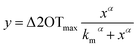Solid phase synthesis of a thrombin binding aptamer on macroporous silica for label free optical quantification of thrombin†
Monica Terraccianoa,
Luca De Stefanoa,
Nicola Borboneb,
Jane Politia,
Giorgia Olivierob,
Fabrizia Nicib,
Maurizio Casalinoa,
Gennaro Picciallib,
Principia Dardanoa,
Michela Varrab and
Ilaria Rea*a
aInstitute for Microelectronics and Microsystems, National Council of Research, Naples, Italy. E-mail: ilaria.rea@na.imm.cnr.it; Fax: +39-0816232598; Tel: +39-0816132594
bDepartment of Pharmacy, University of Naples Federico II, Naples, Italy
First published on 7th September 2016
Abstract
Human α-thrombin (TB) is a serine protease with a crucial role in coagulation and hemostasis. The monitoring of the TB level in blood serum could be of great importance in order to prevent serious damage to human health. In this work, an aptasensor is realized by in situ synthesis of a 17-mer Thrombin Binding Aptamer analogue (TBATT) on silanized macroporous silica (PSi). The interaction between TBATT and TB at different concentrations is monitored by a label-free optical method, spectroscopic reflectometry, and quantified by fast Fourier transform (FFT) analysis. A TBATT-TB affinity constant of 14 ± 8 nM and limit of detection of 1.5 ± 0.3 nM are demonstrated. The selectivity and reversibility of the aptasensor are also proved.
Introduction
Over the last decade biosensor technology has been growing very fast, radically changing human disease diagnosis and environmental monitoring.1 This technology, based on hybrid devices constituted by inorganic transducers coupled to biological probes, can solve some problems related to conventional diagnostic techniques (e.g. molecular assays, microbial culture-based tests), such as low selectivity and sensitivity, long response time and high costs.2 DNA biosensors are a special class of biosensors obtained by immobilization of single-, double- or multi-stranded oligonucleotide (ON) bioprobes on the transducer surface.3 The molecular interactions between bioprobes and their ligands are then transduced into analytical signals by electrochemical, optical or electrical devices, depending on the specific technology used.4–7 DNA aptamers are an emerging class of ONs because of their noteworthy therapeutic and diagnostic potential.8,9 Aptamers are obtained primarily using the Systematic Evolution of Ligands by EXponential Enrichment (SELEX) technique, a process allowing the selection of ONs endowed with high selectivity for a target molecule within large nucleic acids libraries (millions of ONs obtained by combinatorial chemistry), or by in silico- or SAR-guided chemical synthesis.10–12 Aptamers are able to specifically bind their target molecules with high affinity by folding into well-defined secondary or tertiary structures.13 Due to their unique properties, aptamers provide more advantages than conventional bioprobes (e.g. antibody): they can be easily obtained by solid-phase ON synthesis with good reproducibility, whereas the production of antibodies need in vivo immunization of animals;14 they are more resistant to heat, pH change and organic solvents than antibodies or proteins; their binding affinities and specificities can be easily manipulated and improved by proper design.15 Several aptasensors (i.e. biosensors where the biological probes are aptamers) have been realized based on fluorescence, electrochemioluminescence, surface plasmon resonance (SPR) and surface enhanced Raman scattering (SERS).16–21 In this work, we describe the realization of a label-free optical aptasensor obtained by in situ synthesis of the 15-mer thrombin binding aptamer (TBA) on silanized macroporous silica for quantitation of human α-thrombin levels.22,23 To ease the formation of the tri-dimensional structure by TBA, required for thrombin recognition, a short TT spacer was inserted between the silicon support and the 3′-end of TBA, thus obtaining the TBA analogue 5′-GGTTGGTGTGGTTGGTT-3′ (TBATT). Porous silicon is a nanostructured material largely used as transducer due to its sponge-like morphology – characterized by a specific surface area of about 500 m2 cm−3 – that assures an efficient and rapid interaction with the species to detect. Several porous silicon-based sensors have been proposed so far for the electrical or optical recognition of both small and big molecules.24,25 In particular, when it comes to the biosensing of macromolecules, the use of macroporous silicon (namely, pores size greater than 50 nm) is required. A PSi