A review of mechanisms for fluorescent ‘‘turn-on’’ probes to detect Al3+ ions
Ankush Gupta* and
Naresh Kumar*
Department of Chemistry, DAV University, Jalandhar-144012, Punjab, India. E-mail: chemankush@yahoo.co.in; naresh.chem.ia@gmail.com
First published on 2nd November 2016
Abstract
The adverse effect of Al3+ ions on human health as well as the environment makes it desirable to develop sensitive and specific techniques for the detection of Al3+ ions. Detection methodology based on the fluorescence technique in combination with fluorescent chemosensors has shown its credibility attributed to its highly sensitive nature as well as to its applicability to the biological samples. Following a brief introduction to the principle of fluorescent sensors, various sensing methods which have been employed for the detection of Al3+ ions are explored. In addition, the outputs of various research groups from the last two years have been discussed. The objective of the present review is to highlight the recent progress in the field of fluorescent chemosensors related to Al3+ detection. The discussion comprises structural designing and mechanistic approaches used to develop selective fluorescent sensors for Al3+ ions. More emphasis has been given to describing the coordination modes and photophysical processes responsible for an effective detection process.
1. Introduction
Chemical sensing is the interaction between an analyte and a receptor molecule which is signalled by an easily detectable change.1 The sensors, especially optical chemosensors are designed in such a way that the detection event is easily measurable by observing photophysical changes.2 Based on this principle, many chemosensors have been designed and synthesized for the detection of different kinds of species, including cations, anions, biomolecules and neutral species.3,4 Among various biologically and environmentally important metal ions, detection of aluminium earned great scientific interest because of its toxic effects to the environment and human health.5–11 Aluminium is the third most abundant element present in the outermost layer of the earth i.e. crust. The widespread practice of aluminium in our day to day life, such as aluminium foil, vessels and cookie sheets, medicines, bleached flour, the paper industry, food additives and aluminium-based pharmaceuticals makes its exposure to the environment.5,6 During acid rain, the level of Al3+ ions (also known as a neurotoxic agent) is increased in the soil or water sources, which is harmful for growing plants.7 Adverse effect of Al3+ ions not only inhibits the plant growth, but also affects the central nervous system of humans and induces Alzheimer's disease, Parkinson's disease and amyotrophic lateral sclerosis.8–11 Further, it is understood that the aluminium-based drugs such as antiperspirants, antiulcer agents and antacids are more harmful to human health than that of food contaminated with Al3+ ions.12 In addition, Al3+ ions can stay inside the cells and tissues for longer time before being excreted by the body.13 According to WHO, the admissible value of weekly intake of Al3+ ions by humans is about 7 mg kg−1 of the body weight.14,15 Thus, detection of Al3+ ions is important to control its impact on the human health as well as to the environment.In view of the above discussion, many fluorescent chemosensors which are specific for various metal ions or other transition metals have been reported in the past.16 However, the growth related to the fluorescent chemosensors for the detection of Al3+ ions is still less advanced. This may be due to the less availability of complexing units for Al3+ as well as the poor coordination ability of Al3+ as compared to other metal ions. Taking into account the prompt need of Al3+ ion sensors, many research groups are engaged in the development of chemosensors to detect Al3+ ions, indicating the scope of development of fluorescent chemosensors for this cation.
Various analytical methods such as atomic absorption and emission spectroscopy, electrochemiluminscence and electrochemical have been used for the detection of Al3+ ions.17–20 However, these methods cannot be used in the field because of high cost instrumentations, need of trained persons, complicated procedures and lack of selectivity. The aforementioned problems make these methods difficult for routine monitoring and applications. In this context, fluorescence signaling is one of the first choices due to its functional simplicity, real-time and high sensitive detection over the other methods.21 Indeed, the chemosensors which show fluorescence enhancement by interacting with the target analytes are more proficient as there is less probability to give false signals.22–25 To develop selective fluorescence chemosensors for Al3+ ions, one should prefer hard donor sites like N and O in its coordination sphere as Al3+ is hard acid. Therefore, most of the reported Al3+ ions sensors are incorporated with nitrogen and oxygen donor sites.
Based on the photophysical processes, number of signal mechanisms like photo-induced electron transfer (PET), intramolecular charge transfer (ICT) and electronic energy transfer (EET) or resonance energy transfer (RET) have been used to develop efficient fluorescent systems to detect Al3+ ions. Different types of non-covalent interactions between chemosensor and Al3+ are employed to introduce the above stated photophysical mechanisms. In addition to this, in recent years, new sensing mechanisms like aggregation-induced emission (AIE), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and excited-state intramolecular proton transfer (ESIPT) have also been emerged for the detection of Al3+ ions. The research in the field of Al3+ ions detection has increased considerably in the past few years. We believe that the present article will be able to provide an overview of fluorescent chemosensors with respect to their design, complexation units and applications to detect Al3+ ions. Following is the discussion related to the detection of Al3+ ions categorized according to their photophysical processes.
N isomerization and excited-state intramolecular proton transfer (ESIPT) have also been emerged for the detection of Al3+ ions. The research in the field of Al3+ ions detection has increased considerably in the past few years. We believe that the present article will be able to provide an overview of fluorescent chemosensors with respect to their design, complexation units and applications to detect Al3+ ions. Following is the discussion related to the detection of Al3+ ions categorized according to their photophysical processes.
2. PET-based Al3+ detection
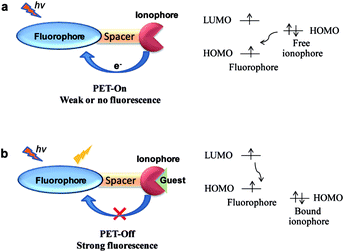
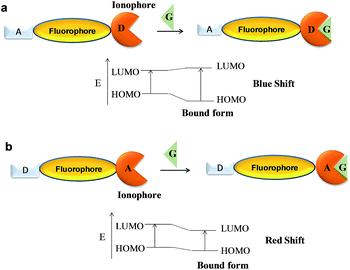
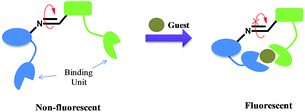
The PET phenomenon based sensors are generally associated with fluorophore connected via spacer to an ionophore unit having non-bonding electron pair. During excitation process of a fluorophore, electron from the lowest unoccupied molecular orbital (LUMO) goes to the highest occupied molecular orbital (HOMO) followed by charge recombination and returns to the ground state along with the release of excess energy as fluorescence emission.However, if the HOMO orbital of another part of the molecule, i.e. ionophore has energy between the energy levels of the fluorophore, then there is electron transfer from the HOMO orbital of ionophore to the half occupied HOMO of the fluorophore, subsequently responsible for the non-radiative decay process, thus, fluorescence quenching (Fig. 1a). On the other hand, in the bound state with a guest molecule, the energy of HOMO of ionophore decreases than that of fluorophore due to increase in the redox potentials (Fig. 1b). This process restricts the electron transfer and enables the fluorescence from the fluorophore.
Based on the PET mechanism, a number of examples have been reported for the detection of Al3+ ions. For instance, Alici et al. synthesized a biphenylcarbonitrile based derivative 1 for the detection of Al3+ ions in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) medium.26 Sensor 1 is non-fluorescent when excited at 365 nm due to the PET process from the imino nitrogen to the biphenylcarbonitrile unit. However, upon addition of Al3+ ions, sensor 1 showed significant increment in fluorescence emission at 516 nm. This shift in fluorescence was due to the interaction of Al3+ with the imino nitrogen and phenolic oxygen which restrict the PET process responsible for the strong emission. However, no change in fluorescence was observed with the addition of other competitive metal ions. Based on the fluorescence titration of 1 with Al3+, the binding constant evaluated was 5.36 ± 0.24 × 109 M−1 while the detection limit was 13.7 ± 0.17 × 10−7 M. Following a similar strategy, Kumar et al. reported a chemosensor 2 based on the naphthyl unit having imino appended phenyl moiety.27 In CH3CN
1; v/v) medium.26 Sensor 1 is non-fluorescent when excited at 365 nm due to the PET process from the imino nitrogen to the biphenylcarbonitrile unit. However, upon addition of Al3+ ions, sensor 1 showed significant increment in fluorescence emission at 516 nm. This shift in fluorescence was due to the interaction of Al3+ with the imino nitrogen and phenolic oxygen which restrict the PET process responsible for the strong emission. However, no change in fluorescence was observed with the addition of other competitive metal ions. Based on the fluorescence titration of 1 with Al3+, the binding constant evaluated was 5.36 ± 0.24 × 109 M−1 while the detection limit was 13.7 ± 0.17 × 10−7 M. Following a similar strategy, Kumar et al. reported a chemosensor 2 based on the naphthyl unit having imino appended phenyl moiety.27 In CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) mixture, compound 2 (1 × 10−5 M) showed weak emission at 430 nm upon excitation at 310 nm. The quenched emission was ascribed to the PET from the nitrogen atom to the naphthyl unit. However, the interaction of Al3+ ions with 2 resulted in a significant emission enhancement at 430 nm (Fig. 2b) ascribed to the blocking of PET process. The fluorescence response of 2 towards Al3+ ions was selective even in the presence of other metal ions except copper ions.
1; v/v) mixture, compound 2 (1 × 10−5 M) showed weak emission at 430 nm upon excitation at 310 nm. The quenched emission was ascribed to the PET from the nitrogen atom to the naphthyl unit. However, the interaction of Al3+ ions with 2 resulted in a significant emission enhancement at 430 nm (Fig. 2b) ascribed to the blocking of PET process. The fluorescence response of 2 towards Al3+ ions was selective even in the presence of other metal ions except copper ions.
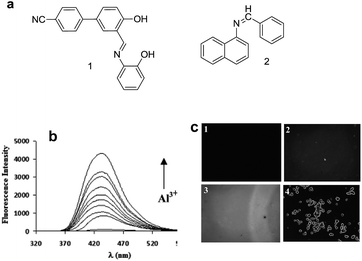 | ||
| Fig. 2 (a) Structure of chemosensor 1 and 2; (b) fluorescence changes in 2 (1 × 10−5 M) upon addition of (one third equivalent) Al3+ ions; (c) fluorescence image of (1) HEK cell solution; (2) HEK cell with Al3+ ions; (3) HEK cell with ligand 2; (4) HEK cell with ligand 2 and Al3+. (b) and (c) reproduced from ref. 27 with permission from The Royal Society of Chemistry, copyright 2015. | ||
The detection limit of 2 for Al3+ ions was 5 × 10−5 M. Sensor 2 interacts with Al3+ through imino nitrogen atoms and showed stoichiometry of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
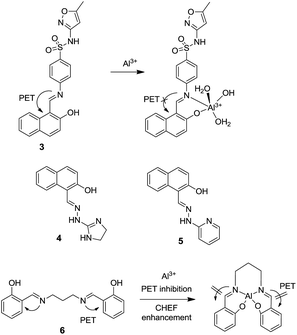
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 for the metal–ligand complexation. In addition, chemosensor 2 has also been employed for the detection of Al3+ ions in the cellular system (Fig. 2c). Mondal et al. synthesized a naphthyl appended sulfonamide based sensor 3 for the selective fluorescence turn-on detection of Al3+ in aqueous medium.28 The chemosensor 3 upon excitation at 370 nm showed weak emission at 520 nm which upon addition of incremental amount of Al3+ ions shifted to 425 nm with emission enhancement. These changes were attributed to the inhibition of PET because of the coordination of azomethine-N and phenolic-O with Al3+ as shown in Fig. 3. The binding constant for the interaction of Al3+ ions and ligand 3 was 1.04 ± 0.01 × 104 M with 1
3 for the metal–ligand complexation. In addition, chemosensor 2 has also been employed for the detection of Al3+ ions in the cellular system (Fig. 2c). Mondal et al. synthesized a naphthyl appended sulfonamide based sensor 3 for the selective fluorescence turn-on detection of Al3+ in aqueous medium.28 The chemosensor 3 upon excitation at 370 nm showed weak emission at 520 nm which upon addition of incremental amount of Al3+ ions shifted to 425 nm with emission enhancement. These changes were attributed to the inhibition of PET because of the coordination of azomethine-N and phenolic-O with Al3+ as shown in Fig. 3. The binding constant for the interaction of Al3+ ions and ligand 3 was 1.04 ± 0.01 × 104 M with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry of metal–ligand complex. Similarly, Mukherjee et al. synthesized naphthalene and hydrazone based sensors 4 and 5 for the selective detection of Al3+ ions in DMSO
1 stoichiometry of metal–ligand complex. Similarly, Mukherjee et al. synthesized naphthalene and hydrazone based sensors 4 and 5 for the selective detection of Al3+ ions in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HEPES (5
HEPES (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95; v/v).29 The fluorescence spectra of 4 (1 × 10−5 M) and 5 (1 × 10−5 M) showed significant emission enhancement upon gradual addition of Al3+ ions (5.0 × 10−5 M for 4 and 15.0 × 10−5 M for 5). Sensor 4 exhibited 5 fold, while sensor 5 showed 45 fold emission enhancements in the presence of Al3+ ions. These fluorescence changes were devoted to the suppression of PET assisted by the chelation-enhanced fluorescence (CHEF) process. However, sensor 5 displayed more sensitivity towards Al3+ ions as compared to 4 because of the presence of pyridine moiety instead of imidazoline. In another example, Huerta-Aguilar et al. reported fluorescent organic nanoparticles (FONPs) based on sensor 6 for the detection of Al3+ in aqueous solution.30 FONPs of 6 exhibited quenched emissions due to the PET process from imino nitrogen donors to the π-conjugated system.
95; v/v).29 The fluorescence spectra of 4 (1 × 10−5 M) and 5 (1 × 10−5 M) showed significant emission enhancement upon gradual addition of Al3+ ions (5.0 × 10−5 M for 4 and 15.0 × 10−5 M for 5). Sensor 4 exhibited 5 fold, while sensor 5 showed 45 fold emission enhancements in the presence of Al3+ ions. These fluorescence changes were devoted to the suppression of PET assisted by the chelation-enhanced fluorescence (CHEF) process. However, sensor 5 displayed more sensitivity towards Al3+ ions as compared to 4 because of the presence of pyridine moiety instead of imidazoline. In another example, Huerta-Aguilar et al. reported fluorescent organic nanoparticles (FONPs) based on sensor 6 for the detection of Al3+ in aqueous solution.30 FONPs of 6 exhibited quenched emissions due to the PET process from imino nitrogen donors to the π-conjugated system.
However, addition of Al3+ (0.45 mM) to the solution of FONPs of 6 (0.1 mM) resulted in a strong fluorescence enhancement at 490 nm. This emission enhancement was attributed to the suppression of PET process upon coordination of FONPs with Al3+ ions. Theoretical calculations showed that there is formation of cavity facilitated by aromatic rings of 6 that allows the coordination of Al3+ ion (Fig. 3). A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
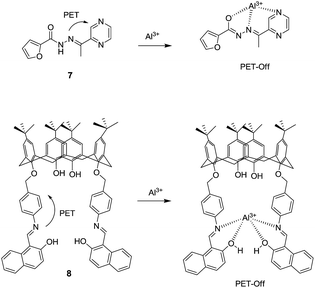
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry between FONPs of 6 and Al3+ was confirmed by Job's plot with detection limit of 1.24 × 10−3 mM. Li et al. have reported a pyrazine based derivative 7 bearing a furan unit for the selective fluorescence turn-on detection of Al3+ in ethanol (Fig. 4).31 In the UV-vis studies, chemosensor 7 (100 μM) in ethanol exhibited absorption band at 306 nm with a shoulder at 382 nm, which is characteristic band of pyrazine. Addition of Al3+ ions (5 equiv.) to solution of 7 showed a significance decrease in the absorption band at 306 nm with the formation of a new band at 382 nm. These changes lead to formation of two isosbestic points at 237 and 336 nm, indicating the complexation between 7 and Al3+ ions via pyrazine unit. On the other hand, upon excitation at 382 nm, the fluorescence spectrum of 7 (50 μM) exhibited quenched emission, indicating the role of PET process. However, addition of Al3+ ions (5 equiv.) resulted in CHEF effect and inhibition of PET process from the Schiff-base nitrogen atom to pyrazine, resulting in the enhancement of emission intensity at 517 nm. A 1
1 stoichiometry between FONPs of 6 and Al3+ was confirmed by Job's plot with detection limit of 1.24 × 10−3 mM. Li et al. have reported a pyrazine based derivative 7 bearing a furan unit for the selective fluorescence turn-on detection of Al3+ in ethanol (Fig. 4).31 In the UV-vis studies, chemosensor 7 (100 μM) in ethanol exhibited absorption band at 306 nm with a shoulder at 382 nm, which is characteristic band of pyrazine. Addition of Al3+ ions (5 equiv.) to solution of 7 showed a significance decrease in the absorption band at 306 nm with the formation of a new band at 382 nm. These changes lead to formation of two isosbestic points at 237 and 336 nm, indicating the complexation between 7 and Al3+ ions via pyrazine unit. On the other hand, upon excitation at 382 nm, the fluorescence spectrum of 7 (50 μM) exhibited quenched emission, indicating the role of PET process. However, addition of Al3+ ions (5 equiv.) resulted in CHEF effect and inhibition of PET process from the Schiff-base nitrogen atom to pyrazine, resulting in the enhancement of emission intensity at 517 nm. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry between 7 and Al3+ was confirmed by Job's plot experiment and the binding constant of 7 with Al3+ was 2.36 × 107 M−1. Apart from naphthalene and pyrazine based systems, calix[4]arene have also been confirmed as suitable candidates for PET-based sensors. For example, Memon et al. have reported a naphthalene appended calix[4]arene based chemosensor 8 for the sensing of Al3+ in THF/water mixture.32 A similar PET mechanism occurs between the naphthalene and the imino moieties of 8 responsible for the quenched emission. However, the coordination of Al3+ ions inhibits the PET process which results in the remarkable fluorescence enhancement at 400 nm.
