Optimization of magnetic hyperthermia effect for breast cancer stem cell therapy†
Yong-Su Kwon‡
a,
Kyunjong Sim‡b,
Taeyoon Seo‡c,
Jin-Kyu Leeb,
Youngwoo Kwon*c and
Tae-Jong Yoon*a
aNanopharmacy Lab., College of Pharmacy and Research Institute of Pharmaceutical Science and Technology (RIPST), Ajou University, 206 Worldcup-ro, Yeongtong-gu, Suwon, Gyeonggi-do 16499, South Korea. E-mail: tjyoon@ajou.ac.kr
bDepartment of Chemistry, Seoul National University, Seoul, 151-747, South Korea
cDepartment of Electrical and Computer Engineering, Institute of New Media and Communications, Seoul National University, Seoul, 151-747, South Korea. E-mail: ykwon@snu.ac.kr
First published on 4th November 2016
Abstract
We successfully prepared magnetic nanoclusters (MNCs) with regular size distribution and high solution stability. To increase the therapeutic effectiveness of MNCs, various factors such as the size distribution of particles, external field frequency and strength should be optimized. The correlation between heat generation ability and the various factors was evaluated by measuring the specific absorption rate. The 60 nm MNC showed the highest heat capacity ability in the various field conditions. Breast cancer stem cells (bCSCs) with chemo/radio-resistance were exposed to the optimized alternating magnetic field after treating with the MNC. The MNC promoted an appropriate temperature increase within 10 min and the particles influenced directly heat shock protein 70 and 90 damage during the magnetic hyperthermia. Thus, the treated CSCs were progressed to the thermal-mediated apoptotic cell death. Our optimization for magnetic hyperthermia will be useful for a variety of research on cancer cell therapy.
Introduction
Cancer stem cells (CSCs) are tumor cells that enhance disease progression, and cause the expansion of primary tumors contributing to metastasis.1,2 Importantly, cancer recurrence could occur because a small population of CSCs is resistant to chemo- or radio-therapy.3,4 The CSC research has been challenged and has important implications for cancer therapy. Meanwhile, magnetic hyperthermia is proposed as a promising clinical tool for a CSC therapy and useful for treating small or deep-seated cancer by supplying heat to tumor cells via magnetic nanoparticle (MNP).5,6 To apply MNPs in magnetic hyperthermia, the MNPs must satisfy a few main conditions such as having a large heating power and good stability.7 The heating power needs to be maximized and optimized since the optimal size of MNPs for the maximum power loss varies with the amplitude of the applied magnetic field. With regard to the stability, superparamagnetic MNPs that can be allowed by the nanometer size are preferred to maintain good colloidal stability and increased bio-accessibility through surface modification with chemicals or polymers.8 Moreover, to be applicable, the used concentration of MNPs should be minimized to limit side effects such as heavy metal ion toxicity or nanotoxicity.9 Hence, it is essential to optimize the power dissipation or heat efficiency of MNPs. The specific absorption rate (SAR) of MNPs represents the heating efficiency and depends on the size of the particles, saturation magnetization (Ms) and magnetic anisotropy of the MNP.10 For a given iron oxide MNP, the mean size of MNPs with varying applied magnetic field (AMF) is an important parameter for optimizing SAR.11,12 The heating efficiency (SAR), also referred to as the specific loss power (SLP), is measured in watts per gram of MNPs as follows:
 | (1) |
Cancer stem cell (CSC) are a subpopulation of cancer cells with stem cell-like properties and are thought to be responsible for tumor drug resistance and relapse.13 Recently, researchers have suggested the therapeutic application of CSC elimination by optical or magnetic heating hyperthermia. They reported only the therapeutic effect using a magnetic particle with one size and not optimized the crystal size under the various external magnetic fields or frequencies yet.14 In this study, as a proof-of-concept, we showed the therapeutic magnetic hyperthermia effect on breast CSCs (bCSCs), and the cell death mechanism was established by biological analysis.