optical transducer responds to binding of inorganic/organic matter on pore walls with change of its average refractive index.26 PSi layer optically acts as a Fabry–Perot interferometer: replacement of air in pores increases average refractive index, thus resulting in a change of reflectivity spectrum, not always trivial to appreciate and precisely quantify.27 The analysis of optical spectra by fast Fourier transform (FFT) could represent a simple method to evaluate refractive index variation.28,29Human thrombin (MW ≈ 37 kDa) is a serine protease, also known as coagulation factor II, that converts soluble fibrinogen (factor I) into insoluble strands of fibrin (factor Ia), with crucial role in coagulation and haemostasis. The equilibrium between production and inhibition of this enzyme prevents haemorrhagic or thrombosis phenomena. The concentration of thrombin in healthy subject can be almost absent until getting up from nM to μM levels in the coagulation process.30 High level of thrombin in the blood (beyond the normal coagulation phenomenon) induces pathological coagulation disorders, such as thromboembolism or ischemic stroke, potentially fatal to human health.31 Moreover, the deregulation of neuronal PAR1-activation by thrombin has been associated to many disorders of the central nervous system (SNC), including the Alzheimer and Parkinson diseases, and the role of thrombin in cancer diseases is well known.32,33 The broad role of thrombin in a variety of (patho)physiological processes has triggered the interest towards the discovery of novel selective thrombin inhibitors (e.g. TBA and its analogues) as well as towards the development of new devices capable of measuring blood thrombin concentration characterized by low cost, high selectivity and very low limit of detection.12,34–38 In this work, we present the synthesis of a new hybrid device, based on TBA-functionalised macroporous silica, which allows the detection and quantitation of thrombin by label free optical reflectrometry. The high sensitivity, selectivity and reversibility of the obtained aptasensor are also demonstrated.
Experimental
Porous silica (PSi) fabrication
Porous silicon structure was fabricated by electrochemical etching of n-type crystalline silicon (0.01–0.02 Ω cm resistivity, 〈100〉 oriented and 500 μm thick) in hydrofluoric acid (HF, 5% in weight)/ethanol solution at room temperature (RT). A current density of 20 mA cm−2 was applied for 90 s to produce a single layer of porous silicon with a porosity of 61% (nPSi = 1.83 at λ = 1.2 μm) and a thickness, L, of 2.1 μm. Porosity and thickness of the porous silicon layer were calculated by spectroscopic ellipsometry (data not shown here).39 Before the etching procedure, the silicon substrate was immersed in HF solution for 2 min to remove the native oxide layer. Porous layer was thermally oxidized in pure O2 by a two-step process: pre-oxidation at 400 °C for 30 min followed by oxidation at 900 °C for 15 min. Spectroscopic ellipsometry, performed on the oxidized sample, revealed a refractive index decreased to 1.69 (data not shown here). After oxidation, porous silica (PSi) was treated in Piranha solution (H2O2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2SO4 = 1
H2SO4 = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; 30 min at RT) in order to generate Si–OH bonds on its surface; the structure was then amino-modified by immersion in 5% (v/v) 3-aminopropyltriethoxysilane (APTES, from Sigma-Aldrich, USA) solution in anhydrous toluene (Sigma-Aldrich, USA) for 30 min at RT and baked at 100 °C for 10 min.39,40
4; 30 min at RT) in order to generate Si–OH bonds on its surface; the structure was then amino-modified by immersion in 5% (v/v) 3-aminopropyltriethoxysilane (APTES, from Sigma-Aldrich, USA) solution in anhydrous toluene (Sigma-Aldrich, USA) for 30 min at RT and baked at 100 °C for 10 min.39,40
In situ synthesis of TBATT
The 17-mer ON 5′-GGTTGGTGTGGTTGGTT-3′ (TBATT) was synthesized in situ on an APTES-modified porous silicon support by solid phase ON synthesis using a PerSeptive Biosystems Expedite 8909 automated DNA synthesizer. The silanization of the PSi support with APTES and the in situ ON synthesis were performed according to the method described elsewhere.39 The obtained TBA-modified PSi aptasensor was then immersed in a solution of dry ammonia in dry methanol (overnight at RT) to deprotect the functional groups of nucleobases. The use of dry ammonia in dry methanol was preferred to the standard deprotection procedure (i.e. 33% aqueous ammonia at 55 °C for 17 h) to avoid the corrosion of the PSi matrix.41Thrombin recognition