1 stoichiometry between 7 and Al3+ was confirmed by Job's plot experiment and the binding constant of 7 with Al3+ was 2.36 × 107 M−1. Apart from naphthalene and pyrazine based systems, calix[4]arene have also been confirmed as suitable candidates for PET-based sensors. For example, Memon et al. have reported a naphthalene appended calix[4]arene based chemosensor 8 for the sensing of Al3+ in THF/water mixture.32 A similar PET mechanism occurs between the naphthalene and the imino moieties of 8 responsible for the quenched emission. However, the coordination of Al3+ ions inhibits the PET process which results in the remarkable fluorescence enhancement at 400 nm.
The competitive metal-ion selectivity experiments confirmed the selectivity of 8 toward Al3+ ions, however, interference was observed with mercury ions. Ligand 8 with Al3+ forms 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric complex with binding constant of 2.49 × 105 M−1. Ghorai et al. reported a fluorescent sensor 9 based on the combination of chemical units pyridine, imine and phenyl moieties for the detection of Al3+ in mixed aqueous medium.33 Chemosensor 9 (10 μM) in MeOH
1 stoichiometric complex with binding constant of 2.49 × 105 M−1. Ghorai et al. reported a fluorescent sensor 9 based on the combination of chemical units pyridine, imine and phenyl moieties for the detection of Al3+ in mixed aqueous medium.33 Chemosensor 9 (10 μM) in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (2
H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) exhibited absorption bands at 240, 287 and 368 nm. Upon incremental addition Al3+ ions, there was decrease in absorption intensity at 368 nm while absorption bands at 240 nm and 287 nm showed absorption enhancement. In the fluorescence studies, ligand 9 presented a weak emission as a result of PET from pyridyl nitrogen atoms to the central π-conjugated system involving imino units as well as aromatic moieties. However, the addition of Al3+ showed a strong fluorescence enhancement at 373 nm attributed to the complexation of Al3+ ions with the pyridyl nitrogen atoms which hinders the PET process (Fig. 5). The sensor form a 1
1; v/v) exhibited absorption bands at 240, 287 and 368 nm. Upon incremental addition Al3+ ions, there was decrease in absorption intensity at 368 nm while absorption bands at 240 nm and 287 nm showed absorption enhancement. In the fluorescence studies, ligand 9 presented a weak emission as a result of PET from pyridyl nitrogen atoms to the central π-conjugated system involving imino units as well as aromatic moieties. However, the addition of Al3+ showed a strong fluorescence enhancement at 373 nm attributed to the complexation of Al3+ ions with the pyridyl nitrogen atoms which hinders the PET process (Fig. 5). The sensor form a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
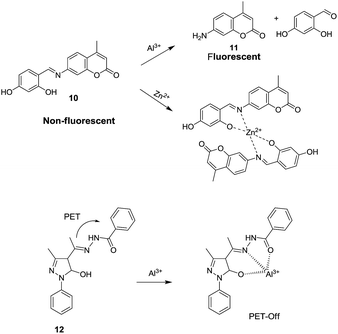
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ligand–metal complex with Al3+ ions as confirmed by Job's plot and mass analysis. Based on the fluorescence titration of 9 with Al3+ ions, the calculated binding constant was 1.26 × 105 M−1 with detection limit of 0.90 μM. In addition to the coordination methods, other detection strategies such as reaction between the analyte and sensor molecule can be utilized for the development of sensors for Al3+ ion. Based on this approach, Qin et al. reported a sensor 10 by employing coumarin unit as a fluorophore.34
2 ligand–metal complex with Al3+ ions as confirmed by Job's plot and mass analysis. Based on the fluorescence titration of 9 with Al3+ ions, the calculated binding constant was 1.26 × 105 M−1 with detection limit of 0.90 μM. In addition to the coordination methods, other detection strategies such as reaction between the analyte and sensor molecule can be utilized for the development of sensors for Al3+ ion. Based on this approach, Qin et al. reported a sensor 10 by employing coumarin unit as a fluorophore.34
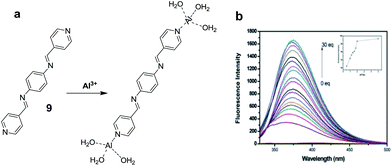 | ||
| Fig. 5 (a) Structure and binding mode in 9; (b) fluorescence spectra of 9 after addition of increasing amounts of Al3+ ions. Reproduced from ref. 33 with permission from The Royal Society of Chemistry, copyright 2015. | ||
Solution of 10 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (95
H2O (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5; v/v) mixture was non-fluorescent, attributed to the PET from imino nitrogen atom to the coumarin unit. However, the addition of Al3+ or Zn2+ ions resulted in the fluorescence enhancements at 427 (λex = 357 nm) and 496 nm (λex = 405 nm), respectively. The fluorescence response in the case of aluminium was ascribed to the hydrolysis of imine unit of 10 resulted in formation of free coumarin 11 which exhibits fluorescence at 427 nm (Fig. 6). In the case of zinc ions, coordination as displayed in Fig. 6 was taking place with inhibition of the PET process which lead to fluorescence enhancement at 496 nm. The sensor showed detection limit of 3.7 × 10−6 M for Al3+ ions. The same group further developed chemosensor 12 for the detection of Al3+ ions35 with detection limit of 2 × 10−7 M. In another report, chemosensor 13 has been designed based on acridine for the detection of Al3+ ions.36 The fluorescence intensity of 13 was enhanced at 425 nm upon addition of Al3+ ions, attributed to the suppression of PET process as a result of electrostatic interactions between COOH and Al3+ ions (Fig. 7). The chemosensor exhibited binding constant and detection limit of 1.7 × 104 M−1 and 5.8 × 10−7 M for the Al3+ ions, respectively.
5; v/v) mixture was non-fluorescent, attributed to the PET from imino nitrogen atom to the coumarin unit. However, the addition of Al3+ or Zn2+ ions resulted in the fluorescence enhancements at 427 (λex = 357 nm) and 496 nm (λex = 405 nm), respectively. The fluorescence response in the case of aluminium was ascribed to the hydrolysis of imine unit of 10 resulted in formation of free coumarin 11 which exhibits fluorescence at 427 nm (Fig. 6). In the case of zinc ions, coordination as displayed in Fig. 6 was taking place with inhibition of the PET process which lead to fluorescence enhancement at 496 nm. The sensor showed detection limit of 3.7 × 10−6 M for Al3+ ions. The same group further developed chemosensor 12 for the detection of Al3+ ions35 with detection limit of 2 × 10−7 M. In another report, chemosensor 13 has been designed based on acridine for the detection of Al3+ ions.36 The fluorescence intensity of 13 was enhanced at 425 nm upon addition of Al3+ ions, attributed to the suppression of PET process as a result of electrostatic interactions between COOH and Al3+ ions (Fig. 7). The chemosensor exhibited binding constant and detection limit of 1.7 × 104 M−1 and 5.8 × 10−7 M for the Al3+ ions, respectively.
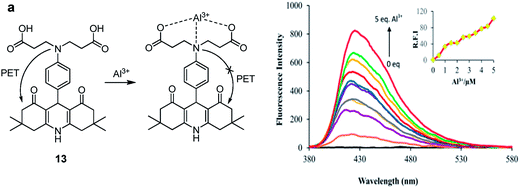 | ||
| Fig. 7 (a) Structure and binding mode of 13; (b) fluorescence spectra of 13 upon addition of Al3+ ions. Reproduced from ref. 36 with permission from The Royal Society of Chemistry, copyright 2015. | ||
Das and co-workers reported a cyanine appended imidazole based fluorescent chemosensor 14 for the near-IR (NIR; emission range 650–900 nm) detection of Al3+ ions.37 Chemosensor 14 in MeOH–HEPES (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2; v/v), at pH 7.2 showed absorption band at 410 nm. The addition of Al3+ resulted in the decrease of absorption at 410 nm along with the appearance of new absorption bands at 343, 539 and 710 nm (Fig. 8b). These changes were ascribed to the internal charge transfer process upon interaction of 14 with Al3+ ions. The fluorescence spectrum of 14 exhibited quenched emission because of the PET from the imidazole unit to the cyanine fluorophore (Fig. 8a). However, upon addition of Al3+ ions an emission band appeared at 730 nm due to the inhibition of PET process (Fig. 8c). The binding mode of 14 involves interaction of imidazole nitrogen atoms with the Al3+ ions. Moreover, the fluorescence behaviour of 14 was selective toward Al3+ ions, although Ga3+ also showed small emission enhancement. A 1
2; v/v), at pH 7.2 showed absorption band at 410 nm. The addition of Al3+ resulted in the decrease of absorption at 410 nm along with the appearance of new absorption bands at 343, 539 and 710 nm (Fig. 8b). These changes were ascribed to the internal charge transfer process upon interaction of 14 with Al3+ ions. The fluorescence spectrum of 14 exhibited quenched emission because of the PET from the imidazole unit to the cyanine fluorophore (Fig. 8a). However, upon addition of Al3+ ions an emission band appeared at 730 nm due to the inhibition of PET process (Fig. 8c). The binding mode of 14 involves interaction of imidazole nitrogen atoms with the Al3+ ions. Moreover, the fluorescence behaviour of 14 was selective toward Al3+ ions, although Ga3+ also showed small emission enhancement. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
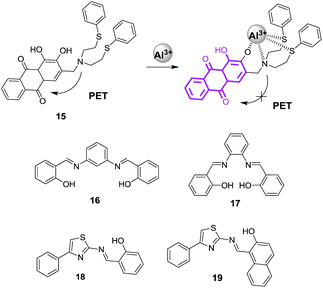
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding stoichiometry between 14 and Al3+ was confirmed by mass analysis with binding constant of 8.4 × 105 M−1. Chemosensor 14 showed detection limit of 1.95 × 10−8 M for Al3+ ions. In addition, the reported sensor 14 was employed to detect Al3+ ions in the intracellular system, thus, revealed the biological applicability of 14 (Fig. 8d). Zeng et al. reported a PET based chemosensor 15 by attaching a pod-and unit having nitrogen and sulphur atoms to the dihydroxyanthraquinone moiety.38 The pod-and unit acts as a complexation moiety for Al3+ ions while anthraquinone performs the role of a signalling unit. Ligand 15 in EtOH
1 binding stoichiometry between 14 and Al3+ was confirmed by mass analysis with binding constant of 8.4 × 105 M−1. Chemosensor 14 showed detection limit of 1.95 × 10−8 M for Al3+ ions. In addition, the reported sensor 14 was employed to detect Al3+ ions in the intracellular system, thus, revealed the biological applicability of 14 (Fig. 8d). Zeng et al. reported a PET based chemosensor 15 by attaching a pod-and unit having nitrogen and sulphur atoms to the dihydroxyanthraquinone moiety.38 The pod-and unit acts as a complexation moiety for Al3+ ions while anthraquinone performs the role of a signalling unit. Ligand 15 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) mixture showed a strong absorption band at 446 nm which upon addition of Al3+ ions displayed red shift to 504 nm with enhancement in absorption.
1; v/v) mixture showed a strong absorption band at 446 nm which upon addition of Al3+ ions displayed red shift to 504 nm with enhancement in absorption.
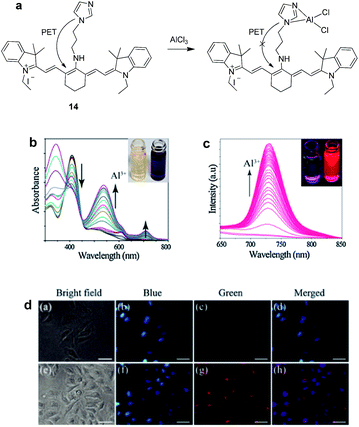 | ||
| Fig. 8 (a) Structure and binding mode of 14; (b) absorption and (c) fluorescence spectra of 14 upon addition of Al3+ ions; (d) fluorescence images: (a–d) HeLa cells treated with 5 mM of 14; (e–h) HeLa cells treated with 5 mM of 14 + 10 mM of Al3+. Reproduced from ref. 37 with permission from Elsevier, copyright 2015. | ||
This process accompanied the appearance of three isosbestic points at 446, 579 and 650 nm, which revealed the direct formation of 15–Al3+. The formation of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric complex between 15 and Al3+ ions was confirmed by Job's plot as well as by mass analysis of the complex. The fluorescence spectrum of 15 shows quenched fluorescence attributed to the PET form the nitrogen and sulphur atoms of pod-and arm to the anthraquinone unit. Upon addition of Al3+, the fluorescence spectrum of 15 exhibited significant emission enhancement at ∼600 nm due to the removal of the PET process as Al3+ ion complexes with the pod-and arm (Fig. 9). The association constant for the binding of Al3+ by 15 was 8.84 × 103 M−1 while the detection limit was 5.0 × 10−7 M. In addition, it was observed that the 15 exhibited good selectivity for Al3+ over the other alkali and transition metal ions. Similarly, Gupta and co-workers used PET based strategy in examples 16, 17, 18 and 19 for the fluorescence turn-on detection of Al3+ ions.39,40
1 stoichiometric complex between 15 and Al3+ ions was confirmed by Job's plot as well as by mass analysis of the complex. The fluorescence spectrum of 15 shows quenched fluorescence attributed to the PET form the nitrogen and sulphur atoms of pod-and arm to the anthraquinone unit. Upon addition of Al3+, the fluorescence spectrum of 15 exhibited significant emission enhancement at ∼600 nm due to the removal of the PET process as Al3+ ion complexes with the pod-and arm (Fig. 9). The association constant for the binding of Al3+ by 15 was 8.84 × 103 M−1 while the detection limit was 5.0 × 10−7 M. In addition, it was observed that the 15 exhibited good selectivity for Al3+ over the other alkali and transition metal ions. Similarly, Gupta and co-workers used PET based strategy in examples 16, 17, 18 and 19 for the fluorescence turn-on detection of Al3+ ions.39,40
It could be easily inferred from the above discussed examples that PET is an advantageous photophysical mechanism for the development of fluorescent chemosensors. Indeed, to have fluorescence turn-on response, PET is certainly one of the best approaches for the detection of ions. Although, various fluorescent chemosensors have been reported by employing PET for the detection of Al3+ ions, still there is need of improvement, like working in the pure aqueous environment.
3. ICT-based fluorescent sensors
In ICT based sensors the fluorophores and the ionophores are directly connected with each other forming a single species. These two functionalities in ICT based sensors behave as either an electron donor or an electron acceptor.In such system, HOMO of the sensor has electron density near to the electron-donating group while the LUMO of sensor has electron density close to the electron-acceptor group, which creates dipoles with ICT from the donor to the acceptor unit upon excitation.41 However, the complexation of a guest species either with the donor or acceptor part results in the alteration in dipole strength of donor–acceptor species which lead to change in photophysical behaviour. In the first instance, the interaction of guest with the donor part results in the decrease in the electron-donating character of the donor, hence, blue-shift in the emission spectrum (Fig. 10a). On the other hand, if acceptor group interacts with the guest species, the electron-withdrawing nature of acceptor group increases, hence, there is red-shift in the emission spectrum (Fig. 10b).