Experimental
Materials
Iron chloride hexahydrate (FeCl3·6H2O, 97%), sodium acetate anhydrous (NaOAc, 98.5%), ethylene glycol (EG, 99.5%), poly(acrylic acid) (PAA, Mw 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, 35 wt% in H2O), ammonia (NH4OH, 29%) were purchased from Sigma-Aldrich Korea without purification. Deionized (DI) water was purified using the Milli-Q system and used for all experiments. Preparing the mixed chalcogenide stock solutions.
000, 35 wt% in H2O), ammonia (NH4OH, 29%) were purchased from Sigma-Aldrich Korea without purification. Deionized (DI) water was purified using the Milli-Q system and used for all experiments. Preparing the mixed chalcogenide stock solutions.
Preparation of the magnetic nanocluster particles (MNCs)
We prepared the MNC particles with various size distribution by previous reported method and slightly modified the preparation procedure for large particle synthesis.15For the 60 nm MNCs, FeCl3·6H2O (24 g), sodium acetate (100 g), and DI water were completely dissolved in 1.5 L of EG by vigorous mechanical stirring for 30 min, which turned the color into a yellow-brown turbid solution. The solution was heated to 70 °C and the temperature was maintained for 1 h, followed by continuous refluxing for 24 h. When the solution turned to a black color, it was cooled to room temperature, precipitated by centrifugation (17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm), and the black precipitates were washed with ethanol (three times) to remove excess chemicals. The prepared MNCs were dispersed in ethanol. For other sizes of MNCs (20, 30, 40, 80, and 140 nm), the same preparation procedure was followed as described above with the 60 nm sized MNC, except different concentrations of FeCl3·6H2O and sodium acetate were used. The different ratio of the chemicals is listed in ESI (Fig. S2c†). The prepared MNCs (25 g) were precipitated by centrifugation and re-dispersed in DI water (1 L). The MNC solution (4 mL) was mixed with 300 mg of PAA and sonicated for 30 min at room temperature. The PAA-coated MNCs were precipitated down by centrifugation (17
000 rpm), and the black precipitates were washed with ethanol (three times) to remove excess chemicals. The prepared MNCs were dispersed in ethanol. For other sizes of MNCs (20, 30, 40, 80, and 140 nm), the same preparation procedure was followed as described above with the 60 nm sized MNC, except different concentrations of FeCl3·6H2O and sodium acetate were used. The different ratio of the chemicals is listed in ESI (Fig. S2c†). The prepared MNCs (25 g) were precipitated by centrifugation and re-dispersed in DI water (1 L). The MNC solution (4 mL) was mixed with 300 mg of PAA and sonicated for 30 min at room temperature. The PAA-coated MNCs were precipitated down by centrifugation (17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 10 min), and excess PAA was washed by centrifugation and re-dispersed in DI water (three times). Finally, the purified MNC–PAA particles (100 mg) were dispersed in DI water (4 mL) and 1 mL of NH4OH solution was added. After shaking for 5 min, the solution was centrifuged at 17
000 rpm, 10 min), and excess PAA was washed by centrifugation and re-dispersed in DI water (three times). Finally, the purified MNC–PAA particles (100 mg) were dispersed in DI water (4 mL) and 1 mL of NH4OH solution was added. After shaking for 5 min, the solution was centrifuged at 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min to obtain the particles, followed by washing three times with DI water according to the above purification procedure.
000 rpm for 10 min to obtain the particles, followed by washing three times with DI water according to the above purification procedure.
