Guanidine based task specific ionic liquids for the synthesis of biolubricant range esters under solvent-free condition
Jyoti Porwala,
Subodh Kumarb,
Savita Kaula and
Suman L. Jain*b
aBiofuel Division, CSIR-Indian Institute of Petroleum, Dehradun-248005, India
bChemical Sciences Division, CSIR-Indian Institute of Petroleum, Dehradun-248005, India. E-mail: suman@iip.res.in; Fax: +91-135-2660202; Tel: +91-135-2525788
First published on 23rd September 2016
Abstract
Guanidine-based task specific ionic liquids (ILs) were synthesized from the reaction of 1,1,3,3-tetramethyl guanidine with protic acids and used for the synthesis of higher alcohol esters of fatty acids as biolubes under solvent free condition. The synthesized 1,1,3,3-tetramethylguanidinium hydrogen sulphate (TMG·HSO4) was found to be most effective among the different ILs including 1,1,3,3-tetramethylguanidinium acetate (TMG·Ac), 1,1,3,3-tetramethylguanidinium hydrogen phosphate (TMG·H2PO4) and 1,1,3,3-tetramethylguanidinium trifluoro acetate (TMG·TFA). The effect of various reaction parameters such as reaction temperature, reaction time, catalyst amount etc. has been studied. After completion the reaction the esterification product was isolated and the recovered IL was reused for several runs without loss in catalytic activity.
1. Introduction
There are several industrial application possibilities for fatty acid esters, as natural compounds.1 Plant oils mainly consist of oleic acid and other long-chain fatty acids (12–20 carbon atoms). Thus the esterification of plant oils involves the reaction of long-chain fatty acids with alcohols. Chain length of the alcohol substrate determines the usage of the product. Long-chain alcohol (5 to 12 carbon atoms) reactions give lubricants.2 The demands against biolubricants are that they should provide maximal protection during the usage, do not pollute the environment and do not accumulate. Usage of biolubricants can reduce the chemical risk, because of the possibility of biodegradability.3 Important physical effect of ester group is to reduce the volatility and increase the flash point,4 as well as providing a good lubricating ability, because the polar ester group facilitates the bonding to the metal surface. Good viscosity–temperature characteristic is their typical property and due to the high molecular weight, low evaporation loss, as well. General expectations over against lubricants that tolerate the mechanical effects and stress, biolubricants can resist these criteria. The disadvantages of biolubricants are low oxidative stability and poor cold flow properties.5 Conventionally these reactions are catalyzed by homogeneous acid catalysts such as sulphuric acid, which are corrosive, non-recyclable and produce undesirable wastes. The disposal of these undesired materials not only makes the overall process expensive but also leads to an environmental pollution.6 Thus, the use of solid catalysts has become an area of current research interest.7,8 The use of solid catalysts offer several advantages such as facile recovery of the catalyst, cheaper production costs due to the reusable nature of the catalyst and also reduce the corrosion and environmental problems.7 In this regard, several solid acid catalysts such as metal oxides,9,10 sulfonic acid functionalized mesoporous materials,11,12 polymers,13 zeolites,14 clays15 and carbon based catalysts16 have been reported for the esterification of fatty acids. Recently sulfonic acid functionalized ionic liquids;17 sulphated zirconia18 and sulfonated graphene oxides19 have been reported for the esterification of fatty acids. In the recent decades, ionic liquids owing to their attractive properties such as negligible vapor pressure, high thermal stability, and excellent solubility with number of reactants, and tunable acidity and basicity have been receiving a lot of attention as green catalyst and medium for a variety of reactions.20–23 Brønsted acidic ionic liquids particularly, imidazole and pyridine derivatives have widely been used for esterification of fatty acids.20 However, these ionic liquids are generally synthesized in two or more steps using expensive materials.Protic ionic liquids, especially those based on the 1,1,3,3-tetramethylguanidine (TMG) are readily synthesized in one step via simple neutralization of equimolar TMG with acids have recently been established as promising catalysts for various organic transformations including Henry reaction,24 one-pot synthesis of pyran,25 synthesis of 3,4-dihydropyridin-2-(1H)-ones26 and direct aldol reaction.27 Recently, we have reported 1,1,3,3-tetramethylguanidinium hydrogen sulphate (TMG·HSO4) IL as an efficient acid catalyst for the hydrolysis of cellulose in CO2 enriched water as reaction media.21 However to the best of our knowledge guanidine derived ionic liquids have never been used for the esterification reactions.
In the present paper we report novel tetramethyl-guanidine-based task specific ionic liquids and effect of different anions on their catalytic activity for esterification of fatty acids with higher alcohols (Scheme 1).
2. Experimental
2.1. Materials
The reactant oleic acid and all fatty alcohols were procured from Merck, India and acid oil has been procured local vendor Parshvanath associates, Dehradun, India. Tetramethylguanidine, ethanol and sulphuric acid were purchased from Merck, India and basic alumina from SD fine chemicals.2.2. Synthesis and characterization of the ILs21
Four tetramethylguanidine based ILs i.e. 1,1,3,3-tetramethylguanidinium hydrogen sulfate (TMG·HSO4) 1, 1,1,3,3-tetramethylguanidinium acetate (TMG·Ac) 2, 1,1,3,3-tetramethylguanidinium-hydrogen phosphate (TMG·H2PO4) 3 and 1,1,3,3-tetramethylguanidinium trifluoroacetate (TMG·TFA) 4 were synthesized by following the literature procedure (Scheme 2).21 In a typical experiment, TMG (20 mmol) was added in 100 mL of ethanol followed by the drop wise addition of H2SO4 (20 mmol in 30 mL of ethanol) at 0 °C under stirring. The stirring was continued for another 2 h. A white semi-solid material was isolated by decantation and washed thoroughly with ethanol, dried under vacuum for 6 h and then characterized by elemental analysis and 1H and 13C NMR spectroscopy as mentioned in our previous report.212.3. Esterification of oleic acid
The esterification experiments were carried out in a 25 mL round bottomed flask equipped with a magnetic stirrer and reflux condenser. In a typical experiment, oleic acid (1 mmol), octanol (1 mmol) and IL (5 wt% of total weight) was taken and the resulting mixture was stirred and heated at 150 °C under neat conditions. At higher temperature, the mixture became homogeneous and thus the reaction occurred in a single phase. The progress of the reaction was monitored by TLC (SiO2). After completion of the reaction, the reaction mixture was allowed to cool at room temperature. On cooling IL layer was separated and the resulting product was isolated by decantation. Subsequently, the IL layer was washed with petroleum ether to remove the organic ester. The combined organic layer was dried over anhydrous MgSO4 and concentrated under reduced pressure to give the product. The recovered ionic liquid was dried at 60 °C in vacuum and reused for recycling experiments. Conversion of fatty acid to the corresponding ester was calculated by means of the acid value (AV). The acid value of the reaction mixture was determined by the acid base titration technique. The conversion of FFA (reduction in acid value) was calculated using the following equation:| XFFA = ai − at/ai |
2.4. Esterification of fatty acid mixtures of acid oil
To explore the activity of the IL catalyst, acid oil, a high FFA (149 mg KOH per g) raw material obtained as a residue after refining of vegetable oil(s) has been taken. The acid oil was esterified with different fatty alcohols i.e. octanol, 2-ethyl hexanol, hexanol, decanol, oleyl alcohol, 2-methyl propane-1-ol, 2,6-dimethylheptan-4-ol under optimized conditions i.e. (IL: 5 wt%, reaction time: 6 h and reaction temperature 150 °C). Conversion of fatty acid to the corresponding esters was calculated by acid base titration technique. Further, the corresponding ester products were purified by column chromatography using basic alumina and analyzed by 1H and 13C NMR.3. Results and discussion
The initial studies were carried out by choosing the esterification of oleic acid with 1-octanol in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio as a representative reaction. The synthesized 1,1,3,3-tetramethylguanidinium hydrogen sulfate (TMG·HSO4) was found to have the highest activity among the different ILs including 1,1,3,3-tetramethylguanidinium acetate (TMG·Ac), 1,1,3,3-tetramethylguanidinium-hydrogen phosphate (TMG·H2PO4) and 1,1,3,3-tetramethylguanidinium trifluoroacetate (TMG·TFA) as shown in Fig. 1. Thus we have considered IL 1 for further studies.