On the basis of ICT process, various chemosensors have been designed and reported for the detection of Al3+ ions. For instance, Li et al. reported a fluorescent chemosensor 20 designed on phenolphthalein for the detection of Al3+ ions.42 The sensor 20 (10 μM) showed a weak emission at 520 nm in EtOH ascribed to the ICT process (Fig. 11a). On the other hand, in the presence of Al3+ (5 equiv.) a strong emission enhancement at 478 nm with 42 nm blue shift was observed (Fig. 11). This fluorescence response as explained by authors was due to the blocking of ICT as well as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization upon coordination of 20 with Al3+ via nitrogen and oxygen atoms. Chemosensor 20 is selective towards Al3+, although Cu2+ induces fluorescence quenching at 520 nm due to ligand–metal charge transfer process arising from 20 to the open-shell d-orbitals of Cu2+. A complex of 1
N isomerization upon coordination of 20 with Al3+ via nitrogen and oxygen atoms. Chemosensor 20 is selective towards Al3+, although Cu2+ induces fluorescence quenching at 520 nm due to ligand–metal charge transfer process arising from 20 to the open-shell d-orbitals of Cu2+. A complex of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ligand–metal stoichiometry was observed for the binding of 20–Al3+ involving the imino and hydroxyl units with binding constant of 2.63 × 104 M−1 and detection limit of 15 nM for Al3+ ions.
2 ligand–metal stoichiometry was observed for the binding of 20–Al3+ involving the imino and hydroxyl units with binding constant of 2.63 × 104 M−1 and detection limit of 15 nM for Al3+ ions.
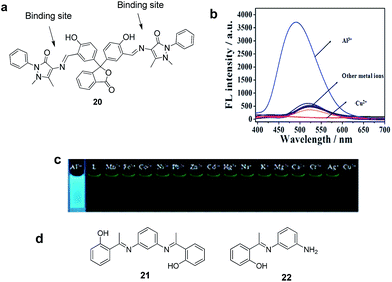 | ||
| Fig. 11 (a) Structure of chemosensor 20; (b) fluorescence spectra of 20 upon addition of different metal ions as their nitrate salts (5 equiv. each); (c) photographs of 20 with metal ions in EtOH under a hand-held UV lamp. Reproduced from ref. 42 with permission from The Royal Society of Chemistry, copyright 2016. (d) Structure of compounds 21 and 22. | ||
New fluorescent sensors 21 and 22 having imino and hydroxy moieties have been reported for turn-on detection of Al3+ ions.43 The UV-vis spectrum of 21 and 22 in methanol showed two absorption bands at 324 and 254 nm. However, the addition of Al3+ to the solution of 21 and 22 resulted in the appearance of a new absorption band at 380 and 324 nm, respectively. Upon excitation at 375, the fluorescence spectrum of 21 and 22 showed no emission, which upon addition of Al3+ ions gave strong fluorescence at 465 and 464 nm, respectively. This increase in fluorescence was due to the binding of Al3+ ions to the charge donating sites of the receptors which hindered the ICT process. The other tested metal ions did not show any significant change in fluorescence emission of 21 and 22, indicating the high selectivity of these sensors for Al3+ ions. The calculated binding constants for 21 and 22 were 6.64 × 103 M−1 and 7.29 × 103 M−1 with detection limits of 5.25 × 10−7 M and 2.38 × 10−6 M, respectively.
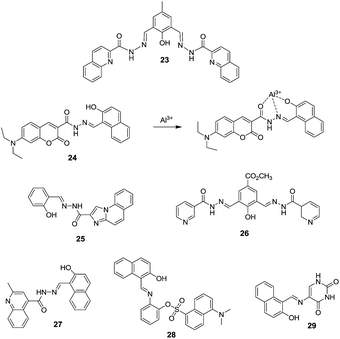
Datta et al. reported a dialdehyde–quinolone based chemosensor 23 for different type of ions including Al3+, Zn2+ and F− ions.44 Ligand 23 exhibited a weak fluorescence emission attributed to the less ICT character and rotation regarding the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond. However, upon interaction with the Al3+ or Zn2+ ions, the rotation around the imino bond become restricted which promotes the ICT process, resulting in emission enhancement. On the other hand, the addition of F− ions to 23 resulted in the deprotonation of hyrdroxy group, which triggered the ICT process, hence, emission enhancement. In another example, Qin et al. synthesized a chemosensor 24 based on the combination of coumarin and naphthalene units.45 Chemosensor 24 in EtOH
N bond. However, upon interaction with the Al3+ or Zn2+ ions, the rotation around the imino bond become restricted which promotes the ICT process, resulting in emission enhancement. On the other hand, the addition of F− ions to 23 resulted in the deprotonation of hyrdroxy group, which triggered the ICT process, hence, emission enhancement. In another example, Qin et al. synthesized a chemosensor 24 based on the combination of coumarin and naphthalene units.45 Chemosensor 24 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) showed ratiometric effect in presence of Al3+ ions. Free 24 (10.0 μM) exhibited emission band at 481 nm which upon addition of Al3+ ions (10 equiv.) shifted to 525 nm (about 45 nm red shift). This shift was ascribed to the enhanced charge transfer process on complexation of Al3+ ions with the acceptor (–C
1; v/v) showed ratiometric effect in presence of Al3+ ions. Free 24 (10.0 μM) exhibited emission band at 481 nm which upon addition of Al3+ ions (10 equiv.) shifted to 525 nm (about 45 nm red shift). This shift was ascribed to the enhanced charge transfer process on complexation of Al3+ ions with the acceptor (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) which caused decrease in electron density of acceptor group as displayed in Fig. 12. The addition of other metal ions did not introduced fluorescence behaviour as observed in the case of Al3+. Chemosensor 24 exhibited detection limit of 3.3 × 10−6 M and binding constant of 1.03 × 104 M−1 for the Al3+ ions.
O) which caused decrease in electron density of acceptor group as displayed in Fig. 12. The addition of other metal ions did not introduced fluorescence behaviour as observed in the case of Al3+. Chemosensor 24 exhibited detection limit of 3.3 × 10−6 M and binding constant of 1.03 × 104 M−1 for the Al3+ ions.
Sun et al. synthesized a fluorescence turn-on chemosensor 25 for Al3+ based on the charge transfer process.46 Chemosensor 25 showed a weak fluorescence band at 375 nm attributed to the ICT. However, with incremental addition of Al3+ ions, there is significant red shift with new emission band at 450 nm. These changes were attributed to the alteration in the ICT process in 25 upon coordination with Al3+, which was supported by DFT studies. In line of this, Qin et al. reported imino-based chemosensors 26 and 27 for the detection of Al3+ ions.47,48 Similarly, in case of chemosensor 28, the combination of naphthalene and dansyl moieties along with the imine and hydroxyl functionalities has been employed for the coordination of Al3+ with similar fluorescence performance.49 Singh et al.50 reported a tridentate fluorescent sensor 29 for the selective turn-on determination of Al3+ in acetonitrile solution. Derivative 29 exhibited a weak fluorescence emission attributed to the ICT character. The stable coordination of 29 with Al3+ restricts the ICT process, which increases the rigidity of the molecular assembly, hence, increases the fluorescence emission. The binding constant of 29 for Al3+ was 3.148 × 104 M−1 with detection limit of 3.2 × 10−7 M.
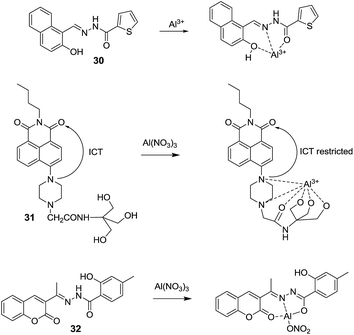
Tiwari et al.51 synthesized a derivative 30 for the selective and sensitive detection of Al3+ ions. The solution of 30 (20 μM) in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; v/v) showed absorption bands at 327 and 365 nm. Upon addition of Al3+ ions (0–40 μM), two new red shifted bands at 339 nm 407 nm were observed. The fluorescence spectrum of 30 (0.5 μM) was characterized by weak fluorescence emissions at 475 and 450 nm (λex 407 nm), which upon addition of two equivalents of Al3+ ions exhibited significant fluorescence enhancement. These changes in fluorescence and absorption were attributed to the restriction of ICT process in 30 upon forming complex with Al3+ ions. FT-IR, 1H NMR titration and ESI-MS experiments confirmed the formation of 1
4; v/v) showed absorption bands at 327 and 365 nm. Upon addition of Al3+ ions (0–40 μM), two new red shifted bands at 339 nm 407 nm were observed. The fluorescence spectrum of 30 (0.5 μM) was characterized by weak fluorescence emissions at 475 and 450 nm (λex 407 nm), which upon addition of two equivalents of Al3+ ions exhibited significant fluorescence enhancement. These changes in fluorescence and absorption were attributed to the restriction of ICT process in 30 upon forming complex with Al3+ ions. FT-IR, 1H NMR titration and ESI-MS experiments confirmed the formation of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex of metal and ligand. The chemosensor 30 showed binding constant and detection limit in the range of 7.06 × 106 M−1 and 1.35 × 10−9 M, respectively.
1 complex of metal and ligand. The chemosensor 30 showed binding constant and detection limit in the range of 7.06 × 106 M−1 and 1.35 × 10−9 M, respectively.
Kang et al.52 reported a naphthalimide based chemosensor 31 for the selective detection of Al3+ ions over the other metal ions. In the chemosensor design, piperazine ring was attached to the 4th position of the 1,8-naphthalimide unit to develop ICT based molecular system. The fluorescence spectrum of 31 in CH3OH showed weak emission at 529 nm (λex = 400 nm) due to ICT process. However, upon addition of Al3+ ions, significant increase in fluorescence emission was observed with blue shift from 529 to 512 nm. This change was due to the formation of a rigid system upon binding with Al3+ ions which suppressed the ICT process by decreasing the electron-donating ability of piperazine nitrogen atom linked to the naphthalene ring (Fig. 13). Recently, Hossain et al.53 reported a coumarin based chemosensor 32 for the coordination of Al3+ ions. The reported sensor was weakly fluorescent in DMF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) mixture which upon coordination with Al3+ ions showed remarkable increase in fluorescence emission. This increase in fluorescence intensity was owing to the binding of Al3+ ions with the 32 which promoted the ICT process via increasing the electron withdrawing ability of the coordinating carbonyl oxygen atom of the coumarin moiety and nitrate group (Fig. 13).
1; v/v) mixture which upon coordination with Al3+ ions showed remarkable increase in fluorescence emission. This increase in fluorescence intensity was owing to the binding of Al3+ ions with the 32 which promoted the ICT process via increasing the electron withdrawing ability of the coordinating carbonyl oxygen atom of the coumarin moiety and nitrate group (Fig. 13).
As we can see in the above examples that population of the ICT excited state can be altered by displacement of electron density of donor–acceptor system with specific metal ions, thus, ICT is an efficient approach to develop sensitive fluorescent chemosensors for detection of various analytes.
4. Resonance energy transfer and rhodamine ring-opening reaction based sensors
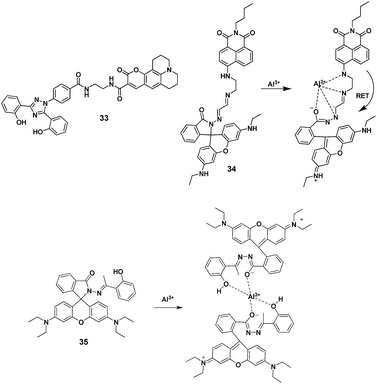
Resonance energy transfer (RET) is a photophysical event in which a chemical moiety in its excited state transfers its excitation energy to a closely located another chemical moiety in a non-radiative way which then releases energy in a radiative fashion (Fig. 14).54A pair of molecules that interact in such a manner so that RET occurs is designated as energy donor–acceptor pair. Generally, the energy transfer depends upon the donor to acceptor distance and extent of spectral overlap between emission and absorption spectra of donor and acceptor components, respectively. In literature, a number of systems based on RET have been developed for sensing and are employed to explore biological mechanisms.55–59 For instance, Arduini et al. designed a RET based chemosensor 33 with combination of coumarin (acceptor) and bis(o-hydroxyphenyl) having triazole unit (donor) for the detection of Al3+ ions (Fig. 15).60 Solution of chemosensor 33 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v; pH 5) showed emission enhancement of coumarin moiety at 489 nm (excitation at 350 nm) in the presence of Al3+ ions. On other hand, no change in fluorescence of 33 was observed upon addition of Al3+ ions when 33 is excited at 445 nm (typical excitation wavelength for the Coumarin 343 nm dye). These results showed that the RET from the donor bis(o-hydroxyphenyl) unit to the acceptor coumarin upon coordination with Al3+ ions is responsible for fluorescence enhancement of coumarin. In addition, 33 exhibited selectivity for Al3+ in the presence of other competitive ions such as Mg2+, Ca2+, Ni2+ and Zn2+ ions. However, there are interferences from Fe3+ and Cu2+ ions for the detection of Al3+ ions.
1; v/v; pH 5) showed emission enhancement of coumarin moiety at 489 nm (excitation at 350 nm) in the presence of Al3+ ions. On other hand, no change in fluorescence of 33 was observed upon addition of Al3+ ions when 33 is excited at 445 nm (typical excitation wavelength for the Coumarin 343 nm dye). These results showed that the RET from the donor bis(o-hydroxyphenyl) unit to the acceptor coumarin upon coordination with Al3+ ions is responsible for fluorescence enhancement of coumarin. In addition, 33 exhibited selectivity for Al3+ in the presence of other competitive ions such as Mg2+, Ca2+, Ni2+ and Zn2+ ions. However, there are interferences from Fe3+ and Cu2+ ions for the detection of Al3+ ions.
Further, rhodamine fluorophore has gained great research interest from the chemist's points of view due to its excellent photophysical properties.62 In general, rhodamine fluorophores are non-fluorescent and colourless because of its ring-closed structure. However, when rhodamine exists in its ring-opened form, it shows strong fluorescence and pink color. In line of this, Qin et al. designed and synthesized a rhodamine–naphthalene conjugate 34 for the ratiometric fluorescent detection of Al3+ ions.61 In chemosensor 34 rhodamine moiety acts as an energy acceptor while naphthalene unit acts as an energy donor.
Chemosensor 34 in EtOH exhibited fluorescence band at 520 nm attributed to the donor naphthalene unit. While characteristic emission of rhodamine at 550 nm was not observed, indicating the ring closed isomeric form. However, addition of Al3+ ions resulted in appearance of a new emission band at 550 nm, which corresponds to the ring-opened rhodamine moiety. This was ascribed to the RET from donor naphthalene to the rhodamine upon complexation with Al3+ ions as it generates ring-opened rhodamine, which acted as energy acceptor. Further, response of 34 was selective towards Al3+ ions, which was confirmed by competitive metal-ions selectivity experiments with detection limit of 8.06 × 10−8 M. Zuo and co-workers reported a rhodamine based chemosensor 35 for the recognition of Al3+ ions.63 In UV-vis studies, solution of 35 in EtOH–HEPES (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v; pH 7.2) in the presence of Cu2+, Fe3+, Ni2+ and Al3+ ions showed a absorption band at 560 nm attributed to the ring-opened form of the rhodamine upon complexation of these metal ions. However, in the fluorescence studies, the addition of only Al3+ ions resulted in an emission enhancement at 578 nm. The binding mode was found to 2
1; v/v; pH 7.2) in the presence of Cu2+, Fe3+, Ni2+ and Al3+ ions showed a absorption band at 560 nm attributed to the ring-opened form of the rhodamine upon complexation of these metal ions. However, in the fluorescence studies, the addition of only Al3+ ions resulted in an emission enhancement at 578 nm. The binding mode was found to 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for the ligand–Al3+ complexation. Moreover, the system works in a wide pH range (4.9–8.5) with a detection limit of 1.63 × 10−7 M for the recognition of Al3+ ions. Based on same strategy, Chatterjee et al. synthesized a rhodamine based chemosensor 36 for Al3+ ions.64 Chemosensor 36 in EtOH
1 for the ligand–Al3+ complexation. Moreover, the system works in a wide pH range (4.9–8.5) with a detection limit of 1.63 × 10−7 M for the recognition of Al3+ ions. Based on same strategy, Chatterjee et al. synthesized a rhodamine based chemosensor 36 for Al3+ ions.64 Chemosensor 36 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (2
H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3; v/v) mixture showed negligible absorption which in the presence of Al3+ ions showed the appearance of a new absorption band at 565 nm, attributed to the ring-opened form of rhodamine unit.
3; v/v) mixture showed negligible absorption which in the presence of Al3+ ions showed the appearance of a new absorption band at 565 nm, attributed to the ring-opened form of rhodamine unit.