Characterization of MNCs
The size and morphology of the prepared MNCs were examined by transmission electron microscopy (TEM, Hitachi-7600) at an acceleration voltage of 100 kV. The crystal phase of the MNCs was determined by a power X-ray diffractometer (XRD, D8 Advance, Bruker) with a Cu Kα (1.5406 Å) source. Magnetization curves of the MNCs were measured by a magnetic property measurement system (MPMS-5XL, Quantum Design) at 300 K with an applied field of up to 50 kOe. The infrared spectra were determined by KBr pellet using a Fourier transform infrared spectrometer (FT-IR, Nicoloet™iS™10, Thermo Scientific). Thermogravimetric analysis (TGA) was performed using a TA instrument SDT Q600 analyzer. The concentration of iron in the samples was measured by inductively coupled plasma emission spectrometry (ICP-ES, ICP730ES, Varian).SAR measurement
The magnetic heating ability (SAR) was measured by conducting time-dependent temperature studies with the MNC solutions after inserting the sample tube into the coil (100B, Magnetic Science) of the easyheat induction heating system (EasyHeat 0224, Ambrell). The device can control magnetic conditions with various frequencies (290–780 kHz) and field strengths (7.1–78.6 kA m−1). The sample tube was thermally insulated with a water-cooled magnetic induction coil (diameter 16 mm). The MNC particle concentration in agarose gel was 5 mg mL−1. The SAR of the MNCs under the various AMF conditions was measured by the rate of absorbed energy per unit mass of particle and the value was calculated using eqn (2):
 | (2) |
Preparation of cell culture and mammosphere
The human breast cancer cell line BT-474 was obtained from the American Type Culture Collection (ATCC). Cells were grown as monolayer, routinely cultured in Hybri-Care Medium (ATCC), and maintained in MEM medium (GIBCO, Invitrogen) supplemented with 10% FBS (GIBCO, Invitrogen), 10 μg mL−1 insulin (Sigma), and penicillin/streptomycin (GIBCO, Invitrogen). For mammosphere culture, cells were suspended at a density of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per mL and seeded into 6-well plates (2.5 mL per well) in DMEM/F12 (1
000 cells per mL and seeded into 6-well plates (2.5 mL per well) in DMEM/F12 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) media containing 5 μg mL−1 bovine insulin (Sigma), 0.4% bovine serum albumin (Sigma), 2% B-27 supplement (Invitrogen), 20 ng mL−1 basic fibroblast growth factor (Peprotech), and 10 ng mL−1 epidermal growth factor (Sigma). One milliliter of fresh media was added to each well every 2 days (without removing the old media). Mammospheres were collected on day 9 by gentle centrifugation and were dissociated to single cell suspensions by treatment with 0.05% trypsin–EDTA for 10 min. For mammosphere formation, the single cells (1000 cells per mL) were cultured in suspension (60 mm-Petri dish flask) to generate mammospheres of the next generation. The percentage and size of the wells with mammospheres were analyzed at the indicated times. For each passage cultured cells, ALDH1 expression was determined with the ALDEFLUOR™ fluorescent reagent system (Stem Cell Technology, Canada), to identify cells expressing ALDH1 activity as a biomarker for bCSCs by using flow cytometry (BD Biosciences).16 For the detection of CD44+/CD24−, cells were stained in 96-well plates in a volume of 50 μL, with 2 μL of each monoclonal antibody per well: CD24-Alexa648, CD44-FITC, and ESA-PcPCy5 (BD Biosciences). Isotype-matched labeled controls were also used in the analysis. Cells were labeled on ice for 30 min and washed twice before analysis with a cytometer.
1) media containing 5 μg mL−1 bovine insulin (Sigma), 0.4% bovine serum albumin (Sigma), 2% B-27 supplement (Invitrogen), 20 ng mL−1 basic fibroblast growth factor (Peprotech), and 10 ng mL−1 epidermal growth factor (Sigma). One milliliter of fresh media was added to each well every 2 days (without removing the old media). Mammospheres were collected on day 9 by gentle centrifugation and were dissociated to single cell suspensions by treatment with 0.05% trypsin–EDTA for 10 min. For mammosphere formation, the single cells (1000 cells per mL) were cultured in suspension (60 mm-Petri dish flask) to generate mammospheres of the next generation. The percentage and size of the wells with mammospheres were analyzed at the indicated times. For each passage cultured cells, ALDH1 expression was determined with the ALDEFLUOR™ fluorescent reagent system (Stem Cell Technology, Canada), to identify cells expressing ALDH1 activity as a biomarker for bCSCs by using flow cytometry (BD Biosciences).16 For the detection of CD44+/CD24−, cells were stained in 96-well plates in a volume of 50 μL, with 2 μL of each monoclonal antibody per well: CD24-Alexa648, CD44-FITC, and ESA-PcPCy5 (BD Biosciences). Isotype-matched labeled controls were also used in the analysis. Cells were labeled on ice for 30 min and washed twice before analysis with a cytometer.