Thrombin (TB, human α-Thrombin, MW ∼ 37 kDa) was solubilized in PBS (20 mM, pH 7.4) at final concentrations of 13, 27, 54 and 109 nM. The TBATT-TB biomolecular recognition was performed incubating the PSi-aptasensor with the thrombin solutions for 2 h at RT. After incubation, the sample was rinsed twice in PBS (20 mM, pH 7.4) and deionized water, and then dried by nitrogen gas. A control experiment was performed monitoring the interaction between TBATT and 109 nM Human Serum Albumin (HSA). The PSi-aptasensor was regenerated by immersing the device in water at 55 °C for 1 h, in order to unfold the TBA and release the thrombin. Chemicals and solvents were purchased from Sigma-Aldrich, USA. Reagents and phosphoramidites for DNA synthesis were purchased from Glen Research, USA.Porous silicon characterization
The morphology of porous silicon layer before and after thermal oxidation was investigated by scanning electron microscopy (SEM) using a Zeiss-Supra 35 field emission instrument. Samples were mounted on a double-faced conductive adhesive tape. Images were acquired at 5 kV accelerating voltage and 30 μm wide aperture. Specific surface area (SSA) of porous silicon was determined by N2 adsorption performed at 77 K using a Micromeritics ASAP 2020 apparatus. Each sample was previously heated at 100 °C under vacuum in the sample cell to complete degassing. The N2 adsorption isotherm was elaborated using the Brunauer–Emmett–Teller (BET) method for surface area estimation.42Spectroscopic reflectometry
Reflectivity spectra of PSi optical structure were obtained by a simple experimental setup: a white light was sent on PSi through a Y optical fiber (Avantes). The same fiber was used to guide the output signal to an optical spectrum analyser (Ando AQ6315A). The spectra were acquired at normal incidence over the range 800–1600 nm with a resolution of 2 nm. Reflectivity spectra shown in the work are the average of three measurements.Results and discussion
Fabrication and surface silanization of PSi matrix
A key prerequisite for the use of functionalized PSi in biosensing is the chemical stability of the PSi matrix in the solvents and conditions used for biological investigations. Recently, we demonstrated two common routes of passivation/functionalization by grafting alkylsilanes (APTES and APDMES) through the formation of covalent Si–O–Si bonds between silanol groups induced on oxidized porous silicon surface and hydrolysed organosilane molecules.39 The silanization process, conferring chemical resistance to the PSi matrix, allows the ONs synthesis and deprotection on the PSi surface without harming the structure. We successfully demonstrated the development of DNA label free optical biosensors through the in situ synthesis of a 13-mer polythymine ON strand, deprotection and hybridization with a complementary ON, opening the prospective of PSi solid phase ONs synthesis without any restriction on the ON composition sequence.39 The advantages of in situ synthesis compared to ex situ immobilized bioprobes are not only the increasing of their density on the surface transducer, which results in a greater sensitivity with respect to devices using purchased probes, but also the fabrication process automation, and the possibility of surface local functionalization in microarrays or similar devices.43–45However, this technology can be limited by small average pores size: in case of 19-mer mixed DNA sequence for biomedical applications, synthetized on mesoporous PSi microcavities, the process showed low yield of functionalization, mainly due to the about 20 nm pores of PSi structure which did not allow proper arrangement of larger molecules because of the steric hindrance of pores.30 In this work, we explored the use of macroporous silica (pores size > 50 nm) for realization of sensitive, selective and reversible label free optical sensor of thrombin; the device was obtained by in situ synthesis of the TBA analogue TBATT on macroporous silica support. Morphological characterization of porous sample before thermal oxidation is reported in Fig. 1: SEM top view image (A) together with the histogram of pores size distribution (B) revealed macropores with dimensions included between 50 and 250 nm with a maximum close to 100 nm. Specific surface area of macroporous silicon was measured via nitrogen adsorption using Brunauer–Emmett–Teller (BET) method. BET isotherm is reported in Fig. 1(C); a surface area of 199 (3) m2 g−1 was evaluated.