On the other hand, the emission spectrum of 36 was characterized by a weak fluorescence, as the rhodamine unit is in spirolactam form. However, in the presence of Al3+ ions a significant fluorescence enhancement at 580 nm was observed (λex = 520 nm; Fig. 16b1). A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry between 36 and Al3+ was confirmed by Job's plot data while the association constant was 6.5 × 103 M−1. The biological application of the 36 was also studied by the authors. The chemosensor 36 was observed to exhibit no emission in the cellular system (Fig. 16c1). In contrast to this, 36 showed red fluorescence in the presence of Al3+ ions in the cellular system (Fig. 16c1), thus, an additional tool for detection of cellular Al3+ ions.
1 stoichiometry between 36 and Al3+ was confirmed by Job's plot data while the association constant was 6.5 × 103 M−1. The biological application of the 36 was also studied by the authors. The chemosensor 36 was observed to exhibit no emission in the cellular system (Fig. 16c1). In contrast to this, 36 showed red fluorescence in the presence of Al3+ ions in the cellular system (Fig. 16c1), thus, an additional tool for detection of cellular Al3+ ions.
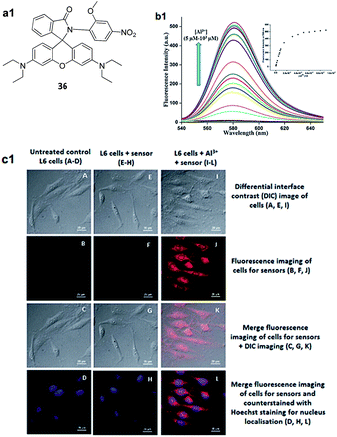 | ||
| Fig. 16 (a1) Structure of chemosensor 36; (b1) fluorescence spectra of 36 with Al3+ ions; (c1) confocal imaging of control and treatment sequences of L6 cells for 36. Reproduced from ref. 64 with permission from The Royal Society of Chemistry, copyright 2016. | ||
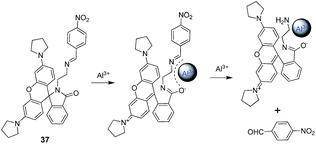
Ding et al. reported a rhodamine based chemosensor 37 for the fluorescence turn-on detection of Al3+ ions.65 The reported sensor in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (95
H2O (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5; v/v) mixture undergo significant emission enhancement at 582 nm upon complexation with Al3+ followed by the subsequent Al3+-promoted hydrolysis of the imino unit as displayed in the Fig. 17. The mechanism was further supported by IR, 1H NMR, and reversibility experiments.
5; v/v) mixture undergo significant emission enhancement at 582 nm upon complexation with Al3+ followed by the subsequent Al3+-promoted hydrolysis of the imino unit as displayed in the Fig. 17. The mechanism was further supported by IR, 1H NMR, and reversibility experiments.
In another example, the combination of rhodamine and naphthalic anhydride units (38) has been employed for the detection of metal ions including Al3+, Cr3+ and Fe3+ ions.66 The absorption and fluorescence response of 38 towards Al3+ is presented in Fig. 18. The absorption spectrum of 38 in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (6
H2O (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; v/v) showed the emergence of a new absorption band at 558 nm on the addition of Al3+ ions (Fig. 18a). In the fluorescence studies, free probe 38 showed an emission band at 375 related to naphthalic unit. The addition of Al3+ ions (1 equiv.) resulted in the appearance of a new emission band at 582 nm related to the rhodamine unit (λex = 330 nm; Fig. 18b). However, the excess addition of Al3+ did not affect the fluorescence intensity at 582 nm. On the other hand, the addition of metal ions such as Cr3+ or Fe3+ ions (1 equiv.) also introduced fluorescence enhancement at 582 nm. However, the fluorescence enhancement with Cr3+ or Fe3+ ions was less as compared to the Al3+ ions which revealed strong complexation of 38 with Al3+. The binding mode of 38 with Al3+ is presented in Fig. 18 while the binding constant for this complexation was 1 × 104 M−1 higher than the binding constant of 38 with Cr3+/Fe3+ (2.6 × 102 M−1/1.2 × 102 M−1). The detection limit of 38 to recognize Al3+ ions was 1.74 nM.
4; v/v) showed the emergence of a new absorption band at 558 nm on the addition of Al3+ ions (Fig. 18a). In the fluorescence studies, free probe 38 showed an emission band at 375 related to naphthalic unit. The addition of Al3+ ions (1 equiv.) resulted in the appearance of a new emission band at 582 nm related to the rhodamine unit (λex = 330 nm; Fig. 18b). However, the excess addition of Al3+ did not affect the fluorescence intensity at 582 nm. On the other hand, the addition of metal ions such as Cr3+ or Fe3+ ions (1 equiv.) also introduced fluorescence enhancement at 582 nm. However, the fluorescence enhancement with Cr3+ or Fe3+ ions was less as compared to the Al3+ ions which revealed strong complexation of 38 with Al3+. The binding mode of 38 with Al3+ is presented in Fig. 18 while the binding constant for this complexation was 1 × 104 M−1 higher than the binding constant of 38 with Cr3+/Fe3+ (2.6 × 102 M−1/1.2 × 102 M−1). The detection limit of 38 to recognize Al3+ ions was 1.74 nM.
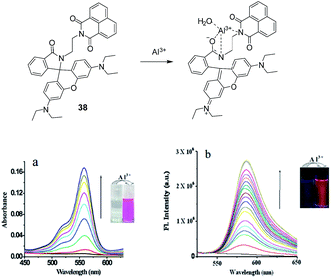 | ||
| Fig. 18 Binding mode in chemosensor 38; (a) UV-vis; (b) fluorescence spectra of 38 in the presence of presence of Al3+ ions. Reproduced from ref. 66 with permission from The Royal Society of Chemistry, copyright 2015. | ||
Based on the similar phenomenon, Bao et al. developed a rhodamine based chemosensor 39 for the detection of Al3+ via color as well as fluorescence changes (Fig. 19).67 Chemosensor 39 showed a weak fluorescence band at 580 nm upon excitation at 560 nm in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) mixture which in the presence of Al3+ exhibited strong emission enhancement at 582 nm. Recently, Gupta et al.68 reported a new rhodamine based chemosensor 40 for Al3+ ions with high selectivity and sensitivity. In UV-vis spectrum, solution of 40 in MeOH
1; v/v) mixture which in the presence of Al3+ exhibited strong emission enhancement at 582 nm. Recently, Gupta et al.68 reported a new rhodamine based chemosensor 40 for Al3+ ions with high selectivity and sensitivity. In UV-vis spectrum, solution of 40 in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (99
DMSO (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) showed two absorption bands at 273 and 322 nm. However the addition of Al3+ ions to 40 resulted in a new absorption band at 555 nm. Upon excitation at 520 nm, derivative 40 did not show any fluorescence emission while the presence of Al3+ ions showed a strong fluorescence at 587 nm, indicating the formation of rhodamine ring opened 40·Al3+ complex.
1; v/v) showed two absorption bands at 273 and 322 nm. However the addition of Al3+ ions to 40 resulted in a new absorption band at 555 nm. Upon excitation at 520 nm, derivative 40 did not show any fluorescence emission while the presence of Al3+ ions showed a strong fluorescence at 587 nm, indicating the formation of rhodamine ring opened 40·Al3+ complex.
Mass analysis and Job's plot experiment confirmed the formation of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
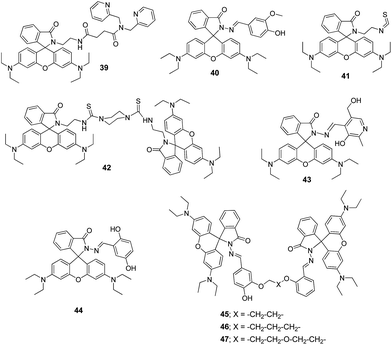
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex. In addition, authors have also developed paper test strips of 40 for the rapid on-site detection of Al3+ ions providing an easy and efficient method for the detection of Al3+ ions. Similarly, the rhodamine ring-opening strategy has also employed in examples 41–47 for the fluorescence turn-on detection of Al3+ ions.69–72 In view of this, alteration in rhodamine fluorophore and derivatives engineering will attract many organic, analytical and biochemists for sensing of various metal ions, anions and biological targets.
1 complex. In addition, authors have also developed paper test strips of 40 for the rapid on-site detection of Al3+ ions providing an easy and efficient method for the detection of Al3+ ions. Similarly, the rhodamine ring-opening strategy has also employed in examples 41–47 for the fluorescence turn-on detection of Al3+ ions.69–72 In view of this, alteration in rhodamine fluorophore and derivatives engineering will attract many organic, analytical and biochemists for sensing of various metal ions, anions and biological targets.
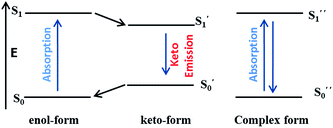
5. C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/h3_char_e001.gif) N isomerization based fluorescent sensors
N isomerization based fluorescent sensors
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization mechanism originated from the study on photophysical properties of conformational restricted compounds. It was observed that C
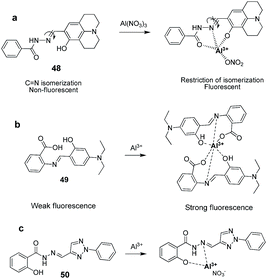
N isomerization mechanism originated from the study on photophysical properties of conformational restricted compounds. It was observed that C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization is a non-radiative decay process of excited states in compounds, hence, non-fluorescent.73–75 This non-radiative process can be inhibited by the complexation with metal cations and thus, offers a way to design fluorescent-sensing molecule (Fig. 20). Based on this hypothesis, a variety of sensors have been reported by various research groups. For instance, Kim and co-workers reported a chemosensor 48 having julolidine chromophore and activated amide functionality for the detection of Al3+ and CN− ions in a mixed aqueous solution.76 The fluorescence spectrum of 48 in a mixture of H2O
N isomerization is a non-radiative decay process of excited states in compounds, hence, non-fluorescent.73–75 This non-radiative process can be inhibited by the complexation with metal cations and thus, offers a way to design fluorescent-sensing molecule (Fig. 20). Based on this hypothesis, a variety of sensors have been reported by various research groups. For instance, Kim and co-workers reported a chemosensor 48 having julolidine chromophore and activated amide functionality for the detection of Al3+ and CN− ions in a mixed aqueous solution.76 The fluorescence spectrum of 48 in a mixture of H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (7
MeOH (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3; v/v) showed a weak fluorescence emission (λex = 410 nm) attributed to the C
3; v/v) showed a weak fluorescence emission (λex = 410 nm) attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process resulting in the emission quenching. However, the additions of Al3+ to the solution of 48 induce a strong fluorescence enhancement at 485 nm. This emission enhancement was attribute to the restriction of the C
N isomerization process resulting in the emission quenching. However, the additions of Al3+ to the solution of 48 induce a strong fluorescence enhancement at 485 nm. This emission enhancement was attribute to the restriction of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and excited state proton transfer mechanisms upon complexation of Al3+ with receptor 48 (Fig. 21a). Chemosensor 48 exhibited good selectivity towards Al3+ ions over the other metal ions with a detection limit of 0.193 μM for these ions. The same research group further reported a chemosensor 49 having imino unit for the detection of Al3+ ions via fluorescence turn-on response.77 Chemosensor 49 in MeOH
N isomerization and excited state proton transfer mechanisms upon complexation of Al3+ with receptor 48 (Fig. 21a). Chemosensor 48 exhibited good selectivity towards Al3+ ions over the other metal ions with a detection limit of 0.193 μM for these ions. The same research group further reported a chemosensor 49 having imino unit for the detection of Al3+ ions via fluorescence turn-on response.77 Chemosensor 49 in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v; pH 7) exhibited weak fluorescence emission at 460 nm when excited at 410 nm, due to isomerization of the C
1; v/v; pH 7) exhibited weak fluorescence emission at 460 nm when excited at 410 nm, due to isomerization of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond in the excited state. The addition of Al3+ ions resulted in the significance emission enhancement at 460 nm. This fluorescence response was attributed to the interaction of Al3+ with sensor 49 as displayed in the Fig. 21b, resulting in the restriction of C
N double bond in the excited state. The addition of Al3+ ions resulted in the significance emission enhancement at 460 nm. This fluorescence response was attributed to the interaction of Al3+ with sensor 49 as displayed in the Fig. 21b, resulting in the restriction of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization as well as initiation of chelation enhanced fluorescence. The binding constant for the complexation of 49 with Al3+ was 3.1 × 108 M−1 while detection limit was 290 nM.
N isomerization as well as initiation of chelation enhanced fluorescence. The binding constant for the complexation of 49 with Al3+ was 3.1 × 108 M−1 while detection limit was 290 nM.
Additionally, chemosensor 49 works well for the detection of Al3+ ions in a wide pH range (5–10). Ma et al. reported a acylhydrazone-based chemosensor 50 which exhibited selectivity for Al3+ ions over the other metal ions including Cu2+ and Fe3+ ions.78 Sensor 50 showed fluorescence enhancement at 442 nm with Al3+ ions in ethanol attributed to the interaction of Al3+ via imino nitrogen and oxygen of hydroxyl group (Fig. 21c). The stoichiometry of the complex was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 while the detection limit of 50 for Al3+ ions was 1.2 × 10−8 M. Liang et al. reported a salicylimine based chemosensor 51 where the combination of imino unit as well as phenolic oxygen atoms is employed for the complexation of Al3+ ions.79 The fluorescence spectrum of 51 was characterized by quenched emission in HEPES buffer containing 1% EtOH (pH = 7.04). This was attributed to the C
1 while the detection limit of 50 for Al3+ ions was 1.2 × 10−8 M. Liang et al. reported a salicylimine based chemosensor 51 where the combination of imino unit as well as phenolic oxygen atoms is employed for the complexation of Al3+ ions.79 The fluorescence spectrum of 51 was characterized by quenched emission in HEPES buffer containing 1% EtOH (pH = 7.04). This was attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization, which facilitate the non-radiative decay of the excited states. However, among various ions tested, the complexation of Al3+ ions with 51 resulted in a significant emission enhancement at 497 nm, ascribed to the restriction of C
N isomerization, which facilitate the non-radiative decay of the excited states. However, among various ions tested, the complexation of Al3+ ions with 51 resulted in a significant emission enhancement at 497 nm, ascribed to the restriction of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process (Fig. 22(1a)). For the coordination of Al3+ ions, two hydroxyl groups as well as imino nitrogen were involved. A 1
N isomerization process (Fig. 22(1a)). For the coordination of Al3+ ions, two hydroxyl groups as well as imino nitrogen were involved. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry was observed between 35 and Al3+ ions. The detection limit of 51 was 2.94 × 10−8 M for Al3+ ions. Additionally, 51–Al3+ complex was used for the detection of pyrophosphate (PPi) based on the fluorescence turn-off response. The biological applications of 51 and 51–Al3+ complex were also reported to detect Al3+ and PPi ions in the cellular system, respectively, by confocal imaging experiments (Fig. 22(1b)).
1 stoichiometry was observed between 35 and Al3+ ions. The detection limit of 51 was 2.94 × 10−8 M for Al3+ ions. Additionally, 51–Al3+ complex was used for the detection of pyrophosphate (PPi) based on the fluorescence turn-off response. The biological applications of 51 and 51–Al3+ complex were also reported to detect Al3+ and PPi ions in the cellular system, respectively, by confocal imaging experiments (Fig. 22(1b)).
