Phase transition and negative thermal expansion in orthorhombic Dy2W3O12†
Weigang Cao,
Qiang Li,
Kun Lin,
Zhanning Liu,
Jinxia Deng,
Jun Chen and
Xianran Xing*
Department of Physical Chemistry, University of Science and Technology Beijing, Beijing 100083, China. E-mail: xing@ustb.edu.cn; Fax: +86-10-62332525; Tel: +86-10-62334200
First published on 29th September 2016
Abstract
New materials with suitable negative thermal expansion (NTE) are much desired to control thermal expansion in solids. In the present study, we determined the phase transition temperature of Dy2W3O12 from the monoclinic phase to the orthorhombic phase at 996 °C. The orthorhombic phase could be retained by quenching, and further high temperature XRD and synchrotron radiation X-ray powder diffraction (SXRD) experiments revealed that Dy2W3O12 shows NTE (−2.6 × 10−5 °C−1) in the temperature range of 150–500 °C, which is the largest negative thermal expansion in the A2W3O12 family (A = rare earth element). A possible NTE mechanism and enhanced NTE were elucidated by the transverse thermal motion of the bridge oxygen in A–O–W linkages accompanied by distortion of the polyhedra with large Dy3+ on the A site.
Introduction
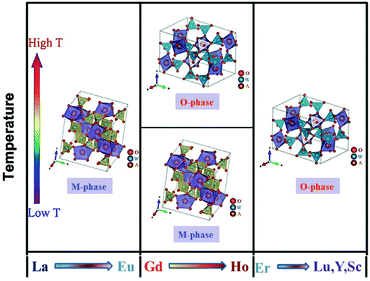
Among the NTE materials, the open-framework structure with NTE property is an important research branch, which includes siliceous faujasite,1 AlPO4,2 ScF3,3–5 ZrW2O8,6–8 A2M3O12 (with A as a trivalent metal cation and M as Mo6+ or W6+),9–16 Zn(CN)2 (ref. 17) and so on. In particular, A2M3O12 is a very large family among these. They have an open-framework structure with corner-sharing polyhedra, i.e., AO6 octahedra share all corners with MO4 tetrahedra, while MO4 tetrahedra connect through vertices to AO6 octahedra. This structure determines that A2M3O12 have high chemical flexibility for highly tunable properties. So far, many studies on A2M3O12 have been carried out, including studies into the mechanism of the NTE,18,19 the adjustment of thermal expansion,20–24 the decrease of hydration,20,25–27 and research on the high pressure behavior.28,29 Among these researches, it was found that both the thermal expansion and the hydration can be adjusted by ionic substitution.In a previous report, the phase diagrams of A2W3O12 with different rare earth elements have been investigated by Nassau.30 These tungstates can be classified into three groups according to their structure change with changes in the ionic radius and temperature, and these are shown in Fig. 1. In group I, A2W3O12 (A = La to Eu) presents the monoclinic phase without transformation to the orthorhombic phase. This group exhibits positive thermal expansion (PTE). In group III, A2W3O12 (A = Er to Lu) adopts an orthorhombic structure and has no phase transition above room temperature, which shows NTE. Group II is a special group, which presents a monoclinic phase at low temperature and transforms to an orthorhombic phase at high temperature. This means that this group shows PTE at low temperature and possesses NTE at high temperature. Owing to difficulty controlling the high temperature phase, seldom have studies on group II investigated the thermal expansion.
Dy2W3O12 is a typical example of group II. Although some studies have been carried out on the photoluminescence properties,31 electrical transport properties32 and magnetic properties33 of its monoclinic phase, there are few investigations into orthorhombic Dy2W3O12. Furthermore, previous studies have shown that the size of the A3+ ion is correlated to thermal expansion in orthorhombic A2W3O12.25 A larger size of A3+ leads to more enhanced NTE, and the cation radius of Dy3+ is large in the orthorhombic A2W3O12 family. So it is significant to investigate orthorhombic Dy2W3O12, which possibly exhibits stronger NTE.
