Graphitic carbon nitride nanosheets with tunable optical properties and their superoxide dismutase mimetic ability†
Xiangling Ren,
Xianwei Meng*,
Jun Ren and
Fangqiong Tang
Laboratory of Controllable Preparation and Application of Nanomaterials, Center for Micro/nanomaterials and Technology, Key Laboratory of Photochemical Conversion and Optoelectronic Materials, Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Beijing, 100190, China. E-mail: mengxw@mail.ipc.ac.cn; Fax: +86-10-62554670; Tel: +86-10-82543521
First published on 23rd September 2016
Abstract
Fluorescent graphitic carbon nitride nanosheets (g-CNNSs) have attracted extensive attention because of their new properties and promising practical application. The design and construction of g-CNNSs with novel optical properties is highly desirable. Herein, g-CNNSs with tunable optical properties were synthesized through a simple, nontoxic and one-pot solid-phase thermal treatment method. The g-CNNSs exhibited a strong absorption at 410 nm in a UV-vis absorption spectrum and had a fluorescence emission band at around 525 nm over a wide range of excitation wavelengths. The optical properties of g-CNNSs exhibited a regular change in storage or alkaline solution. The g-CNNSs showed low cytotoxicity and high stability of fluorescence intensity under high-salt conditions or in a broad pH range (1–14). Interestingly, the g-CNNSs possess intrinsic superoxide dismutase (SOD)-like activity which is related to their structure.
Introduction
Graphitic carbon nitrides (g-C3N4) as layer-structured semiconductors have been widely used in organic pollutant degradation, hydrogen evolution, and solar energy transfer.1–7 Recently, fluorescent graphitic carbon nitrides, such as fluorescent carbon nitride nanosheets (g-CNNSs) and carbon nitride quantum dots (g-CNQDs), have attracted worldwide attention as novel fluorescent probes for biological and environment detection due to their good water solubility, excellent chemical stability, high stability toward photo-bleaching and blinking, and good biocompatibility compared with heavy-metal containing semiconductor quantum dots.8–10 However, most of these reported fluorescent g-C3N4 emit blue fluorescence under ultraviolet, which limits their application in many fields, such as bioimaging in vivo, full-color displays, and so on.11,12 Therefore, the development of g-C3N4 with longer wave emission by simple and effective method is still highly desired. On the other hand, the main factor to control the emission of fluorescent carbon material is particle size.13 There has been no report so far about changing the optical properties of fluorescent g-C3N4 by adjusting their structures.In addition to fluorescence properties, a number of carbon nanomaterials, including graphene oxide (GO), carbon nanotubes (CNTs), carbon dots (CDs) and graphene quantum dots (GQDs), have been recognized as nanomaterial-based artificial enzymes (nanozyme) in recent years.14,15 These carbon nanomaterials own the advantages of natural enzyme, such as facile production on large scale, low costs, long-term storage and high stability in harsh environments. Some metal or metal compound doped g-C3N4 have also exhibited peroxidase-like activity.16,17 Although our group have report the laccase-like activity of the active copper-containing carbon dots (Cu-CDs),18 most of the carbon nanozymes' catalytic reactions are based on peroxidase reactions. The carbon nanozymes, especially g-C3N4, with other type enzyme activity are still scarce.
Herein, we present a low-temperature solid-phase method to synthesize fluorescent g-CNNSs. The prepared g-CNNSs exhibited excellent water-solubility, yellow emission, high stability under salt conditions (positive and negative ions), and low cytotoxicity. The g-CNNSs differed from the previous g-C3N4 on the following points: (i) the g-CNNSs had fluorescence emission band around at 525 nm over a wide range of excitation wavelengths (from 230 to 490 nm). (ii) The UV-vis absorption spectrum of g-CNNSs exhibited a strong absorption at 410 nm, which has never been reported in the fluorescent g-CNQDs or g-CNNSs. (iii) The UV-vis and fluorescence spectra of g-CNNSs were tunable by the change of their structures in storage or alkaline solution. (iv) The g-CNNSs exhibited superoxide dismutase (SOD)-like catalytic activity especially after structural changes. All these unique performance of as-prepared g-CNNSs provides new alternative for the development of applications.
