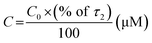DOI:
10.1039/C6RA22124F
(Paper)
RSC Adv., 2016,
6, 110610-110621
Features of partial encapsulation of an ESIPT probe 3-hydroxy-2-naphthoic acid (3HNA) in the nano cavities of β- and γ-cyclodextrin: comparative study with sequestered 3HNA in micelles and reverse micelle†
Received
3rd September 2016
, Accepted 14th November 2016
First published on 15th November 2016
Abstract
The role of intramolecular H-bonding within an excited state intramolecular proton transfer (ESIPT) probe, the intermolecular H-bonding ability of the microenvironment with the probe in different organized assemblies, the polarity of the medium after encapsulation and the motional restriction of the probe have been investigated by monitoring the ESIPT emission as well as the localized π–π* emission of 3-hydroxy-2-naphthoic acid (3HNA) in various confined media like cyclodextrins, micelles and reverse micelle. The steady state and time-resolved fluorescence and also the time resolved anisotropy measurements of the encapsulated probe indicate the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex between the β- and γ-cyclodextrin hosts and the guest. The values of the binding constants and different thermodynamic parameters for complexation have also been reported. The results in various confined media have been correlated using control experiments on the ESIPT emission of 3HNA in mixed solvents consisting of dimethyl sulfoxide (DMSO) and water (mixture of an aprotic polar and a protic polar solvent) and also of acetonitrile (ACN) and dichloromethane (DCM) (mixture of an aprotic polar and a non-polar solvent). Theoretical calculation on the orientation of 3HNA within β- and γ-cyclodextrins has been carried out in order to explain the observed rotational correlation time of the probe in confined environments.
1 complex between the β- and γ-cyclodextrin hosts and the guest. The values of the binding constants and different thermodynamic parameters for complexation have also been reported. The results in various confined media have been correlated using control experiments on the ESIPT emission of 3HNA in mixed solvents consisting of dimethyl sulfoxide (DMSO) and water (mixture of an aprotic polar and a protic polar solvent) and also of acetonitrile (ACN) and dichloromethane (DCM) (mixture of an aprotic polar and a non-polar solvent). Theoretical calculation on the orientation of 3HNA within β- and γ-cyclodextrins has been carried out in order to explain the observed rotational correlation time of the probe in confined environments.
1. Introduction
Proton transfer, particularly the excited state intramolecular proton transfer (ESIPT) process, is very important for studying fundamental photophysical and photochemical processes.1–12 In various hydrogen-bonded molecules, the reversible transfer of a proton between the proton donor–acceptor systems within a particular distance provides wide applications of the ESIPT processes. These include development of dye lasers,13–15 optical switches, sensors, photostabilisers,16–21 biological probes,1,10–12 metal-cation chelates,22 energy/data storage devices15,20 and proton-transfer materials with future applications in electroluminescence.23–25 The importance of the ESIPT process was first viewed by Weller26 on salicylic acid (SA) and methyl salicylate (MS) and thereafter the diverse applications of the excited state photophysics deliver the most constructive and useful applications in present day research (Scheme 1).
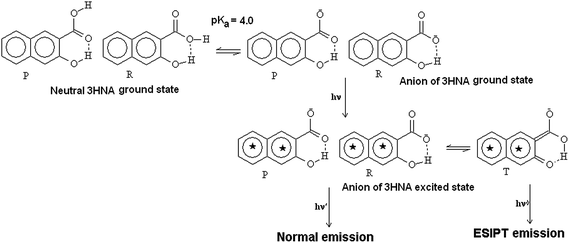 |
| | Scheme 1 Structural representation of 3HNA in its neutral and anionic form in ground and excited states. | |
3-Hydroxy 2-naphthoic acid (3HNA) exhibits dual emission having emission maxima at around 418 nm and 495–515 nm regions representing the normal localized π–π* and the ESIPT emissions respectively.16,18,27 The intensity of these two emissions depend on the nature of the solvent and the temperature.16,28 The ESIPT emission of 3HNA gets enhanced when sequestered within serum albumins at physiological pH10,11 and also in water soluble polymers (poly N-vinyl 2-pyrrolidone).29 The present work reports the steady state, time resolved fluorescence and time resolved anisotropy monitoring the ESIPT emission and also the π–π* emission of the versatile probe 3HNA in β-cyclodextrin, γ-cyclodextrin, in cationic, anionic and neutral micelles as well as in reverse micelle. The surfactants chosen for the micelles are CTAB (cationic), TTAB (cationic), and Triton-X (neutral). The stoichiometry and the binding constants along with thermodynamic parameters of the complexes with cyclodextrins have been estimated. In order to reveal the microenvironment of the probe molecule in different confined media we also present the ESIPT processes of 3HNA in mixed solvents comprising of DMSO and water (a mixture of aprotic and protic polar solvents) and also in ACN/DCM (a mixture of aprotic polar and non-polar solvent). The role of intramolecular hydrogen bonding of the probe with the environment and the intramolecular hydrogen bonding within the probe has been explored. Theoretical calculation of the optimized geometry of the complexes of 3HNA with β-cyclodextrin and γ-cyclodextrin has been carried out in order to explain the motional restriction of 3HNA within the nano-cavity of cyclodextrins as revealed by rotational correlation time of 3HNA.
2. Materials & methods
2.1 Materials
3-HNA, cyclodextrins and all the surfactants were purchased from Sigma Aldrich, Fluka and Sisco. All the solvents used were of spectral grade and further dried by standard procedures. The 3HNA was purified in ethyl alcohol by repeated crystallization. Phosphate buffer of pH 7 was prepared in doubly distilled water and used for making experimental solutions. For the preparation of reverse micelle solutions, double distilled water was injected into 0.1 M solutions of AOT in n-hexane (E Merck Germany) using a desired water content W0 = [H2O]/[AOT].
2.2 Steady-state absorption and fluorescence measurements
UV-Vis absorption spectra were recorded on a Hitachi U-4100 spectrophotometer at 298 K. The steady-state fluorescence measurements were carried out using a Hitachi Model F-7000 spectrofluorimeter equipped with a 150 W xenon lamp, at 298 K using a stopper cell of 1 cm path length. Fluorescence quantum yield was determined in each case by comparing the corrected emission spectrum of the samples with that of quinine sulfate in 0.1 N H2SO4 (ΦD = 0.54)10,30 considering the total area under the emission curve.
The measurement of steady state anisotropy was also performed in Hitachi Model F-7000 spectrofluorimeter with manual Glen Thompson polarizer. The steady state anisotropy (r) is defined by
| | |
r = (IVV − G × IVH)/(IVV + 2G × IVH)
| (1) |
where
IVV and
IVH are the intensities obtained with the excitation polarizer orientated vertically and the emission polarizer oriented vertically and horizontally, respectively. The
G factor is defined as
‘
I’ terms refer to the parameters similar to those mentioned above for the horizontal position of the excitation polarizer and vertical and horizontal position of the emission polarizer, respectively.
2.3 Time-resolved fluorescence measurements
Singlet state lifetimes were measured by Time Master Fluorimeter from Photon Technology International (PTI), USA. The system measures the fluorescence lifetime using PTI's patented strobe technique and gated detection for N2 flash lamp. The system consists of a pulsed laser driver of a PDL series i.e., PDL-800-B (from PicoQuant, Germany) with interchangeable sub nano second pulsed LEDs (370 nm having pulse width 600 ps [full width at half-maximum]) (PicoQuant, Germany) with a TCSPC set up (PTI, USA) operating at high repetition rate of 10 MHz driven by PDL 800-B driver, PicoQuant, Germany. The software Felix 32 controls all acquisition modes and data analyses of the Time Master system.31 The sample was excited using a thyratron gated nitrogen flash lamp FWHM 1.2 ns that is capable of measuring fluorescence time resolved acquisition at a flash rate of 25 kHz.
