N,N′-Diaryl-perylene-3,9-diamine derivatives: synthesis, characterization and electroluminescence properties†
Érica Torresab,
Rafal Bogel-Lukasik c,
Mário Nuno Berberan-Santos
c,
Mário Nuno Berberan-Santos b,
Stefan Höfle*d,
Alexander Colsmannd and
Maria João Brites*a
b,
Stefan Höfle*d,
Alexander Colsmannd and
Maria João Brites*a
aLNEG – UES – Unidade de Energia Solar, Estrada do Paço do Lumiar, 22, 1649-038 Lisboa, Portugal. E-mail: mjoao.brites@lneg.pt; Fax: +351-217127195; Tel: +351-210924600
bCQFM-IST – CQFM – Centro de Química-Física Molecular, IN – Institute of Nanoscience and Nanotechnology, Instituto Superior Técnico, Universidade de Lisboa, 1049-001 Lisboa, Portugal
cLNEG – UB – Unidade de Bioenergia, Estrada do Paço do Lumiar, 22, 1649-038 Lisboa, Portugal
dLight Technology Institute, Karlsruhe Institute of Technology (KIT), Engesserstrasse 13, 76131 Karlsruhe, Germany. E-mail: stefan.hoefle@kit.edu
First published on 3rd November 2016
Abstract
In this work, N-N′-diaryl-perylene-3,9-diamine (PDA) derivatives featuring different alkoxyl substituents at the para position of the N-aryl rings were synthesized and employed as emitters in solution processed organic light emitting diodes. Their physical properties were studied by UV-Vis absorption, fluorescence, differential scanning calorimetry, photo-electron yield spectroscopy and cyclic voltammetry, and were correlated with the OLED performance. Changing the length of the alkoxyl groups influences the PDAs' thermal properties, their bulk crystallinities and affects the performance of the respective OLEDs. The OLED emission spectra are red-shifted up to wavelengths of 800 nm, covering a spectral emission regime formerly inaccessible by other PDA derivatives.
Introduction
Although vacuum-deposited organic light emitting diodes (OLEDs) have been introduced to displays and TVs in the consumer market, the strong cost pressure inspires researchers to look for less expensive fabrication methods, such as device printing from solution. Commonly, polymers are considered more suitable for solution processing than low-molecular weight materials due to their better film forming properties. Low-molecular weight materials, however, have the advantage of higher purity and hence superior optoelectronic performance. Since most low-molecular weight materials were designed for vacuum processing, the prospect of future device printing will require new material engineering to solubilize such molecules.Perylenes are an important class of organic chromophores. Although first developed as dyes, their applications have reached far beyond the original purpose.1,2 Among all members of the perylene family, perylene diimides (PDI), monoimides (PMI) and monohydrides (PMA) have been investigated as active components in organic electronic devices such as light emitting diodes or solar cells.3–6 In addition to their exceptional chemical, thermal and photochemical stability, PDIs exhibit strong optical absorption in the visible to near-infrared spectral range and fluorescence with quantum yields near unity. Moreover, they show good charge carrier transport properties.7
Perylene derivatives with electron donating substituents, such as diphenylamino (DPA) groups, were also reported as organic electroluminescent materials, and molecular design of these compounds with or without methyl or styryl-substituents on DPA groups proved useful for controlling color emission from blue to red.8 To obtain compounds with red-shifted absorption, two strategies are adopted: extending the π-conjugation length of the molecules or introducing an additional donor (AD) to the main donor.
In this work, we report on the synthesis and characterization of N,N′-diaryl-perylene-3,9-diamine derivatives (PDA) with different alkoxyl substituents (methoxyl or hexyloxyl) at para position of N-aryl rings, in order to increase the donor strength of the DPA group. With this chemical modification we yielded an emission at longer wavelengths than previously reported for DPA-substituted perylenes,8,9 and also improved the compounds' solubility in common organic solvents and hence their processability from solution. The effect of the alkyl chain length in alkoxy groups attached to the DPA donors on the PDAs' photophysical, electrochemical and thermal properties were investigated. Furthermore, their impact on the optoelectronic properties of solution processed organic light emitting diodes (OLEDs) was studied.
Results and discussion
Synthesis
The molecules were synthesized as outlined in Scheme 1. The diarylamines bis(4-methoxy-phenyl)amine 3, 4-(hexyloxy-N-4(-methoxy-phenyl)aniline) 4 and bis-(4-hexyloxy-phenyl)-amine 5 were synthesized following the route previously described in the literature.10 | ||
| Scheme 1 (i) Br2 (1.0 equiv.), benzene, 2 h 30 min, −5 °C; (ii) diarylamine 3/4/5 (2.0 equiv.), Pd(OAc)2 (0.020 equiv.), P(t-Bu)3 (0.04 equiv.), NaOt-Bu (1.5 equiv.), anhydrous toluene, 120 °C. | ||
The synthesis starts with dibromation of 1 with bromine in benzene at −5 °C to give 3,9(10)-dibromoperylene 2 as a yellow-green solid in high yield (92%) which was used in the next step without further purification.11 Coupling of 2 with diarylamines 3, 4 and 5 under Buchwald–Hartwig reaction conditions [Pd(OAc)2/P(t-Bu)3/NaOt-Bu] in toluene yielded N,N′-diaryl-perylene-3,9-diamine derivatives 6a, 6b and 6c as red solids. These target compounds were isolated with 88%, 68% and 67% yields, respectively, by rapid vacuum filtration through a pad of silica in lieu of column chromatography. The chemical structures and purities of these materials were verified through the use of NMR spectroscopy and high-resolution mass spectrometry. The perylene arylamine derivatives 6a–c are soluble in most organic solvents [e.g. toluene, N,N-dimethylformamide (DMF), ethanol (EtOH), methanol (MeOH), dichloromethane (DCM), diethyl ether (Et2O)] enabling thin-film deposition from solution.