In this paper, we synthesized Dy2W3O12 with an orthorhombic phase by high temperature quenching, investigated the mechanism of synthesis (see in Fig. S1†) and characterized the sample by XRD and TG-DSC. The as-synthesized orthorhombic Dy2W3O12 can be stabilized by 500 °C. The thermal expansion was studied by high temperature XRD and synchrotron radiation X-ray powder diffraction (SXRD). The photoluminescence and the magnetic properties of hydrated orthorhombic Dy2W3O12 were also examined (see the ESI†), which is helpful for the further study of orthorhombic Dy2W3O12.
Experimental
Orthorhombic Dy2W3O12 was prepared by a conventional solid state reaction followed by high temperature quenching. Stoichiometric ratios of Dy2O3 and WO3 were weighed and ground thoroughly in a mortar after they were preheated at 600 °C for 6 h. The obtained mixture was calcined at 1100 °C for 10 h. The calcined powder was milled again and recalcined at 1100 °C for another 10 h, then cooled down slowly to room temperature. The quenched sample had a similar synthesis process as above, but was quenched with cold water at the final calcination.Phase identification and high temperature X-ray powder diffraction (XRD) were carried out on a PANalytical diffractometer with Cu Kα radiation. In order to exhibit the thermal expansion of the quenched sample, high temperature XRD data were collected using an HTK1200 accessory, and recorded in the temperature range from 150 °C to 500 °C at intervals of 50 °C, and between 10° and 80°. SXRD was also used to study the thermal expansion. Variable temperature SXRD data were collected at the BL44B2 beamline of the Spring-8 synchrotron radiation facility, Japan, using a constant wavelength of λ = 1.08017 Å. TG-DSC (SETARAM, Labsys Evo) data of the quenched sample were collected under an air atmosphere to investigate the weight change and the phase transition from room temperature to 1000 °C at a rate of 10 °C min−1. Meanwhile, high temperature XRD was also used to investigate the phase transition from 200 °C to 800 °C. Photoluminescence was investigated with a F-4500 spectrometer, and the magnetic data was recorded by SQUID in the temperature range from −268 °C to 127 °C.
Results and discussions
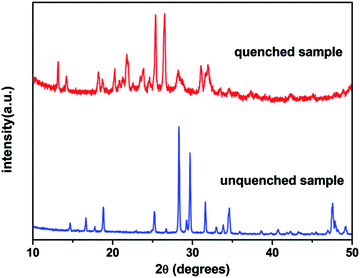
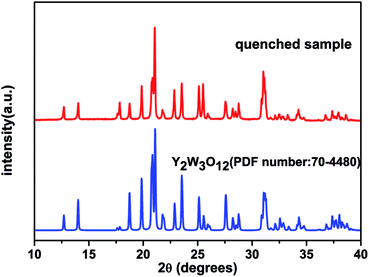
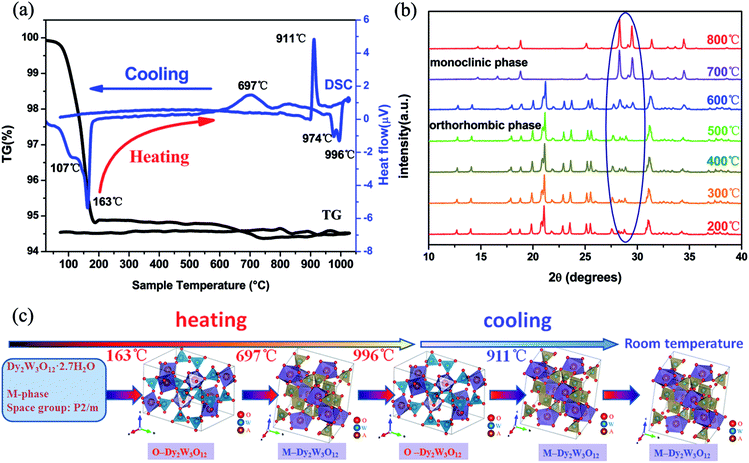
The XRD patterns of Dy2W3O12 with and without quenching can be viewed in Fig. 2. It is obvious that the quenched sample has a different structure to the unquenched one. The unquenched sample can be indexed to the monoclinic phase with the space group of C2/c, while the quenched one has the same XRD pattern as hydrated Y2W3O12 except for some peaks, and belongs to the monoclinic phase with the space group of P2/m. Furthermore, the high background of the quenched sample reveals that the sample coexists with an amorphous phase due to hydration. In an attempt to elucidate the phase of quenched sample, the high temperature XRD method was carried out. Fig. 3 shows the XRD pattern of the quenched sample at 200 °C, which could be indexed to the orthorhombic phase and matches well with the PDF card of orthorhombic Y2W3O12. The quenched sample has an orthorhombic