Experimental
Materials
All reagents were of analytical reagent grade and used as received throughout the work. Pyrogallol were purchased from Sigma. Urea, sodium citrate, citric acid and other reagents were obtained from Beijing Shiji Company. Hep G2 cell (Human hepatocellular liver carcinoma cell line) were obtained from American type culture collection (ATCC, USA). Ultrapure water was produced using a Millipore-Q water system with a 0.22 μm filter membrane.Instrumentation
Transmission electron microscopy (TEM) was performed on a JEOL JEM 2010F electron microscope operating at 200 kV. Fourier transform infrared spectrometry (FT-IR, Varian, Model 3100 Excalibur) was employed to examine the g-CNNSs. X-ray diffraction patterns (XRD) of the samples were measured on a Bruker D8 Focus powder X-ray diffractometer using CuKα radiation (λ = 1.5418 Å). UV-vis spectra were recorded using a JASCO V-570 spectrophotometer at room temperature. Fluorescence measurements were carried out on a Cary Eclipse fluorescence spectrophotometer (Varian, Inc.). The emission spectra of g-CNNSs were recorded upon excitation at 410 nm. The exciting slit and the emission slit were 2.5 and 5 nm, respectively. The samples for the fluorescence measurements were placed in a 10 mm optical path length quartz fluorescence cuvette. The X-ray photoelectron spectroscopy (XPS) measurements were performed on an ESCALAB-MKII 250 photoelectron spectrometer with Al/Kα as the X-ray source for excitation.Synthesis of g-CNNSs
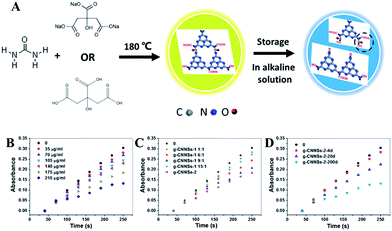
The g-CNNSs were prepared by solid-phase thermal treatment method with urea, sodium citrate and citric acid. Briefly, 0.202 g urea and 0.11 g sodium citrate were mixed in an agate mortar and grounded to a uniform powder. The mixture was placed in a crucible and heated to 180 °C for 1 h in a vacuum drying oven. Then the crucible was cooled down to room temperature. The obtained yellowish-brown powders were dissolved with ultrapure water and then purificated by high speed centrifugation. The sample was designated as g-CNNSs-1.The g-CNNSs-2 was synthesized with 0.202 g urea, 0.037 g sodium citrate and 0.048 g citric acid. Other operations were the same as g-CNNSs-1.
Cytotoxicity assay
The cytotoxicity of g-CNNSs against Hep G2 cells was determined by a methylthiazoleterazolium (MTT) assay with standard protocol.19 All experiments were carried out for six replicates.Analytical procedures for the SOD activity of g-CNNSs
A substrate pyrogallol HCl solution (10 mM) was freshly prepared before use. The g-CNNSs with different concentrations were mixed with Tris–HCl buffer solution (pH = 8.2, containing 1 mM EDTA-2Na). The timer started after 10 μL of pyrogallol solution was added. The total volume of the mixture was 4 mL. The SOD activities were analysed by monitoring the absorbance change at 325 nm.Results and discussion
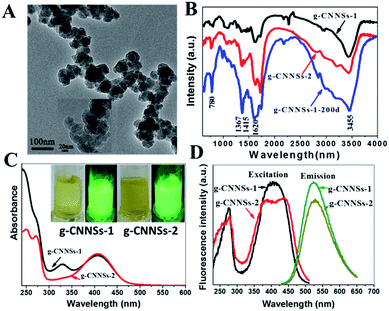
The g-CNNSs were synthesized by the solid mixture of urea and sodium citrate without (designated as g-CNNSs-1) or with (designated as g-CNNSs-2) citric acid. The obtained yellowish-brown powders were dissolved with ultrapure water and then purificated by high speed centrifugation. The g-CNNSs were characterized by means of TEM, EDS, XRD and FTIR. Typical TEM image of g-CNNSs-1 (Fig. 1A) showed a mean diameter of 50 nm. The high resolution TEM (HRTEM) (Fig. 1A, insets) exhibited a layers structure of g-CNNSs-1. The electron diffraction spectroscopy (EDS) result as shown in Fig. S1† confirmed the presence of carbon (C), nitrogen (N), oxygen (O) and sodium (Na) in the g-CNNSs-1 sample. The structural features of