Optical and photophysical properties
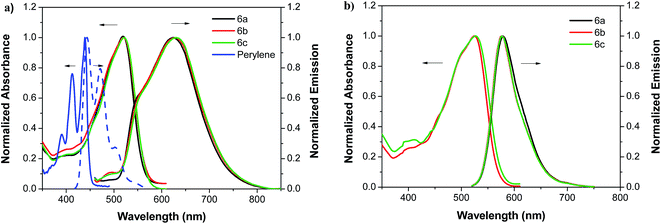
The UV-Vis absorption and photoluminescence (PL) spectra of N,N′-diaryl-perylene-3,9-diamine derivatives 6a, 6b and 6c in toluene solution and in Zeonex films are shown in Fig. 1.The perylene diarylamine derivatives show intensive charge-transfer absorption bands in the visible region with a unique peak at 519 nm for 6a and 6b and 521 nm for 6c, respectively. The molar absorption coefficients of 6a–c range between 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 43
000 and 43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 at the absorption maximum (Table 1).
000 M−1 cm−1 at the absorption maximum (Table 1).
| λmaxa (nm) (ε/M−1 cm−1) | λemb (nm) | Stokes shiftd (Δλ/nm) | ΦFe | τFf (ns) | kr/107h (s−1) | knr/107i (s−1) | |
|---|---|---|---|---|---|---|---|
| a Wavelength of the absorption spectrum maximum and the maximum molar absorption coefficient.b Wavelength of the emission spectrum maximum in toluene solution.c Wavelength of the emission spectrum maximum in Zeonex film.d Stokes shift.e Fluorescence quantum yield.f Fluorescence lifetime in solution and in Zeonex film.g Fluorescence lifetime in Zeonex film.h Radiative rate constant.i Non-radiative rate constant. | |||||||
| 1 | 440 (34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) 000) |
444 | 4 | 0.95 | 4.0 | 24 | 1.3 |
| 6a | 519 (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300) 300) |
625 (578)c | 106 | 0.72 | 5.1 (6.0)g | 14 | 5.5 |
| 6b | 519 (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) 000) |
627 (576)c | 108 | 0.76 | 3.6 (6.8)g | 21 | 6.7 |
| 6c | 521 (42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 870) 870) |
629 (576)c | 108 | 0.84 | 5.1 (7.6)g | 17 | 3.1 |
Upon photoexcitation, the compounds 6a–c show strong orange emission in both solution and solid state. The solution photoluminescence (PL) spectra of 6a, 6b and 6c show a maximum emission at 625, 627 and 629 nm, respectively, and a high energy shoulder at 547 nm, the latter disappearing in the Zeonex thin-film PL emission spectra. We attribute this observation to an excited-state rotamer with weak intramolecular charge-transfer (ICT) formed in solution that disappears in solid state since excited state intramolecular rotation of bulky groups is suppressed in a rigid medium.
The compounds show large Stokes shifts of more than 100 nm and their fluorescence quantum yields (ΦF) measured in toluene solution, range from 0.72 to 0.84. The average fluorescence lifetimes τ of 6a–c measured in toluene at room temperature are determined from fits with a sum of three exponentials. The radiative rate constants (kr) of all compounds are close to the kr of the perylene molecule 1, however the absorption and emission spectra are significantly red-shifted (about 80 nm and 180 nm respectively) due to the introduction of additional electro-donor substituents on DPA groups.12
Electrochemical properties
The redox properties of the compounds 6a–c were studied by cyclic voltammetry (CV) in DMF solution containing 0.1 M of TBAPF6 as supporting electrolyte in a three electrode cell and potentiostat assembly. The redox potentials were measured against an Ag/Ag+ reference electrode and the CV curves (see ESI†) were calibrated using the ferrocene/ferrocenium (Fc/Fc+) redox couple as an external standard which was measured under the same condition after the measurement of samples. The half-wave potential of Fc/Fc+ in DMF was 0.488 V.The N,N′-diaryl-perylene-3,9-diamine derivatives 6a–c showed irreversible oxidation with a single oxidation potentials (versus Fc/Fc+ system) of 0.292 V, 0.272 V and 0.242 V, respectively (Table 2). The oxidation potentials of 6a–c decreased slightly with increasing size of the alkyl groups.
The ionization potential (IP) energy (EIP) was estimated from the onset of the first oxidation peak (Table 2) and calibrated versus Fc/Fc+ (−4.8 eV).13,14 The IP of the compounds 6a–c in thin-films were determined by photo-electron yield spectroscopy in air (PESA).
Although PESA measurements on solid thin-films yielded higher ionisation potentials for all three compounds compared to CV measurements in solution (difference of about 0.3 eV),15 similar trends were observed with both techniques, with 6a showing the lowest and 6c the highest IP. The substitution of methyl (Me) by hexyl (Hex) chains in the alkoxy groups causes a slight increase in the IP (about 0.02–0.03 eV by each Me replaced) which may originate from interactions between neighboring side chains that can modify the charge distribution in the molecule.