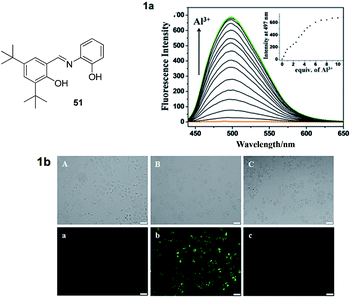 | ||
| Fig. 22 Structure of chemosensor 51; (1a) fluorescence spectra of 51 with Al3+ ions; (1b) brightfield (A–C) and fluorescence (a–c) images of HeLa cells: (A, a) cells treated with 51; (B, b) cells treated with 51 + Al3+; (C, c) cells treated with 51 + Al3+ + PPi. Reproduced from ref. 79 with permission from The Royal Society of Chemistry, copyright 2015. | ||
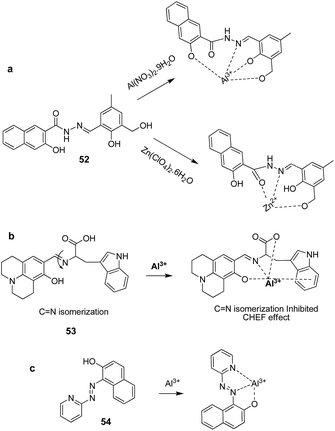
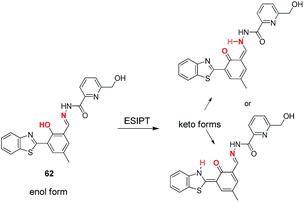
Chemosensor 52 was reported by Ali and co-workers based on the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT mechanisms.80 The fluorescence spectra of 52 showed quenched emission at 543 nm in THF
N isomerization and ESIPT mechanisms.80 The fluorescence spectra of 52 showed quenched emission at 543 nm in THF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (6
H2O (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; v/v) mixture ascribed to the both C
4; v/v) mixture ascribed to the both C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT processes. However, the addition of Al3+ and Zn2+ resulted in the fluorescence enhancement with blue shift to 486 and 498 nm, respectively. This was due to the inhibition of both C
N isomerization and ESIPT processes. However, the addition of Al3+ and Zn2+ resulted in the fluorescence enhancement with blue shift to 486 and 498 nm, respectively. This was due to the inhibition of both C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT processes upon complexation with Al3+ and Zn2+ ions. The binding mode of these ions with 52 is presented in Fig. 23. The two phenolate oxygen atoms, one imino nitrogen and hydroxymethyl group of 52 were participated to complex Al3+. However, in case of the Zn2+ ions, only hydroxymethyl group, one imine N atom and amido O atom were involved in binding. No phenolate O atoms were involved in binding with Zn2+ ions. The detection limits of 52 for Al3+ and Zn2+ ions were 0.92 and 3.1 nM, respectively. Kim and co-workers reported a chemosensor 53 by utilizing the combination of julolidine and tryptophan units for the detection of Al3+ in aqueous medium.81 The reported sensor exhibited quenched fluorescence due to the C
N isomerization and ESIPT processes upon complexation with Al3+ and Zn2+ ions. The binding mode of these ions with 52 is presented in Fig. 23. The two phenolate oxygen atoms, one imino nitrogen and hydroxymethyl group of 52 were participated to complex Al3+. However, in case of the Zn2+ ions, only hydroxymethyl group, one imine N atom and amido O atom were involved in binding. No phenolate O atoms were involved in binding with Zn2+ ions. The detection limits of 52 for Al3+ and Zn2+ ions were 0.92 and 3.1 nM, respectively. Kim and co-workers reported a chemosensor 53 by utilizing the combination of julolidine and tryptophan units for the detection of Al3+ in aqueous medium.81 The reported sensor exhibited quenched fluorescence due to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization. However, upon complexation with Al3+, there is emission enhancement at 432 nm ascribed to the inhibition of C
N isomerization. However, upon complexation with Al3+, there is emission enhancement at 432 nm ascribed to the inhibition of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process. The binding mode of 53 involved the participation of imine N, phenolic O, carboxylate O and indole moiety as displayed in Fig. 23b. Chemosensor 53 showed good selectivity along with detection limit of 6.4 μM for Al3+ ions. In another example, C
N isomerization process. The binding mode of 53 involved the participation of imine N, phenolic O, carboxylate O and indole moiety as displayed in Fig. 23b. Chemosensor 53 showed good selectivity along with detection limit of 6.4 μM for Al3+ ions. In another example, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization based chemosensor 5482 has been also employed for the sensitive fluorescence turn-on detection (detection limit: 1.81 × 10−8 M) of Al3+ ions. The binding mode for the complexation of Al3+ ions involves the interaction of Al3+ ions with hydroxyl group, nitrogen atoms of the azo and pyridine units as shown in Fig. 23.
N isomerization based chemosensor 5482 has been also employed for the sensitive fluorescence turn-on detection (detection limit: 1.81 × 10−8 M) of Al3+ ions. The binding mode for the complexation of Al3+ ions involves the interaction of Al3+ ions with hydroxyl group, nitrogen atoms of the azo and pyridine units as shown in Fig. 23.
Liu et al. synthesized a colorimetric and fluorescent chemosensor 55 for Al3+ ions in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) solution.83 In the UV-vis studies, addition of Al3+ ions to the solution of 55 (50 μM) showed the appearance of a new absorption band at 402 nm. The changes in absorption were observed with significant color change from colourless to blue which is visible by naked-eye. On the other hand, upon excitation at 402 nm, the fluorescence spectrum of 55 (50 μM) showed weak emission, indicating the role of C
1; v/v) solution.83 In the UV-vis studies, addition of Al3+ ions to the solution of 55 (50 μM) showed the appearance of a new absorption band at 402 nm. The changes in absorption were observed with significant color change from colourless to blue which is visible by naked-eye. On the other hand, upon excitation at 402 nm, the fluorescence spectrum of 55 (50 μM) showed weak emission, indicating the role of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization which favours the non-radiative decay process in the excited states. This fact is supported by the lifetime study of 55 which showed that the non-radiative decay process is the predominant process in the excited states of 55. However, the addition of Al3+ ions (50 μM) to 55 showed emission enhancement at 468 nm. This increase in emission intensity was due to the inhibition of C
N isomerization which favours the non-radiative decay process in the excited states. This fact is supported by the lifetime study of 55 which showed that the non-radiative decay process is the predominant process in the excited states of 55. However, the addition of Al3+ ions (50 μM) to 55 showed emission enhancement at 468 nm. This increase in emission intensity was due to the inhibition of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process upon binding of Al3+ ions. The formation of 55–Al3+ complex was confirmed by mass analysis which also indicated the 1
N isomerization process upon binding of Al3+ ions. The formation of 55–Al3+ complex was confirmed by mass analysis which also indicated the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric complex. Regarding the coordination mode, the 1H NMR study showed that Al3+ binds to 55 via phenolic OH, carbonyl group and the imine unit.
1 stoichiometric complex. Regarding the coordination mode, the 1H NMR study showed that Al3+ binds to 55 via phenolic OH, carbonyl group and the imine unit.
Chen et al. reported a reversible fluorescence turn-on sensor 56 for Al3+ ions84 which functioned through inhibition of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process. Addition of Al3+ ions to the solution of 56 in EtOH
N isomerization process. Addition of Al3+ ions to the solution of 56 in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (95
H2O (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5; v/v) resulted in a highly emissive complex 56
5; v/v) resulted in a highly emissive complex 56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al3+, which showed blue shift in emission from 510 to 480 nm. Upon binding with Al3+ ions, compound 56 forms a rigid system which restricted the C
Al3+, which showed blue shift in emission from 510 to 480 nm. Upon binding with Al3+ ions, compound 56 forms a rigid system which restricted the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process, hence, increment in fluorescence emission was observed.
N isomerization process, hence, increment in fluorescence emission was observed.
Similarly, the inhibition of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process has also employed in examples 57–61 for the fluorescence turn-on detection of Al3+ ions in aqueous medium or aqueous mixture (Fig. 24).85–88
N isomerization process has also employed in examples 57–61 for the fluorescence turn-on detection of Al3+ ions in aqueous medium or aqueous mixture (Fig. 24).85–88
The overhead discussed examples confirmed that the restriction in conformation of organic molecules is an important and efficient method for controlling the fluorescence of a molecule. Thus, this method can be used to detect various analytes with high selectivity and sensitivity in particular environments. In addition, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization strategy can also be used for C
N isomerization strategy can also be used for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) P isomerization and C
P isomerization and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C isomerization, thus, providing a new interesting prospect for further research.
C isomerization, thus, providing a new interesting prospect for further research.
6. ESIPT-based fluorescent sensors
ESIPT is a phenomenon that occurs in fluorophores having the possibility of proton transfer between two sites of the molecule, typically keto–enol tautomers. In such systems, tautomer in ground state is different from the one in the excited state. In the ground state, enol-form is stabilized by intramolecular hydrogen bonding. Upon excitation, the excited enol-form is quickly converted into the excited keto-form by transfer of a proton, which gives rise to an emission band at higher wavelength than that expected in the absence of the process. After this the keto-form reverts to the original enol-form via reverse proton transfer and decaying to the ground state. However, if ESIPT process is inhibited by guest which restricts the proton transfer, then emission band appears at shorter wavelengths than that appears in ESIPT process (Fig. 25).Various sensors have been reported for the detection of Al3+ ions based on ESIPT phenomenon. To extend ESIPT based sensors, Das et al. reported a fluorescence ratiometric sensor 62 based on the hydroxybenzothiazole (HBT) unit for the detection of Al3+ ions.13 The free 62 probe in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9; v/v) showed two broad emission bands at 435 and 565 nm, attributed to the ESIPT through keto–enol tautomerism as displayed in Fig. 26. However, in the presence of Al3+ ions, there is a ratiometric fluorescence change with the emergence of a new emission band at 480 nm. This ratiometric change was ascribed to the inhibition of the ESIPT process upon complexation of Al3+ followed by the chelation enhanced fluorescence effect. The presence of other metal ions including transition and alkali ions did not introduce this type of fluorescence change. The stoichiometry of the 62–Al3+ complex was 1
9; v/v) showed two broad emission bands at 435 and 565 nm, attributed to the ESIPT through keto–enol tautomerism as displayed in Fig. 26. However, in the presence of Al3+ ions, there is a ratiometric fluorescence change with the emergence of a new emission band at 480 nm. This ratiometric change was ascribed to the inhibition of the ESIPT process upon complexation of Al3+ followed by the chelation enhanced fluorescence effect. The presence of other metal ions including transition and alkali ions did not introduce this type of fluorescence change. The stoichiometry of the 62–Al3+ complex was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with binding constant of 1.24 × 105 M−1. The detection limit of 62 for Al3+ ions was 6.72 × 10−8 M.
1 with binding constant of 1.24 × 105 M−1. The detection limit of 62 for Al3+ ions was 6.72 × 10−8 M.
Sukwattanasinitt and co-workers reported a series of hydrazide based chemosensors 63–65 for the fluorescence turn-on detection of Al3+ ions.89 Chemosensors 63–65 in aqueous media exhibited weak emission attributed to the ESIPT and PET from amino nitrogen atom. Upon addition of Al3+ ions to the aqueous solutions of 63–65, emission enhancements were observed at 458, 524 and 601 nm, respectively (Fig. 27b). We can clearly see the large Stokes shifts while moving from 63–Al3+ complex to 65–Al3+ complex ascribed to the increased π-conjugation as well as of an additional hydroxyl substituent. The emission enhancement in all cases was attributed to the formation of ligand–Al3+ complex which restricts the ESIPT and PET mechanisms, thus, responsible for the chelation enhanced fluorescence. The addition of other metal ions was not able to introduce fluorescence response as observed in the case of Al3+ ions. Chemosensor 63 and 64 formed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexes with Al3+ ions, while 65 form 2
1 complexes with Al3+ ions, while 65 form 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 metal–ligand complex. The binding constants for the complexation of 63–65 for the Al3+ were 1.6 × 105, 5.4 × 105 and 2.0 × 1010 M−1, respectively. In addition, chemosensor 65 was further employed to develop paper based sensor for the fluorescence detection of Al3+ and Zn2+ ions as showed in Fig. 27c. The paper strips containing 65 on addition of Al3+ and Zn2+ ions resulted in the yellow and orange coloured emission.
1 metal–ligand complex. The binding constants for the complexation of 63–65 for the Al3+ were 1.6 × 105, 5.4 × 105 and 2.0 × 1010 M−1, respectively. In addition, chemosensor 65 was further employed to develop paper based sensor for the fluorescence detection of Al3+ and Zn2+ ions as showed in Fig. 27c. The paper strips containing 65 on addition of Al3+ and Zn2+ ions resulted in the yellow and orange coloured emission.
 | ||
| Fig. 27 (a) Structure of chemosensors 63–65; (b) fluorescence spectra of 63–65 in the presence of metal ions including Na+, K+, Ag+, Mg2+, Ca2+, Hg2+, Ba2+, Pb2+, Cd2+, Mn2+, Ni2+, Co2+, Cu2+, Fe2+, Zn2+, Cr3+, Fe3+, and Al3+ ions; (c) image of metal-ion detection by chemosensor 65 on filter paper (irradiated wavelength 365 ± 50 nm). Reprinted with permission from ref. 89. Copyright 2016 American Chemical Society. | ||
Alam et al. reported a chemosensor 66 incorporated with two imino units for the detection of Al3+ ions.90 Free 66 exhibited weak emission band at 520 nm (λex = 370 nm) in MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (8
H2O (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2; v/v; pH 7.2) ascribed to both C
2; v/v; pH 7.2) ascribed to both C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT process operating between phenolic OH and imino nitrogen. However, addition of Al3+ ions to 66 inhibits the ESIPT and C
N isomerization and ESIPT process operating between phenolic OH and imino nitrogen. However, addition of Al3+ ions to 66 inhibits the ESIPT and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization process and causes a blue-shift of 34 nm with fluorescence enhancement at 486 nm. Additionally, sensor 66 was also employed for the detection of pyrophosphate. A 1
N isomerization process and causes a blue-shift of 34 nm with fluorescence enhancement at 486 nm. Additionally, sensor 66 was also employed for the detection of pyrophosphate. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding mode between 66 and Al3+ was confirmed by theoretical calculations. The detection limit of 66 for Al3+ ions was 7.55 μM. Based on pyrene scaffold, Mahapatra et al. reported fluorescence chemosensors 67–69 for the detection of Al3+ ions with detection limits of 3.60, 2.13 and 2.16 μM, respectively.91 The interaction of Al3+ with chemosensor 67–69 showed significant fluorescence enhancement ascribed to the restriction of both ESIPT and C
1 binding mode between 66 and Al3+ was confirmed by theoretical calculations. The detection limit of 66 for Al3+ ions was 7.55 μM. Based on pyrene scaffold, Mahapatra et al. reported fluorescence chemosensors 67–69 for the detection of Al3+ ions with detection limits of 3.60, 2.13 and 2.16 μM, respectively.91 The interaction of Al3+ with chemosensor 67–69 showed significant fluorescence enhancement ascribed to the restriction of both ESIPT and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization processes. Based on similar strategy, Kim et al. reported a fluorescence turn-on chemosensor 70 having electron-donating tert-butyl group for the detection of Al3+ ions.92 The reported sensor 70 was weakly fluorescent due to the ESIPT mechanism and upon coordination with Al3+ ions, the C
N isomerization processes. Based on similar strategy, Kim et al. reported a fluorescence turn-on chemosensor 70 having electron-donating tert-butyl group for the detection of Al3+ ions.92 The reported sensor 70 was weakly fluorescent due to the ESIPT mechanism and upon coordination with Al3+ ions, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ESIPT processes were inhibited thus, a significant fluorescence increase is observed. The binding of 70 with Al3+ was further supported by 1H NMR experiments. In addition, sensor 70 was successfully employed for simultaneous detection and quantification of Al3+ ions in living cells.
N isomerization and ESIPT processes were inhibited thus, a significant fluorescence increase is observed. The binding of 70 with Al3+ was further supported by 1H NMR experiments. In addition, sensor 70 was successfully employed for simultaneous detection and quantification of Al3+ ions in living cells.