g-CNNSs were verified by two characteristic peaks in XRD pattern at 2θ of 27.6 and 13.1°, which originate from the (002) interlayer diffraction of C–N layers and an in-plane structural packing motif indexed as the (100) peaks, respectively (Fig. S2†). The g-CNNSs-2 had a similar structure and both of them were consistent with the previous reports on g-CNNSs.20 In addition, FT-IR spectra were used to identify the functional groups of g-CNNSs. As shown in Fig. 1B, the two samples have similar spectra and the broad band at 3000 to 3700 cm−1 was attributed to the stretching mode of –NH2 and –OH group. The stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O was observed according to the peaks around 1620 cm−1. The peaks at 1367 and 1415 cm−1 were the typical stretching vibration modes of C
O was observed according to the peaks around 1620 cm−1. The peaks at 1367 and 1415 cm−1 were the typical stretching vibration modes of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and C–N heterocycles. The absorption bands at 780 cm−1 can be assigned to the breathing mode of the triazine units.
N and C–N heterocycles. The absorption bands at 780 cm−1 can be assigned to the breathing mode of the triazine units.
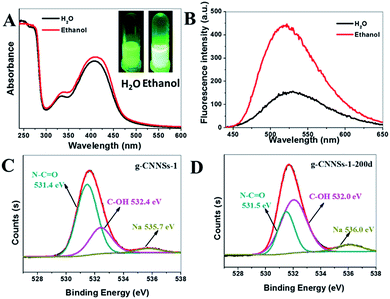
The g-CNNSs aqueous solution present a yellowish (g-CNNSs-2 color slightly deeper than g-CNNSs-1) in daylight, which changes to yellow luminescence under UV excitation (Fig. 1C, insets). The optical properties of the g-CNNSs were investigated by the UV-vis absorption and fluorescent spectra. As shown in Fig. 1C, the UV-vis absorption spectrum exhibited some weak peaks between 200 to 270 nm and the peaks of g-CNNSs-2 were more obvious. These peaks were due to π–π* electronic transitions for carbon nitrides containing s-triazine rings. The UV-vis absorption spectrum of g-CNNSs-1 showed an obvious peak centered at 330 nm which is similar to the absorption peak for graphene carbon nitride quantum dots (g-CNQDs).21 However, the UV-vis absorption spectrum of g-CNNSs-2 did not show any peak around 330 nm. To our surprise, a strong absorption at 410 nm was observed in both samples, which presumably belong to the n–π* transitions involving lone pairs of oxygen or nitrogen atoms.22 To the best of our knowledge, this absorption peak has never been reported in the fluorescent g-CNQDs or g-CNNSs. This absorption peak meant that our g-CNNSs has a unique structure.
As shown in excitation spectrum of g-CNNSs (Fig. 1D), there was a main excitation peak around 400 nm and a sharp peaked at 270 nm. The as-prepared g-CNNSs fluoresce with a wide range of excitation wavelengths (from 230 to 490 nm), and the maximum excitation wavelengths were located at 410 nm (Fig. S3A†) and they have a strong fluorescence emission band centered at 525 nm (Fig. 1D). The fluorescence emission peak of g-CNNSs-2 was less affected by the excitation wavelengths compared with g-CNNSs-1 (Fig. S3B†). It may be due to the excitation peaks of g-CNNSs-2 were wider than g-CNNSs-1 (Fig. 1D). The PL lifetime of g-CNNSs-1 and g-CNNSs-2 were measured to be 6.23 and 8.00 ns, respectively (Fig. S4†). These results were comparable to that of other g-CNNSs in the previous reported.23
The influence of the representative ionic strength on the fluorescence intensity of g-CNNSs has been confirmed. As shown in Fig. S5A and B,† the fluorescence change of g-CNNSs-1 presented only a very slight quenching effect in both solutions at a high ionic strength of 500 μM sodium salt or at 50 μM of different metal ions. These results indicate that the fluorescence of g-CNNSs is very stable in ionic solution.