Measurement of PSi optical spectra by spectroscopic reflectometry is a very useful technique in order to investigate PSi stability, chemical functionalization and sensing properties.46,47 In particular, the optical thickness (i.e. OT = nPSiL) of the PSi layer was calculated from the reflectivity spectrum by FFT, which displayed a peak whose position along the x-axis corresponded to two times the optical thickness (2OT) of the layer.28,48 In this work, the porous silica structure was first silanized using APTES, in order to protect the device against corrosion on exposure to chemicals and introduce functional amino groups onto the PSi surface for further covalent conjugation with TBA bioprobes, synthesized in situ.39,44 Normal incidence reflectivity spectra of PSi layer before and after APTES modification (PSi-APTES) are shown in Fig. S1A (ESI†) together with the corresponding FFTs (Fig. S1B, ESI†). Since the physical thickness L (2100 nm) of the PSi layer was fixed, the FFT peak shift of about 30 nm was due to an increase of the average refractive index equal to 0.007 (0.001), as result of APTES functionalization.39
Realization of PSi aptasensor
A tricky step in fabrication of PSi biosensor is biofunctionalization (i.e. covalent immobilization of the bioprobe) of its surface with the biological probe in order to make the device selective for the species of interest.49 TBA bioprobes for thrombin detection were covalently conjugated with the amino groups of PSi-APTES surface during TBATT solid phase in situ synthesis. APTES-modified PSi structures were used as solid phase supports during the synthesis of TBA. Coupling yield of each synthesis cycle was quantified by ultraviolet (UV) spectroscopy after detritylation step of 5′-dimethoxytritil (DMT) group released in solution;44 results are reported in Fig. 2. After synthesis of first four nucleotides, coupling yield kept almost constant value, quite differently to previously observed behaviour in case of 19-mer mixed synthesis.41 The data could be ascribed to less steric hindrance of macropores compared to mesopores, allowing correct growth of bioprobes.41 Surface functionalization of PSi with TBA, FTBA, was quantified using the Lambert–Beer formula (DMT molar absorptivity ε = 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 M−1 cm−1, λ = 500 nm) and the value of UV intensity corresponding to the last synthetized nucleotide, IN17 = 0.055 ± 0.001. Considering a weight of 0.2 mg for the PSi sample, FTBA was estimated to be (1.92 ± 0.03) × 10−5 mol g−1. In order to calculate the surface functionalization in terms of nmol cm−2, the SSA of porous silica (SSAPSi) is required. SSAPSi was obtained considering pores size reduction of 20 nm, estimated by SEM imaging after oxidation (Fig. S2 in ESI†), with respect to SSA of as-etched sample (199 m2 g−1 evaluated by BET analysis, see Fig. 1C). Applying a simplified model based on the cylindrical shape of pores, SSAPSi of 153 m2 g−1 was quantified. Surface sample functionalization of 0.0125 ± 0.0002 nmol cm−2 was calculated as the ratio FTBA/SSAPSi.
700 M−1 cm−1, λ = 500 nm) and the value of UV intensity corresponding to the last synthetized nucleotide, IN17 = 0.055 ± 0.001. Considering a weight of 0.2 mg for the PSi sample, FTBA was estimated to be (1.92 ± 0.03) × 10−5 mol g−1. In order to calculate the surface functionalization in terms of nmol cm−2, the SSA of porous silica (SSAPSi) is required. SSAPSi was obtained considering pores size reduction of 20 nm, estimated by SEM imaging after oxidation (Fig. S2 in ESI†), with respect to SSA of as-etched sample (199 m2 g−1 evaluated by BET analysis, see Fig. 1C). Applying a simplified model based on the cylindrical shape of pores, SSAPSi of 153 m2 g−1 was quantified. Surface sample functionalization of 0.0125 ± 0.0002 nmol cm−2 was calculated as the ratio FTBA/SSAPSi.
 | ||
| Fig. 2 Coupling yield calculated by means of DMT measurements during TBATT synthesis on silanized macroporous silica. | ||
TBA synthesis was also monitored by spectroscopic reflectometry. Fig. 3 shows reflectivity spectra (A) with corresponding FFTs (B) of PSi-APTES before and after the in situ synthesis process. A calculated FFT peak shift of 36 nm, corresponding to a refractive index variation of 0.009, confirmed successful of TBA growth, thus making the PSi structure sensitive to thrombin detection. The target recognition by a DNA-biosensor required the deprotection of each base constituting the ONs grown on the support in order to allow biomolecular interaction with the ligand.50 Standard deprotection process of mixed ON sequence is based on sample treatment in 33% aqueous ammonia (55 °C for 2 h), which can be very dangerous in terms of PSi corrosion on exposure to alkaline solutions due to basic hydrolysis of PSi surface.51 The PSi-aptasensor (i.e. PSi-APTES after TBA synthesis) was deprotected by ammonia solution under anhydrous condition (NH3/dry methanol), a valid alternative to conventional ON deprotection, as demonstrated in our previus work.41 NH3/dry methanol promoted effective deprotection of phosphate and exocyclic amino groups of nucleobases without inducing basic hydrolysis on PSi support, thus preserving structural integrity of PSi matrix.41,52 After deprotection, no shift was observed in the reflectivity spectrum of device.
