2010, 6, 3337–3345 RSC.
- P. Bairi, B. Roy and A. K. Nandi, J. Phys. Chem. B, 2010, 114, 11454–11461 CrossRef CAS PubMed.
- A. Saha, S. Manna and A. K. Nandi, Chem. Commun., 2008, 3732–3734 RSC.
- A. Saha, S. Manna and A. K. Nandi, Langmuir, 2007, 23, 13126–13135 CrossRef CAS PubMed.
- S. Manna, A. Saha and A. K. Nandi, Chem. Commun., 2006, 4285–4287 RSC.
- G. M. Sheldrick, SADABS: a program for absorption correction with the Siemens SMART system, University of Gӧettingen, Germany, 1996 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122 CrossRef CAS PubMed.
- Y. Wang, W. Li and L. Wu, Langmuir, 2009, 25, 13194–13200 CrossRef CAS PubMed.
- X. Z. Luo, X. J. Jia, J. H. Deng, J. L. Zhong, H. J. Liu, K. J. Wang and D. C. Zhong, J. Am. Chem. Soc., 2013, 135, 11684–11687 CrossRef CAS PubMed.
- X. J. Jia, J. Wang, J. Wu, Y. Du, B. Zhao and D. den Engelsen, RSC Adv., 2015, 5, 72321–72330 RSC.
- H. Liang, Z. Zhang, Q. Yuan and J. Liu, Chem. Commun., 2015, 51, 15196–15199 RSC.
- J. W. Steed, Chem. Soc. Rev., 2010, 39, 3686–3699 RSC.
- L. M. Lacroix, R. Arenal and G. Viau, J. Am. Chem. Soc., 2014, 136, 13075–13077 CrossRef CAS PubMed.
- L. E. Buerkle and S. J. Rowan, Chem. Soc. Rev., 2012, 41, 6089–6102 RSC.
- M. A. Ouimet, R. Fogaça, S. S. Snyder, S. Sathaye, L. H. Catalani, D. J. Pochan and K. E. Uhrich, Macromol. Biosci., 2015, 15, 342–350 CrossRef CAS PubMed.
- P. Stallforth and J. Clardy, Nature, 2013, 495, 456–457 CrossRef CAS PubMed.
- J. P. Zhang, P. Q. Liao, H. L. Zhou, R. B. Lin and X. M. Chen, Chem. Soc. Rev., 2014, 43, 5789–5814 RSC.
- P. Gilli, V. Bertolasi, L. Pretto, V. Ferretti and G. Gilli, J. Am. Chem. Soc., 2004, 126, 3845–3855 CrossRef CAS PubMed.
- S. Liokatis, A. Dose, D. Schwarzer and P. Selenko, J. Am. Chem. Soc., 2010, 132, 14704–14705 CrossRef CAS PubMed.
- B. Escuder, M. LLusar and J. F. Miravet, J. Org. Chem., 2006, 71, 7747–7752 CrossRef CAS PubMed.
- S. Yagai, M. Higashi, T. Karatsu and A. Kitamura, Chem. Mater., 2004, 16, 3582–3585 CrossRef CAS.
- J. Makarević, M. Jokić, B. Perić, V. Tomišić, B. Kojić-Prodić and M. Žinić, Chem.–Eur. J., 2001, 7, 3328–3341 CrossRef.
- S. Dong, B. Zheng, D. Xu, X. Yan, M. Zhang and F. Huang, Adv. Mater., 2012, 24, 3191–3195 CrossRef CAS PubMed.
- J. H. Jung, S. Shinkai and T. Shimizu, Chem.–Eur. J., 2002, 8, 2684–2690 CrossRef CAS.
- G. Yu, X. Yan, C. Han and F. Huang, Chem. Soc. Rev., 2013, 42, 6697–6722 RSC.
- L. Wang, L. Jiang, D. Su, C. Sun, M. Chen, K. Goh and Y. Chen, J. Colloid Interface Sci., 2014, 430, 121–128 CrossRef CAS PubMed.
- S. Tang, X. Huang, X. Chen and N. Zheng, Adv. Funct. Mater., 2010, 20, 2442–2447 CrossRef CAS.
- V. Abbate, A. R. Bassindale, K. F. Brandstadt and P. G. Taylr, J. Catal., 2011, 284, 68–76 CrossRef CAS.
- V. Krishnakumar and R. Mathammal, J. Raman Spectrosc., 2009, 40, 264–271 CrossRef CAS.
- B. Jürgens, E. Irran, J. Senker, P. Kroll, H. Müller and W. Schnick, J. Am. Chem. Soc., 2003, 125, 10288–10300 CrossRef PubMed.
- L. Liu, P. Z. Li, L. Zhu, R. Zou and Y. Zhao, Polymer, 2013, 54, 596–600 CrossRef CAS.
- M. G. Schwab, B. Fassbender, H. W. Spiess, A. Thomas, X. Feng and K. Müllen, J. Am. Chem. Soc., 2009, 131, 7216–7217 CrossRef CAS PubMed.
- O. Lebel, M. È. Perron, T. Maris, S. F. Zalzal, A. Nanci and J. D. Wuest, Chem. Mater., 2006, 18, 3616–3626 CrossRef CAS.
- P. Terech, N. M. Sangeetha and U. Maitra, J. Phys. Chem. B, 2006, 110, 15224–15233 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: CCDC 1504096. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra23761d |
| This journal is © The Royal Society of Chemistry 2016 |