The cytotoxicity of g-CNNSs was evaluated by viability of Hep G2 cells in vitro through the MTT assay to assess the biocompatibility of g-CNNSs. As shown in Fig. S6,† the viability of cells remained unchanged when the cells were exposed to g-CNNSs-1 in the range of 3.9–1000 μg mL−1. Even after incubation for 24 h at the highest tested concentration of 1000 μg mL−1, no apparent loss of cell viability was observed. This result suggests the excellent biocompatibility of g-CNNSs.
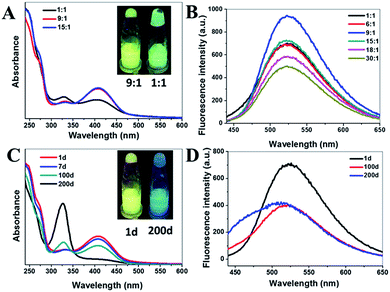
Notably, the optical properties of g-CNNSs were changed by adjusting the molar ratio of urea to sodium citrate. When increasing the amount of sodium citrate, a distinct change of the UV-vis absorption spectrum of g-CNNSs can be observed (Fig. 2A). The peak centered at 330 nm was significantly enhanced and became higher than the peak centered at 410 nm when the molar ratio of urea to sodium citrate was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. At the same time, the luminescence of g-CNNSs aqueous solution changed from yellow to green under UV excitation (Fig. 2A, insets). The fluorescence emission peak of g-CNNSs changed little, and the widening of peak only showed when the molar ratio was 1
1. At the same time, the luminescence of g-CNNSs aqueous solution changed from yellow to green under UV excitation (Fig. 2A, insets). The fluorescence emission peak of g-CNNSs changed little, and the widening of peak only showed when the molar ratio was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Fig. 2B). The fluorescence intensity of g-CNNSs was increased when the molar ratio was less than 9
1 (Fig. 2B). The fluorescence intensity of g-CNNSs was increased when the molar ratio was less than 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and decreased gradually with the increase of the molar ratio.
1, and decreased gradually with the increase of the molar ratio.
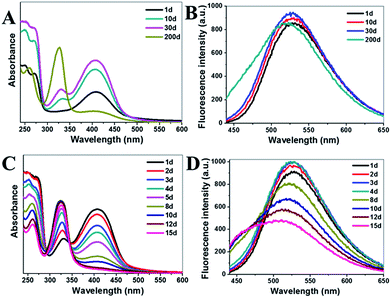
Interestingly, the effect of storage time on the optical properties of g-CNNSs was also observed. With the extension of storage time at room temperature, a regular change of the UV-vis absorption spectrum of g-CNNSs-1 was shown in Fig. 2C. The peak centered at 330 nm was enhanced continuously and the peak centered at 410 nm was decreased gradually until disappear. The luminescence of g-CNNSs-1 aqueous solution changed from yellow to blue under UV excitation (Fig. 2C, insets) after 200 days. The fluorescence emission peak of g-CNNSs-1 was widening as storage time and obvious blue shift (Fig. 2D). The optical properties of g-CNNSs-2 changes with the storage time were more slightly than g-CNNSs-1 (Fig. 3A). Although the peak centered at 330 nm increased to higher than the peak centered at 410 nm, but the peak centered at 410 nm was not disappear completely after 200 days. The fluorescence emission peak of g-CNNSs-2 showed a little broadening and blue shift after 200 days (Fig. 3B). In order to accelerate the effect of storage time on optical properties of g-CNNSs-2, the UV-vis absorption spectra and fluorescence emission spectra of g-CNNSs-2 which stored at 70 °C was measured continuously. As shown in Fig. 3C and D, the peak centered at 330 nm was enhanced continuously and the peak centered at 410 nm was decreased gradually until disappear. The fluorescence emission peak of g-CNNSs-2 was enhanced at first then decreased, and gradually broadening and blue shift. This distinctly demonstrated that the effect of storage time on g-CNNSs-2 was as same as g-CNNSs-1.