 | ||
| Fig. 3 Reflectivity spectra (A) and corresponding Fourier transforms (B) of PSi-APTES before (black line) and after TBA synthesis (red line). | ||
Reversibility, sensitivity, and limits of detection in human thrombin monitoring
TBA is an aptamer able to inhibit thrombin activity; due to this specific feature, TBA structure was extensively studied in order to better understand the binding mode with thrombin.53 In presence of thrombin and/or monovalent cations, TBA folds into an antiparallel unimolecular G-quadruplex dictating its thrombin-binding affinity.36,53 Its three-dimensional chair-like structure consists of two G-tetrads connected by TT and TGT loops.54After deprotection process, the PSi-aptasensor was exposed to different molar concentrations of thrombin (13, 27, 54 and 109 nM). TBA–thrombin interaction was monitored by a label-free optical method based on spectroscopic reflectometry. Fig. 4 shows reflectivity spectra of PSi-aptasensor for each thrombin concentration (A) with the corresponding FFT (B). FFT peak shift, namely Δ2OT, vs. thrombin concentration is reported in Fig. 5. Experimental data were fitted using Hill equation:
 | ||
| Fig. 4 Reflectivity spectra (A) and corresponding Fourier transforms (B) of PSi-aptasensor after exposure to different thrombin concentrations (13, 27, 54 and 109 nM). | ||
In order to verify the specificity of the PSi-aptasensor for the human thrombin, the device was exposed to 109 nM of albumin; no peak shift was registered in this case (Fig. S3 in ESI†).
Reversibility of a biosensor is an advantageous feature of device allowing several sensing measurements with benefits both in terms of cost and time consuming.26,64 In order to demonstrate the reversibility of PSi-aptasensor, the device was regenerated by incubation in water at 53 °C, which was above the melting temperature of TBA.65 At 53 °C, TBA lost the typical G-quadruplex structure and, as consequence, the affinity with thrombin. However, the process was reversible: by exposing TBA-PSi to thrombin and/or monovalent cations, the aptamer folded again into G-quadruplex and allowed molecular interaction.66 Fig. 6 reports reflectivity spectra (A) with corresponding FFTs (B) of PSi-aptasensor before regeneration (i.e. surface saturated with 109 nM of thrombin), after regeneration, and after a new exposure to 109 nM of thrombin. Regeneration induced a decrease of optical thickness (about 14 nm) due to releasing of thrombin from the PSi pores. An increase of 38 nm was recorded after exposure to 109 nM of thrombin solution, demonstrating proper working functionality of the biosensor to recognize the analyte.
 | ||
| Fig. 6 Reflectivity spectra (A) and corresponding Fourier transforms (B) of PSi-aptasensor before regeneration (black line), after regeneration (red line), and after exposure to 109 nM of thrombin. | ||
Conclusions
In this work, fabrication and characterization of an aptasensor, obtained by in situ synthesis of TBATT on macroporous silica (pores size > 50 nm), were described. Before synthesis, porous silica surface was stabilized/functionalized by grafting an alkylsilane to prevent its corrosion by aggressive chemicals used for the solid phase synthesis and oligonucleotide deprotection. TBA growth was monitored both by standard technique, i.e. analysis of DMT released in solution after each synthesis cycle, and by label-free optical method based on spectroscopic reflectometry followed by FFT. In order to allow the thrombin recognition, TBA was deprotected by dry ammonia in methanol thus preserving structural integrity of porous support. PSi-aptasensor was demonstrated to recognize thrombin at different concentrations up to 0.11 mM; the sensing mechanism was investigated by spectroscopic reflectometry and FFT analysis. Reversibility of the PSi-aptasensor was also proved. The results reported in this work endorsed macroporous silica as suitable substrate for the development of a wide range of aptasensors for macromolecules detection. Moreover, the label-free optical PSi-aptasensor, simple to use and sensitive, represents a good starting point for the realization of Lab-on-Chip (LoC) devices for early diagnosis of vascular diseases.Acknowledgements
Authors would like to thank Prof. D. Caputo and Dr N. Gargiulo of University of Naples Federico II, Italy, for BET measurements.References
- J. Kirsch, C. Siltanen, Q. Zhou, A. Revzin and A. Simonian, Chem. Soc. Rev., 2013, 42, 8733–8768 RSC.