Instrument response function (IRF) were measured at the respective excitation wavelengths, namely, 356 nm (in case of nitrogen flash lamp) and 370 nm (in case of LED) using slits with a band pass of 3 nm using Ludox as the scatterer. The decay parameters were recovered using a non-linear iterative fitting procedure based on the Marquardt algorithm.31 A deconvolution technique was used to determine the lifetime up to 300–350 ps with a nitrogen flash lamp while the time resolution is 150–200 ps with sub nano-second pulsed LED. The quality of fit has been assessed over the entire decay, including the rising edge, and tested with a plot of weighted residuals and other statistical parameters e.g. the reduced χ2 ratio and the Durbin–Watson (DW) parameters.31
Anisotropy decay measurements were also carried out in TCSPC from PTI, USA using motorized Glen Thompson polarizer. The anisotropy, r(t) is defined as
| | |
r(t) = [IVV(t) − G × IVH(t)]/[IVV(t) + 2 × G × IVH(t)]
| (3) |
where
I(
t) terms are defined as intensity decay of ESIPT emission of 3HNA with excitation polarizer orientated vertically and the emission polarizer oriented vertically and horizontally, respectively:
where
G is the correction term for the relative throughput of each polarization through the emission optics. The entire data analysis was done with the software Felix 32 which analyses the raw data
IVV and
IVH simultaneously by global multi-exponential program and then the deconvolved curves (ID
VV and ID
VH) are used to construct
r(
t)
31 and from the fitted curve the rotational correlation time (
θc) can be recovered.
2.4 Theoretical calculations
All theoretical calculations were performed using GAUSSIAN 09 software package. A Quantum chemical ab initio calculations were carried out to investigate the structures and properties for the inclusion complexes of 3-HNA into β-cyclodextrin and into γ-cyclodextrin. The structural parameters known from the X-ray studies were used as a starting point for β-cyclodextrin and γ-cyclodextrin. Firstly full geometry optimization of β-cyclodextrin and γ-cyclodextrin molecules was performed at the program default conditions without any symmetry constrain imposed using DFT at M06-2X/6-31G(d,p)32 level of theory in the gas phase. The optimised structure of β-CD and γ-CD is used as an initial condition of (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) inclusion complex with 3HNA for further optimisation.
1) inclusion complex with 3HNA for further optimisation.
3. Results and discussion
3.1 Absorption studies at 298 K
The absorption spectra of free 3HNA (20 μM) and 3HNA (20 μM) in the presence of β-CD (6 mM) in aqueous medium at 298 K are provided in the Fig. 1. Similar spectra for 3HNA (20 μM) were observed in the presence of γ-CD. The absorption spectra in all the cases show two absorption bands.16,33,34 One is the intense 1La band of naphthalene moiety in the region 250–300 nm and the other is a relatively weak 1Lb band in the region 320–400 nm. With the addition of CD the absorbance is found to increase to a small extent with no significant shift in the λmax (Fig. 1). This is some indication of the formation of a host–guest inclusion complex as observed in different cyclodextrin–probe complexes.35,36 The absorption spectra of 3HNA (20 μM) in CTAB is also shown in Fig. 1. The absorption spectra of 3HNA in TTAB and other neutral micelles are similar.
 |
| | Fig. 1 Absorption spectra of 3HNA (20 μM) in (a) aqueous buffer, (b) β-CD (6 mM), (c) CTAB (1 mM) at 298 K. | |
3.2 Steady state emission and fluorescence lifetime studies of 3HNA in aqueous medium at 298 K
3.2.1 In the cyclodextrins. The room temperature (298 K) emission spectra of 3HNA (20 μM) as a function of increasing concentration of aqueous β-CD (0.2–6.0 mM) are shown in Fig. 2A. The emission profile indicates that there has been a steady increase in the ESIPT emission of 3HNA in the presence of β-CD with a small blue shift of the λmax of the emission maxima (from 510 nm in water to 504 nm) (Table 1) without any emission from the localized π–π* state. Similar phenomena are also observed for γ-CD. The quantum yield (ΦD) of the ESIPT emission of 3HNA (20 μM) in β-CD is found to be higher than that in the γ-CD (Table 1). The changes of quantum yields of ESIPT emission with the concentration of cyclodextrin are shown in Fig. 3. The blue shift of the λmax probably indicates the inclusion of 3HNA into the non-polar or hydrophobic interior of cyclodextrins.
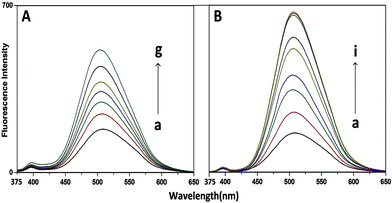 |
| | Fig. 2 [A] Fluorescence spectra of 3HNA (20 μM) at 298 K with varying concentration of β-CD. Curves (a–g) represent 0, 0.2, 0.4, 0.7, 1, 2, 6, mM β-CD respectively; λexc = 350 nm; excitation band pass 10 nm, and emission band pass = 5 nm. [B] Fluorescence spectra of 3HNA (20 μM) at 298 K with varying concentration of CTAB; curves (a–i) represents 0, 0.06, 0.08, 0.12, 0.2, 0.4, 0.8, 1, 2 mM CTAB, respectively; excitation band pass = 10 nm and emission band pass = 5 nm. | |
Table 1 Quantum yield, average lifetime and rotational correlation times of ESIPT emission of 3HNA in different systems
| 3HNA (20 μM) in |
λmax (nm) |
Quantum yield (ΦF) |
Average singlet state lifetime (〈τ〉) (ns) |
Rotational correlation time, θc (ns) using anisotropy decay |
| Water |
510 |
0.08 |
1.7 (100%) |
— |
| β-CD (6 mM) |
504 |
0.23 |
3.2 |
1.0 |
| γ-CD (30 mM) |
504 |
0.16 |
2.3 |
1.8 |
| CTAB (1 mM) |
508 |
0.32 |
5.2 |
0.5 |
| TTAB (4 mM) |
508 |
0.31 |
5.0 |
0.5 |
| Triton-X (20 mM) |
505 |
0.32 |
4.7 |
0.6 |
 |
| | Fig. 3 Variation of (i) fluorescence quantum yield (Φ) (ii) fluorescence anisotropy (r) (iii) singlet state average lifetime 〈τ〉 of 3HNA (20 μM) in aqueous buffer monitoring the ESIPT emission with increasing concentrations of (A) β-CD and (B) γ-CD. | |
The decay of the ESIPT emission of 3HNA in aqueous buffer was best fitted with a single exponential function and the lifetime recovered as 1.7 ns (100%)10 (Table 2 and Fig. 4). One component fit suggests the presence of a single fluorescent species in a particular environment. However, the existence of multiple environments exchanging rapidly within the lifetime cannot be ruled out.37,38 In the CD environments the decay of the ESIPT emission becomes biexponential (Table 2 and Fig. 4). The biexponential decays are found to best fit with a fixed shorter component (similar to the lifetime value of the guest in the bulk aqueous medium) and a relatively longer component (Table 2). With the varying concentration of cyclodextrins, the relative contribution of the shorter component decreases progressively with concomitant increase of the contribution of the longer component in both the cyclodextrins (Table 2). The average lifetimes of 3HNA are found to increase with the gradual increase in the concentration of β-CD (Fig. 4) and γ-CD which are consistent with the steady state fluorescence data (Table 2).
Table 2 Singlet state lifetime data of 3HNA–β-cyclodextrin complex monitoring the ESIPT emission of 3HNA (20 μM) at 298 K
| [β-CD] (μM) |
[3 HNA] (μM) |
τ1 (%) (ns) |
τ2 (%) (ns) |
〈τ〉 (ns) |
C0 (μM) |
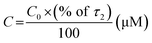
|
| 0 |
20 |
1.66 (100%) |
— |
1.66 |
20 |
— |
| 200 |
20 |
1.66 (83.0%) |
3.70 (17.0%) |
2.05 |
20 |
3.4 |
| 400 |
20 |
1.66 (71.5%) |
3.90 (28.5%) |
2.31 |
20 |
5.8 |
| 700 |
20 |
1.66 (62.0%) |
3.90 (38.0%) |
2.53 |
20 |
7.6 |
| 1000 |
20 |
1.66 (53.6%) |
3.87 (46.4%) |
2.67 |
20 |
92.5 |
| 2000 |
20 |
1.66 (40.5%) |
3.86 (59.5%) |
2.96 |
20 |
11.9 |
| 6000 |
20 |
1.66 (28.7%) |
3.85 (71.3%) |
3.22 |
20 |
14.3 |
 |
| | Fig. 4 Fluorescence decay of 3HNA (20 μM) monitoring the ESIPT emission at 298 K in aqueous buffer (pH 7) with varying concentration of β-CD, (a–h) represents 0, 200, 400, 700,1000, 2000, 6000, 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM β-CD; λexc = 370 nm; excitation and emission band pass = 10 nm each. 000 μM β-CD; λexc = 370 nm; excitation and emission band pass = 10 nm each. | |
For β-CD–probe complex the contribution of the shorter component is 29% (Table 2) at the saturation level whereas for γ-CD–probe complexes the contribution is about ∼55%. Also the average lifetime values recovered were 3.2 ns for the β-CD–probe complex and 2.3 ns for the γ-CD–probe complexes at their saturation level (Table 1). This is indicative of a higher degree of encapsulation of the probe 3HNA in the β-CD environment as compared to that in the γ-CD environment. This may be due to the fact that most of the probe molecules experience greater degrees of freedom in the bulk aqueous environment causing a decrease in the intramolecular H-bond formation via ESIPT emission and increased intermolecular H-bond with the solvent molecules for γ-CD–probe complex than that in the case of the β-CD–probe complex. The fluorescence quantum yield values obtained in both the cases strongly support this contention (Table 1).