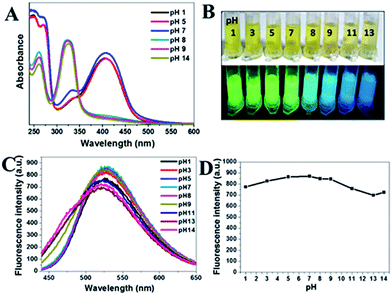
Another factor affecting optical properties of g-CNNSs was pH values. The UV-vis absorption spectrum of g-CNNSs-2 in different pH solution from 1.0 to 14.0 has been investigated (Fig. 4A). Different from the effect of storage time, the peak centered at 330 nm was increased sharply and the peak centered at 410 nm was decreased sharply until disappear when pH higher than 8. The luminescence of g-CNNSs-2 aqueous solution with different pH were shown in Fig. 4B. The colors changed from yellow to blue under UV excitation at pH 8. The fluorescence emission peak of g-CNNSs-2 was widening as storage time and obvious blue shift (Fig. 4C). The fluorescence emission peak of g-CNNSs-2 showed obvious broadening when pH higher than 9. To our delight, the fluorescence intensity was little change in the pH range of 1–14 (Fig. 4D). The lowest fluorescence intensity still retained the highest value of 80%.
Further studies revealed that the fluorescence intensity of g-CNNSs was greatly enhanced in ethanol. The g-CNNSs-2 emitted much brighter yellow fluorescence in ethanol than water (Fig. 5A, insets). The UV-vis absorption spectra of g-CNNSs-2 in ethanol was similar to it in water (Fig. 5A). However, the fluorescence intensity of g-CNNSs-2 in ethanol was 2.8 times higher than it in water and the emission peak was slight blue shift (Fig. 5B). The fluorescence emission of g-CNNSs in ethanol focused on 513 nm with a very wide range of excitation wavelengths (from 230 to 470 nm, Fig. S7†).
In general, the optical properties of g-CNNSs can be affected by the following four factors: the molar ratio of urea to sodium citrate, storage time, pH values and ethanol. Both prolonged storage time and high pH values change the luminescence of g-CNNSs aqueous solution from yellow to blue. At the same time, the change of UV-vis absorption spectra of g-CNNSs was similar. The peak centered at 330 nm which are associated with a characteristic absorption peak for g-CNQDs was increased. Meanwhile, the peak centered at 410 nm which has been reported rarely in the fluorescent g-CNQDs or g-CNNSs was decreased and disappear. The excitation and emission peaks of g-CNNSs-1 in pH 13 solution or storage after 200 days were also recorded (Fig. S8†). The excitation and emission peaks of two samples were very similar and both blue shift compared with the g-CNNSs-1. There was a main excitation peak around 370 nm and a sharp peaked at 290 nm and the emission peak were located at 454 nm with the excitation wavelength at 370 nm. The fluorescence peak of g-CNNSs in pH 13 solution and storage after 200 days shifted to long wave when the aqueous solution of g-CNNSs was excited at a wavelength ranging between 330 nm and 510 nm (Fig. S9†). Similar excitation dependent behavior is reported in the literature for fluorescent g-CNQDs or CDs. On the basis of the above results, it can be assumed that the structure of g-CNNSs have same change with prolonged storage time or high pH values.