- A. P. Turner, Chem. Soc. Rev., 2013, 42, 3184–3196 RSC.
- G. Shtenberg and E. Segal, Handbook of Porous Silicon, Springer, 2014, pp. 857–868 Search PubMed.
- A. Sassolas, B. D. Leca-Bouvier and L. J. Blum, Chem. Rev., 2008, 108, 109–139 CrossRef CAS PubMed.
- L. De Stefano, I. Rea, A. Caliò, J. Politi, M. Terracciano and G. Korotcenkov, Porous Silicon: From Formation to Application: Biomedical and Sensor Applications, CRC Press, 2016, p. 69 Search PubMed.
- L. Canham, Properties of Porous Silicon, Inspec/IEE, 1997 Search PubMed.
- W. Ma, L. Xu, L. Wang, H. Kuang and C. Xu, Biosens. Bioelectron., 2016, 79, 220–236 CrossRef CAS PubMed.
- X. Ni, M. Castanares, A. Mukherjee and S. E. Lupold, Curr. Med. Chem., 2011, 18, 4206–4214 CrossRef CAS PubMed.
- D. Musumeci, C. Riccardi and D. Montesarchio, Molecules, 2015, 20, 17511–17532 CrossRef CAS PubMed.
- G. Oliviero, M. Stornaiuolo, V. D'Atri, F. Nici, A. M. Yousif, S. D'Errico, G. Piccialli, L. Mayol, E. Novellino, L. Marinelli, P. Grieco, A. Carotenuto, S. Noppen, S. Liekens, J. Balzarini and N. Borbone, Anal. Chem., 2016, 88, 2327–2334 CrossRef CAS PubMed.
- V. D'Atri, G. Oliviero, J. Amato, N. Borbone, S. D'Errico, L. Mayol, V. Piccialli, S. Haider, B. Hoorelbeke, J. Balzarini and G. Piccialli, Chem. Commun., 2012, 48, 9516–9518 RSC.
- N. Borbone, M. Bucci, G. Oliviero, E. Morelli, J. Amato, V. D'Atri, S. D'Errico, V. Vellecco, G. Cirino, G. Piccialli, C. Fattorusso, M. Varra, L. Mayol, M. Persico and M. Scuotto, J. Med. Chem., 2012, 55, 10716–10728 CrossRef CAS PubMed.
- S. D. Jayasena, Clin. Chem., 1999, 45, 1628–1650 CAS.
- T. Hermannand and D. J. Patel, Science, 2000, 287, 820–825 CrossRef.
- M. Mascini, Aptamers in bioanalysis, John Wiley & Sons, 2009 Search PubMed.
- Y. Zhang and X. Sun, Chem. Commun., 2011, 47, 3927–3929 RSC.
- X. Zhao, S. Li, L. Xu, W. Ma, X. Wu, H. Kuang and C. Xu, Biosens. Bioelectron., 2015, 70, 372–375 CrossRef CAS PubMed.
- X. Y. Wang, A. Gao, C. C. Lu, X. W. He and X. B. Yin, Biosens. Bioelectron., 2013, 48, 120–125 CrossRef CAS PubMed.
- G. Pelossof, R. Tel-Vered, X. Q. Liu and I. Willner, Chem.–Eur. J., 2011, 17, 8904–8912 CrossRef CAS PubMed.
- J. Feng, X. Wu, W. Ma, H. Kuang, L. Xu and C. Xu, Chem. Commun., 2015, 51, 14761–14763 RSC.
- L. Xu, S. Zhao, W. Ma, X. Wu, S. Li, H. Kuang and C. Xu, Adv. Funct. Mater., 2016, 26, 1602–1608 CrossRef CAS.
- L. C. Bock, L. C. Griffin, J. A. Latham, E. H. Vermaas and J. J. Toole, Nature, 1992, 355, 564–566 CrossRef CAS PubMed.