3.2.2 In the micelle. In the presence of cationic surfactants (water medium) the ESIPT emission of 3HNA increases and gets saturated at the critical micelle concentration (CMC) value of surfactants. Fig. 2B represents the emission spectra of 3HNA (20 μM) as a function of increasing concentration of CTAB in water. The quantum yield (Φ) of the ESIPT emission of 3HNA (20 μM) increases by four times compared to that in the pure aqueous solution (Fig. 2B, Table 3). From the plot of quantum yield (Φ) of the ESIPT emission vs. added concentration of cationic surfactant, the CMC value has been evaluated which is found to match well the standard value39 (Fig. 5A). The maxima (λmax) of the ESIPT emission spectra remain same. The enhancement of the ESIPT emission was also reflected in the singlet state decay (Fig. 6). In the presence of CTAB, the decay exhibits two components of ∼5.5 ns and 1.7 ns (Table 3). As the concentration of the CTAB increases the percentage contribution of the shorter component gradually decreases with the simultaneous increase of that of the longer component (Table 3). The longer component may thus be assigned as the lifetime of 3HNA upon its interaction with CTAB. The plots of the quantum yield and the average lifetime of 3HNA vs. concentration of CTAB are shown in Fig. 5A and C respectively. The quantum yield and the life time value of the ESIPT emission of 3HNA in 1 mM CTAB match well with that in the 4 mM poly N-vinyl-2-pyrrolidone (PVP) polymer in aqueous medium.29 Thus it may be concluded that the microenvironment of 3HNA in the CTAB is quite similar to that in the PVP. A similar phenomenon is observed with TTAB though the relative quantum yield of the ESIPT emission of 3HNA is slightly greater (∼1.03 times) in CTAB compared to that in TTAB as shown in the Fig. S1† (Table 1). However, with anionic surfactants (SDS), no significant change in the ESIPT emission spectra of 3HNA was observed (data not shown). In case of neutral surfactants ESIPT emission may increase or remain same depending upon the nature of the surfactant used. With Triton-X, ESIPT emission increases by same amount as that of CTAB (Fig. S1†). But the difference is that in the case of CTAB and TTAB, the ESIPT emission increases up to the CMC values where as for Triton-X this ESIPT emission increases even beyond the CMC value. The ESIPT emission of 3HNA in 0.02 M Triton-X is shown in Fig. S1.† The ESIPT emission gets saturated at a high concentration of Triton-X which is almost 80 times greater than its CMC value. The maxima (λmax) of the emission spectra gradually shift towards blue region (from 510.0 nm in water to 505.0 nm in Triton-X) (Table 1). This indicates that the local environment of the 3HNA is more hydrophobic in the presence of Triton-X. There is no significant change in the ESIPT emission of 3HNA with Tween-60, used as neutral surfactant.
Table 3 3HNA–CTAB interaction; as a function of concentration of CTAB monitoring the ESIPT emission of 3HNA (20 μM) at 298 K
| Concn of CTAB (mM) |
Quantum yielda (ΦF) |
τ1 (ns) |
τ2 (ns) |
〈τ〉 (ns) |
Steady state anisotropy (r) |
| Error in the measurement = ±2%. |
| 0.00 |
0.080 |
1.66 (100%) |
— |
1.66 |
0.003 |
| 0.06 |
0.116 |
1.66 (82%) |
5.6 (18%) |
2.4 |
0.007 |
| 0.08 |
0.159 |
1.66 (56%) |
5.6 (44%) |
3.4 |
0.009 |
| 0.12 |
0.187 |
1.66 (27%) |
5.6 (73%) |
4.5 |
0.010 |
| 0.20 |
0.247 |
1.66 (14%) |
5.7 (86%) |
5.1 |
0.012 |
| 0.40 |
0.282 |
1.66 (4.4%) |
5.5 (95.6%) |
5.3 |
0.013 |
| 0.60 |
0.304 |
1.66 (2%) |
5.3 (98%) |
5.2 |
0.013 |
| 1.00 |
0.320 |
1.66 (1%) |
5.2 (99%) |
5.2 |
0.014 |
| 2.00 |
0.320 |
1.66 (1%) |
5.2 (99%) |
5.2 |
0.014 |
 |
| | Fig. 5 (A) Variation of fluorescence quantum yield (Φ); (B) fluorescence anisotropy; (C) singlet state average lifetime (s) of 3HNA (20 μM) in aqueous medium monitoring the ESIPT emission with increasing concentration of CTAB at 298 K. | |
 |
| | Fig. 6 Fluorescence decay of 20 μM 3HNA monitoring the ESIPT band at 510 nm in (a) 0 mM, (b) 0.04 mM, (c) 0.06 mM, (d) 0.12 nM (e) 0.2 mM (f) 1 mM CTAB in aqueous medium at 298 K; λexc = 370 nm; excitation band pass = 5 nm and emission band pass = 5 nm. | |
3.2.3 In the reverse micelle. The photophysical properties of 3HNA in the sodium di-2-ethylhexylsulfosuccinate (AOT)/n-hexane/water reverse micelle having W0 = 2, 5, 7 were investigated. In the n-hexane medium 3HNA exhibits a structured emission spectra (Fig. 7) from locally excited lowest 1(π–π*) state peaking around 400–450 nm. This fluorescence emission is well resolved exhibiting (0,0) band at 404 nm and other vibronic bands at 428, 453 and 486 nm. In the presence of 0.1 M AOT in n-hexane, 3HNA exhibits both ESIPT emission and emission from the local excited state. The presence of ESIPT emission can be explained due to the fact that, in the presence of AOT, the n-hexane medium becomes polar to some extent (as AOT is an ionic surfactant) leading to intramolecular hydrogen bonding in the ESIPT state. In the AOT/n-hexane/water reverse micelle, 3HNA exhibits both the local and ESIPT emissions. These emission intensities gradually decreases as one moves from W0 = 2 to W0 = 7 (Fig. 7). The position of the local band remains same in all the cases. The decays of the local band of 3HNA in different systems nicely fitted with two components of 1 ns and 0.6 ns (Fig. 8 and Table 4). The average lifetime value of the local band slightly decreases in the reverse micelles as one moves from W0 = 2 to W0 = 7 (Table 4). However the position of the λmax of the ESIPT band was difficult to locate owing to the overlapping of the two bands (Fig. 8). The decrease in the intensity of the ESIPT band (in reverse micelles having W0 = 2 to W0 = 7) is also reflected in the life time values (Table 4). The decay profiles monitored at 515 nm were found to fit nicely with two components (having a fixed component of ∼1 ns). From Table 4, it is clear that the fixed component is the contribution of the local band. The time resolved emission spectra (TRES) of 3HNA in 0.1 M AOT were measured in n-hexane under different conditions. Fig. 7 (inset) represents the TRES obtained at delay of 1 ns and 10 ns following excitation. The TRES at longer times (10 ns) shows that the intensity of the ESIPT emission increases compared to that of the local emission. This clearly indicates that the species emitting at higher wavelength is predominant at longer times. As the local emission is predominant in the reverse micelles, we may conclude that 3HNA interacts with the hydrophobic region of AOT.