To better understand the structure change of g-CNNSs, the XRD (Fig. S2†) and FTIR spectrum (Fig. 1B) of g-CNNSs-1 which preserved at room temperature over 200 days (g-CNNSs-200d) were recorded. There were no significant differences with g-CNNSs. Furthermore, the XPS measurement was performed to investigate the surface state and elemental composition of g-CNNSs. There were three peaks in each curve (g-CNNSs-1 and g-CNNSs-200d) at 284.8, 399.9, and 531.7 eV in the XPS spectrum corresponding to C 1s, N 1s, and O 1s, respectively (Fig. S10†). The atom ratio of C/N/O in three samples was shown in Table S1,† of which the content of O in g-CNNSs-200d was obviously higher than that of g-CNNSs-1. This prove that some oxygen-containing groups generated during storage. So the high-resolution O 1s spectrum was further investigated. The O 1s spectrum of g-CNNSs-1 (Fig. 5C) showed two peaks detected at 531.4 eV and 532.4 eV, which were attributed to N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–OH groups, respectively.23 Correspondingly, the peak of N–C
O and C–OH groups, respectively.23 Correspondingly, the peak of N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group (located at 531.5 eV) decreased significantly and the peak of C–OH group (located at 532.0 eV) increased obviously the in the high-resolution O 1s spectrum of g-CNNSs-200d (Fig. 5D). This indicated some N–C
O group (located at 531.5 eV) decreased significantly and the peak of C–OH group (located at 532.0 eV) increased obviously the in the high-resolution O 1s spectrum of g-CNNSs-200d (Fig. 5D). This indicated some N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds were disappeared in g-CNNSs-200d.
O bonds were disappeared in g-CNNSs-200d.
Considering the changes of g-CNNSs in storage or alkaline solution, a proposed mechanism of structural changes of g-CNNSs was depicted in Fig. 6A. The g-CNNSs prepared by solid phase thermal reaction with urea and sodium citrate might be have the connection with N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds unlike other g-CNNSs or g-CNQDs synthesized by hydrothermal method. This can be proved by the strong absorption at 410 nm formed by the n–π* transitions. When the g-CNNSs preserved at room temperature or in alkaline solution, the N–C
O bonds unlike other g-CNNSs or g-CNQDs synthesized by hydrothermal method. This can be proved by the strong absorption at 410 nm formed by the n–π* transitions. When the g-CNNSs preserved at room temperature or in alkaline solution, the N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds was decomposed into COOH and N–H which similar to the hydrolysis process of amide bond. So the peak centered at 330 nm was increased and the peak centered at 410 nm was disappear. This assumption was confirmed by the reduction of N–C
O bonds was decomposed into COOH and N–H which similar to the hydrolysis process of amide bond. So the peak centered at 330 nm was increased and the peak centered at 410 nm was disappear. This assumption was confirmed by the reduction of N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds and the increase of C–OH bands in XPS results.
O bonds and the increase of C–OH bands in XPS results.
Further studies revealed that g-CNNSs exhibit SOD-like catalytic activity. SODs are known to be an important kind of metalloprotein enzyme and are common in living cells.24 They can specifically catalyze the dismutation of the superoxide anion (O2˙−) to O2 and H2O2, protecting organisms from oxidative damages from the reactive oxygen species. For this reason, more and more researchers have shown interest in development of SOD mimics.25 The SOD activity of g-CNNSs has been investigated by inhibiting the autoxidation of pyrogallol.26 Under alkaline conditions, pyrogallol rapidly oxidize and release the superoxide (O2˙−), and then generate an intermediate product with color. The UV-vis absorption values of the intermediate product (325 nm) has a good linear relationship with the reaction time. The superoxide dismutase (SOD) added in the autoxidation of pyrogallol reaction system catalyze the dismutation reaction of O2˙− and produces O2 and H2O2 which would blocked the production of colored intermediate products. This led to the decrease of autoxidation rate of pyrogallol and the UV-vis absorption values of the intermediate product. So the low absorption values of the intermediate product means the high activity of SOD. The absorption of the pyrogallol buffer solution (pH = 8.2) with or without g-CNNSs was tested to demonstrate the SOD-like activity of the nanozymes, respectively (Fig. 6B). As can be seen, there was good linear relationship between the difference of absorption values of the autoxidation of pyrogallol system and reaction time. The values was obviously decreased after adding the g-CNNSs. Furthermore, the absorption values decreased successively with increasing g-CNNSs concentration. This proved that the reaction rate of autoxidation of pyrogallol was reduced with g-CNNSs. All these observations indicated that g-CNNSs exhibit SOD-like catalytic activity.