We note that the differences in the IP energies as determined by CV and PESA has already been reported in the literature and can be attributed to solvation effects in the cyclic voltammetry setup.16,17
Thermophysical properties
For OLED applications, the thermal stability of materials and their molecular order is crucial for the device stability and lifetime. For example, low glass transition temperatures Tg of an amorphous organic phase lead to faster degradation and eventually device failure, fostered by the elevated temperatures that can occur during device operation. Also, the crystallinity of the emitter can affect its electronic properties. Here, the crystallinity of the planar perylene core may be affected by long alkyl chains attached to the 4-alkoxy-phenylamino groups.Therefore, we investigated the influence of the methoxy (OMe) and hexyloxy (OHex) groups on the thermal properties of the N,N′-diaryl-perylene-3,9-diamine derivatives 6a–c by Differential Scanning Calorimetry (DSC). This technique allows the detection of phase transitions as characteristic endothermic or exothermic peaks of melting or crystallization and to trace effects stemming from changes in heat capacity of the materials (step-like changes), such as glass transitions. The thermal properties of 6a–c are summarized in Table 3, and the respective DSCs curves are depicted in Fig. 2.
| Compound | Tga [°C] | Tcb [°C] | Tmc [°C] | ΔHmd [J g−1] | Ttre [°C] | ΔHtrf [J mol−1] |
|---|---|---|---|---|---|---|
| a Glass transition temperature (Tg).b Crystallization temperature (Tc).c Melting temperature (Tm).d Melting enthalpy (ΔHm).e Solid–solid phase transition temperature (Ttr).f Enthalpy of solid–solid phase transition (ΔHtr). | ||||||
| 6a | 86 | 123 | 137 | 0.40 | — | — |
| 6b | — | 33 | 54 | 0.96 | — | — |
| 6c | — | 3 | 60 | 22.97 | 35.07 | 1.16 |
 | ||
| Fig. 2 DSC thermograph of (a) 6a and (b) 6b and 6c, measured at a heating rate of 10 °C min−1 in air (1st heating cycle). | ||
DSC measurements reveal that the phase transition temperatures of the N,N′-diaryl-perylene-3,9-diamine derivatives 6a–c strongly depend on the alkyl chain attached to the 4-alkoxy-phenylamino group in the perylene core.
In the DSC analysis presented in Fig. 2a, compound 6a comprising four OMe groups exhibits a glass transition temperature Tg = 86 °C and a melting point Tm = 137 °C. At a temperature of Tc = 123 °C, negligible crystallization (exothermic peak) was observed. In the cooling cycle, no further transitions were observed besides a small exothermic peak at Tm = 137 °C due to re-crystallization of 6a. In all subsequent heating/cooling runs, 6a exhibits similar thermal behavior. Noteworthy, the melting enthalpy of ΔHm = 0.4 J g−1 (0.0005 J mol−1) is significantly lower than typical melting enthalpies of molecular compounds which are in the order of tens of kJ mol−1.18 This indicates that, right below Tm and above Tc, the organized (semicrystalline) structure of 6a can be easily disturbed (low energy required) to produce an amorphous structure. Similar thermal properties (e.g., a pronounced glass transition peak at low Tg) have already been reported for N-annelated perylene derivatives and fluorene arylamine derivatives with OMe substituents.19,20
The crystallization and melting transition temperatures of compound 6c with four OHex groups are considerably lower than those obtained for 6a and the compound does not show glass transition. The DSC thermograph shows an exothermic peak of crystallization at Tc = 3 °C followed by a solid–solid phase transition at Ttr = 35 °C and a melting transition at Tm = 60 °C. In subsequent heating/cooling runs similar thermal properties were observed (see Fig. S2 in the ESI†). From the low Tc we conclude that 6c is predominantly crystalline at room temperature.
As depicted in Fig. 2b, compound 6b with asymmetric alkoxy groups (OMe and OHex) does not show glass transition but only crystallization at Tc = 33 °C and melting at Tm = 54 °C. In subsequent heating/cooling runs, a change of the melting enthalpy ΔHm was observed depending on the sample holdup time at −20 °C (Fig. S3 and S4, ESI†). For a holdup time of zero (2nd run and 4th run), the melting enthalpy is only 10% of the melting enthalpy obtained in the 1st run, whereas for a holdup time of 20 min (3rd run) the melting enthalpy increases again to its original value. These results indicate that the crystallization kinetics of 6b are slow, i.e., longer induction times are needed for nucleation.
We found that the crystallization kinetics of 6b do not only depend on the holdup time but also on the starting temperature. We performed DSC measurements of heating/cooling runs of 6a, 6b and 6c starting somewhat below room temperature, at 15 °C (Fig. 3). Here, we did not observe any crystallization of 6b, while 6a and 6c exhibit the thermal behavior analogous to previously observed (Fig. 2).