 |
| | Fig. 7 Fluorescence spectra of 3HNA (20 μM) at 298 K in n-hexane medium; (a) in reverse micelle having W0 = 2, (b) in reverse micelle having W0 = 5, (c) in reverse micelle having W0 = 7, (d) 100 mM AOT, (e) in pure n-hexane; excitation band pass = 10 nm and emission band pass = 5 nm. Inset: time resolved emission spectra (TRES) of 20 μM 3HNA in 0.1 M AOT in n-hexane at delay of (a) 1 ns and (b) 10 ns following excitation. λexc = 356 nm. Excitation band-pass = 10 nm, emission bandpass = 10 nm. | |
 |
| | Fig. 8 Fluorescence decay of 20 mM 3HNA in n-hexane monitoring the local band at 427 nm in (a) pure n-hexane, (b) reverse micelle with Wo = 2, and monitoring the ESIPT band at 515 nm in (c) reverse micelle with Wo = 7, (d) reverse micelle with Wo = 5, (e) reverse micelle with Wo = 2 (f) 100 mM AOT; λexc = 370 nm; excitation band pass = 5 nm and emission band pass = 3 nm at 298 K. | |
Table 4 Singlet state life time values of 20 μM 3HNA in reverse micelle of AOT/n-hexane/water system
| System |
λmonitored (nm) |
τ1 (%) (ns) |
τ2 (%) (ns) |
〈τ〉 (ns) |
χ2 |
| 3HNA in n-hexane |
427 |
1.1 (52%) |
0.60 (48%) |
0.85 |
1.12 |
| 3HNA + 0.1 M AOT in n-hexane |
427 |
1.1 (54%) |
0.60 (46%) |
0.87 |
1.10 |
| 515 |
1.2 (69%) |
6.01 (31%) |
2.70 |
1.18 |
| 3HNA in reverse micelle with W0 = 2 |
427 |
1.0 (89%) |
0.56 (11%) |
0.97 |
1.07 |
| 515 |
1.1 (75%) |
4.40 (25%) |
1.92 |
1.05 |
| 3HNA in reverse micelle with W0 = 5 |
427 |
1.0 (80%) |
0.60 (20%) |
0.92 |
1.14 |
| 515 |
1.1 (75%) |
3.12 (25%) |
1.60 |
1.14 |
| 3HNA in reverse micelle with W0 = 7 |
427 |
1.0 (75%) |
0.60 (25%) |
0.90 |
0.95 |
| 515 |
1.1 (71%) |
2.91 (29%) |
1.61 |
1.09 |
3.3 Cyclodextrin–3HNA binding study from steady state emission spectra at 298 K
The mode of encapsulation between the cyclodextrins and the probe 3HNA was quantitatively estimated by modified Benesi–Hildebrand equation40 for determining the stoichiometry of the complexes, binding constant (K) and associated free energy change changes (ΔG0) of the process using the eqn (1) for the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host
1 host![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) guest complexes
guest complexes| | |
1/ΔF = 1/ΔFmax + (1/K[L])(1/ΔFmax)
| (5) |
where ΔF = Fx − F0 and ΔFmax = F∞ − F0 and F0, Fx and F∞ are the fluorescence intensities of the particular species of 3HNA considered in the absence of β-CD and γ-CD, at an intermediate β-CD and γ-CD concentration and at a concentration of β-CD and γ-CD for fluorescence saturation, respectively. K is the binding constant and [L] is the β-CD and γ-CD concentration. Rearrangement of eqn (5) leads to the following simpler form:| | |
(F∞ − F0)/(Fx − F0) = 1 + (K1[L])−1
| (6) |
Similarly, for 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest complex formation the equation takes the form
1 host–guest complex formation the equation takes the form
| | |
(F∞ − F0)/(Fx − F0) = 1 + (K2[L])−2
| (7) |
K1 and
K2 are the binding constants for the 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
1 and the 2
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
1 host–guest complexes respectively. Linearity in the plot of (
F∞ −
F0)/(
Fx −
F0) against [L]
−1 [from
eqn (6)] confirms a 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
1 interaction of β-CD and γ-CD with 3HNA (Fig. S2A
†).
Eqn (7) shows the nonlinear regression with a curvature.
The binding constant values have been determined from the slope of the plot of (F∞ − F0)/(Fx − F0) against [L]−1 (Table 5). The binding constant for the complex of β-CD is found to be greater than that for the complex of γ-CD in our case (Table 5).
Table 5 Binding constants (Kb) and the thermodynamic parameters (ΔG, ΔH and ΔS) associated with binding following different methods monitoring the ESIPT emission of 3HNA (20 μM) in the complex with β-CD (6000 μM) and γ-CD (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM) at 298 K
000 μM) at 298 K
| System |
Method |
Binding constant (Kb) (M−1) |
ΔG0 (kJ mol−1) |
ΔH0 (kJ mol−1) |
ΔS0 (kJ mol−1 K−1) |
3HNA–β-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300) 300) |
Modified Benesi–Hildebrand equation |
1.000 × 103 |
−17.1 |
−3.80 |
0.0446 |
| Steady state fluorescence anisotropy |
1.200 × 103 |
−17.6 |
0.0463 |
| Time resolved fluorescence decay |
1.000 × 103 |
−17.1 |
0.0446 |
3HNA–γ-CD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1500) 1500) |
Modified Benesi–Hildebrand equation |
0.040 × 103 |
−9.0 |
−5.76 |
0.0109 |
| Steady state fluorescence anisotropy |
0.030 × 103 |
−8.3 |
0.0085 |
| Time resolved fluorescence decay |
0.035 × 103 |
−8.7 |
0.0098 |
The binding constants determined from steady state anisotropy of the ESIPT emission and also from the time resolved emission studies are provided in Table 5 [see eqn (S1)–(S3) and Fig. S2B and C† for detail study].
3.4 Effect of temperature on the fluorescence behavior of the cyclodextrin–guest complexes: thermodynamic parameter of binding
Temperature variation of the ESIPT emission of 20 μM 3HNA in 6 mM β-CD and 30 mM γ-CD in water has been carried out (Fig. S3†). Though the local 1(π–π*) emission remains practically constant, the quantum yield of the ESIPT emission gradually decreases with increase in temperature. Variation of emission intensity with temperature is utilized to calculate the change in enthalpy (ΔH0) and the change in entropy (ΔS0) of the binding of 3HNA with cyclodextrins using the Van't Hoff reaction isotherm (assuming ΔH is temperature independent in the temperature range studied). The plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T gives a straight line (Fig. S3,† inset); from the slope of the line ΔH0 can be calculated. The values of ΔG0, ΔH0 and ΔS0 for binding are provided in Table 5.
K vs. 1/T gives a straight line (Fig. S3,† inset); from the slope of the line ΔH0 can be calculated. The values of ΔG0, ΔH0 and ΔS0 for binding are provided in Table 5.
3.5 Control experiments
3.5.1 Steady state fluorescence data of 3HNA in water/DMSO mixtures. The emission spectra of 3HNA in the mixed solvents of water and a polar aprotic solvent DMSO are shown in Fig. S4.† The λmax of the ESIPT emission band of 3HNA (20 μM) in water/DMSO mixtures are presented in Table 6. Fig. S5A† represents a plot of transition energy (ET in kcal mol−1) of ESIPT emission of 3HNA (20 μM) in mixed solvents against the solvent polarity index ET(30)-scale (kcal mol−1) by Reichardt.41 The plot is found to be linear. The plot clearly indicates that the transition energy of ESIPT emission decreases with the increase in ET(30) value41,42 of the mixed solvents. This implies that the stabilization of the ESIPT state is associated with an increase in solvent polarity index and consequently the emission shifts towards higher wavelength (Table 6).
Table 6 Solvent and photophysical parameters of the 3HNA (20 μM) in water/DMSO mixtures at 298 K
| χH2O |
ET(30) (kcal mol−1) |
π* |
α |
β |
λem (nm) |
Δ (cm−1) |
Φ |
〈τ〉 ns |
| 1 |
63.1 |
1.09 |
1.17 |
0.47 |
510.0 |
8963 |
0.08 |
01.7 |
| 0.940 |
60.6 |
1.12 |
0.94 |
0.54 |
507.2 |
8854 |
0.14 |
03.1 |
| 0.886 |
56.8 |
1.12 |
0.70 |
0.57 |
502.8 |
8682 |
0.25 |
04.9 |
| 0.725 |
53.6 |
1.11 |
0.49 |
0.59 |
498.8 |
8522 |
0.42 |
08.2 |
| 0.497 |
49.9 |
1.05 |
0.32 |
0.66 |
495.2 |
8377 |
0.70 |
12.8 |
| 0.305 |
47.6 |
1.01 |
0.20 |
0.69 |
493.6 |
8311 |
0.87 |
14.1 |
| 0 |
45.0 |
0.98 |
0.00 |
0.76 |
490.0 |
8162 |
0.94 |
16.0 |
Fig. S5B† represents a plot of transition energy of ESIPT emission of 3HNA (20 μM) in different composition of water/DMSO mixed solvents against the hydrogen bond donating ability (α, Table 6) of the mixed solvents. The plot indicates the fact that the hydrogen bond donating ability (α), of the solvents increases the stability of the ESIPT state. In order to determine the influence of solvent polarity (π*), hydrogen bond donating (α) and hydrogen bond accepting (β) ability of the mixed solvents of water/DMSO having different composition on ESIPT emission of 3HNA, Kamlet–Taft multi-parameter approach43 is employed. These parameters for mixed solvents43–46 along with Stokes shift of ESIPT emission of 3HNA (20 μM) in those mixed solvents are given in Table 6. A linear regression analysis was done taking into account all the three solvent parameters along with Stokes shift (Δ) in the mixed solvents and this reduces to the relation (Fig. S5C†):
| | |
Δ = 7058.75 + 618.84π* + 790.07α + 657.18β
| (8) |
An excellent connection between the Stokes shift (Δ) of ESIPT emission and the solvent parameters of Kamlet–Taft42 is found when all the solvent parameters are considered simultaneously (Fig. S3C†). This clearly indicates that the polarity, the hydrogen bond donating ability as well as the hydrogen bond accepting ability of the solvents have an insightful control on the ESIPT emission of 3HNA.