phase with the space group Pnca, and no other phase is detected. On the other hand, the conclusion could be drawn that the quenched sample is easy to hydrate at room temperature like orthorhombic Y2W3O12. The lattice constants of orthorhombic Dy2W3O12 at 200 °C were calculated by the Le Bail fit of the XRD pattern, which can be viewed in the ESI (see in Fig. S2†), and are a = 10.0846(1) Å, b = 13.9955(1) Å, and c = 10.0042(1) Å.Fig. 4(a) displays the TG-DSC curves of the quenched sample with one thermal cycle. According to the slope of the weight loss, it can be identified that there are two regions of weight loss between 100 °C and 200 °C. The first stage of the weight loss is attributed to desorption of H2O, which is adsorbed on the surface. The second weight loss is due to the crystal water, which is equivalent to 44 g mol−1. The formula of the quenched sample is described as Dy2W3O12·2.7H2O by calculating the weight loss of crystal water. There is nearly no weight change above 200 °C in the heating process. Furthermore, there is also no obvious weight change in the cooling process. The DSC curve and the high temperature XRD measurements obviously show the phase transformation as a function of temperature, which is explicitly illustrated in Fig. 4(c). In the DSC curve, the endothermic peaks of 107 °C and 163 °C are in compliance with the weight loss, indicating that Dy2W3O12·2.7H2O changes to orthorhombic Dy2W3O12. The broad exothermic peak at 697 °C is ascribed to the phase transition from the orthorhombic phase to the monoclinic phase, which is identified by the high temperature XRD measurement shown in Fig. 4(b). The high temperature XRD measurement indicated that the phase transition occurs between 500 °C and 600 °C, and total change to the monoclinic phase occurs at 700 °C. This change trend is consistent with the DSC results. The peak at 996 °C is attributed to the phase transition from the monoclinic phase to the orthorhombic phase, which corresponds with the previous report.30 In addition, the peak at 974 °C may be due to the recrystallization of the amorphous phase. Furthermore, the exothermic peak at 911 °C is due to the phase transition from the orthorhombic to monoclinic phase during cooling. At low temperature, the monoclinic phase of Dy2W3O12 is the thermodynamically stable phase. From the TG-DSC and high temperature XRD, we can conclude that the orthorhombic phase is a metastable phase, and exists stably below 500 °C.
The thermal expansion of the quenched sample is investigated by the high temperature XRD and high temperature SXRD methods. Based on the conclusion of the TG-DSC results, the quenched sample was measured below 500 °C. Meanwhile, the hydrated sample doesn’t exhibit negative thermal expansion due to H2O hindering the rocking motion of the bridge oxygen. So the thermal behaviour of the quenched sample is studied above 150 °C. Therefore, high temperature XRD data were collected in the range from 150 °C to 500 °C, in which the quenched sample retains the orthorhombic phase. Furthermore, the quenched sample totally excludes water until 250 °C in the process of collecting SXRD data, which is due to operating in a sealed environment.