Cell viability assay
The percentage of cell viability was determined with the WST-1 (4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate) assay kit (Thermofisher) as described earlier.17 BT-474 bCSC cells were seeded in a 96-well plate (1 × 105 cells per well) and incubated overnight. Cells were treated with various concentrations (0–10 mg mL−1) of MNCs of different sizes. After 3 h of incubation, the supernatants were aspirated to remove the excess particles, cell monolayers were washed with PBS, and WST-1 reagent (200 μL) was added to each well. Cells were incubated for 1 h and the absorbance was measured at 450 nm using a Bio-Rad Benchmark microplate reader.Results and discussion
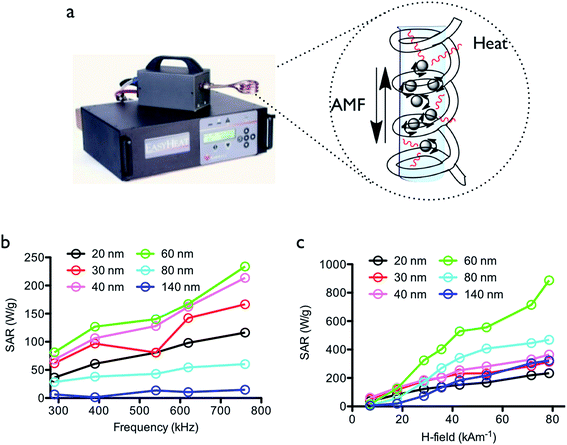
We prepared MNCs using FeCl3 as the iron source and EG as both a solvent and a reductant in the presence of sodium acetate and deionized water. The size of the prepared MNCs could be precisely tuned from 20 nm to 140 nm by deliberately adjusting the concentrations of FeCl3 and sodium acetate. In the TEM analysis (Fig. 1a), the synthesized MNCs showed high regular size distributions. Moreover, the surface was modified with poly(acrylic acid) (PAA) to improve their stability in aqueous solution. The carboxylic acid functional group of PAA was directly coordinated onto the iron metal of MNC, and the amount of PAA and the changes of the chemical functional group were characterized by FT-IR and thermogravimetric analysis (TGA), as shown in Fig. S1.† The magnetic properties and structural properties of the various sized MNCs were evaluated by X-ray diffraction (XRD) and the magnetic property measurement system (MPMS-5XL, Quantum Design) (Fig. S2†). All samples were revealed typical inverse spinel ferrite structure and the crystal size of MNCs in the [220] peak was calculated. According to the peak analysis, the MNCs of 20, 30, and 40 nm in size showed the same crystalline domain size of 10 nm and exhibited increasing crystal size as the whole size of the MNC increased (Fig. S3†). The formation of MNC composed of small nanocrystals could be demonstrated through two-stage growth model.18 Primary nanocrystal was formed by nucleation and aggregated to secondary structure in order to minimize their surface energy. The sizes of primary and secondary particle could be influenced by the concentration of precursors in reaction condition. The sizes of primary crystals and secondary particles were changed by different reaction conditions and increased with higher concentration of iron precursor. The increase of iron precursor concentration was introduced accelerating the rate of nucleation and grain growth through the increase of hydrolysis/condensation rate of FeCl3 and then, the bigger nano-crystalline could be formed. And also, a higher concentration of sodium acetate was chelated more onto the surface of primary crystal and prevented the aggregation of the crystals by stabilizing and reducing surface energy. Therefore, the 20 and 30 nm MNCs were shown different secondary particle sizes. To summarize, the crystal size was increased with whole particle size owing to the presence of additives metal ions or surface chelating molecules.The prepared MNCs displayed similar Ms value with an average of 66.3 emu g−1 (±3.0) and coercivity (Hc) of 12.2 Oe (±1.4), except for the MNC of 140 nm in size (26 Oe) at 300 K. However, we thought that all the prepared MNCs demonstrated superparamagnetic behavior in the solution. The produced MNCs are summarized the physical properties and preparation conditions in Fig. 1b and S2c.