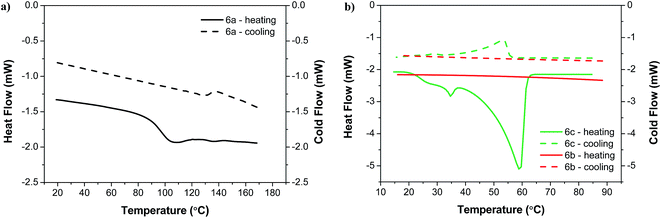 | ||
| Fig. 3 DSC thermographs of heating and cooling runs at 10 °C min −1 performed in air (1st heating and cooling cycles): (a) 6a (15–170 °C) and (b) 6b and 6c (15–90 °C). | ||
Electroluminescence properties
To investigate the electroluminescence (EL) properties of N,N′-diaryl-perylene-3,9-diamine derivatives 6a, 6b and 6c, solution processed OLEDs, henceforth referred to as OLED-A, OLED-B and OLED-C, were fabricated employing the device architecture ITO/ZnO/PEI/emission layer/MoO3/Ag.21,22 The emission layers were deposited from 6a, 6b or 6c solution by spin coating, yielding low surface roughnesses of Rq = 1.5 nm as determined by atomic force microscopy.Fig. 4 shows the optoelectronic characteristics of typical OLEDs. Despite the same functional perylene core of 6a, 6b and 6c, we find the J–V curves of the respective OLEDs shifted to higher or lower voltages. The superior performance of OLED-B and OLED-C over OLED-A may be attributed to the longer alkyl chains of 6b and 6c which enhance the molecular order and thus improve charge carrier transport. This higher molecular order is reflected in the thermophysical properties in Table 3 and, in particular, in the crystallization temperatures Tc: at room temperature, the degree of crystallinity increases from 6a to 6b to 6c. 6a can be considered amorphous (highest Tc = 123 °C), whereas 6c exhibits the highest degree of crystallinity (lowest Tc = 3 °C). 6b exhibits an intermediate Tc, with the precise degree of crystallinity of 6b being difficult to anticipate, since Tc = 33 °C is close to room temperature and OLEDs intrinsically operate at somewhat elevated temperatures. The thermophysical analysis also shows an additional phase transition of compound 6c at 35 °C, and hence within the device operation temperature regime, which may be the origin for OLED-B outperforming OLED-C. The different device performances are also reflected in the onset voltages: OLED-A shows an onset voltage of Von = 5.1 V (at a luminance L = 1 cd m−2), whereas the onset voltage of OLED-B is Von = 2.7 V and the onset voltage of OLED-C is Von = 3.3 V. Besides the lowest onset voltage, OLED-B also shows the highest luminance of all three devices, exceeding 1000 cd m2, which is commonly considered suitable for consumer display applications. Furthermore, we observed a broadening of the electroluminescence spectra from OLED-A to OLED-C in Fig. 4b. The higher the molecular order as induced by the alkyl chains, the better is the inter-molecular interaction of π-orbitals which may lead to a red-shift of the long-wavelength slope of the emission spectrum. This effect is not visible in the photoluminescence spectra in Fig. 1 that were recorded in solution where the molecular order is much lower.
Conclusions
Three novel N,N′-diaryl-perylene-3,9-diamine derivatives were synthesized, characterized and employed as emitter layers in OLEDs. We found that these materials exhibit similar electro-optical properties, independently of the alkoxy-substituted arylamino groups present in the molecule. However, the alkyl chains of the 4-alkoxy-phenylamino groups in 6a–c affect the PDAs' thermal properties, affecting the degree of crystallization in thin-films and the performance of the respective OLEDs. Solution processed, inverted OLEDs comprising 6a–c emit in the orange/red color regime (maximum peak wavelength 620 nm). Compound 6b with asymmetric alkyl chains (Me and Hex) in 4-alkoxy-phenylamino group yielded the best device performance.Experimental section
Materials
All building blocks were used as purchased without further purifications. Solvents were dried and distilled immediately prior to use by standard procedures.23 Vacuum filtration purification was performed using silica gel (230–400 mesh) as the stationary phase in a filter funnel with porous plate. Tetrabutylammonium hexafluorophosphate (TBAPF6-98%) used for cyclic voltammetry (CV) measurements was purified by recrystallization from ethanol and dried under vacuum. The polymeric films were prepared from cyclic polyolefin Zeonex 480R (average Mw = 480![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, pellets) from Zeon Chemicals L.P.
000, pellets) from Zeon Chemicals L.P.
Synthesis
The diarylamines bis(4-methoxy-phenyl)amine 3, 4-(hexyloxy-N-4(-methoxy-phenyl)aniline) 4 and bis-(4-hexyloxy-phenyl)-amine 5 were synthesized by following reported procedure.103,9(10)-dibromoperylene
Perylene (1.00 g, 3.96 mmol) was dissolved in benzene (30 mL). Bromine (406.00 μL, 7.92 mmol) was slowly added to the mixture at −5 °C under nitrogen atmosphere. The reaction mixture was stirred for 2.5 h. The solvent was removed under vacuum. The mixture was washed with HCl solution (0.10 M) and extracted with chloroform (2 × 20 mL). The organic layer was dried over anhydrous MgSO4. After filtration, the solvent was removed under reduced pressure. The remaining solvent was fully evaporated using a vacuum pump to afford compound 2 as a yellow-green solid (1.49 g, 92%). Mp 223–225 °C;11 IR νmax (KBr): 1501, 1491 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic ring), 820, 811, 807 (C–H aromatic ring), 782, 760, 759 (C–Br) cm−1; 1H NMR d (400 MHz; CDCl3): 7.53 (1H, d, J = 8.0 Hz), 7.57 (1H, d, J = 8.0 Hz), 7.72 (2H, t, J = 8.0 Hz), 7.90 (1H, d, J = 8.0 Hz), 7.94 (1H, d, J = 8.0 Hz), 8.07 (2H, d, J = 8.0 Hz), 8.14 (1H, d, J = 7.6 Hz), 8.18 (1H, d, J = 8.0 Hz) ppm. HRMS (ESI): found [M]+ 408.9142; molecular formula C20H10Br2 requires [M]+ 408.9129.
C aromatic ring), 820, 811, 807 (C–H aromatic ring), 782, 760, 759 (C–Br) cm−1; 1H NMR d (400 MHz; CDCl3): 7.53 (1H, d, J = 8.0 Hz), 7.57 (1H, d, J = 8.0 Hz), 7.72 (2H, t, J = 8.0 Hz), 7.90 (1H, d, J = 8.0 Hz), 7.94 (1H, d, J = 8.0 Hz), 8.07 (2H, d, J = 8.0 Hz), 8.14 (1H, d, J = 7.6 Hz), 8.18 (1H, d, J = 8.0 Hz) ppm. HRMS (ESI): found [M]+ 408.9142; molecular formula C20H10Br2 requires [M]+ 408.9129.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 8
1 to 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as eluent.
2) as eluent.
N,N,N′,N′-Tetrakis-(4-methoxy-phenyl)-perylene-3,9-diamine (6a). The general method was used with 3,9(10)-dibromoperylene 2 (0.100 g, 0.245 mmol) and bis(4-methoxyphenyl)amine 3 (0.112 g, 0.490 mmol) to afford 6a (0.153 g, 88%) as a red solid. Mp: 133–134 °C; IR νmax (KBr): 2922, 2852 (C–H alkyl), 1508, 1500 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic ring), 1265 (–C–O–C–), 1240 (C–N), 1034, 826, 809 (C–H aromatic ring) cm−1; 1H NMR d (400 MHz; DMSO): 3.68 (12H, OCH3, s), 6.75 (8H, d, J = 8.0 Hz), 6.90 (8H, d, J = 8.0 Hz), 7.12 (2H, d, J = 8.0 Hz), 7.38 (1H, d, J = 8.0 Hz), 7.40 (1H, d, J = 8.0 Hz), 7.73 (2H, t, J = 8.0 Hz), 8.16 (1H, d, J = 8.0 Hz), 8.19 (1H, d, J = 8.0 Hz), 8.26 (1H, d, J = 8.0 Hz), 8.29 (1H, d, J = 8.0 Hz) ppm; 13C NMR δ (400 MHz, DMSO): 155.1, 149.9, 144.1, 142.5, 131.6, 131.5, 129.8, 128.1, 127.4, 126.9, 124.8, 124.5, 123.8, 122.0, 121.6, 121.1, 119.0, 116.6, 115.3, 55.8 ppm; HRMS (ESI): found [M]+ 706.2814; molecular formula C48H38N2O4 requires [M]+ 706.2826.
C aromatic ring), 1265 (–C–O–C–), 1240 (C–N), 1034, 826, 809 (C–H aromatic ring) cm−1; 1H NMR d (400 MHz; DMSO): 3.68 (12H, OCH3, s), 6.75 (8H, d, J = 8.0 Hz), 6.90 (8H, d, J = 8.0 Hz), 7.12 (2H, d, J = 8.0 Hz), 7.38 (1H, d, J = 8.0 Hz), 7.40 (1H, d, J = 8.0 Hz), 7.73 (2H, t, J = 8.0 Hz), 8.16 (1H, d, J = 8.0 Hz), 8.19 (1H, d, J = 8.0 Hz), 8.26 (1H, d, J = 8.0 Hz), 8.29 (1H, d, J = 8.0 Hz) ppm; 13C NMR δ (400 MHz, DMSO): 155.1, 149.9, 144.1, 142.5, 131.6, 131.5, 129.8, 128.1, 127.4, 126.9, 124.8, 124.5, 123.8, 122.0, 121.6, 121.1, 119.0, 116.6, 115.3, 55.8 ppm; HRMS (ESI): found [M]+ 706.2814; molecular formula C48H38N2O4 requires [M]+ 706.2826.
N,N′-Bis-(4-hexyloxy-phenyl)-N,N′-bis-(4-methoxy-phenyl)-perylene-3,9-diamine (6b). The general method was used with 3,9(10)-dibromoperylene 2 (0.200 g, 0.490 mmol) and 4-(hexyloxy-N-4(-methoxy-phenyl)aniline) 4 (0.293 g, 0.980 mmol) to afford 6b (0.280 g, 68%) as a red solid. Mp: 57–60 °C; IR νmax (KBr): 2928, 2859 (C–H alkyl), 1508, 1501 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic ring), 1298 (–C–O–C–), 1474, 1466, 1389 (C–H alkyl), 1239 (C–N), 1032, 829, 809 (C–H aromatic ring) cm−1, 1H NMR d (400 MHz; DMSO): 0.88 (6H, m), 1.30 (6H, t, J = 8.0 Hz), 1.26–1.30 (8H, m), 1.40–1.44 (4H, m), 1.66–1.70 (4H, m), 3.69 (6H, OCH3, s), 3.87 (4H, t, J = 8.0 Hz), 6.77 (8H, d, J = 8.0 Hz), 6.93 (8H, d, J = 8.0 Hz), 7.17 (2H, d, J = 8.0 Hz), 7.42 (1H, t, J = 8.0 Hz), 7.48 (2H, d, J = 8.0 Hz), 7.75 (1H, t, J = 8.0 Hz), 8.25 (2H, d, J = 8.0 Hz), 8.32 (2H, d, J = 8.0 Hz) ppm; 13C NMR δ (400 MHz, CDCl3): 154.8, 154.5, 131.6, 144.5, 142.7, 142.6, 142.5, 142.3, 140.5, 136.1, 134.6, 134.0, 131.7, 130.0, 129.3, 128.2, 126.7, 126.3, 123.8, 123.6, 68.3, 55.5, 31.6, 30.6, 29.4, 27.5, 25.8, 22.6, 14.1 ppm; HRMS (ESI): found [M]+ 846.4381; molecular formula C58H58N2O4 requires [M]+ 846.4391.
C aromatic ring), 1298 (–C–O–C–), 1474, 1466, 1389 (C–H alkyl), 1239 (C–N), 1032, 829, 809 (C–H aromatic ring) cm−1, 1H NMR d (400 MHz; DMSO): 0.88 (6H, m), 1.30 (6H, t, J = 8.0 Hz), 1.26–1.30 (8H, m), 1.40–1.44 (4H, m), 1.66–1.70 (4H, m), 3.69 (6H, OCH3, s), 3.87 (4H, t, J = 8.0 Hz), 6.77 (8H, d, J = 8.0 Hz), 6.93 (8H, d, J = 8.0 Hz), 7.17 (2H, d, J = 8.0 Hz), 7.42 (1H, t, J = 8.0 Hz), 7.48 (2H, d, J = 8.0 Hz), 7.75 (1H, t, J = 8.0 Hz), 8.25 (2H, d, J = 8.0 Hz), 8.32 (2H, d, J = 8.0 Hz) ppm; 13C NMR δ (400 MHz, CDCl3): 154.8, 154.5, 131.6, 144.5, 142.7, 142.6, 142.5, 142.3, 140.5, 136.1, 134.6, 134.0, 131.7, 130.0, 129.3, 128.2, 126.7, 126.3, 123.8, 123.6, 68.3, 55.5, 31.6, 30.6, 29.4, 27.5, 25.8, 22.6, 14.1 ppm; HRMS (ESI): found [M]+ 846.4381; molecular formula C58H58N2O4 requires [M]+ 846.4391.
N,N,N′,N′-Tetrakis-(4-hexyloxy-phenyl)-perylene-3,9-diamine (6c). The general method was used with 3,9(10)-dibromoperylene 2 (0.200 g; 0.490 mmol) and bis-(4-hexyloxy-phenyl)-amine 5 (0.359 g; 0.973 mmol) to afford 6c (0.320 mg; 67%) as a red solid. Mp: 48–50 °C; IR νmax (KBr): 2929, 2858 (C–H alkyl), 1503 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic ring), 1474, 1467, 1468, 1390 (C–H alkyl), 1266 (–C–O–C–), 1239 (C–N), 1030, 825, 808 (C–H aromatic ring) cm−1, 1H NMR δ (400 MHz; CDCl3): 0.86–0.90 (12H, m), 1.29–1.33 (16H, m), 1.40–1.45 (8H, m), 1.70–1.75 (8H, m), 3.90 (8H, t, J = 8.0 Hz), 6.75 (8H, d, J = 8.0 Hz), 6.90 (8H, d, J = 8.0 Hz), 7.19 (1H, brs), 7.29–7.32 (2H, m), 7.50–7.53 (1H, m), 7.67–7.71 (2H, m), 8.09–8.13 (4H, m) ppm; 13C NMR δ (400 MHz, CDCl3): 154.4, 149.8, 148.2, 144.5, 142.5, 142.6, 142.5, 136.1, 133.0, 134.0, 131.6, 130.9, 130.0, 129.6, 128.3, 127.9, 127.7, 127.4, 127.0, 126.7, 126.5, 126.3, 124.4, 123.7, 122.6, 122.2, 121.6, 121.3, 121.1, 120.5, 120.2, 115.2, 68.3, 31.6, 30.6, 29.4, 25.7, 25.8, 22.6, 14.0 ppm; HRMS (ESI): found [M]+ 986.5939; molecular formula C68H78N2O4 requires [M]+ 986.5956.
C aromatic ring), 1474, 1467, 1468, 1390 (C–H alkyl), 1266 (–C–O–C–), 1239 (C–N), 1030, 825, 808 (C–H aromatic ring) cm−1, 1H NMR δ (400 MHz; CDCl3): 0.86–0.90 (12H, m), 1.29–1.33 (16H, m), 1.40–1.45 (8H, m), 1.70–1.75 (8H, m), 3.90 (8H, t, J = 8.0 Hz), 6.75 (8H, d, J = 8.0 Hz), 6.90 (8H, d, J = 8.0 Hz), 7.19 (1H, brs), 7.29–7.32 (2H, m), 7.50–7.53 (1H, m), 7.67–7.71 (2H, m), 8.09–8.13 (4H, m) ppm; 13C NMR δ (400 MHz, CDCl3): 154.4, 149.8, 148.2, 144.5, 142.5, 142.6, 142.5, 136.1, 133.0, 134.0, 131.6, 130.9, 130.0, 129.6, 128.3, 127.9, 127.7, 127.4, 127.0, 126.7, 126.5, 126.3, 124.4, 123.7, 122.6, 122.2, 121.6, 121.3, 121.1, 120.5, 120.2, 115.2, 68.3, 31.6, 30.6, 29.4, 25.7, 25.8, 22.6, 14.0 ppm; HRMS (ESI): found [M]+ 986.5939; molecular formula C68H78N2O4 requires [M]+ 986.5956.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 counts were accumulated at the maximum channel. Solutions of the samples were contained in 1 cm path-length quartz cells at the optical density below 0.1 for fluorescence studies. All CV measurements were performed on potentiostat/galvanostat Gamry Instrument reference 600 at room temperature using a three electrode cell. The scan rate was 100 mV s−1. A platinum disk, platinum wire and Ag/AgCl (3 M KCl solution) were used as working (WE), counter (CE) and reference electrodes, respectively. The supporting electrolyte was a solution of 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) in anhydrous N,N-dimethylformamide, with concentration of HTMs fixed at 1 × 10−3 M. Ionization potentials were referenced versus the ferrocene (Fc) ionization potential 4.8 eV, calibrated with E1/2 (Fc/Fc+) = 0.488 V. Photoelectron yield spectroscopy in air (PESA) measurements of solid films on glass were performed with a Riken Keiki AC2-E. A power number of 0.5 was used for the calculation of the photoelectron yield and subsequently to extract the ionization potentials.