The activities of g-CNNSs with adjusting the molar ratio of urea to sodium citrate have been studied (Fig. 6C). The activities of g-CNNSs increased gradually with decrease the amount of sodium citrate and then decreased when the molar ratio was more than 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The g-CNNSs-2 showed the highest activity. The dependence of the activities of g-CNNSs-2 on the storage time was also investigated. Fig. 6D showed that the storage time affect the SOD-like activities of g-CNNSs-2 and the activities increased gradually with the prolonging of storage time. Some carbon nanomaterials, such as derivatives of the fullerene C60 molecule or the poly(ethylene glycolated) hydrophilic carbon cluster (PEG-HCC), have been reported to show catalytic activity in quenching of O2˙−.25,27 The activity of fullerene believed to be attributed to the structure of the C3 (tris malonic acid C60 derivative). The g-CNNSs after storage would have similar structure to C3 (see Fig. 6A) that may be the reason for the SOD-like activity of g-CNNSs. Moreover, it was usually considered that a destabilizing radical prefers a planar conformation. So g-CNNSs would have good catalytic activity. The activities enhancement of g-CNNSs during the storage time indirectly proved the proposal of the structure of g-CNNSs.
1. The g-CNNSs-2 showed the highest activity. The dependence of the activities of g-CNNSs-2 on the storage time was also investigated. Fig. 6D showed that the storage time affect the SOD-like activities of g-CNNSs-2 and the activities increased gradually with the prolonging of storage time. Some carbon nanomaterials, such as derivatives of the fullerene C60 molecule or the poly(ethylene glycolated) hydrophilic carbon cluster (PEG-HCC), have been reported to show catalytic activity in quenching of O2˙−.25,27 The activity of fullerene believed to be attributed to the structure of the C3 (tris malonic acid C60 derivative). The g-CNNSs after storage would have similar structure to C3 (see Fig. 6A) that may be the reason for the SOD-like activity of g-CNNSs. Moreover, it was usually considered that a destabilizing radical prefers a planar conformation. So g-CNNSs would have good catalytic activity. The activities enhancement of g-CNNSs during the storage time indirectly proved the proposal of the structure of g-CNNSs.
Conclusions
We successfully synthesized bright fluorescent yellow g-CNNSs with tunable optical properties by a low-temperature solid-phase method. The g-CNNSs showed high stability under ion conditions and low cytotoxicity. The luminescence of g-CNNSs aqueous solution changed from yellow to blue under UV excitation in storage or alkaline solution. This was a result of the N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds change of g-CNNSs, which has been demonstrated by the UV-vis absorption and XPS spectra. The g-CNNSs possess intrinsic SOD-like activity and have great potential applications in enzyme catalysis, biological medicine and detection.
O bonds change of g-CNNSs, which has been demonstrated by the UV-vis absorption and XPS spectra. The g-CNNSs possess intrinsic SOD-like activity and have great potential applications in enzyme catalysis, biological medicine and detection.
Acknowledgements
We acknowledge financial support from the National Natural Science Foundation of China (61571426, 61671435, 81671845, 81630053) and Beijing Natural Science Foundation (project No. 4161003).Notes and references
- S. Cao, J. Low, J. Yu and M. Jaroniec, Adv. Mater., 2015, 27, 2150–2176 CrossRef CAS PubMed.
- H. Zhang, L. Guo, L. Zhao, B. Wan and Y. Yang, J. Phys. Chem. Lett., 2015, 6, 958–963 CrossRef CAS PubMed.
- D. J. Martin, K. Qiu, S. A. Shevlin, A. D. Handoko, X. Chen, Z. Guo and J. Tang, Angew. Chem., Int. Ed., 2014, 53, 9240–9243 CrossRef CAS PubMed.
- Y. Hou, Z. Wen, S. Cui, X. Feng and J. Chen, Nano Lett., 2016, 16, 2268–2277 CrossRef CAS PubMed.
- J. K. Gao, J. P. Wang, X. F. Qian, Y. Y. Dong, H. Xu, R. J. Song, C. F. Yan, H. C. Zhu, Q. W. Zhong, G. D. Qian and J. M. Yao, J. Solid State Chem., 2015, 228, 60–64 CrossRef CAS.
- J. Q. Tian, Q. L. Liu, A. M. Asiri, K. A. Alamry and X. P. Sun, ChemSusChem, 2014, 7, 2125–2130 CrossRef CAS PubMed.