3.5.2 Steady state fluorescence data of 3HNA in acetonitrile (ACN)/dichloromethane (DCM) mixtures. Fig. S6† represents the emission spectra of 3HNA (20 mM) in mixed solvent containing a polar aprotic solvent ACN and DCM. In pure DCM, only the local π–π* emission of 3HNA is observed.10 As the percent volume of ACN in the mixed solvent increases, the quantum yield of the ESIPT emission gradually increases with a simultaneous blue shift in the emission maxima. The plot of the quantum yield of the ESIPT emission of 20 μM 3HNA against dielectric constant (ε) of the mixed solvents composed of ACN and DCM is shown in Fig. S6† (inset).
3.5.3 Effect of micro polarity around the fluorophore in various systems. The polarity of the solvent molecule plays an important role in determining the dynamics of ESIPT. Furthermore, the ESIPT emission is highly sensitive to external H-bonding perturbation with the solvent molecule which can compete with that of the intramolecular H-bonding of the probe.10,29 The quantum yield and the position of the λmax of the ESIPT emission of 3HNA with the different non-polar and polar (protic and aprotic) solvents had been ascribed in our previous work.10,29 S. S. Maity et al. reported that the polarity and the hydrogen bond donating ability of the polar protic solvents play a balancing role in determining the extent of quantum yield of polar protic solvents and for polar aprotic solvents the quantum yield is governed only by the polarity of the solvents. The maximum quantum yield observed in DMSO could be attributed to the aprotic nature of the solvent where intramolecular hydrogen bonding in 3HNA is most favorable. In protic solvents, intermolecular hydrogen bonding with the solvents is responsible for lower quantum yield.10,29During formation of the inclusion complexes in aqueous medium, the internal environments of the cyclodextrin cavities are not completely free from water molecules. In fact, the cyclodextrins (particularly β-CD) are known to be in a strained configuration due to the presence of water under these conditions though in the pure state the CD cavity is free from the water molecules. Hence, the encapsulation of the guest 3HNA molecules in the hydrophobic pocket by removing some of the water molecules out of the CD cavity are accompanied by the release of strain from the CD. This results help to minimize the intermolecular H-bonding interaction of 3HNA with water molecules keeping the intramolecular H-bond in 3HNA intact. Therefore, solvent perturbation of ESIPT through intermolecular H-bonding interaction47 is minimized in the inclusion complex of 3HNA with CDs and hence the resultant enhancement of the ESIPT emission.
3.6 Anisotropy studies
3.6.1 Steady state fluorescence anisotropy study. In the biochemical and biophysical research, steady state fluorescence anisotropy imparts a great deal of idea about the microenvironment around the fluorophore. The fluorescence anisotropy is dependent on the rotational diffusion of the fluorophore and the measure of the degree of restriction imposed by the microenvironment of the fluorophore10,29,48 and thus it helps to get information about the probable location of the guest molecule in the confined environment.35,36 Fig. 3(ii) illustrates the variation of fluorescence anisotropy (r) of 3HNA with increasing concentration of the cyclodextrin solutions. The steady state anisotropy value of 3HNA in water is 0.003.10 In presence of CD, the anisotropy values gradually increases with an increase in the CD concentration, then reaches to a saturation value (0.016 for β-CD and 0.040 for γ-CD). It implies motional restriction on the fluorophore in the complexes. This observation also confirms the incorporation of 3HNA into the nanocavities β-CD and γ-CD environment. In case of γ-CD, a higher value of anisotropy at the saturation concentrations indicates that the probe molecule is experiencing a more rigid environment in the γ-CD cavity which is a clear implication of either stronger binding or tightness of the fit of 3HNA molecule in γ-CD environment than that of the β-CD environment (see Section 3.6.2).The steady state anisotropy of the ESIPT emission was also measured in the surfactant medium. Change in steady state anisotropy of the ESIPT emission of 3HNA (20 μM) as a function of increasing concentration of CTAB is shown in Fig. 5B. In all the systems (CTAB, TTAB, Triton-X), the saturated values of steady state anisotropy of the ESIPT emission of 3HNA (20 μM) were found to be ∼0.014.
3.6.2 Time resolved anisotropy. In order to calculate the rotational correlation time (θc) of 3HNA in different systems, time resolved anisotropy decay monitoring the ESIPT emission has been carried out. The rotational correlation times (θc) of 3HNA (20 μM) in β-CD–3HNA complex and in γ-CD–3HNA complex were recovered to be 1 ns and 1.8 ns, respectively (Fig. 9). Since the binding constant of 3HNA with γ-CD is less than that of 3HNA with β-CD, the larger θc value in γ-CD indicates more rotational restriction of 3HNA in γ-CD (see theoretical calculation, Section 3.7) compared to that in β-CD. The hydrodynamic volume of the rotating species obtained from θc using eqn (9)49 has been presented in Table 7.where, η is the viscosity coefficient of the surroundings (assuming η to be same for both the systems) and V is the hydrodynamic volume of the rotating species. The volume of gyration of free 3HNA and 3HNA–β-CD, 3HNA–γ-CD-complexes obtained from theoretical calculation50 are also presented in the Table 7 for comparison. It is to be noted that the ratio of the experimental hydrodynamic volumes of the rotating species of γ-CD to β-CD complexes (1.80) matches quite well with the ratio for the volume of gyration of the complexes (1.64) obtained from theoretical calculations.
 |
| | Fig. 9 Fluorescence anisotropy decays of 3HNA (20 μM) monitoring the ESIPT emission at 298 K in 6000 μM β-CD IVV and IVH represent decays of ESIPT emission of 3HNA with excitation polarizer at vertical position and emission polarizer at vertical and horizontal position, respectively; λexc = 370 nm; excitation and emission. Band pass = 10 nm each. | |
Table 7 Volume of 3HNA–β-CD and 3HNA–γ-CD complexes
| System |
θc (exp.) (ns) |
Hydrodynamic volume of the rotating species obtained from θc (cm3 mol−1) |
Volume of gyration from theoretical calculation (cm3 mol−1) |
| θc for free 3HNA is beyond our experimental setup. |
| Free 3HNAa |
— |
— |
65.83 |
| 3HNA–β-CD-complex |
1.0 |
2635.72 |
414.28 |
| 3HNA–γ-CD-complex |
1.8 |
4744.29 |
679.51 |
The θc values of 3HNA (20 μM) in the surfactant medium are found to be near 0.5 ns (Table 1) indicating more freedom of rotation of 3HNA in micelles compared to that in cyclodextrin. Due to the overlapping of local band with the ESIPT band, it was difficult to measure the rotational correlation times of 3HNA in the reverse micelle.