The lattice constant and volume of orthorhombic Dy2W3O12 as a function of temperature are shown in Fig. 5. The refined results of the XRD and SXRD are given in Tables S1 and S2, respectively (see the ESI†), and the coefficients of the thermal expansion for the axes and volume were calculated from those data, which are shown in Table 1. From Fig. 5, it is clear to see that the quenched sample shows shrinkage along three axes, and the a-axis and c-axis have stronger negative thermal expansion than the b-axis. The overall volume thermal contraction of the quenched sample is αv = −2.6 × 10−5 °C−1, which is the largest negative thermal expansion in a A2W3O12 system so far, as shown in Table 1. This is because there is a close correlation between the magnitude of the thermal expansion and the size of the A cation, as a larger A cation has a more negative coefficient of thermal expansion. Dy3+ has the largest size in the A2W3O12 family (A = rare earth element) with an orthorhombic phase at present. High temperature quenching is a useful technology to explore the negative thermal expansion.
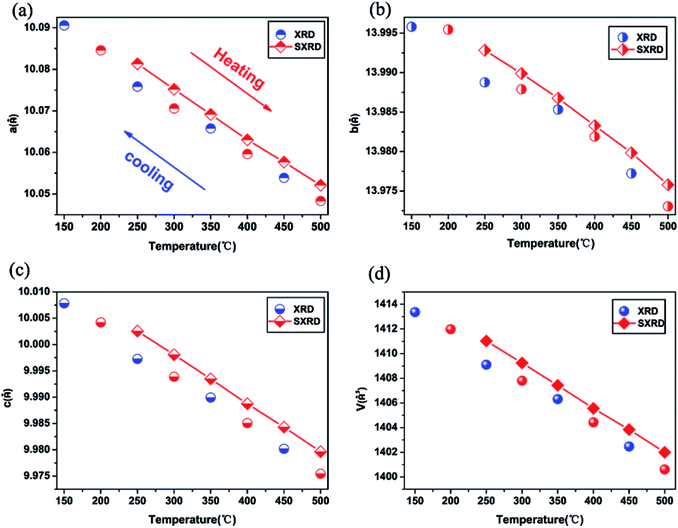 | ||
| Fig. 5 Plots of the evolution of unit cell parameters and cell volume of the quenched sample as a function of temperature. | ||
| Rare earth tungstates | A3+ (Å) | αa (×10−6 °C−1) | αb (×10−6 °C−1) | αc (×10−6 °C−1) | αv (×10−5 °C−1) | Reference |
|---|---|---|---|---|---|---|
| Sc2W3O12 | 0.68 | −6.3 | 7.5 | −5.5 | −0.4 | Evans14 |
| Y2W3O12 | 0.88 | −10.4 | −3.1 | −7.6 | −2.1 | Woodcock15 |
| Lu2W3O12 | 0.848 | −9.7 | −2.9 | −5.7 | −1.8 | Sumithra16 |
| Yb2W3O12 | 0.858 | −10.2 | −2.7 | −6.4 | −1.9 | Sumithra16 |
| Er2W3O12 | 0.881 | −10.1 | −3.4 | −6.7 | −2.0 | Sumithra16 |
| Dy2W3O12 (XRD) | 0.908 | −11.9 | −4.7 | −9.2 | −2.6 | This work |
| Dy2W3O12 (SXRD) | 0.908 | −11.6 | −4.9 | −9.2 | −2.6 | This work |
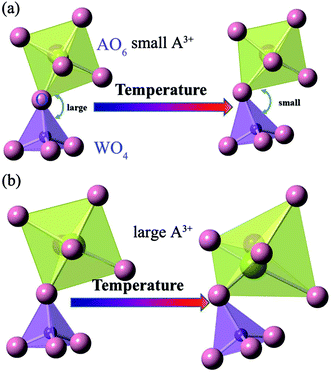
Many detailed and elegant analyses have been carried out on the mechanism of NTE in A2W3O12.13,18,19,25,34–36 The root cause is the transverse thermal motion of the bridge oxygen in A–O–W linkages accompanied by distortion of the polyhedra to shorten the distance between A and W atoms. As far as we know, the magnitude of the thermal expansivity is correlated with the size of the A cation. This is because the rigidity of the polyhedra hinders the rocking motion of the bridging oxygen and then decreases the negative thermal expansion. A detailed description is shown in Fig. 6. With a small A3+, the oxygen–oxygen repulsion makes the polyhedra of AO6 rigid, which decreases the rocking motion of the bridging oxygen. Meanwhile a large A3+ makes the polyhedra of AO6 more flexible. With increasing the ionic size of A3+, the oxygen–oxygen interaction diminishes, and the polyhedra of AO6 appear to be less rigid, which enhances the negative thermal expansion. This is the reason that Dy2W3O12 with the largest cation Dy3+ has the most enhanced thermal expansion in the A2W3O12 (A = rare earth element) family.