Mu and co-workers reported a styryl unit connected quinoline based chemosensor 71 for the fluorescent detection of Al3+ ions (Fig. 28).93 UV-vis spectra of chemosensor 71 exhibited absorption bands at 249, 298 and 360 nm in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (5
H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2; v/v; pH 7). However, the addition of Al3+ ions resulted in formation of new red shifted band at 269 nm, ascribed to the charge transfer from ligand to the Al3+ as well as interruption to the intramolecular H-bond formation. Further, emission spectrum of ligand 71 displayed weak fluorescence at 570 nm, owing to the electron transfer and ESIPT processes. The addition of Al3+ ions resulted in the blue shift to 545 nm with significant fluorescence enhancement. This was due to interaction of Al3+ ions to oxygen atoms and nitrogen atoms of the phenolic and quinoline moiety of 71, which suppressed the ESIPT phenomenon, hence, CHEF. The addition of other metal ions did not introduce this kind of fluorescence changes. The Job's plot experiment confirmed the 1
2; v/v; pH 7). However, the addition of Al3+ ions resulted in formation of new red shifted band at 269 nm, ascribed to the charge transfer from ligand to the Al3+ as well as interruption to the intramolecular H-bond formation. Further, emission spectrum of ligand 71 displayed weak fluorescence at 570 nm, owing to the electron transfer and ESIPT processes. The addition of Al3+ ions resulted in the blue shift to 545 nm with significant fluorescence enhancement. This was due to interaction of Al3+ ions to oxygen atoms and nitrogen atoms of the phenolic and quinoline moiety of 71, which suppressed the ESIPT phenomenon, hence, CHEF. The addition of other metal ions did not introduce this kind of fluorescence changes. The Job's plot experiment confirmed the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding mode for the complexation process while the binding constant was 1.52 × 104 M−1. The complexation mode of 71 with Al3+ ions was also established based on 1H NMR and IR studies which confirmed the participation of quinoline nitrogen and phenolic oxygen atoms. Similarly, chemosensor 72, which comprises of hydroxyquinoline and pyrazole units, exhibited high detection selectivity for Al3+ ions.94 Ligand 72 in HEPES buffer (pH 7.2) presented a weak emission band at 470 nm due to ESIPT process which takes place between –N
1 binding mode for the complexation process while the binding constant was 1.52 × 104 M−1. The complexation mode of 71 with Al3+ ions was also established based on 1H NMR and IR studies which confirmed the participation of quinoline nitrogen and phenolic oxygen atoms. Similarly, chemosensor 72, which comprises of hydroxyquinoline and pyrazole units, exhibited high detection selectivity for Al3+ ions.94 Ligand 72 in HEPES buffer (pH 7.2) presented a weak emission band at 470 nm due to ESIPT process which takes place between –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) and phenol units. Additionally, rotation across the C
and phenol units. Additionally, rotation across the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond also contributed to the observed fluorescence quenching. However, the fluorescence emission of the system at 470 nm was revived upon addition of Al3+ ions which was ascribed to the chelation enhanced fluorescence upon complexation of Al3+ through nitrogen and oxygen atoms of pyrazole, amide, and quinoline units. The 72–Al3+ complex demonstrated 1
N bond also contributed to the observed fluorescence quenching. However, the fluorescence emission of the system at 470 nm was revived upon addition of Al3+ ions which was ascribed to the chelation enhanced fluorescence upon complexation of Al3+ through nitrogen and oxygen atoms of pyrazole, amide, and quinoline units. The 72–Al3+ complex demonstrated 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry which was confirmed by Job's plot as well by mass analysis. Additionally, ligand 72 presented detection limit of 4.29 nM for Al3+ ions.
1 stoichiometry which was confirmed by Job's plot as well by mass analysis. Additionally, ligand 72 presented detection limit of 4.29 nM for Al3+ ions.
Thus, unique feature of ESIPT process like large Stokes shift which avoid interference from other fluorescent materials present in the sample makes it ideal candidate for the construction of fluorescence sensor. However, it has been observed that fluorophores which are required for constructing ESIPT molecules are still rare. In addition, the reported fluorophores have shorter emission wavelength generally in the blue and green regions. In view of this, there is great need to develop new ESIPT-based fluorophores which can offer shifts up to near IR region with their applications in the area of biology and energy.
7. AIE-based fluorescent sensors
In 2001 Tang et al. reported the term “aggregation-induced emission” (AIE) for molecules which behave non-luminescent in the solution state but become strongly emissive when aggregated.95 Attracted by this novel AIE characteristic and its applications, many research groups have utilized it for the development of fluorescence sensors for the sensitive and selective detection of analytes. Typically, AIE active sensors are propeller-shaped in which easily rotatable phenyl rings attached to central core through C–C bond with specific functional groups for analyte. In solution phase, rotation of these rotors are active by which the energy of approaching photon is dissipated with a non-radiative pathway thus, molecule is non-luminescent or less luminescent in solution state.However, upon addition of analyte, the interaction between AIE active molecules and guest molecules lead to formation of aggregates due to poor solubility and restriction in rotation of the rotors. The restriction in rotation of the rotors is because of close packing in aggregated state which blocks the non-radiative pathways, thus, resulting in release of excitation energy as a fluorescence of the molecule (Fig. 29). In view of this, new AIE mechanism provides a new platform for designing and synthesizing sensitive and selective chemosensors. In addition, the sensor formed by AIE strategy offers advantages such as high selectivity, sensitivity, low background interference as well as the simplicity of the fluorescence technique.96 Taking into account the applications of AIE, many research groups have developed fluorescent aggregates for sensing Al3+ ions. As tetraphenylethene (TPE) derivatives are known to show AIE phenomenon. Benefiting from such phenomenon, Gui et al. synthesized a fluorescence turn-on TPE based chemosensor 73 for the detection of Al3+ ions.97 Compound 73 was chemically functionalized by carboxylic group which was expected to complex Al3+ ions through oxygen donor sites of the carboxyl group. Compound 73 in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (2
H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98; v/v) was non-fluorescent due to the fast intramolecular rotations around the C–C bond which facilitate the non-radiative decay process. However, upon addition of Al3+ ions among the various metal ions tested, a strong fluorescence enhancement took place at 470 nm (Fig. 30a). This enhancement at 470 nm was due to the formation of aggregates upon addition of Al3+ ions which restrict the intramolecular rotations, thus, results in emergence of fluorescence. Further, TPE derivative without the carboxyl group was not able to interact with Al3+ ions and hence, no fluorescence change was observed upon addition of Al3+ ions, thus, indicates the role of carboxylic group. The formation of aggregates of 73–Al3+ complex was confirmed by TEM as well as Dynamic light scattering (DLS) studies. In addition, it was evaluated that the four molecules of compound 73 were participated to coordinate an Al3+ ion.
98; v/v) was non-fluorescent due to the fast intramolecular rotations around the C–C bond which facilitate the non-radiative decay process. However, upon addition of Al3+ ions among the various metal ions tested, a strong fluorescence enhancement took place at 470 nm (Fig. 30a). This enhancement at 470 nm was due to the formation of aggregates upon addition of Al3+ ions which restrict the intramolecular rotations, thus, results in emergence of fluorescence. Further, TPE derivative without the carboxyl group was not able to interact with Al3+ ions and hence, no fluorescence change was observed upon addition of Al3+ ions, thus, indicates the role of carboxylic group. The formation of aggregates of 73–Al3+ complex was confirmed by TEM as well as Dynamic light scattering (DLS) studies. In addition, it was evaluated that the four molecules of compound 73 were participated to coordinate an Al3+ ion.
 | ||
| Fig. 30 Structure of chemosensor 73; (a) fluorescence spectra of 73 in the presence of Al3+ ions; (b) confocal images of HeLa cells (1) without any treatment, (2) after sequentially incubation with Al3+ and 73; (3) with 73; (4) monitoring of the complexation process of 73 to Al3+-loaded cells. Reprinted with permission from ref. 97. Copyright 2015 American Chemical Society. | ||
The detection limit of 73 for Al3+ ions was 21.6 nM for Al3+ ions. The biological application of 73 was also evaluated to detect Al3+ in the cellular system. Fig. 30b displayed the images of the cells with 73 and 73–Al3+ complex. The blue coloured fluorescence in the case of cells incubated with 73 followed by Al3+ ions revealed applicability of 73 to detect Al3+ ions in the biological system.
Similarly, Samanta et al. reported a fluorescence turn-on sensor 74 based on AIE for the detection of Al3+ ions (Fig. 31).98 Compound 74 showed weak emission band at 501 nm in CH3OH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v) upon excitation at 390 nm. Addition of Al3+ ions resulted in a significant emission enhancement at 523 nm. Increase in fluorescence intensity was ascribed to the metal chelation which is responsible for the AIE behaviour. Köstereli et al.99 reported a BIDIPY based amphiphile system having disulfonated BODIPY unit as a head group while heptadecyl side chain acted as a tail. Concentration-dependent fluorescence studies showed that compound 75 with short alkyl chain did not exhibit aggregation behaviour in the concentration range between 1 μM and 1 mM while compound 76 with long alkyl chain showed the formation of micellar aggregates at ∼20 μM. Sensing studies of 76 was performed slightly below its critical micelle concentration. Among various metal salts tested, only Al3+ ions showed significant quenching in fluorescence emission of 76 at 505 nm. Very small effect on fluorescence emission of 76 was observed with other metal ions. This was ascribed to the analyte-induced aggregation which resulted in the quenching of emission. However, under identical condition 75 did not show any significant fluorescence quenching with Al3+ ions, thus, indicating the importance of aggregates of 76 in the sensing of Al3+ ions.
1; v/v) upon excitation at 390 nm. Addition of Al3+ ions resulted in a significant emission enhancement at 523 nm. Increase in fluorescence intensity was ascribed to the metal chelation which is responsible for the AIE behaviour. Köstereli et al.99 reported a BIDIPY based amphiphile system having disulfonated BODIPY unit as a head group while heptadecyl side chain acted as a tail. Concentration-dependent fluorescence studies showed that compound 75 with short alkyl chain did not exhibit aggregation behaviour in the concentration range between 1 μM and 1 mM while compound 76 with long alkyl chain showed the formation of micellar aggregates at ∼20 μM. Sensing studies of 76 was performed slightly below its critical micelle concentration. Among various metal salts tested, only Al3+ ions showed significant quenching in fluorescence emission of 76 at 505 nm. Very small effect on fluorescence emission of 76 was observed with other metal ions. This was ascribed to the analyte-induced aggregation which resulted in the quenching of emission. However, under identical condition 75 did not show any significant fluorescence quenching with Al3+ ions, thus, indicating the importance of aggregates of 76 in the sensing of Al3+ ions.
In another example, Dong and co-workers reported an AIE based sensor 77 by employing the combination of diphenylpyrrol with benzoate unit for the selective fluorescence turn-on detection of Al3+ ions.100 Sensor 77 (100 μM) in THF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (25
H2O (25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75; v/v) displayed quenched fluorescence due to the non-radiative decay process because of intramolecular rotations. However, the interaction of Al3+ ions (20 μM) with the benzoate unit resulted in the formation of aggregates responsible for the restricted rotations and emission enhancement at 460 nm. The increment in emission intensity was only upto 28 μM, indicating that each Al3+ cation can interact with three TriPP-COO− anions.
75; v/v) displayed quenched fluorescence due to the non-radiative decay process because of intramolecular rotations. However, the interaction of Al3+ ions (20 μM) with the benzoate unit resulted in the formation of aggregates responsible for the restricted rotations and emission enhancement at 460 nm. The increment in emission intensity was only upto 28 μM, indicating that each Al3+ cation can interact with three TriPP-COO− anions.
Misra et al. employed a pyrene appended imino-phenol unit based sensor 78 for the detection of Al3+ ions via AIE process.101 Free 78 in CH3CN showed a weak fluorescence emission at 426 nm (λex = 364 nm) due to the PET from imino nitrogen to the excited pyrene unit. However, the addition of Al3+ ions was responsible for the fluorescence enhancement at 426 nm. This emission enhancement was attributed to the interaction of Al3+ via imino and hydroxy units with ligand 78 which restricted the PET process causing fluorescence increase. Likewise, the excess addition of Al3+ which was answerable for the aggregate formation, also contributed to the observed fluorescence enhancement. The aggregation state formation was proved by DLS studies which showed the change of particle from 232 to 1753 nm for 78–Al3+ complex system. Additionally, the fluorescence response of 78 with Al3+ ions was stable in the presence of other metal ions. Moreover, the reported sensor exhibited detection limit of 8.6 nM for Al3+ ions and worked well across the wide pH range (2 to 11).
Leary et al. reported a water soluble tetrasulfonated calix[4]arene conjugated with dansyl fluorophores 79 for the AIE based detection of Al3+ ions.102
Under the acidic pH condition (i.e. pH = 6) in lutidine buffer ligand 79 exhibited an emission band at 576 nm ascribed to the high polarity as well as to the hydrogen bonding interactions operating between water and dansyl molecules. The addition of Al3+ ions resulted in the blue shift of 46 nm and appearance of a new emission band at 530 nm with significant fluorescence enhancement (Fig. 32a). These changes are attributed to the interaction of negatively charged ligand 79 with the positively charged Al(OH)3 colloidal particles at pH 6.0, which is responsible for the aggregate formation that reduces the polarity around the dansyl unit and hence, blue shift in the emission (Fig. 32b). The system displayed detection limit of 1.8 μM for Al3+ ions. Likewise, the competitive selectivity experiments revealed the selective nature of ligand 79 for the detection of Al3+ ions. Based on above discussion, we noted that, a variety of derivatives have been used for the detection of Al3+ ions based on AIE phenomenon
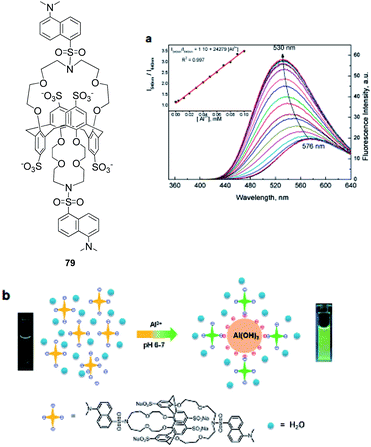 | ||
| Fig. 32 Structure of chemosensor 79; (a) fluorescence spectra of 79 with Al3+ in lutidine buffer solution (pH = 6.0); inset is the ratiometric response I540/I640 against [Al3+]; (b) schematic illustration of interaction between 79 and Al3+. Reproduced from ref. 102 with permission from The Royal Society of Chemistry, copyright 2014. | ||
This AIE-active probe offers the advantage of detection of analytes in aqueous media which make these sensors useful in biological systems. The “turn-on” feature of the AIE based sensors makes them capable for on-site and household testing etc. Further, developments of fluorescent probes to detect various analyte in aqueous media are very rare and research in this area is still in progress. Future work in this area will be the development of probes with incorporation of recognition group for high specificity, sensitivity, solubility in aqueous environment. Further, the probes that can emit in NIR region are of great concern which will provide the favourable condition for fluorescent imaging studies.
8. Miscellaneous examples
In addition to the above chemosensors, a few other interesting chemical systems have been employed for the optical detection of Al3+ ions. For instance, Çoşut and co-workers connected phenanthroline unit with the cyclotriphosphazene ring to develop chemosensor 80 which can detect Al3+ ions with fluorescence turn-on response (Fig. 33).103 In another example, Ma et al. reported a chemosensor 81 for Al3+ ions based on the combination of iridium(III) complex and receptor unit for Al3+ ion.104 Chemosensor 81 exhibited weak luminescence emission at 573 nm in CH3CN (λex = 350 nm) indicating large Stokes shift of near about 230 nm.The addition of Al3+ ions to 81 resulted in the luminescence enhancement. This emission enhancement was ascribed to the complexation of Al3+ with the phenolsalicylimine unit which generates a metal–ligand charge transfer state. Further, the ligand–metal binding constant was 1.14 × 104 M−1. Likewise, sensor 81 is selective towards Al3+ even in the presence of other metal ions and showed detection limit of 1 μM for Al3+ ions. In line of this, Sykes and co-workers reported a reaction based sensor 82 for the detection of Al3+ ions.105 Sensor 82 is non-emissive in DMSO/potassium phosphate buffer (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture. However, the addition of Al3+ results in the appearance of a blue coloured fluorescence which is attributed to the formation of anthracene facilitated via dehydration of secondary alcohols in 82. The formation of anthracene is confirmed by carrying out the 1H NMR analysis of 82 in the presence of Al3+ ions.
1) mixture. However, the addition of Al3+ results in the appearance of a blue coloured fluorescence which is attributed to the formation of anthracene facilitated via dehydration of secondary alcohols in 82. The formation of anthracene is confirmed by carrying out the 1H NMR analysis of 82 in the presence of Al3+ ions.
Wu and co-workers have employed commercially available 2-hydroxy-1-naphthaldehyde for the selective fluorescent turn-on detection of Al3+ ions.106 This compound in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (95
H2O (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5; v/v) mixture exhibited weak fluorescence emission at 435 nm (λex = 317 nm). The addition of Al3+ ions resulted in a significant fluorescence enhancement (12-fold) along with red shift to 445 nm. This fluorescence change was ascribed to the complexation of Al3+ via hydroxy and aldehyde units of 2-hydroxy-1-naphthaldehyde, making a rigid system responsible for the chelation-enhanced fluorescence. The fluorescence response in the presence of Al3+ is stable over a wide pH range (3–9). Based on the fluorescence titration with Al3+, the binding constant was calculated as 8.32 × 106 M−1, while the detection limit for Al3+ ions was 3.28 × 10−6 M. Recently, Mahata et al. employed a two-dimensional metal–organic framework (MOF) for the detection of Al3+ ions in the aqueous medium.107 The reported MOF system showed emission at 407 nm in water which upon addition of Al3+ ions exhibited blue shift of 24 nm with enhancement of luminescence intensity. On the other hand, the addition of metal ions such as Fe3+, Cr3+ and Fe2+ resulted in the quenching of luminescence at 407 nm. However, the MOF system still exhibited good selectivity for Al3+ ions even in the presence of other interfering metal ions and have detection limit of 57.5 ppb for Al3+ ions.