3.7 Theoretical calculations: geometry of the complexes of 3HNA with cyclodextrins
Molecular structure for β-CD and γ-CD were built from their corresponding X-ray coordinates. In the first step we fully optimized these structures using DFT at M06-2X/6-31G(d,p) level of theory in the gas phase. The 3-HNA molecule was then introduced inside the β-CD and γ-CD cavity. The complex was optimized in the same level of theory mentioned above. The optimized structures are depicted in the Fig. 10A and B. All guest inclusion orientations were let to pass through the wider native ring of β-CD and γ-CD. The cavity diameter of β-CD is about 7.3 Å in contrast to the rough width of 3-HNA is 4.8 Å. The final structure of β-CD complex shows that the guest molecule enters the cavity being almost oriented along the XZ-plane whereas in the case of γ-CD complex the guest molecule is almost in XY-plane (Fig. 10A and B). This could result from the larger inner rim diameter (9.8 Å) of γ-CD (Table S1†). The analysis of host–guest inter atomic molecular distances is shown in the Table S2† showing one hydrogen bonding interaction in the case of γ-CD complex and two in the case of β-CD complex (Fig. 10C and D). In the case of β-CD complex the guest molecule is more exposed to solvent compared to that in γ-CD, since the 3HNA molecule is in its short axis in the complex of β-CD. This orientation of 3HNA in β-CD gives rise to lower θc value compared to that observed in γ-CD (Table 1). All DFT calculations were performed using Gaussian 09 package.
 |
| | Fig. 10 Optimized structures of (A) β-cyclodextrin–3HNA and (B) γ-cyclodextrin–3HNA (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) inclusion complex using DFT at M06-2X/6-31G(d,p) level. Closed view of (C) β-cyclodextrin–3HNA and (D) γ-cyclodextrin–3HNA (1 1) inclusion complex using DFT at M06-2X/6-31G(d,p) level. Closed view of (C) β-cyclodextrin–3HNA and (D) γ-cyclodextrin–3HNA (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) inclusion complex showing H-bonding interactions. Inset: schematic view of (A) 3HNA is in XZ-plane inside the β-CD cavity and (B) 3HNA is in XY-plane inside the γ-CD cavity. 1) inclusion complex showing H-bonding interactions. Inset: schematic view of (A) 3HNA is in XZ-plane inside the β-CD cavity and (B) 3HNA is in XY-plane inside the γ-CD cavity. | |
4. Conclusion
The photophysics of an ESIPT probe, 3HNA in presence of the different organized assemblies like β and γ-cyclodextrins, cationic micelle (CTAB) and reverse micelle (AOT) has been explored using steady state and time resolved fluorescence studies and also steady state and time resolved anisotropy monitoring ESIPT emission as well as the localised π–π* emission of 3HNA at room temperature. The formation of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest inclusion complexes of cyclodextrins and 3HNA, the binding constant values obtained by different methods and the rotational correlation time of the probe observed indicate that the mode of encapsulation of the 3HNA is different in the β and γ-CD hosts. In order to realise the environment of the confined guest molecule, control experiments using the mixed solvents like DMSO–H2O and ACN–DCM have been carried out. The role of microenvironments like polarity, H-bonding ability of the solvents/mixed solvents helps understand the homogeneity/heterogeneity inside the cavity of the CDs or micelles and also the rigidity of the respective environment. The marked enhancement of the ESIPT emission in presence of the confined media, the mode of complexation and the motional restriction of the probe were also corroborated by theoretical calculation of the orientation of the encapsulated 3HNA in both the cyclodextrin hosts.
1 host–guest inclusion complexes of cyclodextrins and 3HNA, the binding constant values obtained by different methods and the rotational correlation time of the probe observed indicate that the mode of encapsulation of the 3HNA is different in the β and γ-CD hosts. In order to realise the environment of the confined guest molecule, control experiments using the mixed solvents like DMSO–H2O and ACN–DCM have been carried out. The role of microenvironments like polarity, H-bonding ability of the solvents/mixed solvents helps understand the homogeneity/heterogeneity inside the cavity of the CDs or micelles and also the rigidity of the respective environment. The marked enhancement of the ESIPT emission in presence of the confined media, the mode of complexation and the motional restriction of the probe were also corroborated by theoretical calculation of the orientation of the encapsulated 3HNA in both the cyclodextrin hosts.
Acknowledgements
S. G. gratefully acknowledges Department of Science and Technology (DST), Government of India (Grant No. SB/S1/PC-003/2013) and the Council of Scientific and Industrial Research (CSIR), Government of India (Grant No. 01(2142)/07/EMR-II and 21(0871)/11/EMR-II) for financially supporting this work. PSS thank DST, GOI (Grant No. SB/FT/CS-141/2012) for Fast Track Young Scientist Project and SS thank CSIR, Govt. of India (Grant No. 01(2142)/07/EMR-II and 21(0871)/11/EMR-II) for JRF and SRF fellowship. We acknowledge Prof. Ayan Datta and Titas Kumar Mukhopadhyay of Indian Association for the Cultivation of Science, Jadavpur, Kolkata 700032, for helping in our revised DFT calculation.
References and notes
- A. Sytnik, D. Gormin and M. Kasha, Proc. Natl. Acad. Sci. U. S. A., 1994, 91, 11968–11972 CrossRef CAS.
- A. Sytnik and M. Kasha, Proc. Natl. Acad. Sci. U. S. A., 1994, 91, 8627–8630 CrossRef CAS.
- M. Kasha, J. Chem. Soc., Faraday Trans. 2, 1986, 82, 2379–2392 RSC.
- B. J. Schwartz, L. A. Peteanu and C. B. Harrish, J. Phys. Chem., 1992, 96, 3591–3598 CrossRef CAS.
- F. Laermer, T. Elsaesser and W. Kiser, Chem. Phys. Lett., 1988, 148, 119–124 CrossRef CAS.
- E. M. Kosower and D. Huppert, Annu. Rev. Phys. Chem., 1986, 37, 127–156 CrossRef CAS.
- C. Rambaud, A. Oppenlander, M. Pierre, H. P. Trommsdorff and J. C. Vial, Chem. Phys., 1989, 136, 335–347 CrossRef CAS.
- S. F. Formosinho and L. G. Arnaut, J. Photochem. Photobiol., A, 1993, 75, 21–48 CrossRef CAS.
- S. Scheiner, J. Phys. Chem. A, 2000, 104, 5898–5909 CrossRef CAS.
- S. S. Maity, S. Samanta, P. Saha Sardar, A. Pal, S. Dasgupta and S. Ghosh, Chem. Phys., 2008, 354, 162–173 CrossRef CAS.
- P. Saha Sardar, S. Samanta, S. S. Maity, S. Dasgupta and S. Ghosh, J. Phys. Chem. B, 2008, 112, 3451–3461 CrossRef PubMed.
- S. Kr Ghorai, D. Ranjan Tripathy, S. Dasgupta and S. Ghosh, J. Photochem. Photobiol., B, 2014, 131, 1–15 CrossRef PubMed.
- A. Costela and R. Sastre, Appl. Phys. B, 1995, 60, 383 CrossRef.
- U. Acuna, F. A. Gueni, A. Costela, A. Dauhal, J. M. Figurea, F. Florida and R. Sastre, Chem. Phys. Lett., 1991, 187, 98 CrossRef.
-
(a) P. T. Chou, D. MxMorrow, T. J. Aartsna and M. Kasha, J. Phys. Chem., 1984, 88, 4596 CrossRef CAS;
(b) S. Park, O. H. Kown, S. Kim, S. Park, M. G. Choi, M. Cha, S. Y. Park and D. J. Jang, J. Am. Chem. Soc., 2005, 127, 10070 CrossRef CAS PubMed.
- H. Mishra, H. C. Joshi, H. B. Tripathi, S. Maheshwary, N. Sathyamurthy, M. Panda and J. Chandrasekhar, J. Photochem. Photobiol., A, 2001, 139, 23 CrossRef CAS.
- V. Mishra, H. Mishra, H. C. Joshi and T. C. Pant, Sens. Actuators, B, 2002, 82, 133 CrossRef.
- H. Mishra, V. Mishra, M. S. Mehata, T. C. Pant and H. B. Tripathi, J. Phys. Chem. A, 2004, 108, 2346 CrossRef CAS.
- A. Douhal, J. Dabrio and R. Sastre, J. Phys. Chem. A, 1996, 100, 149 CrossRef CAS.
- A. L. Sobolewski and W. Domcke, Phys. Chem. Chem. Phys., 2006, 8, 3410 RSC.
- T. Sekikawa, T. Kobayashi and T. Inabe, J. Phys. Chem. B, 1997, 101, 10645 CrossRef CAS.
- A. D. Roshal, A. V. Grigorovich, A. O. Doroshenko, V. G. Pivovarenko and A. P. Demchenko, J. Phys. Chem. A, 1998, 102, 5907–5914 CrossRef CAS.
- W. Sun, S. Li and R. Hu, et al., J. Phys. Chem. A, 2009, 113, 5888–5895 CrossRef CAS PubMed.