- R. Macaya, P. Schultze, F. Smith, J. Roe and J. Feigon, Proc. Natl. Acad. Sci. U. S. A., 1993, 90, 3745–3749 CrossRef CAS.
- A. Jane, R. Dronov, A. Hodges and N. H. Voelcker, Trends Biotechnol., 2009, 27, 230–239 CrossRef CAS PubMed.
- S. Dhanekar and S. Jain, Biosens. Bioelectron., 2013, 41, 54–64 CrossRef CAS PubMed.
- K. P. S. Dancil, D. P. Greiner and M. J. Sailor, J. Am. Chem. Soc., 1999, 121, 7925–7930 CrossRef CAS.
- L. De Stefano, P. Arcari, A. Lamberti, C. Sanges, L. Rotiroti, I. Rea and I. Rendina, Sensors, 2007, 7, 214–221 CrossRef CAS.
- I. Rea, L. Sansone, M. Terracciano, L. De Stefano, P. Dardano, M. Giordano, A. Borriello and M. Casalino, J. Phys. Chem. C, 2014, 118, 27301 CAS.
- J. Wu and M. J. Sailor, Adv. Funct. Mater., 2009, 19, 733–741 CrossRef CAS.
- T. C. Chiu and C. C. Huang, Sensors, 2009, 9, 10356–10388 CrossRef CAS PubMed.
- E. Baldrich, A. Restrepo and C. K. O'Sullivan, Anal. Chem., 2004, 76, 7053–7063 CrossRef CAS PubMed.
- M. Ben Shimon, M. Lenz, B. Ikenberg, D. Becker, E. Shavit Stein, J. Chapman, A. Vlachos and N. Maggio, Front. Cell. Neurosci., 2015, 9, 151 Search PubMed.
- S. Danckwardt, M. W. Hentze and A. E. Kulozik, J. Mol. Med., 2013, 91, 1257–1271 CrossRef CAS PubMed.
- J. Zain, Y. Q. Huang, X. Feng, M. L. Nierodzik, J. J. Li and S. Karpatkin, Blood, 2000, 95, 3133–3138 CAS.
- M. Scuotto, M. Persico, M. Bucci, V. Vellecco, N. Borbone, E. Morelli, G. Oliviero, E. Novellino, G. Piccialli, G. Cirino, M. Varra, C. Fattorusso and L. Mayol, Org. Biomol. Chem., 2014, 12, 5235–5242 CAS.
- M. Scuotto, E. Rivieccio, A. Varone, D. Corda, M. Bucci, V. Vellecco, G. Cirino, A. Virgilio, V. Esposito, A. Galeone, N. Borbone, M. Varra and L. Mayol, Nucleic Acids Res., 2015, 43, 7702–7716 CrossRef CAS PubMed.
- D. Musumeci, G. Piccialli, G. Oliviero, G. N. Roviello and E. M. Bucci, Bioconjugate Chem., 2012, 23, 382–391 CrossRef CAS PubMed.
- H. Yang, J. Ji, Y. Liu, J. Kong and B. Liu, Electrochem. Commun., 2009, 11, 38–40 CrossRef CAS.
- L. De Stefano, O. Oliviero, J. Amato, N. Borbone, G. Piccialli, L. Mayol, I. Rendina, M. Terracciano and I. Rea, J. R. Soc., Interface, 2013, 10, 20130160 CrossRef PubMed.
- M. Terracciano, I. Rea, J. Politi and L. De Stefano, Journal of the European Optical Society: Rapid Publications, 2013, 8, 13075 CrossRef.
- M. Terracciano, I. Rea, L. De Stefano, I. Rendina, G. Oliviero, G. Piccialli, F. Nici, S. D'Errico and N. Borbone, Nanoscale Res. Lett., 2014, 9, 1–9 CrossRef CAS PubMed.
- F. Rouquerol, J. Rouquerol and K. Sing, Adsorption by Powders and Porous Solids, Academic Press, 1999, pp. 93–115 Search PubMed.
- L. De Stefano, P. Arcari, A. Lamberti, C. Sanges, L. Rotiroti, I. Rea and I. Rendina, Sensors, 2007, 7, 214–221 CrossRef CAS.
- I. Rea, G. Oliviero, J. Amato, N. Borbone, G. Piccialli, I. Rendina and L. D. Stefano, J. Phys. Chem. C, 2010, 114, 2617–2621 CAS.
- I. Rea, E. Orabona, I. Rendina and L. De Stefano, Porous silicon integrated photonic devices for biochemical optical sensing, InTech Open Access Publisher, 2011 Search PubMed.
- V. S. Y. Lin, K. Motesharei, K. P. S. Dancil, M. J. Sailor and M. R. Ghadiri, Science, 1997, 278, 840–843 CrossRef CAS PubMed.
- I. Rendina, L. Rea, L. Rotiroti and L. De Stefano, Phys. E, 2007, 38, 188–192 CrossRef CAS.
- A. Janshoff, K. P. S. Dancil, C. Steinem, D. P. Greiner, V. S. Y. Lin, C. Gurtner and M. R. Ghadiri, J. Am. Chem. Soc., 1998, 120, 12108–12116 CrossRef CAS.
- J. J. Gooding and S. Ciampi, Chem. Soc. Rev., 2011, 40, 2704–2718 RSC.
- H. Vu, C. McCollum, K. Jacobson, P. Theisen, R. Vinayak, E. Spiess and A. Andrus, Tetrahedron Lett., 1990, 31, 7269–7272 CrossRef.
- T. Jalkanen, E. Mäkilä, Y. I. Suzuki, T. Urata, K. Fukami, T. Sakka and Y. H. Ogata, Adv. Funct. Mater., 2012, 22, 3890–3898 CrossRef CAS.
- R. P. Iyer, D. Yu, J. Xie, W. Zhou and S. Agrawal, Bioorg. Med. Chem. Lett., 1997, 7, 1443–1448 CrossRef CAS.
- I. R. Krauss, A. Merlino, A. Randazzo, E. Novellino, L. Mazzarella and F. Sica, Nucleic Acids Res., 2012, 512, 1–8 Search PubMed.
- T. Coppola, M. Varra, G. Oliviero, A. Galeone, G. D'Isa, L. Mayol and N. Borbone, Bioorg. Med. Chem., 2008, 16, 8244–8253 CrossRef CAS PubMed.
- J. Politi, I. Rea, F. Nici, P. Dardano, M. Terracciano, G. Oliviero, N. Borbone, G. Piccialli and L. De Stefano, J. Sens., 2016, 2016, 1–8 CrossRef.
- M. B. Gu and H. S. Kim, Biosensors based on aptamers and enzymes, Springer, 2014 Search PubMed.
- J. R. Epstein, M. Lee and D. R. Walt, Anal. Chem., 2002, 74, 1836–1840 CrossRef CAS PubMed.
- T. Goda, D. Higashi, A. Matsumoto, T. Hoshi, T. Sawaguchi and Y. Miyahara, Biosens. Bioelectron., 2015, 73, 174–180 CrossRef CAS PubMed.
- V. Ostatná, H. Vaisocherová, J. Homola and T. Hianik, Anal. Bioanal. Chem., 2008, 391, 1861–1869 CrossRef PubMed.
- S. Li, L. Xu, W. Ma, X. Wu, M. Sun, H. Kuang and C. Xu, J. Am. Chem. Soc., 2015, 138, 306–312 CrossRef PubMed.
- L. Xu, W. Yan, W. Ma, H. Kuang, X. Wu, L. Liu, Y. Zhao, L. Wang and C. Xu, Adv. Mater., 2015, 27, 1706–1711 CrossRef CAS PubMed.
- L. Tang, S. Li, F. Han, L. Liu, L. Xu, W. Ma and C. Xu, Biosens. Bioelectron., 2015, 71, 7–12 CrossRef CAS PubMed.
- S. Zhao, W. Ma, L. Xu, X. Wu, H. Kuang, L. Wang and C. Xu, Biosens. Bioelectron., 2015, 68, 593–597 CrossRef CAS PubMed.
- C. R. Yonzon, C. L. Haynes, X. Zhang, J. T. Walsh and R. P. Van Duyne, Anal. Chem., 2004, 76, 78–85 CrossRef CAS PubMed.
- G. W. Lehmanand and J. P. McTague, J. Chem. Phys., 1968, 49, 3170–3179 CrossRef.
- I. Smirnov and R. H. Shafer, Biochemistry, 2000, 39, 1462–1468 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra18401d |
| This journal is © The Royal Society of Chemistry 2016 |