5; v/v) mixture exhibited weak fluorescence emission at 435 nm (λex = 317 nm). The addition of Al3+ ions resulted in a significant fluorescence enhancement (12-fold) along with red shift to 445 nm. This fluorescence change was ascribed to the complexation of Al3+ via hydroxy and aldehyde units of 2-hydroxy-1-naphthaldehyde, making a rigid system responsible for the chelation-enhanced fluorescence. The fluorescence response in the presence of Al3+ is stable over a wide pH range (3–9). Based on the fluorescence titration with Al3+, the binding constant was calculated as 8.32 × 106 M−1, while the detection limit for Al3+ ions was 3.28 × 10−6 M. Recently, Mahata et al. employed a two-dimensional metal–organic framework (MOF) for the detection of Al3+ ions in the aqueous medium.107 The reported MOF system showed emission at 407 nm in water which upon addition of Al3+ ions exhibited blue shift of 24 nm with enhancement of luminescence intensity. On the other hand, the addition of metal ions such as Fe3+, Cr3+ and Fe2+ resulted in the quenching of luminescence at 407 nm. However, the MOF system still exhibited good selectivity for Al3+ ions even in the presence of other interfering metal ions and have detection limit of 57.5 ppb for Al3+ ions.
In another example, Lee and co-workers used carbon-nanodots (C-Dots; energy donor) immobilized with rhodamine fluorophore as an energy acceptor for the detection of Al3+ ions based on fluorescence RET mechanism (Fig. 34).108 The probe showed fluorescence emission at 410 nm attributed to the C-dots when excited at 350 nm in EtOH. However, in the presence of Al3+ ions, the ring-opening reaction of rhodamine moiety takes place which enables the energy transfer from C-dots to the rhodamine unit. This energy transfer process results in the fluorescence emission corresponding to the rhodamine fluorophore at 560 nm. The system is selective for Al3+ ions with the detection limit of 3.8 × 10−5 M for these ions. Likewise, the practical application of the sensor was also demonstrated by employing paper strips containing the probe for the detection of Al3+ ions.
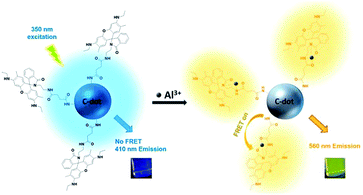 | ||
| Fig. 34 Illustration of FRET-based detection of Al3+ by employing C dots-rhodamine. Reprinted with permission from ref. 108. Copyright 2015 American Chemical Society. | ||
As a final remark, the research in the field of development of Al3+ sensor is fast developing in recent years. Here, in this review, we have discussed selective examples for discussions. In this regard, as compensation to loss, we are providing below a summarized table of recently published papers by different research teams for interested readers (Table 1).
| Chemosensor | Medium | λex/λem (nm) | Detection limit | Binding constant (M−1) | Mechanism | Ref. |
|---|---|---|---|---|---|---|
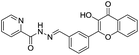 |
DMSO/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
350/453 | 21.7 × 10−6 M | — | ESIPT | 109 |
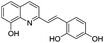 |
CH3CN/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
440/545 | 0.06 × 10−6 M | 5.62 × 104 | PET | 110 |
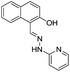 |
EtOH/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
336/454 | 36.6 × 10−9 M | 4.87 × 104 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization N isomerization |
111 |
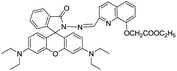 |
EtOH | 345/579 | — | 5.23 × 105 | Rhodamine ring-opening reaction | 112 |
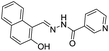 |
H2O | 409/474 | 4 × 10−6 M | 4 × 103 | PET | 113 |
 |
EtOH | 392/479 | 8.2 × 10−7 M | 3.3 × 106 | PET | 114 |
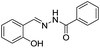 |
H2O | 366/452 | 5.2 × 10−9 M | 5.5 × 104 | PET | 115 |
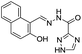 |
MeCN/H2O (1/1) | 360/470 | 9.82 × 10−6 M | 1.26 × 103 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and ICT N isomerization and ICT |
116 |
 |
EtOH | 425/490 | 99.5 × 10−9 M | 2.5 × 104 | PET and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization N isomerization |
117 |
 |
H2O | 389/506 | 6.9 × 10−7 M | 1.67 × 104 | ICT | 118 |
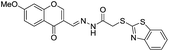 |
EtOH | 410/480 | 3.19 × 10−8 M | 1.21 × 104 | PET | 119 |
 |
H2O/MeOH (99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
400/465 | 1 × 10−5 M | 6.02 × 103 | PET | 120 |
 |
DMSO/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) 4) |
480/590 | 0.19 × 10−6 M | 4.78 × 104 | Rhodamine ring-opening reaction | 121 |
 |
CH3CN/H2O (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
560/584 | 59 × 10−9 M | 6.42 × 104 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization N isomerization |
122 |
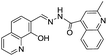 |
EtOH/H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
397/500 | 5.2 × 10−6 M | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = 9.98, n = 2.11 K = 9.98, n = 2.11 |
PET | 123 |
 |
EtOH/H2O (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
422/550 | 6 × 10−7 M | 7.5 × 103 | (PET) coupled with (ICT) | 124 |
 |
DMSO/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) 9) |
380/476 | 9 × 10−9 M | 2.8 × 103 | PET | 125 |
 |
CH3CN/H2O (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
557/568 | — | 2.03 × 103 | Rhodamine ring-opening reaction | 126 |
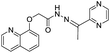 |
EtOH | 350/488 | 10 × 10−67 M | 1.35 × 104 | PET | 127 |
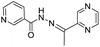 |
EtOH | 382/490 | 10 × 10−7 M | — | PET | 128 |
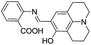 |
DMSO/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
365/478 | 24 × 10−9 M | 1.0 × 104 | ESIPT | 129 |
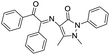 |
MeOH | 375/463 | 0.49 × 10−6 M | 4.58 × 104 | PET | 130 |
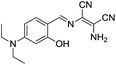 |
EtOH/H2O (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
480/499 or 535 | 5.13 × 10−9 M | 6.8 × 105 | PET, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization and activation of ESIPT and ICT N isomerization and activation of ESIPT and ICT |
131 |
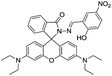 |
DMSO/H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
560/581 | 4.43 × 10−7 M | 2.76 × 104 | Rhodamine ring-opening reaction | 132 |
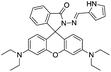 |
DMF/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) 8) |
535/557 | 3.20 × 10−6 M | — | Rhodamine ring-opening reaction | 133 |
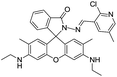 |
CH3CN | 528/553 | 4.28 × 10−9 M | 5.71 × 105 | Rhodamine ring-opening reaction | 134 |
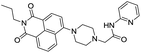 |
H2O/CH3CN (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
409/530 | 3.6 × 10−7 M | 3.5 × 104 | PET | 135 |
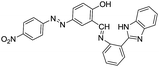 |
DMSO/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8) 8) |
320/474 | 3.41 × 10−7 M | 5.56 × 102 | ICT | 136 |
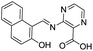 |
EtOH/H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
354/491 | 5.21 × 10−7 M | 2.08 × 106 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N isomerization N isomerization |
137 |
9. Conclusions
The above discussion has shown that how chemists from various backgrounds have constructed fluorescent signalling systems for sensing of Al3+ ions. Various sensing mechanisms (PET, ICT and AIE etc.), optical properties, and other miscellaneous applications have been explained and used for designing fluorescent chemosensors for the sensing of Al3+ ions. In addition, number of fluorophore like naphthalene, coumarin, cyanine, rhodamine, benzothiazole, pyrene etc. has been used as an ideal unit for the development of fluorescent chemosensors for Al3+ ions. Most of the discussed chemosensors fulfil the required WHO detection limit for Al3+ in drinking water (7.41 μM). Among them, few fluorescent sensors were successfully applied to detect Al3+ in the cellular system, thus, revealed applicability of chemosensors in the biological system. Further, restriction in conformation can lead to suppression of non-radiative decay which provides efficient method for controlling the fluorescence and can lead to increases in fluorescent quantum yield by two to three orders of magnitude. On the other hand, carbon nanodots having advantages over other fluorophores such as high solubility in water, low toxicity and good biocompatibility has been used for selective detection of Al3+ ion using FRET based ratiometric sensing mechanism, thus, provides another approach for the development of chemosensor.It can be noted from the discussion that the most of the examples work in the organic solvents like ethanol, acetonitrile, DMSO or in mixed aqueous systems which restrict their biological applicability. In addition, for the real-time applications, the sensor must present high selectivity for Al3+ ions, even in the presence of excess of other cations or other molecules. These are the crucial points and should be addressed to achieve the practical applications of a chemosensor. Moreover, there is need to develop devices for the on-site detection. It has been also observed that most of the reported sensors show either blue or green emission on interacting with Al3+ ions. This also limited their biological applications as chemosensors having red or near-infra red emissions are favoured in biology attributed to their low energy emission. Thus, future work in this field needs to develop sensor having high specificity, sensitivity with low interference from other species, fast reversible chelation, water solubility, fluorescence bio-imaging capacity and a feasible synthetic route. On the other hand, the development of new chemosensor that can emit at long wavelengths upon interacting with analyte should be the key points for further research in this area. Further, by linking quantum dots to the NIR fluorophore and using the FRET approach, ratiometric sensors which can emit at long wavelengths can be built up.
We hope that the information provided herein will encourage the scientific community to explore the novel ideas for structural modification in the design and synthesis of new sensors. Further, AIE mechanism discussed in this review is new emerging field and has wide scope in biological application. In addition, this review might educate the researchers to develop a new field as well as guidance for selection of appropriate sensing modes for future applications.
Acknowledgements
We are thankful to the researchers who contributed towards the development of chemosensors for the detection of aluminium ions. A. G. is highly thankful to DAV University, Jalandhar for providing the research facilities and infrastructure.Notes and references
- J. P. Desvergne and A. W. Czarnik, Chemosensors of Ion and Molecule Recognition, Kluwer Academic Press, Netherlands, 1997 Search PubMed.
- A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher and T. E. Rice, Chem. Rev., 1997, 97, 1515–1566 CrossRef CAS PubMed.
- J. Wu, W. Liu, J. Ge, H. Zhang and P. Wang, Chem. Soc. Rev., 2011, 40, 3483–3495 RSC.
- M. Wang, G. Zhang, D. Zhang, D. Zhu and B. Z. Tang, J. Mater. Chem., 2010, 20, 1858–1867 RSC.
- P. Nayak, Environ. Res., 2002, 89, 101–115 CrossRef CAS PubMed.
- N. Fimreite, O. O. Hansen and H. C. Pettersen, Bull. Environ. Contam. Toxicol., 1997, 58, 1–7 CrossRef CAS PubMed.
- E. Delhaize and P. Ryan, Plant Physiol., 1995, 107, 315–321 CrossRef CAS PubMed.
- A. Salifoglou, Coord. Chem. Rev., 2002, 228, 297–317 CrossRef CAS.
- M. E. Percy, T. P. A. Kruck, A. I. Pogue and W. J. Lukiw, J. Inorg. Biochem., 2011, 105, 1505–1512 CrossRef CAS PubMed.
- G. D. Fasman, Coord. Chem. Rev., 1996, 149, 125–165 CrossRef CAS.
- D. Krewski, R. A. Yokel, E. Nieboer, D. Borchelt, J. Cohen, J. Harry, S. Kacew, J. Lindsay, A. M. Mahfouz and V. Rondeau, J. Toxicol. Environ. Health, Part B, 2007, 10, 1–269 CAS.
- X. Li, J. Chen and E. Wang, Chin. J. Chem., 2014, 32, 429–433 CrossRef CAS.
- S. Das, S. Goswami, K. Aich, K. Ghoshal, C. K. Quah, M. Bhattacharyya and H. K. Fun, New J. Chem., 2015, 39, 8582–8587 RSC.
- J. Barcelo and C. Poschenrieder, Environ. Exp. Bot., 2002, 48, 75–92 CrossRef CAS.
- Z. Krejpcio and R. W. P. Wojciak, Pol. J. Environ. Stud., 2002, 11, 251–254 CAS.
- H. N. Kim, W. X. Ren, J. S. Kim and J. Yoon, Chem. Soc. Rev., 2012, 41, 3210–3244 RSC.
- H. Sang, P. Liang and D. Du, J. Hazard. Mater., 2008, 154, 1127–1132 CrossRef CAS PubMed.
- S. J. Djane, M. Gra and C. Korn, Spectrochim. Acta, Part B, 2000, 55, 389–394 CrossRef.
- S. Abbasi and A. Farmany, Food Chem., 2009, 116, 1019–1023 CrossRef CAS.
- V. K. Gupta, A. K. Jain and G. Maheshwari, Talanta, 2007, 72, 1469–1473 CrossRef CAS PubMed.
- D. T. McQuade, A. E. Pullen and T. M. Swager, Chem. Rev., 2000, 100, 2537–2574 CrossRef CAS PubMed.
- K. Rurack, M. Kollmannsberger, U. Resch-Genger and J. Daub, J. Am. Chem. Soc., 2000, 122, 968–969 CrossRef CAS.
- J. L. Bricks, A. Kovalchuk, C. Trieflinger, M. Nofz, M. Buschel, A. I. Tolmachev, J. Daub and K. Rurack, J. Am. Chem. Soc., 2005, 127, 13522–13529 CrossRef CAS PubMed.
- E. M. Nolan and S. J. Lippard, J. Am. Chem. Soc., 2007, 129, 5910–5918 CrossRef CAS PubMed.
- T. L. Andrew and T. M. Swager, J. Am. Chem. Soc., 2007, 129, 7254–7255 CrossRef CAS PubMed.
- O. Alici and S. Erdemir, Sens. Actuators, B, 2015, 208, 159–163 CrossRef CAS.
- J. Kumar, M. J. Sarma, P. Phukan and D. K. Das, Dalton Trans., 2015, 44, 4576–4581 RSC.
- S. Mondal, A. K. Bhanja, D. Ojha, T. K. Mondal, D. Chattopadhyay and C. Sinha, RSC Adv., 2015, 5, 73626–73638 RSC.
- S. Mukherjee, P. Mal and H. Stoeckli-Evans, J. Lumin., 2016, 172, 124–130 CrossRef CAS.
- C. A. Huerta-Aguilar, P. Raj, P. Thangarasu and N. Singh, RSC Adv., 2016, 6, 37944–37952 RSC.
- C. R. Li, J. C. Qin, G. Q. Wang, B. D. Wang and Z. Y. Yang, Anal. Methods, 2015, 7, 3500–3505 RSC.
- S. Memon, A. A. Bhatti, Ü. Ocak and M. Ocak, Anal. Methods, 2015, 7, 5114–5121 RSC.
- A. Ghorai, J. Mondal, R. Chandra and G. K. Patra, Dalton Trans., 2015, 44, 13261–13271 RSC.
- J. C. Qin, L. Fan, B. D. Wang, Z. Y. Yang and T. R. Li, Anal. Methods, 2015, 7, 716–722 RSC.
- J. C. Qin, X. Y. Cheng, K. C. Yu, R. Fang, M. F. Wang and Z. Y. Yang, Anal. Methods, 2015, 7, 6799–6803 RSC.
- S. Chemate and N. Sekar, RSC Adv., 2015, 5, 27282–27289 RSC.
- B. K. Datta, D. Thiyagarajan, C. Kar, A. Ramesh and G. Das, Anal. Chim. Acta, 2015, 882, 76–82 CrossRef CAS PubMed.
- Y. Lu, S. Huang, Y. Liu, S. He, L. Zhao and X. Zeng, Org. Lett., 2011, 13, 5274–5277 CrossRef CAS PubMed.
- S. K. Shoora, A. K. Jain and V. K. Gupta, Sens. Actuators, B, 2015, 216, 86–104 CrossRef.
- V. K. Gupta, A. K. Singh and L. K. Kumawat, Sens. Actuators, B, 2014, 195, 98–108 CrossRef CAS.
- H. S. Jung, P. Verwilst, W. Y. Kim and J. S. Kim, Chem. Soc. Rev., 2016, 45, 1242–1256 RSC.
- Y. Li, C. Liao, S. Huang, H. Xu, B. Zheng, J. Du and D. Xiao, RSC Adv., 2016, 6, 25420–25426 RSC.
- V. K. Gupta, A. K. Jain and S. K. Shoora, Sens. Actuators, B, 2015, 219, 101 Search PubMed.