- S. Park, E. K. Ji and H. K. Se, et al., J. Am. Chem. Soc., 2009, 131, 14043–14049 CrossRef CAS PubMed.
- S. Kim, D. W. Chang, S. Y. Park, K. Kim and J. I. Jin, Bull. Korean Chem. Soc., 2001, 22, 12 Search PubMed.
- A. Weller, Bunsen-Ges. Phys. Chem., Ber., 1956, 60, 1144–1147 CAS.
- H. Mishra, S. Maheshwary, H. B. Tripathi and N. Sathyamurthy, J. Phys. Chem. A, 2005, 109, 2746–2754 CrossRef CAS PubMed.
- H. Mishra, J. Phys. Chem. B, 2006, 110, 9387–9396 CrossRef CAS PubMed.
- A. Pal, S. S. Maity, S. Samanta, P. Saha Sardar and S. Ghosh, J. Lumin., 2010, 130, 1975–1982 CrossRef CAS.
- J. N. Demas and G. A. Crosby, J. Phys. Chem., 1971, 75, 991 CrossRef.
- FELIX 32, Operation Manual, Version 1.1, Photon Technology International, Inc., NJ, 2003 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian, Inc., Wallingford CT, 2013.
- P. J. Kovi and S. G. Shulman, Anal. Chem., 1973, 45, 989–991 CrossRef CAS.
- P. J. Kovi, C. L. Miller and S. G. Schulman, Anal. Chim. Acta, 1972, 61, 7–13 CrossRef CAS PubMed.
-
(a) B. K. Paul and N. Guchhait, Langmuir, 2010, 26, 3214 CrossRef CAS PubMed;
(b) B. K. Paul, A. Samanta and N. Guchhait, J. Colloid Interface Sci., 2011, 353, 237–247 CrossRef CAS PubMed.
-
(a) E. L. Roberts, P. T. Chou, T. A. Alexander, R. A. Agbaria and I. M. Warner, J. Phys. Chem., 1995, 99, 5431 CrossRef CAS;
(b) E. L. Roberts, J. Dey and I. M. Warner, J. Phys. Chem. A, 1997, 101, 5296 CrossRef CAS;
(c) M. Mukhopadhyay, D. Banerjee and S. Mukherjee, J. Phys. Chem. A, 2006, 110, 12743 CrossRef CAS PubMed;
(d) M. Mukhopadhyay, A. Mandal, R. Misra, D. Banerjee, S. P. Bhattacharyya and S. Mukherjee, J. Phys. Chem. B, 2009, 113, 567 CrossRef CAS PubMed;
(e) B. Cohen, D. Huppert, K. M. Solnstev, Y. Tsfadia, E. Nachliel and M. Gutman, J. Am. Chem. Soc., 2002, 124, 7539 CrossRef CAS PubMed.
- P. Hazra, D. Chakraborty and N. Sarkar, Langmuir, 2002, 18, 7872 CrossRef CAS.
- S. Matzinger, D. M. Hussey and M. D. Fayer, J. Phys. Chem. B, 1998, 102, 7216 CrossRef CAS.
- J. Baxterhammond, C. R. Powley, K. D. Cook and T. A. Nieman, J. Colloid Interface Sci., 1980, 76, 434 CrossRef CAS.
- H. A. Benesi and J. H. Hilderbrand, J. Am. Chem. Soc., 1949, 71, 2703–2707 CrossRef CAS.
- C. Reichardt, in Molecular Interaction, ed. H. Ratajazak and W. J. Orville-Thomas, Wiley, NY, 1982, vol. 3, p. 255 Search PubMed.
- C. Reichardt, Chem. Rev., 1994, 94, 2319 CrossRef CAS.
- M. J. Kamlet, J. L. M. Abboud, M. H. Abraham and R. W. Taft, J. Org. Chem., 1983, 48, 2877–2887 CrossRef CAS.
- C. Reichardt, Solvent and Solvent Effects, in Organic Chemistry, VCH Verlagsgesellschaft, Weinheim, Germany, 1988 Search PubMed.
- Y. Marcus, J. Chem. Soc., Perkin Trans. 2, 1994, 5, 1751 RSC.
- H. C. Joshi, C. Gooijer and G. van der Zwan, J. Phys. Chem. A, 2002, 106, 11422 CrossRef CAS.
- M. K. Singh, H. Pal, A. S. R. Koti and A. V. Sapre, J. Phys. Chem. A, 2004, 108, 1465–1474 CrossRef CAS.
- M. Mukherjee, P. S. Sardar, S. K. Ghorai, S. K. Samanta, A. S. Roy, S. Dasgupta and S. Ghosh, PLoS One, 2013, 8, 1–16 CrossRef.
- J. R. Lakowicz, Principles of Fluorescence Spectroscopy, Kluwer Academic/Plenum Publishers, 2006 Search PubMed.
-
(a) M. Kubinyi, A. Grofcsik, I. Pápai and W. J. Jones, Chem. Phys., 2003, 286, 81–96 CrossRef CAS;
(b) M. Kubinyi, A. Grofcsik, T. Kárpáti and W. J. Jones, Chem. Phys., 2006, 322, 247–253 CrossRef CAS.
Footnote |
† Electronic supplementary information (ESI) available: Binding constant equation obtained from steady state anisotropy data (eqn (S1)). Binding constant equation obtained from the time resolved emission studies (eqn (S2) and (S3)). Cavity diameter and molar volume of β- and γ-cyclodextrin obtained from theoretical calculation (Table S1). Distance table for interactions of the inclusion complex of β-CD–3HNA and γ-CD–3HNA (Table S2A and B). Fluorescence spectra of 3HNA (20 μM) at 298 K in aqueous medium; (a) in pure water, (b) in 4 mM TTAB, (c) in 1 mM CTAB, (d) in 20 mM Triton-X; λexc = 350 nm; excitation band pass 10 nm, and emission band pass = 5 nm (Fig. S1). (A) The plot of (F∞ − F0)/(Fx − F0) against [β-CD]−1; (B) plot of (r − rf)/R (rb − r) against [β-CD]−1; (C) plot of C0/C vs. 1/[β-CD] (Fig. S2). Fluorescence spectra of 3HNA (20 μM) in aqueous buffer solution of β-CD (6 mM) (pH 7) with varying temperature: (a) 15 °C, (b) 24 °C, (c) 34 °C; λexc = 350 nm; excitation band pass 10 nm, and emission band pass = 5 nm. Inset: the plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T (Fig. S3). Fluorescence spectra of 3HNA (20 μM) in DMSO and water mixtures at 298 K. Curves (a–f) represents 20%, 40%, 60%, 80%, 95% and 100% DMSO, respectively. λexc = 350 nm; excitation and emission band pass = 10 nm each. Inset: plot of τ (ns) of 3HNA at 298 K in DMSO–water mixed solvent against the % volume of DMSO in the mixed solvent (Fig. S4). (A) Plot of transition energy (ET in kcal mol−1) of ESIPT emission of 3HNA (20 μM) in different mixed solvents of water and DMSO against the solvent polarity index ET(30)-scale (kcal mol−1): (1) DMSO, (2) 90% DMSO, (3) 80% DMSO, (4) 60% DMSO, (5) 40% DMSO, (6) 20% DMSO, (7) water; λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. (B) Plot of transition energy (kcal mol−1) of ESIPT emission of 3HNA in different mixed solvents of water and DMSO against the hydrogen bond donating acidity parameter alpha (α). (C) Plot of Stokes shift (Δ) (cm−1) vs. eqn (6) for different mixed solvents of water and DMSO (Fig. S5). Fluorescence spectra of 3HNA (20 μM) in DCM–ACN mixed solvents at 298 K. Curves (a–i) represents 0%, 4%, 8%, 12%, 16%, 22%, 27%, 39% and 100% ACN respectively. λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. Inset: plot of dielectric constant of ACN/DCM mixtures against λmax of 3HNA (20 μM) emission on those mixtures (Fig. S6). See DOI: 10.1039/c6ra22124f K vs. 1/T (Fig. S3). Fluorescence spectra of 3HNA (20 μM) in DMSO and water mixtures at 298 K. Curves (a–f) represents 20%, 40%, 60%, 80%, 95% and 100% DMSO, respectively. λexc = 350 nm; excitation and emission band pass = 10 nm each. Inset: plot of τ (ns) of 3HNA at 298 K in DMSO–water mixed solvent against the % volume of DMSO in the mixed solvent (Fig. S4). (A) Plot of transition energy (ET in kcal mol−1) of ESIPT emission of 3HNA (20 μM) in different mixed solvents of water and DMSO against the solvent polarity index ET(30)-scale (kcal mol−1): (1) DMSO, (2) 90% DMSO, (3) 80% DMSO, (4) 60% DMSO, (5) 40% DMSO, (6) 20% DMSO, (7) water; λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. (B) Plot of transition energy (kcal mol−1) of ESIPT emission of 3HNA in different mixed solvents of water and DMSO against the hydrogen bond donating acidity parameter alpha (α). (C) Plot of Stokes shift (Δ) (cm−1) vs. eqn (6) for different mixed solvents of water and DMSO (Fig. S5). Fluorescence spectra of 3HNA (20 μM) in DCM–ACN mixed solvents at 298 K. Curves (a–i) represents 0%, 4%, 8%, 12%, 16%, 22%, 27%, 39% and 100% ACN respectively. λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. Inset: plot of dielectric constant of ACN/DCM mixtures against λmax of 3HNA (20 μM) emission on those mixtures (Fig. S6). See DOI: 10.1039/c6ra22124f |
|
| This journal is © The Royal Society of Chemistry 2016 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex between the β- and γ-cyclodextrin hosts and the guest. The values of the binding constants and different thermodynamic parameters for complexation have also been reported. The results in various confined media have been correlated using control experiments on the ESIPT emission of 3HNA in mixed solvents consisting of dimethyl sulfoxide (DMSO) and water (mixture of an aprotic polar and a protic polar solvent) and also of acetonitrile (ACN) and dichloromethane (DCM) (mixture of an aprotic polar and a non-polar solvent). Theoretical calculation on the orientation of 3HNA within β- and γ-cyclodextrins has been carried out in order to explain the observed rotational correlation time of the probe in confined environments.
1 complex between the β- and γ-cyclodextrin hosts and the guest. The values of the binding constants and different thermodynamic parameters for complexation have also been reported. The results in various confined media have been correlated using control experiments on the ESIPT emission of 3HNA in mixed solvents consisting of dimethyl sulfoxide (DMSO) and water (mixture of an aprotic polar and a protic polar solvent) and also of acetonitrile (ACN) and dichloromethane (DCM) (mixture of an aprotic polar and a non-polar solvent). Theoretical calculation on the orientation of 3HNA within β- and γ-cyclodextrins has been carried out in order to explain the observed rotational correlation time of the probe in confined environments.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) inclusion complex with 3HNA for further optimisation.
1) inclusion complex with 3HNA for further optimisation.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host
1 host![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) guest complexes
guest complexes![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest complex formation the equation takes the form
1 host–guest complex formation the equation takes the form![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and the 2
1 and the 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest complexes respectively. Linearity in the plot of (F∞ − F0)/(Fx − F0) against [L]−1 [from eqn (6)] confirms a 1
1 host–guest complexes respectively. Linearity in the plot of (F∞ − F0)/(Fx − F0) against [L]−1 [from eqn (6)] confirms a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 interaction of β-CD and γ-CD with 3HNA (Fig. S2A†). Eqn (7) shows the nonlinear regression with a curvature.
1 interaction of β-CD and γ-CD with 3HNA (Fig. S2A†). Eqn (7) shows the nonlinear regression with a curvature.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μM) at 298 K
000 μM) at 298 K
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300)
300)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1500)
1500)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T gives a straight line (Fig. S3,† inset); from the slope of the line ΔH0 can be calculated. The values of ΔG0, ΔH0 and ΔS0 for binding are provided in Table 5.
K vs. 1/T gives a straight line (Fig. S3,† inset); from the slope of the line ΔH0 can be calculated. The values of ΔG0, ΔH0 and ΔS0 for binding are provided in Table 5.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest inclusion complexes of cyclodextrins and 3HNA, the binding constant values obtained by different methods and the rotational correlation time of the probe observed indicate that the mode of encapsulation of the 3HNA is different in the β and γ-CD hosts. In order to realise the environment of the confined guest molecule, control experiments using the mixed solvents like DMSO–H2O and ACN–DCM have been carried out. The role of microenvironments like polarity, H-bonding ability of the solvents/mixed solvents helps understand the homogeneity/heterogeneity inside the cavity of the CDs or micelles and also the rigidity of the respective environment. The marked enhancement of the ESIPT emission in presence of the confined media, the mode of complexation and the motional restriction of the probe were also corroborated by theoretical calculation of the orientation of the encapsulated 3HNA in both the cyclodextrin hosts.
1 host–guest inclusion complexes of cyclodextrins and 3HNA, the binding constant values obtained by different methods and the rotational correlation time of the probe observed indicate that the mode of encapsulation of the 3HNA is different in the β and γ-CD hosts. In order to realise the environment of the confined guest molecule, control experiments using the mixed solvents like DMSO–H2O and ACN–DCM have been carried out. The role of microenvironments like polarity, H-bonding ability of the solvents/mixed solvents helps understand the homogeneity/heterogeneity inside the cavity of the CDs or micelles and also the rigidity of the respective environment. The marked enhancement of the ESIPT emission in presence of the confined media, the mode of complexation and the motional restriction of the probe were also corroborated by theoretical calculation of the orientation of the encapsulated 3HNA in both the cyclodextrin hosts.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T (Fig. S3). Fluorescence spectra of 3HNA (20 μM) in DMSO and water mixtures at 298 K. Curves (a–f) represents 20%, 40%, 60%, 80%, 95% and 100% DMSO, respectively. λexc = 350 nm; excitation and emission band pass = 10 nm each. Inset: plot of τ (ns) of 3HNA at 298 K in DMSO–water mixed solvent against the % volume of DMSO in the mixed solvent (Fig. S4). (A) Plot of transition energy (ET in kcal mol−1) of ESIPT emission of 3HNA (20 μM) in different mixed solvents of water and DMSO against the solvent polarity index ET(30)-scale (kcal mol−1): (1) DMSO, (2) 90% DMSO, (3) 80% DMSO, (4) 60% DMSO, (5) 40% DMSO, (6) 20% DMSO, (7) water; λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. (B) Plot of transition energy (kcal mol−1) of ESIPT emission of 3HNA in different mixed solvents of water and DMSO against the hydrogen bond donating acidity parameter alpha (α). (C) Plot of Stokes shift (Δ) (cm−1) vs. eqn (6) for different mixed solvents of water and DMSO (Fig. S5). Fluorescence spectra of 3HNA (20 μM) in DCM–ACN mixed solvents at 298 K. Curves (a–i) represents 0%, 4%, 8%, 12%, 16%, 22%, 27%, 39% and 100% ACN respectively. λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. Inset: plot of dielectric constant of ACN/DCM mixtures against λmax of 3HNA (20 μM) emission on those mixtures (Fig. S6). See DOI: 10.1039/c6ra22124f
K vs. 1/T (Fig. S3). Fluorescence spectra of 3HNA (20 μM) in DMSO and water mixtures at 298 K. Curves (a–f) represents 20%, 40%, 60%, 80%, 95% and 100% DMSO, respectively. λexc = 350 nm; excitation and emission band pass = 10 nm each. Inset: plot of τ (ns) of 3HNA at 298 K in DMSO–water mixed solvent against the % volume of DMSO in the mixed solvent (Fig. S4). (A) Plot of transition energy (ET in kcal mol−1) of ESIPT emission of 3HNA (20 μM) in different mixed solvents of water and DMSO against the solvent polarity index ET(30)-scale (kcal mol−1): (1) DMSO, (2) 90% DMSO, (3) 80% DMSO, (4) 60% DMSO, (5) 40% DMSO, (6) 20% DMSO, (7) water; λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. (B) Plot of transition energy (kcal mol−1) of ESIPT emission of 3HNA in different mixed solvents of water and DMSO against the hydrogen bond donating acidity parameter alpha (α). (C) Plot of Stokes shift (Δ) (cm−1) vs. eqn (6) for different mixed solvents of water and DMSO (Fig. S5). Fluorescence spectra of 3HNA (20 μM) in DCM–ACN mixed solvents at 298 K. Curves (a–i) represents 0%, 4%, 8%, 12%, 16%, 22%, 27%, 39% and 100% ACN respectively. λexc = 350 nm; excitation band pass = 10 nm and emission band pass = 5 nm. Inset: plot of dielectric constant of ACN/DCM mixtures against λmax of 3HNA (20 μM) emission on those mixtures (Fig. S6). See DOI: 10.1039/c6ra22124f