† Heat dissipation experiments were performed by using the EasyHeat device consisting of a sample coil enclosure powered by a AC power supply, generator and power amplifier forming a resonant circuit (Fig. 2a). The system could be monitored by an output from the coil enclosure with an oscilloscope, which allowed the measurement of the coil voltage and the approximate field strength. The cycling frequency was modified by changing the capacitor or coil configuration. Therefore, this device could examine the heating properties over a wide range of frequencies and field values. The prepared MNCs were dispersed in 3.3 wt% agarose gel to prevent physical changes, such as sedimentation, during the application of an external field. First, the heating efficiency (SAR) of the various MNC samples was measured as a function of the frequency from 290 to 760 kHz at a fixed magnetic field strength (7.1 kA m−1). The SAR values increased linearly as the frequency increased (Fig. 2b). In general, the dispersed MNPs showed that heating is dominated by Neel and Brown relaxation.19 According to a previous study, the transition from Neel to Brownian rotation occurred at a particle size of 16 nm.20 In this study, the increase in the SAR mainly resulted from the effect of Brownian relaxation of MNCs, while the relaxation via Neel mechanism was unaffected. The effect of the MNCs of over 80 nm in diameter was not dependent on the frequencies since the mechanical movement of the particles in the solution was neglected, and subsequently the Brownian relaxation was also neglected as a heat generation mechanism.
Interestingly, the 60 nm whole size MNCs significantly increased the SAR value and showed the strongest dependency with the external frequency. We thought that the 60 nm MNC with 20 nm crystalline size might matches well with the applied frequency and it subsequently exhibited marked increases in temperature owing to the faster rotation rate of the MNCs. As a secondary factor, the SAR of the various sized MNC samples was measured by increasing AMF strength from 7.1 to 78.6 kA m−1 at the same frequency (290 kHz). The SAR of all particles increased as the magnetic field strength increased (Fig. 2c). The 60 nm-sized MNC was also significantly associated with the external field strength, displaying the highest SAR value, while MNCs of other sizes increased gradually as the magnetic field strength increased. The specific increasing SAR of the 60 nm MNC was possibly attributed to the crystalline size of 20 nm and an external frequency of 290 kHz, which enhanced the SAR by the field strengths. The 20 nm crystalline sized MNC with 60 nm whole diameter demonstrated the highest effectiveness under the AMF in this study.
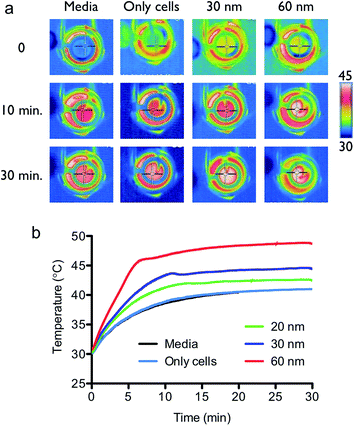
The bCSCs were prepared in accordance with the method of Dontu et. al. with modifications (see the Experimental section in detail).21 The BT-474 cells were incubated for 9 days at 37 °C in a CO2 incubator, and the mammosphere diameters were determined with an inverted microscope using the Image J software according to the typical CSC characterization method (Fig. S4†). As shown in Fig. 3a, the whole size of the cells gradually increased from a single cell (∼20 μm) to the mammosphere form (∼120 μm) in the culture media. As the number of culture passage increases, the capacity of mammosphere to form spheres was markedly hampered but the ALDH1 enzymatic activity and CD44+/CD24− cell surface antigens increased, which are known biomarkers for bCSCs (Fig. 3b and c). The intensity of the bCSC biomarkers was determined by flow cytometry, and the expression levels of ALDH1 and CD44+/CD24− were the highest after 3 times of passage culturing (see the Experimental section). The red fluorescent MNC particles were prepared by RITC and EDC/NHS conjugation method, and the bCSCs were treated with these particles for 3 h, followed by washing three times with culture media to eliminate excess particles. The fluorescence intensity was determined with a confocal microscope and showed that the particles could penetrate into the cell cytosol. The treated cells were responsible for the external point magnet owing to the magnetic property of the internalized MNCs (Fig. 3d). From the results of the SAR, 60 nm sized MNCs and the frequency of 290 kHz were used for thermal therapy to target bCSCs. Moreover, the AMF field strength of 60 kA m−1 was selected for the therapy because the SAR value of the 60 nm MNCs at 60 kA m−1 was significantly different from those of other sized MNCs. To compare the magnetic hyperthermia effect, bCSCs were treated with the 30 and 60 nm sized MNCs (0.2 mg mL−1) for 3 h at 37 °C in a CO2 incubator and the excess particles were removed by washing gently several times with a PBS buffer solution. Then, the cells were treated with trypsin–EDTA solution to detach cells (see the Experimental section). The cells were exposed to a magnetic field after homogeneous mixing with an agarose solution for different durations (Fig. 4a). The media and only cell samples (untreated) acted as controls and exhibited a similar temperature increase (40 °C). The temperature increase was caused by the warming of the magnetic coil during the application of the external field. These results indicated that the cell only sample did not affect the hyperthermia initiated by the external magnetic field. The MNCs-treated cells showed a higher increase in temperature than the controls. Similar with the SAR results, cells treated with the 60 nm MNCs demonstrated the highest and fastest increase in temperature (47 °C) within 10 min (Fig. 4b). From these results, the 60 nm MNC particles appear to be an appropriate magnetic material size for thermal therapy because the heating of cells to temperatures between 42 and 46 °C (315–319 K) resulted in the death of tumor cells. Above this temperature, healthy cells could be affected to induce necrosis. The cell viability was determined by the WST-1 assay after MNCs treatment. Both the 30 and 60 nm sized MNCs did not exert acute cytotoxicity with various concentrations of the particle solutions in the absence of a magnetic field (Fig. 5a). Under the AMF conditions, the 60 nm MNC dramatically reduced cell viability within 10 min, while the 30 nm MNCs did not significantly decrease cell viability, possibly because the produced temperature (43 °C) was not enough to induce cell death. This was consistent with the results observed with the hyperthermia experiments in the tube. At this point, we suggest that magnetic hyperthermia for cancer therapy could induce cell death at temperatures over 43 °C. The changes in the expression levels of the bCSC biomarkers during the magnetic hyperthermia were determined by flow cytometry. The intensity of ALDH1 and CD44+/CD24− expressions decreased, which is consistent with the cell viability results (Fig. 5b and S5a†). To understand the mechanism of cell death induced by thermal therapy, the expression levels of heat shock protein 70 and 90 (HSP70 and HSP90), which respond to thermal stress and provide transient thermotolerance to cells on exposure to heat stress, were determined by western blot analysis (Fig. 5c). The expression levels of HSP70 and 90 were downregulated. The co-inhibition of HSP70/90, which protects against heat stress, accelerated the apoptosis process.22 To further understand the apoptosis process, the treated cells were stained with annexin V-FITC and PI (apoptosis kit, Invitrogen) and subjected to flow cytometry (Fig. S5b†). The results indicated that the 60 nm sized MNC generated enough heat and thermal stress to promote the thermal apoptosis process under the AMF conditions.
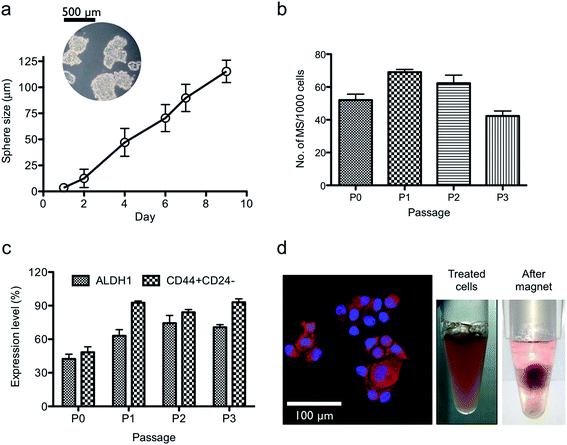 | ||
| Fig. 3 Mammosphere (MS) formation. (a) The size of the cultured MS at different days; diagram showing MS cells on day 9 (Fig. S4A†). (b) The number of MS per 1000 cells as the number of passage increases. (c) The expression level of ALDH1 and CD44+/CD24− as biomarkers for bCSCs at each passage culturing (Fig. S4B†). (d) Confocal microscopic image of the MS cells treated with red fluorescent MNC particles (left) and the floating cells after MNC treatment (right). | ||
 | ||
| Fig. 5 Magnetic hyperthermia effect of the prepared MNCs on bCSCs under the optimized AMF conditions (290 kHz and 60 kA m−1). (a) Cell viability at different concentrations of the MNCs (in the absence of an AMF, left) and cell viability at different treatment times of AMF (right). (b) Expression levels of ALDH1 and CD44+/CD24− during AMF treatment (Fig. S5a†). (c) Expression levels of HSP70 and 90 proteins after thermotherapy characterized by western blot analysis (left) and normalized against β-actin (right). | ||
Conclusions
Malignant cancer cells, especially CSCs, are tumor cells that enhance disease progression, causing the expansion of primary tumor and generation of metastasis. Therefore, several studies have focused on CSC therapy; however, cells can exhibit resistance against radiation and/or chemotherapy. Thermal therapy such as magnetic hyperthermia was proposed as a great tool for CSC treatment. To improve the effect of thermal therapy, the size distribution of materials and external magnetic field properties should be optimized. The MNC particle system is useful for magnetic hyperthermia owing to its sustainable superparamagnetic property. However, the correlation between heat generation ability (SAR) and other factors such as size and applied magnetic field remains to be explored. The SAR of the prepared MNCs was proportional to the external magnetic frequency and field strength. Interestingly, the 60 nm MNC showed the highest heat producing potential and appropriately increased the temperature to induce cell death. As a proof-of-concept, bCSCs were treated with the MNCs, and were exposed to the AMF conditions (290 kHz and 60 kA m−1). The 60 nm MNC exhibited a significant increase in temperature within 10 min for inducing cell death compared to MNCs of different sizes. During magnetic hyperthermia, the particles influenced HSP70/90 damage in the cell cytosol, and the co-inhibition of these heat-shock proteins enhanced the apoptotic cell death response of bCSC. We investigated the important conditions for magnetic hyperthermia for bCSC treatment: (i) the MNC size was optimized under various external magnetic conditions (frequency and strength), (ii) bCSC thermal therapy was established under the optimized MNC and AMF conditions (iii) the mechanism of thermal therapy was established at the protein level. The optimized hyperthermia system will be useful for a variety of studies on magnetic nanomaterials, CSC therapy, clinical magnetic hyperthermia, and other advanced nanobiotechnology applications such as heat-mediated drug/gene delivery and thermal activation of metabolic signals in cells.Acknowledgements
This work was funded by a grant by the Industrial Strategic Technology Development Program (project no. 10047679) of the Ministry of Trade, Industry & Energy (MI, Korea). The Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (2015M3C7A1028393). We thank the Institute for Basic Science-Center for Functional Interfaces of Correlated Electron Systems (IBS-CCES), Department of Physics and Astronomy, Seoul National University for measurements of Magnetic Property Measurement System (MPMS).References
- A. K. Croker and A. L. Allan, J. Cell. Mol. Med., 2008, 12, 374–390 CrossRef CAS PubMed.
- C. Tang, B. T. Anq and S. Pervaiz, FASEB J., 2007, 21, 3777–3785 CrossRef CAS PubMed.
- J. Zhao, Pharmacol. Ther., 2016, 160, 145–158 CrossRef CAS PubMed.
- Z. X. Yang, Y. H. Sun, H. G. He and G. Q. Jiang, Oncol. Lett., 2015, 10, 3443–3449 Search PubMed.
- T. Sadhukha, L. Niu, T. S. Wiedmann and J. Panyam, Mol. Pharm., 2013, 10, 1432–1441 CrossRef CAS PubMed.
- P. T. Yin, S. Shah, N. J. Pasquale, O. B. Garbuzenko, T. Minko and K. B. Lee, Biomaterials, 2016, 81, 46–57 CrossRef CAS PubMed.
- Y. Piñeiro-Redondo, M. Bañobre-López, I. Pardiñas-Blanco, G. Goya, M. A. López-Quintela and J. Rivas, Nanoscale Res. Lett., 2011, 6, 383–389 CrossRef PubMed.
- F. M. Kievit and M. Zhang, Acc. Chem. Res., 2011, 44, 853–862 CrossRef CAS PubMed.
- B. Mehdaoui, A. Meffre, J. Carrey, S. Lachaize, L. Lacroix, M. Gougeon, B. Chaudret and M. Respaud, Adv. Funct. Mater., 2011, 21, 4573–4581 CrossRef CAS.
- M. Ahamed, M. J. Akhtar, M. A. Siddiqui, J. Ahmad, J. Musarrat, A. A. Al-Khedhairy, M. S. AlSalhi and S. A. Alrokayan, Toxicology, 2011, 282, 101–108 CrossRef PubMed.
- B. Mehdaoui, R. P. Tan, A. Meffre, J. Carrey, S. Lachaize, B. Chaudret and M. Respaud, Phys. Rev. B: Solid State, 2013, 87, 174419–174428 CrossRef.
- J. Mohapatra, S. Nigam, J. Gupta, A. Mitra, M. Aslam and D. Bahadur, RSC Adv., 2015, 5, 14311–14321 RSC.
- J. Chandler and E. Lagasse, Stem Cell Res. Ther., 2010, 1, 13–22 CrossRef PubMed.
- T. Sadhukha, L. Niu, T. Wiedmann and J. Panyam, Mol. Pharm., 2013, 10, 1432–1441 CrossRef CAS PubMed.
- J. Cha, Y. Kwon, T. Yoon and J. Lee, Chem. Commun., 2013, 49, 457–459 RSC.
- L. Lin, A. Liu, Z. Peng, H. J. Lin, P. K. Li, C. Li and J. Lin, Cancer Res., 2011, 71, 7226–7237 CrossRef CAS PubMed.
- J. Ranke, K. Molter, F. Stock, U. Bottin-Weber, J. Poczobutt, J. Hoffmann, B. Ondruschka, J. Filser and B. Jastorff, Ecotoxicol. Environ. Saf., 2004, 58, 396–404 CrossRef CAS PubMed.
- X. Hu, J. Gong, L. Zhang and J. C. Yu, Adv. Mater., 2008, 20, 4845–4850 CrossRef CAS.
- A. E. Deatsch and B. A. Evans, J. Magn. Magn. Mater., 2014, 354, 163–172 CrossRef CAS.
- A. P. Khandhar, R. M. Ferguson and K. M. Krishnan, J. Appl. Phys., 2011, 109, 07B310 CrossRef PubMed.
- G. Dontu, W. M. Abdallah, J. M. Foley, K. W. Jackson, M. F. Clarke, M. J. Kawamura and M. S. Wicha, Genes Dev., 2003, 17, 1253–1270 CrossRef CAS PubMed.
- X. B. Cui, Z. Y. Yu, W. Wang, Y. Q. Zheng, W. Liu and L. X. Li, Integr. Cancer Ther., 2012, 11, 61–67 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Characterization data for the magnetic nanoclusters and breast cancer stem cells. See DOI: 10.1039/c6ra22382f |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2016 |