- B. K. Datta, D. Thiyagarajan, A. Ramesh and G. Das, Dalton Trans., 2015, 44, 13093–13099 RSC.
- J. C. Qin and Z. Y. Yang, Anal. Methods, 2015, 7, 2036–2040 RSC.
- J. Sun, Z. Liu, Y. Wang, S. Xiao, M. Pei, X. Zhao and G. Zhang, RSC Adv., 2015, 5, 100873–100878 RSC.
- J. C. Qin and Z. Y. Yang, J. Photochem. Photobiol., A, 2015, 303–304, 99–104 CrossRef CAS.
- J. C. Qin, L. Fan, T. R. Li and Z. Y. Yang, Synth. Met., 2015, 199, 179–186 CrossRef CAS.
- Y. J. Chang, S. S. Wu, C. H. Hu, C. Cho, M. X. Kao and A. T. Wu, Inorg. Chim. Acta, 2015, 432, 25–31 CrossRef CAS.
- V. P. Singh, K. Tiwari, M. Mishra, N. Srivastava and S. Saha, Sens. Actuators, B, 2013, 182, 546–554 CrossRef CAS.
- K. Tiwari, M. Mishra and V. P. Singh, RSC Adv., 2013, 3, 12124–12132 RSC.
- L. Kang, Z.-Y. Xing, X.-Y. Ma, Y.-T. Liu and Y. Zhang, Spectrochim. Acta, Part A, 2016, 167, 59–65 CrossRef CAS PubMed.
- S. M. Hossain, K. Singh, A. Lakma, R. N. Pradhan and A. K. Singh, Sens. Actuators, B, 2017, 239, 1109–1117 CrossRef CAS.
- N. Kumar, V. Bhalla and M. Kumar, Analyst, 2014, 139, 543–558 RSC.
- V. S. Kraynov, C. Chamberlain, G. M. Bokoch, M. A. Schwartz, S. Slabaugh and K. M. Hah n, Science, 2000, 290, 333–337 CrossRef CAS PubMed.
- M. Fehr, W. B. Frommer and S. Lalonde, Proc. Natl. Acad. Sci. U. S. A., 2002, 99, 9846–9851 CrossRef CAS PubMed.
- S. A. Farber, M. Pack, S. Y. Ho, I. D. Johnson, D. S. Wagner, R. Dosch, M. C. Mullins, H. S. Hendrickson, E. K. Hendrickson and M. E. Halpern, Science, 2001, 292, 1385–1388 CrossRef CAS PubMed.
- O. Wichmann, J. Wittbrodt and C. Schultz, Angew. Chem., Int. Ed., 2006, 45, 508–512 CrossRef CAS PubMed.
- I. A. Yudushkin, A. Schleifenbaum, A. Kinkhabwala, B. G. Neel, C. Schultz and P. I. H. Bastiaens, Science, 2007, 315, 115–119 CrossRef CAS PubMed.
- M. Arduini, F. Felluga, F. Mancin, P. Rossi, P. Tecilla, U. Tonellato and N. Valentinuzzi, Chem. Commun., 2003, 1606–1607 RSC.
- J. C. Qin, J. Yan, B. D. Wang and Z. Y. Yang, Tetrahedron Lett., 2016, 57, 1935–1939 CrossRef CAS.
- R. W. Ramette and E. B. Sandell, J. Am. Chem. Soc., 1956, 78, 4872–4878 CrossRef CAS.
- S. Xia, S. Y. Xiao, Q. Q. Hong, J. R. Zou, S. Yang, M. X. Zhang and H. Zuo, RSC Adv., 2015, 5, 5244–5249 RSC.
- N. Chatterjee, S. B. Maity, A. Samadder, P. Mukherjee, A. R. Khuda-Bukhsh and P. K. Bharadwaj, RSC Adv., 2016, 6, 17995–18001 RSC.
- P. Ding, J. Wang, J. Cheng, Y. Zhao and Y. Ye, New J. Chem., 2015, 39, 342–348 RSC.
- S. Paul, S. Goswami and A. Manna, Dalton Trans., 2015, 44, 11805–11810 RSC.
- X. Bao, Q. Cao, Y. Xu, Y. Gao, Y. Xu, X. Nie, B. Zhou, T. Pang and J. Zhu, Bioorg. Med. Chem., 2015, 23, 694–702 CrossRef CAS PubMed.
- V. K. Gupta, N. Mergu, L. K. Kumawat and A. K. Singh, Talanta, 2015, 144, 80–89 CrossRef CAS PubMed.
- S. Chemate and N. Sekar, Sens. Actuators, B, 2015, 220, 1196–1204 CrossRef CAS.
- Q. Huang, Q. Zhang, E. Wang, Y. Zhou, H. Qiao, L. Pang and F. Yu, Spectrochim. Acta, Part A, 2016, 152, 70–76 CrossRef CAS PubMed.
- V. K. Gupta, N. Mergu and L. K. Kumawat, Sens. Actuators, B, 2016, 223, 101–113 CrossRef CAS.
- N. Mergu, A. K. Singh and V. K. Gupta, Sensors, 2015, 15, 9097–9111 CrossRef CAS PubMed.
- G. Q. Yang, F. Morlet-Savary, Z. K. Peng, S. K. Wu and J. P. Fouassier, Chem. Phys. Lett., 1996, 256, 536–542 CrossRef CAS.
- Z. M. Li and S. K. Wu, J. Fluoresc., 1997, 7, 237–242 CrossRef CAS.
- P. F. Wang and S. K. Wu, J. Photochem. Photobiol., A, 1995, 86, 109–113 CrossRef CAS.
- S. A. Lee, G. R. You, Y. W. Choi, H. Y. Jo, A. R. Kim, I. Noh, S.-J. Kim, Y. Kim and C. Kim, Dalton Trans., 2014, 43, 6650–6659 RSC.
- I. H. Hwang, Y. W. Choi, K. B. Kim, G. J. Park, J. J. Lee, L. Nguyen, I. Noh and C. Kim, New J. Chem., 2016, 40, 171–178 RSC.
- J. Ma, W. Shi, L. Feng, Y. Chen, K. Fan, Y. Hao, Y. Hui and Z. Xie, RSC Adv., 2016, 6, 28034–28037 RSC.
- C. Liang, W. Bu, C. Li, G. Men, M. Deng, Y. Jiangyao, H. Sun and S. Jiang, Dalton Trans., 2015, 44, 11352–11359 RSC.
- R. Alam, T. Mistri, R. Bhowmick, A. Katarkar, K. Chaudhuri and M. Ali, RSC Adv., 2016, 6, 1268–1278 RSC.
- Y. W. Choi, J. J. Lee, E. Nam, M. H. Lim and C. Kim, Tetrahedron, 2016, 72, 1998–2005 CrossRef CAS.
- V. K. Gupta, S. K. Shoora, L. K. Kumawat and A. K. Jain, Sens. Actuators, B, 2015, 209, 15–24 CrossRef CAS.
- Z. Liu, H. Xu, L. Sheng, S. Chen, D. Huang and J. Liu, Spectrochim. Acta, Part A, 2016, 157, 6–10 CrossRef CAS PubMed.
- C.-H. Chen, D.-J. Liao, C.-F. Wan and A.-T. Wu, Analyst, 2013, 138, 2527–2530 RSC.
- V. K. Gupta, A. K. Singh and N. Mergu, Electrochim. Acta, 2014, 117, 405–412 CrossRef CAS.
- T. Liu, Y. Dong, X. Wan, W. Li and Y. Yao, RSC Adv., 2015, 5, 76939–76942 RSC.
- S. M. Hossain, A. Lakma, R. N. Pradhan, A. Chakraborty, A. Biswas and A. K. Singh, RSC Adv., 2015, 5, 63338–63344 RSC.
- S. Dey, S. Halder, A. Mukherjee, K. Ghosh and P. Roy, Sens. Actuators, B, 2015, 215, 196–205 CrossRef CAS.
- K. Boonkitpatarakul, J. Wang, N. Niamnont, B. Liu, L. Mcdonald, Y. Pang and M. Sukwattanasinitt, ACS Sens., 2016, 2, 144–150 CrossRef.
- R. Alam, T. Mistri, R. Bhowmicka, A. Katarkar, K. Chaudhuri and M. Ali, RSC Adv., 2015, 5, 53940–53948 RSC.
- A. K. Mahapatra, S. S. Ali, K. Maiti, A. K. Manna, R. Maji, S. Ondal, M. R. Uddin, S. Mandal and P. A. Sahoo, RSC Adv., 2015, 5, 81203–81211 RSC.
- Y. S. Kim, J. J. Lee, Y. W. Choi, G. R. You, L. Nguyen, I. Noh and C. Kim, Dyes Pigm., 2016, 129, 43–53 CrossRef CAS.
- Y. H. Zhao, X. Zeng, L. Mu, J. Li, C. Redshaw and G. Wei, Sens. Actuators, B, 2014, 204, 450–458 CrossRef CAS.
- A. S. M. Islam, R. Bhowmick, H. Mohammad, A. Katarkar, K. Chaudhuri and M. Ali, New J. Chem., 2016, 40, 4710–4719 RSC.
- J. Luo, Z. Xie, J. W. Y. Lam, L. Cheng, H. Chen, C. Qiu, H. S. Kwok, X. Zhan, Y. Liu, D. Zhu and B. Z. Tang, Chem. Commun., 2001, 40, 1740–1742 RSC.
- H. Shi, J. Liu, J. Geng, B. Z. Tang and B. Liu, J. Am. Chem. Soc., 2012, 134, 9569–9572 CrossRef CAS PubMed.
- S. Gui, Y. Huang, F. Hu, Y. Jin, G. Zhang, L. Yan, D. Zhang and R. Zhao, Anal. Chem., 2015, 87, 1470–1474 CrossRef CAS PubMed.
- S. Samanta, U. Manna, T. Ray and G. Das, Dalton Trans., 2015, 44, 18902–18910 RSC.
- Z. Köstereli and K. Severin, Org. Biomol. Chem., 2015, 13, 252–257 Search PubMed.
- T. Han, X. Feng, B. Tong, J. Shi, L. Chen, J. Zhi and Y. Dong, Chem. Commun., 2012, 48, 416–418 RSC.
- M. Shyamal, P. Mazumdar, S. Maity, G. P. Sahoo, G. Salgado-Morán and A. Misra, J. Phys. Chem. A, 2016, 120, 210–220 CrossRef CAS PubMed.
- Y. B. Ruan, A. Depauw and I. Leray, Org. Biomol. Chem., 2014, 12, 4335–4341 CAS.
- E. Özcan, S. O. Tümay, H. A. Alidaği, B. Çoşut and S. Yeşilot, Dyes Pigm., 2016, 132, 230–236 CrossRef.
- W. Wang, Z. Mao, M. Wang, L. J. Liu, D. W. J. Kwong, C. H. Leung and D. L. Ma, Chem. Commun., 2016, 52, 3611–3614 RSC.
- M. Alaparthi, K. Mariappan, E. Dufek, M. Hoffmana and A. G. Sykes, RSC Adv., 2016, 6, 11295–11302 RSC.
- Y.-W. Liu, C.-H. Chen and A.-T. Wu, Analyst, 2012, 137, 5201–5203 RSC.
- D. K. Singha and P. Mahata, Inorg. Chem., 2015, 54, 6373–6379 CrossRef CAS PubMed.
- Y. Kim, G. Jang and T. S. Lee, ACS Appl. Mater. Interfaces, 2015, 7, 15649–15657 CAS.
- J. Zhao, Y. Zhao, S. Xu, N. Luo and R. Tang, Inorg. Chim. Acta, 2015, 438, 105–111 CrossRef CAS.
- Y. H. Zhao, X. Zeng, L. Mua, J. Li, C. Redshaw and G. Wei, Sens. Actuators, B, 2014, 204, 450–458 CrossRef CAS.
- F. Yu, L. J. Hou, L. Y. Qin, J. B. Chao, Y. Wang and W. J. Jin, J. Photochem. Photobiol., A, 2016, 315, 8–13 CrossRef CAS.
- M. Yu, R. Yuan, C. Shi, W. Zhou, L. Wei and Z. Li, Dyes Pigm., 2013, 99, 887–894 CrossRef CAS.
- J. C. Qin, Z. Y. Yang and P. Yang, Inorg. Chim. Acta, 2015, 432, 136–141 CrossRef CAS.
- J. C. Qin, T. R. Li, B. D. Wang, Z. Y. Yang and L. Fan, Spectrochim. Acta, Part A, 2014, 133, 38–43 CrossRef CAS PubMed.
- H. Wang, B. Wang, Z. Shi, X. Tang, W. Dou, Q. Han, Y. Zhang and W. Liu, Biosens. Bioelectron., 2015, 65, 91–96 CrossRef CAS PubMed.
- S. Erdemir and S. Malkondu, J. Lumin., 2015, 158, 401–406 CrossRef CAS.
- P. Torawane, K. Tayade, S. Bothra, S. K. Sahoo, N. Singh and A. Borse, Sens. Actuators, B, 2016, 222, 562–566 CrossRef CAS.
- J. C. Qin and Z. Y. Yang, J. Photochem. Photobiol., A, 2015, 303–304, 99–104 CrossRef CAS.
- C. J. Liu, Z. Y. Yang, L. Fan, X. L. Jin, J. M. An, X. Y. Cheng and B. D. Wang, J. Lumin., 2015, 158, 172–175 CrossRef CAS.
- G. T. Selvan, M. Kumaresan, R. Sivaraj, I. V. M. V. Enoch and P. M. Selvakumar, Sens. Actuators, B, 2016, 229, 181–189 CrossRef CAS.
- S. Sahana, S. Bose, S. K. Mukhopadhyay and P. K. Bharadwaj, J. Lumin., 2016, 169, 334–341 CrossRef CAS.
- A. Rai, N. Kumari, A. K. Srivastava, S. K. Singh, S. Srikrishna and L. Mishra, J. Photochem. Photobiol., A, 2016, 319, 78–86 CrossRef.
- J. C. Qin and Z. Y. Yang, Synth. Met., 2015, 209, 570–576 CrossRef CAS.
- J. C. Qin, T. R. Li, B. D. Wang, Z. Y. Yang and L. Fan, Synth. Met., 2014, 195, 141–146 CrossRef CAS.
- M. Mukherjee, B. Sen, S. Pal, A. Maji, D. Budhadev and P. Chattopadhyay, Spectrochim. Acta, Part A, 2016, 157, 11–16 CrossRef CAS PubMed.
- R. Manjunath, E. Hrishikesan and P. Kannan, Spectrochim. Acta, Part A, 2015, 140, 509–515 CrossRef CAS PubMed.
- C. R. Li, J. C. Qin, G. Q. Wang, B. D. Wang, A. K. Fu and Z. Y. Yang, Inorg. Chim. Acta, 2015, 430, 91–95 CrossRef CAS.
- C. R. Li, Z. C. Liao, J. C. Qin, B. D. Wang and Z. Y. Yang, J. Lumin., 2015, 168, 330–333 CrossRef CAS.
- J. J. Lee, G. J. Park, Y. S. Kim, S. Y. Lee, H. J. Lee, I. Noh and C. Kim, Biosens. Bioelectron., 2015, 69, 226–229 CrossRef PubMed.
- L. K. Kumawat, N. Mergu, M. Asif and V. K. Gupta, Sens. Actuators, B, 2016, 231, 847–859 CrossRef.
- V. Kumar, A. Kumar, U. Diwan, S. Ramesh, S. K. Srivastava and K. K. Upadhyay, Sens. Actuators, B, 2015, 207, 650–657 CrossRef CAS.
- H. S. Kim, S. Angupillai and Y. A. Son, Sens. Actuators, B, 2016, 222, 447–458 CrossRef CAS.
- H. Kim, B. A. Rao, J. W. Jeong, S. Mallick, S. M. Kang, J. S. Choi, C. S. Lee and Y. A. Son, Sens. Actuators, B, 2015, 210, 173–182 CrossRef CAS.
- J. W. Jeong, B. A. Rao, J. Y. Lee, J. Y. Hwang and Y. A. Son, Sens. Actuators, B, 2016, 227, 227–241 CrossRef CAS.
- U. Janakipriya, N. R. Chereddy, P. Korrapati, S. Thennarasu and A. B. Mandal, Spectrochim. Acta, Part A, 2016, 153, 465–470 CrossRef PubMed.
- M. Iniya, D. Jeyanthi, K. Krishnaveni and D. Chellappa, J. Lumin., 2015, 157, 383–389 CrossRef CAS.
- P. J. Hung, J. L. Chir, W. Ting and A. T. Wu, J. Lumin., 2015, 158, 371–375 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |