Improving energy relay dyes for dye-sensitized solar cells by use of a group of uniform materials based on organic salts (GUMBOS)†
Abstract
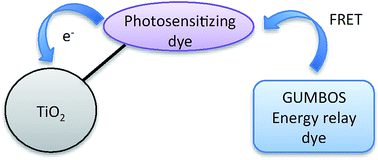
In this study, GUMBOS (a group of uniform materials based on organic salts) derived from rhodamine B chloride, 1,1′-diethyl-2,2′-carbocyanine iodide, 3,3′-diethylthiacarbocyanine iodide, and meso-tetra(4-carboxyphenyl)porphine were synthesized and characterized for application as energy relay dyes (ERDs) in dye-sensitized solar cells (DSSCs). A facile ion exchange reaction was employed for synthesis of GUMBOS. These GUMBOS exhibited improved characteristics in comparison to their respective parent dyes, including increased solubility, thermal stability, molar extinction coefficient, and fluorescence quantum yield. In addition, excellent spectral overlap integral and Förster resonance energy transfer efficiency were observed between various GUMBOS based-ERDs (donors) and the photosensitizing dye, N719 [di-tetrabutylammonium cis-bis(isothiocyanato)bis(2,2′-bipyridyl-4,4′-dicarboxylato)-ruthenium(II)] (acceptor). DSSC devices were fabricated and solar efficiency was evaluated in the absence and presence of ERDs using N719 as the photosensitizing dye. DSSCs in the presence of GUMBOS-based ERDs exhibited increased solar efficiencies in comparison to DSSCs in the absence of ERDs. Moreover, increases in solar efficiencies were found to be dependent on the counterions used in GUMBOS synthesis.

 Please wait while we load your content...
Please wait while we load your content...