Conclusions
The orthorhombic phase of Dy2W3O12 has been successfully synthesized by high temperature quenching, and its structure, phase transition, and thermal expansion have been investigated carefully. The orthorhombic phase is stable below 500 °C and exhibits negative thermal expansion along three axes in the temperature range from 150 °C to 500 °C. The volume thermal contraction is −2.6 × 10−5 °C−1, which is calculated from XRD and SXRD. This is the largest negative thermal expansion in a A2W3O12 (A = rare earth element) system so far. High temperature quenching is an effective technology to explore novel negative thermal expansion materials.Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 21590793, 91422301, and 21231001) and the Fundamental Research Funds for the Central Universities, China (No. FRF-SD-13-008A). The various-temperature XRD experiments for orthorhombic Dy2W3O12 were performed at the BL44B2 of Spring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI) (Proposal No. 2015B1127 and No. 2016A1060).Notes and references
- M. P. Attfield, Chem. Commun., 1998, 601–602 RSC.
- M. P. Attfield and A. W. Sleight, Chem. Mater., 1998, 10, 2013–2019 CrossRef CAS.
- L. Hu, J. Chen, L. Fan, Y. Ren, Y. Rong, Z. Pan, J. Deng, R. Yu and X. Xing, J. Am. Chem. Soc., 2014, 136, 13566–13569 CrossRef CAS PubMed.
- L. Hu, J. Chen, A. Sanson, H. Wu, C. Guglieri Rodriguez, L. Olivi, Y. Ren, L. Fan, J. Deng and X. Xing, J. Am. Chem. Soc., 2016, 138(27), 8320–8323 CrossRef CAS PubMed.
- J. Chen, L. Hu, J. Deng and X. Xing, Chem. Soc. Rev., 2015, 44, 3522–3567 RSC.
- T. Mary, J. Evans, T. Vogt and A. Sleight, Science, 1996, 272, 90 CAS.
- J. Evans, T. Mary, T. Vogt, M. Subramanian and A. Sleight, Chem. Mater., 1996, 8, 2809–2823 CrossRef CAS.
- W. Cao, Q. Huang, Y. Rong, Y. Wang, J. Deng, J. Chen and X. Xing, Inorg. Chem. Front., 2016, 3, 856–860 RSC.
- L. H. Rimmer and M. T. Dove, J. Phys.: Condens. Matter, 2015, 27, 185401 CrossRef PubMed.
- M.-Y. Wu, Y. Jia and Q. Sun, Comput. Mater. Sci., 2016, 111, 28–33 CrossRef CAS.
- B. A. Marinkovic, M. Ari, R. R. de Avillez, F. Rizzo, F. F. Ferreira, K. J. Miller, M. B. Johnson and M. A. White, Chem. Mater., 2009, 21, 2886–2894 CrossRef CAS.
- C. P. Romao, F. A. Perras, U. Werner-Zwanziger, J. A. Lussier, K. J. Miller, C. M. Calahoo, J. W. Zwanziger, M. Bieringer, B. A. Marinkovic, D. L. Bryce and M. A. White, Chem. Mater., 2015, 27, 2633–2646 CrossRef CAS.
- C. P. Romao, K. J. Miller, M. B. Johnson, J. W. Zwanziger, B. A. Marinkovic and M. A. White, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 90(2), 024305 CrossRef.
- J. Evans, T. Mary and A. Sleight, J. Solid State Chem., 1998, 137, 148–160 CrossRef CAS.
- D. A. Woodcock, P. Lightfoot and C. Ritter, J. Solid State Chem., 2000, 149, 92–98 CrossRef CAS.