- J. P. Wang, H. Xu, X. F. Qian, Y. Y. Dong, J. K. Gao, G. D. Qian and J. M. Yao, Chem.–Asian J., 2015, 10, 1276–1280 CrossRef CAS PubMed.
- Q. Cui, J. Xu, X. Wang, L. Li, M. Antonietti and M. Shalom, Angew. Chem., Int. Ed., 2016, 55, 3672–3676 CrossRef CAS PubMed.
- X. Zhang, X. Xie, H. Wang, J. Zhang, B. Pan and Y. Xie, J. Am. Chem. Soc., 2013, 135, 18–21 CrossRef CAS PubMed.
- J. Q. Tian, Q. Liu, A. M. Asiri, A. O. Al-Youbi and X. P. Sun, Anal. Chem., 2013, 85, 5595–5599 CrossRef CAS PubMed.
- J. Zhou, Y. Yang and C. Zhang, Chem. Commun., 2013, 49, 8605–8607 RSC.
- S. K. Bhunia, A. Saha, A. R. Maity, S. C. Ray and N. R. Jana, Sci. Rep., 2013, 3, 1473–1477 Search PubMed.
- K. Jiang, S. Sun, L. Zhang, Y. Lu, A. Wu, C. Cai and H. Lin, Angew. Chem., Int. Ed., 2015, 54, 5360–5363 CrossRef CAS PubMed.
- H. Sun, A. Zhao, N. Gao, K. Li, J. Ren and X. Qu, Angew. Chem. Int. Ed., 2015, 54, 7176–7180 CrossRef CAS PubMed.
- M. Shamsipur, A. Safavi and Z. Mohammadpour, Sens. Actuators, B, 2014, 199, 463–469 CrossRef CAS.
- J. Mu, J. Li, X. Zhao, E. Yang and X. Zhao, RSC Adv., 2016, 6, 35568–35576 RSC.
- J. Q. Tian, Q. Liu, A. M. Asiri, A. H. Qusti, A. O. Al-Youbi and X. P. Sun, Nanoscale, 2013, 5, 11604–11609 RSC.
- X. L. Ren, J. Liu, J. Ren, F. Q. Tang and X. W. Meng, Nanoscale, 2015, 7, 19641–19646 RSC.
- T. L. Liu, H. Y. Liu, C. H. Fu, L. L. Li, D. Chen and Y. Q. Zhang, J. Colloid Interface Sci., 2013, 400, 168–174 CrossRef CAS PubMed.
- H. Zhang, Y. Huang, S. Hu, Q. Huang, C. Wei, W. Zhang, L. Kang, Z. Huang and A. Hao, J. Mater. Chem. C, 2015, 3, 2093–2100 RSC.
- S. Barman and M. Sadhukhan, J. Mater. Chem., 2012, 22, 21832–21837 RSC.
- V. N. Khabashesku, J. L. Zimmerman and J. L. Margrave, Chem. Mater., 2000, 12, 3264–3270 CrossRef CAS.
- M. Rong, L. Lin, X. Song, T. Zhao, Y. Zhong, J. Yuan, Y. Wang and X. Chen, Anal. Chem., 2015, 87, 1288–1296 CrossRef CAS PubMed.
- Y. Bi, Z. Huang and Y. Zhao, Biosens. Bioelectron., 2006, 21, 1350–1354 CrossRef CAS PubMed.
- E. L. G. Samuel, D. C. Marcano, V. Berka, B. R. Bitner, G. Wu, A. Potter, R. H. Fabian, R. G. Pautler, T. A. Kent, A. Tsai and J. M. Tour, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 2343–2348 CrossRef CAS PubMed.
- S. Marklund and G. Marklund, Eur. J. Biochem., 1974, 47, 469–474 CrossRef CAS PubMed.
- S. S. Ali, J. I. Hardt, K. L. Quick, J. S. Kim-Han, B. F. Erlanger, T. Huang, C. J. Epstein and L. L. Dugan, Free Radical Biol. Med., 2004, 37, 1191–1202 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra21624b |
| This journal is © The Royal Society of Chemistry 2016 |