000 counts were accumulated at the maximum channel. Solutions of the samples were contained in 1 cm path-length quartz cells at the optical density below 0.1 for fluorescence studies. All CV measurements were performed on potentiostat/galvanostat Gamry Instrument reference 600 at room temperature using a three electrode cell. The scan rate was 100 mV s−1. A platinum disk, platinum wire and Ag/AgCl (3 M KCl solution) were used as working (WE), counter (CE) and reference electrodes, respectively. The supporting electrolyte was a solution of 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) in anhydrous N,N-dimethylformamide, with concentration of HTMs fixed at 1 × 10−3 M. Ionization potentials were referenced versus the ferrocene (Fc) ionization potential 4.8 eV, calibrated with E1/2 (Fc/Fc+) = 0.488 V. Photoelectron yield spectroscopy in air (PESA) measurements of solid films on glass were performed with a Riken Keiki AC2-E. A power number of 0.5 was used for the calculation of the photoelectron yield and subsequently to extract the ionization potentials.6b was cooled to −20 °C followed by heating ramp to 90 °C with a rate of 10 °C min−1, and next cooled down to −20 °C with the same rate. Subsequently, the sample was maintained at −20 °C for determining the period of time between 2 min and 90 min and two identical previously described cycles were repeated. The different holdup time (between 2 and 90 min) at −20 °C was studied to see the effect of time and low temperature on the molecular conformation of 6b. 6c was examined following the same procedure, however, no holdup time between cycles was applied.
Prior to each DSC measurement, the instrument (Mettler Toledo DSC 823e) was calibrated with a sample of indium, with 99.9999 mol% purity. The calorimeter was calibrated before each experiment. The calorimetric accuracy was ±1% and the precision was ±0.5% to the uncertainty of melting points was estimated to about ±0.5 K.
The ZnO layer was spin cast from nanoparticle dispersion (Nanograde N-10, 0.55 wt% in isopropanol, 4000 rpm, 30 s, h = 30 nm) and then annealed (10 min, 150 °C).
PEI was spin cast from a 2-methoxyethanol solution (0.4 wt%, 5000 rpm, 50 s) according to the process described in the literature.21 Afterwards, the samples were annealed in ambient atmosphere (10 minutes, 100 °C) and rinsed with water in order to remove any PEI surplus.
The emitters (6a, 6b and 6c) were spin cast from toluene solution (20 g L−1, 1000 rpm, 45 s, 40 nm). A MoO3/silver (10 nm/100 nm) counter electrode was thermally evaporated in high vacuum (10−6 mbar), altogether yielding the device architecture depicted in Fig. 5. The intersection of the top anode and the ITO cathode defined an active device area of 5 × 5 mm2.
The OLED current density–voltage (J–V) characteristics were recorded with a source measure unit (Keithley 238). The device luminance was calculated from the emission spectrum in forward direction. The spectrometer had been calibrated with a secondary standard calibration halogen lamp (Philips FEL-1000 W) before.
Acknowledgements
This work was supported by National Funds through FCT—Foundation for Science and Technology under the Project PTDC/CTM-CER/111590/2009. We would like to thank FCT for the PhD grant (SFRH/BD/73999/2010). S. H. and A. C. acknowledge fruitful discussions with Tanja Schneider and Stefan Gärtner.References
- H. Zollinger, Color Chemistry, VCH, Weinheim, 3rd edn, 2003 Search PubMed.
- W. Herbst, K. Hunger, G. Wilker, H. Ohleier and R. Winter, Industrial Organic Pigments: Production, Properties, Applications, Wiley-VCH Verlag GmbH & Co. KGaA, Germany, 3rd edn, 2004 Search PubMed.
- Y. Geerts, H. Quante, H. Platz, R. Mahrt, M. Hopmeier, A. Böhm and K. Müllen, Quaterrylenebis(dicarboximide)s: near infrared absorbing and emitting dyes, J. Mater. Chem., 1998, 8(11), 2357–2369 RSC.
- L. Schmidt-Mende, A. Fechtenkötter, K. Müllen, E. Moons, R. H. Friend and J. D. MacKenzie, Self-organized discotic liquid crystals for high-efficiency organic photovoltaics, Science, 2001, 293, 1119–1122 CrossRef CAS PubMed , http://www.sciencemag.org/content/293/5532/1119.full.pdf.
- X. Zhan, Z. Tan, B. Domercq, Z. An, X. Zhang, S. Barlow, Y. Li, D. Zhu, B. Kippelen and S. R. Marder, A high-mobility electron-transport polymer with broad absorption and its use in field-effect transistors and all-polymer solar cells, J. Am. Chem. Soc., 2007, 129, 7246–7247 CrossRef CAS PubMed.
- C. Li, J. Schöneboom, Z. Liu, N. G. Pschirer, P. Erk, A. Herrmann and K. Müllen, Rainbow perylene monoimides: easy control of optical properties, Chemistry, 2009, 15(4), 878–884 CrossRef CAS PubMed , http://www.ncbi.nlm.nih.gov/pubmed/19072796.
- C. Li and H. Wonneberger, Perylene imides for organic photovoltaics: Yesterday, today, and tomorrow, Adv. Mater., 2012, 24, 613–636 CrossRef CAS PubMed.