1 molar ratio as a representative reaction. The synthesized 1,1,3,3-tetramethylguanidinium hydrogen sulfate (TMG·HSO4) was found to have the highest activity among the different ILs including 1,1,3,3-tetramethylguanidinium acetate (TMG·Ac), 1,1,3,3-tetramethylguanidinium-hydrogen phosphate (TMG·H2PO4) and 1,1,3,3-tetramethylguanidinium trifluoroacetate (TMG·TFA) as shown in Fig. 1. Thus we have considered IL 1 for further studies.
To evaluate the effect of the reaction temperature, esterification of oleic acid with octanol was performed by varying the temperature from 75 to 175 °C (Fig. 2). As shown, the reaction was found to be increased with temperature and at 150 °C the maximum conversion to corresponding ester (96.83%) was achieved. Further increase in reaction temperature did not show any significant change in the reaction rate and the conversion remained almost same. Thus we have chosen 150 °C as the optimum reaction temperature for further study.
 | ||
| Fig. 2 Effect of temperature on reaction conversion for the esterification of oleic acid with 1-octanol; TMG·HSO4 catalyst 5 wt%, time 6 h. | ||
The effect of reaction time on the esterification reaction was studied by collecting the samples at regular interval of time and analyzing the ester peaks at 4.0 ppm by 1H NMR spectroscopy. From Fig. 3 it is clear that initially the reaction is very fast with 42.11% conversion in 1 h and as the reaction proceeds, maximum conversion 96.83% is observed in 6 h afterwards no significant change is seen.
 | ||
| Fig. 3 Effect of time on reaction conversion for the esterification of oleic acid with 1-octanol using 5 wt% TMG·HSO4 catalyst and 150 °C temperature. | ||
In order to extent the scope of the reaction, we further studied the esterification of acid oil. Fatty acid composition of acid oil has been determined as C18:1 as major component and other component as C18:2, C18:0, C16:0 by gas chromatography (Table 1). The acid oil was esterified with different normal (hexanol, octanol, decanol, oleyl alcohol) and branched alcohols (i.e. isobutyl alcohol, 2-ethyl hexanol, 2,6-dimethyl heptanol) further purified and analysed by IR, 1H and 13C NMR (Fig. 4). Yields: 85.5–96.5% (Table 1); IR (neat, cm−1): 2933 (C–H), 1739 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1173 (C–C(
O), 1173 (C–C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)–O); 1H NMR (CDCl3, δ ppm): 0.90 [t, (–C
O)–O); 1H NMR (CDCl3, δ ppm): 0.90 [t, (–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 3)], 1.2–1.3 [m, (–C
3)], 1.2–1.3 [m, (–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2)], 1.6 [m, (–CO–CH2–C
2)], 1.6 [m, (–CO–CH2–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2)], 2.0 [m, –C
2)], 2.0 [m, –C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2–CH
2–CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH–C
CH–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2], 2.29 [t, (–CO–C
2], 2.29 [t, (–CO–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2)], 4.0 [t, (–O–C
2)], 4.0 [t, (–O–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2)], 5.3 [b, (–C
2)], 5.3 [b, (–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) –)]; 13C NMR (CDCl3, δ ppm): 14.1 [2 × (
–)]; 13C NMR (CDCl3, δ ppm): 14.1 [2 × (![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) H3)], 22.7 [2 × (C
H3)], 22.7 [2 × (C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2–CH3)], 32 [(
2–CH3)], 32 [(![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –C
–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–
C–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) )], 34.3 [(
)], 34.3 [(![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) –CO)], 64 (–O–C
–CO)], 64 (–O–C![[H with combining low line]](https://www.rsc.org/images/entities/char_0048_0332.gif) 2–), 130 [2 (
2–), 130 [2 (![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif)
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif)
![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) )], 173[(–
)], 173[(–![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) OO)].
OO)].
| Entry | Fatty acids | Fatty alcohol | Conv.b (%) | Yieldc (%) |
|---|---|---|---|---|
| a Reaction conditions: [TMG·HSO4] IL (5 wt%), reaction time 6 h, temperature 150 °C.b Conversion of fatty acid to ester was calculated by means of the acid value (AV). The acid value of the reaction mixture was determined by the acid base titration technique. The conversion of FFA (reduction in acid value) was calculated using the following equation: XFFA = ai − at/ai; where ai is the initial acidity and at is the acidity at time t (6 h).c Isolated yield. | ||||
| 1 |  |
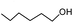 |
95.78 | 95.2 |
| 2 |  |
 |
96.83 | 96.1 |
| 3 |  |
 |
95.67 | 95.4 |
| 4 | 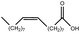 |
 |
96.89 | 96.5 |
| 5 |  |
 |
90.10 | 89.8 |
| 6 | Acid oil (major component-C18:1; other component-C18:1, C18:0, C16:0) |  |
96.86 | 96.2 |
| 7 | Acid oil (major component-C18:1; other component-C18:1, C18:0, C16:0) |  |
96.77 | 96.2 |
| 8 | Acid oil (major component-C18:1; other component-C18:2, C18:0, C16:0) |  |
97.10 | 96.8 |
| 9 | Acid oil (major component-C18:1; other component-C16:0, C18:0, C18:2) |  |
86.23 | 85.5 |
| 10 | Acid oil (major component-C18:1; other component-C16:0, C18:0, C18:2) |  |
88.34 | 88 |
| 11 | Acid oil (major component-C18:1; other component-C16:0, C18:0, C18:2) |  |
90.99 | 90 |
One desirable property of the IL catalyst is its reusability. Thus the recycling performance of the TMG·HSO4 IL was investigated for esterification of oleic acid with 1-octanol by performing several runs under the optimum reaction conditions. After completion of the reaction, the product was isolated from the ionic liquid by decantation. The recovered ionic liquid catalyst was then washed with petroleum ether to remove the organic ester. The results of recycling experiments as summarized in Fig. 5 clearly show that the IL catalyst exhibited high stability and the conversion remained unchanged even after seventh recycling instances.
4. Conclusions
We have demonstrated for the first time the application of tetramethylguanidine (TMG) derived ionic liquids as efficient and reusable acid catalyst for the esterification of fatty acids with higher alcohols to give higher alcohol esters of fatty acids for biolubricant applications. These ionic liquids can readily be synthesized by neutralizing equimolar amount of TMG with corresponding acids. The synthesized 1,1,3,3-tetramethylguanidinium hydrogen sulfate (TMG·HSO4) was found to have the highest activity among the different ILs including 1,1,3,3-tetramethylguanidinium acetate (TMG·Ac), 1,1,3,3-tetramethylguanidinium-hydrogen phosphate (TMG·H2PO4) and 1,1,3,3-tetramethylguanidinium trifluoroacetate (TMG·TFA). After the reaction, product was easily separated by decantation and the recovered IL could be recycled for several runs without any significant decrease in the catalytic activity.Acknowledgements
We kindly acknowledge Director IIP for his kind permission to publish these results. We are thankful to the Analytical Science Division of IIP for providing help in the analyses of the samples. SK kindly acknowledges CSIR, New Delhi for providing fellowship.References
- I. Diaz, C. Marquez-Alvarez, F. Mohino, J. Perez-Pariente and E. Sastre, J. Catal., 2000, 193, 295–302 CrossRef CAS.
- A. Demirbas, Biodiesel fuels from plant oils via catalytic and non-catalytic supercritical alcohol transesterifications and other methods: a survey, Energy Convers. Manage., 2003, 44, 2093–2109 CrossRef CAS.
- A. Campanella, E. Rustoy, A. Baldessari and M. A. Baltanas, Lubricants from chemically modified plant oils, Bioresour. Technol., 2010, 101, 245–254 CrossRef CAS PubMed.
- S. Z. Erhant, B. K. Sharma and J. M. Perez, Ind. Crops Prod., 2006, 24, 292–299 CrossRef.
- S. Jumat, S. Nadia and Y. Emad, J. King Saud Univ., Sci., 2012, 24, 11–17 CrossRef.
- J. M. Marchetti and A. F. Errazu, Biomass Bioenergy, 2008, 32, 892–895 CrossRef CAS.
- Z. Helwani, M. R. Othman, N. Aziz, J. Kim and W. J. N. Fernando, Appl. Catal., A, 2009, 363, 1–10 CrossRef CAS.
- H. Schuster, L. A. Rios, P. P. Wekjes and W. F. Hoelderich, Appl. Catal., A, 2009, 348, 266–270 CrossRef.
- M. J. Da Silva and A. L. Cardoso, J. Catal., 2013, 510509 Search PubMed , 11 pages.
- V. M. Mello, G. P. A. G. Pousa, M. S. C. Pereira, I. M. Dias and P. A. Z. Suarez, Fuel Process. Technol., 2011, 92, 53–57 CrossRef CAS.
- W. D. Bassaert, D. E. De Vos, W. M. Van Rhijin, J. Bullen, P. J. Grobet and P. A. Jacobs, J. Catal., 1999, 182, 156–164 CrossRef.
- I. Diaz, F. Mohino, J. P. Pariente and E. Sastre, Appl. Catal., A, 2001, 205, 19–30 CrossRef CAS.
- Y. Leng, P. Jiang and J. A. Wang, Catal. Commun., 2012, 25, 41–44 CrossRef CAS.
- A. S. Abbas and R. N. Abbas, Iraqi Journal of Chemical and Petroleum Engineering, 2013, 14, 35–43 Search PubMed.
- D. Konwar, P. K. Gogoi, G. Borah, R. Baruah, N. Hazarika and R. Borgohain, Indian J. Chem. Technol., 2008, 15, 75–78 CAS.
- L. Geng, Y. Wang, G. Yu and Y. Zhu, Catal. Commun., 2011, 13, 26–30 CrossRef CAS.
- B. Aghabarari, N. Dorostkar, M. Ghiaci, S. G. Amini, E. Rahimi and M. V. J. Mar-tinez-Huerta, J. Taiwan Inst. Chem. Eng., 2014, 45, 431–435 CrossRef CAS.
- J. Oh, S. Yang, C. Kim, I. Choi, J. H. Kim and H. Lee, Appl. Catal., A, 2013, 455, 164–171 CrossRef CAS.
- B. Garg, T. Bisht and Y. C. Ling, RSC Adv., 2014, 4, 57297–57307 RSC.
- J. H. Clark, T. J. Farmer, D. J. Macquarrie and J. Sherwood, Sustainable Chem. Processes, 2013, 1(23), 1–3 Search PubMed.
- S. Kumar, R. K. Singh and S. L. Jain, RSC Adv., 2014, 4, 58238–58242 RSC.
- T. Welton, Chem. Rev., 1999, 99, 2071–2083 CrossRef CAS PubMed.
- J. Dupont, T. Itoh, P. Lozano and S. Malhotra, Environmentally Friendly Syntheses Using Ionic Liquids, CRC Press, Taylor & Francis group, 2015 Search PubMed.
- T. Jiang, H. X. Gao, B. X. Han, G. Y. Zhao, Y. H. Chang, W. Z. Wu, L. Gao and G. Y. Yang, Tetrahedron Lett., 2004, 45, 2699–2701 CrossRef CAS.
- A. Shaabani, S. Samadi, Z. Badri and A. Rahmati, Catal. Lett., 2005, 104, 39–43 CrossRef CAS.
- A. Shaabani and A. Rahmati, Catal. Lett., 2005, 100, 177–179 CrossRef CAS.
- A. Zhu, T. Jiang, B. Han, J. Huang, J. Zhang and X. Ma, New J. Chem., 2006, 30, 736–740 RSC.
- H. Muthu, V. SathyaSelvabala, T. K. Varathachary, D. Kirupha Selvaraj, J. Nandagopal and S. Subramanian, Braz. J. Chem. Eng., 2010, 27(4), 601–608 CAS.
| This journal is © The Royal Society of Chemistry 2016 |