- S. Sumithra, A. K. Tyagi and A. M. Umarji, Mater. Sci. Eng., B, 2005, 116, 14–18 CrossRef.
- K. W. Chapman, P. J. Chupas and C. J. Kepert, J. Am. Chem. Soc., 2005, 127, 15630–15636 CrossRef CAS PubMed.
- A. K. Pryde, K. D. Hammonds, M. T. Dove, V. Heine, J. D. Gale and M. C. Warren, J. Phys.: Condens. Matter, 1996, 8, 10973 CrossRef CAS.
- B. A. Marinkovic, M. Ari, R. R. de Avillez, F. Rizzo, F. F. Ferreira, K. J. Miller, M. B. Johnson and M. A. White, Chem. Mater., 2009, 21, 2886–2894 CrossRef CAS.
- Z. Yu, J. Peng, H. Wang, M. Wu, Y. Cheng, Z. Hu and D. Chen, Sci. China, Ser. E: Technol. Sci., 2008, 51, 25–32 CrossRef CAS.
- J. Peng, X. Liu, F. Guo, S. Han, Y. Liu, D. Chen and Z. Hu, Int. J. Miner., Metall. Mater., 2010, 17, 786–790 CrossRef CAS.
- M.-M. Wu, J. Peng, Y. Zu, R.-D. Liu, Z.-B. Hu, Y.-T. Liu and D.-F. Chen, Chin. Phys. B, 2012, 21, 116102 CrossRef.
- H. Liu, Z. Zhang, W. Zhang, X. Zeng and X. Chen, Ceram. Int., 2013, 39, 2781–2786 CrossRef CAS.
- X. Ge, Y. Mao, X. Liu, Y. Cheng, B. Yuan, M. Chao and E. Liang, Sci. Rep., 2016, 6, 24832 CrossRef CAS PubMed.
- Z. Y. Li, W. B. Song and E. J. Liang, J. Phys. Chem. C, 2011, 115, 17806–17811 CAS.
- Q.-J. Li, B.-H. Yuan, W.-B. Song, E.-J. Liang and B. Yuan, Chin. Phys. B, 2012, 21, 046501 CrossRef.
- M.-Y. Wu, L. Wang, Y. Jia, Z.-X. Guo and Q. Sun, AIP Adv., 2015, 5, 027126 CrossRef.
- R. Secco, H. Liu, N. Imanaka and G. Adachi, J. Mater. Sci. Lett., 2001, 20, 1339–1340 CrossRef CAS.
- S. Achary, G. Mukherjee, A. Tyagi and S. Vaidya, J. Mater. Sci., 2002, 37, 2501–2509 CrossRef CAS.
- K. Nassau, H. Levinstein and G. Loiacono, J. Phys. Chem. Solids, 1965, 26, 1805–1816 CrossRef CAS.
- W. Zhang, C. Lu, Y. Ni, J. Song, W. Huang, J. Tao and Z. Xu, J. Wuhan Univ. Technol., Mater. Sci. Ed., 2013, 5, 002 Search PubMed.
- K. Maurya, A. Thakur and S. Yadav, Asian J. Chem., 2011, 23, 3089 CAS.
- R. Shen, C. Wang, T. Wang, C. Dong, X. Chen and J. Liang, Rare Met., 2003, 1, 49 Search PubMed.
- S. Sumithra, U. V. Waghmare and A. M. Umarji, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76(2), 024307 CrossRef.
- L. H. Rimmer and M. T. Dove, J. Phys.: Condens. Matter, 2015, 27, 185401 CrossRef PubMed.
- L. Wang, F. Wang, P.-F. Yuan, Q. Sun, E.-J. Liang, Y. Jia and Z.-X. Guo, Mater. Res. Bull., 2013, 48, 2724–2729 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: The mechanism of quenching, Le Bail fit of XRD data of the quenched sample at 200 °C, the cell parameters of orthorhombic Dy2W3O12 versus temperature from XRD results and SXRD results, the photoluminescence and the magnetic properties of the quenched sample. See DOI: 10.1039/c6ra21136d |
| This journal is © The Royal Society of Chemistry 2016 |