- S. Toguchi, Y. Morioka, H. Ishikawa, A. Oda and E. Hasegawa, Novel red organic electroluminescent materials including perylene moiety, Synth. Met., 2000, 111, 57–61 CrossRef.
- S. H. Kim, J. Choi, C. Sakong, J. W. Namgoong, W. Lee, D. H. Kim, B. Kim, M. J. Ko and J. P. Kim, The effect of the number, position, and shape of methoxy groups in triphenylamine donors on the performance of dye-sensitized solar cells, Dyes Pigm., 2015, 113, 390–401 CrossRef CAS , http://www.linkinghub.elsevier.com/retrieve/pii/S0143720814003672.
- B. Gigante, M. A. Esteves, N. Pires, M. L. Davies, P. Douglas, S. M. Fonseca, H. D. Burrows, R. A. E. Castro, J. Pina and J. S. Melo, Synthesis, spectroscopy, photophysics and thermal behaviour of stilbene-based triarylamines with dehydroabietic acid methyl ester moieties, New J. Chem., 2009, 33, 877–885 RSC.
- T. Uchida, K. Kozawa, Y. N. and T. Misonoo, The Crystal and Molecular Strucure of an Isomer of Dibromoperylenes, Bull. Chem. Soc. Jpn., 1979, 52, 1547–1548 CrossRef CAS.
- E. F. S. Torres, Síntese e Fotoquímica de Novos Compostos Orgânicos para Células Solares Híbridas, Universidade Técnica de Lisboa - Instituto Superior Técnico, 2015 Search PubMed.
- M. Planells, L. Pellejà, J. N. Clifford, M. Pastore, F. De Angelis, N. López, S. R. Marder and E. Palomares, Energy levels, charge injection, charge recombination and dye regeneration dynamics for donor–acceptor π-conjugated organic dyes in mesoscopic TiO2 sensitized solar cells, Energy Environ. Sci., 2011, 4(5), 1820–1829 CAS , http://www.xlink.rsc.org/?DOI=c1ee01060c.
- C. M. Cardona, W. Li, A. E. Kaifer, D. Stockdale and G. C. Bazan, Electrochemical considerations for determining absolute frontier orbital energy levels of conjugated polymers for solar cell applications, Adv. Mater., 2011, 23(20), 2367–2371 CrossRef CAS PubMed , http://www.ncbi.nlm.nih.gov/pubmed/21462372.
- R. J. Davis, M. T. Lloyd, S. R. Ferreira, M. J. Bruzek, S. E. Watkins, L. Lindell, P. Sehati, M. Fahlman, J. E. Anthony and J. W. P. Hsu, Determination of energy level alignment at interfaces of hybrid and organic solar cells under ambient environment, J. Mater. Chem., 2011, 21, 1721–1729 RSC.
- J. T. Bloking, X. Han, A. T. Higgs, J. P. Kastrop, L. Pandey, J. E. Norton, C. Risko, C. E. Chen, J. L. Brédas, M. D. McGehee and A. Sellinger, Solution-processed organic solar cells with power conversion efficiencies of 2.5% using benzothiadiazole/imide-based acceptors, Chem. Mater., 2011, 23, 5484–5490 CrossRef CAS.
- P. Gratia, A. Magomedov, T. Malinauskas, M. Daskeviciene, A. Abate, S. Ahmad, M. Grätzel, V. Getautis and M. K. Nazeeruddin, A Methoxydiphenylamine-Substituted Carbazole Twin Derivative: An Efficient Hole-Transporting Material for Perovskite Solar Cells, Angew. Chem., Int. Ed., 2015, 54, 11409–11413 CrossRef CAS PubMed.
- R. Bogel-Łukasik, L. M. Nobre Gonçalves and E. Bogel-Łukasik, Phase equilibrium phenomena in solutions involving tannins, flavonoids and ionic liquids, Green Chem., 2010, 12(11), 1947 RSC.
- A. Bucinskas, D. Volyniuk, Y. Danyliv, J. V. Grazulevicius, G. Baryshnikov, B. Minaev, K. Ivaniuk, V. Cherpak and P. Stakhira, N-Annelated perylenes as effective green emitters for OLEDs RSC Advances for OLEDs, RSC Adv., 2015, 5, 78150–78159 RSC.
- T. Leijtens, I.-K. Ding, T. Giovenzana, J. T. Bloking, M. D. Mcgehee and A. Sellinger, Hole transport materials with low glass transition temperatures and high solubility for application in solid-state dye sensitized solar cells, ACS Nano, 2012, 6, 1455–1462 CrossRef CAS PubMed.
- S. Hoefle, A. Schienle, C. Bernhard, M. Bruns, U. Lemmer and A. Colsmann, Solution processed, white emitting tandem organic light-emitting diodes with inverted device architecture, Adv. Mater., 2014, 26, 5155–5159 CrossRef CAS PubMed.
- S. Hoefle, A. Schienle, M. Bruns, U. Lemmer and A. Colsmann, Enhanced electron injection into inverted polymer light-emitting diodes by combined solution-processed zinc oxide/polyethylenimine interlayers, Adv. Mater., 2014, 26, 2750–2754 CrossRef CAS.
- W. L. F. Armarego and C. L. L. C. Chai, Purification of Laboratory Chemicals, ed. O. Butterworth-Heinemann, Butterworth-Heinemann, Oxford, 7th edn, 2012 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra20126a |
| This journal is © The Royal Society of Chemistry 2016 |