Rapid and complete degradation of sulfur mustard adsorbed on M/zeolite-13X supported (M = 5 wt% Mn, Fe, Co) metal oxide catalysts with ozone
Chowdam Ramakrishna*,
Bijendra Kumar Saini,
Krishna Racharla,
Swetha Gujarathi,
Chandra Shekar Sridara,
Arvind Gupta,
Gopi Thakkallapalli and
P. V. L. Rao
Chemical Defence Equipments Evaluation Facility (CDEF), Defence R & D Establishment, Jhansi Road, Gwalior, MP 474002, India. E-mail: ramakrishna.drde@gmail.com; Fax: +91-7103-280610; Tel: +91-7103-280702 Tel: +91-8551-894451
First published on 15th September 2016
Abstract
Degradation of sulfur mustard (HD) adsorbed on zeolite-13X as well as M/zeolite-13X metal oxide materials (M = 5 wt% Mn, Fe, Co) was studied with and without gaseous ozone as the oxidizing agent under ambient reaction conditions. In the absence of ozone gas HD degradation was very slow over bare zeolite-13X as well as on the metal oxide impregnated zeolite-13X catalysts. Introduction of ozone gas remarkably enhanced the rate of degradation of HD and complete oxidation of HD was achieved on M/zeolite-13X materials in less than 10 minutes of reaction time. The influence of metal content, and catalyst quantity on the distribution of HD degradation products was also investigated. The results revealed that slightly longer reaction times (>5 minutes) and higher catalyst quantity favor the complete oxidation of HD. Carbon oxides were identified as the major degradation products of HD in the presence of ozone. GC-MS techniques were used for the analysis of the gas phase, condensed and surface extracted products. The HD degradation mechanism in the presence and absence of ozone was proposed based on GC-MS analysis results. The M/zeolite-13X catalysts were prepared using a wet impregnation method and characterized by different characterization techniques.
1. Introduction
Protection against harmful chemicals is of major social and military concern. Particularly, protection against chemical warfare agents (CWA's) such as sarin (GB), sulphur mustard (HD), etc. is essential. The chemical protective equipment widely utilized for individual protection like gas-mask filters, protective over garments, canisters, etc. contain activated carbon or metal impregnated carbon systems as the adsorbent material. On the other hand, collective protection from contaminated field shelters, concealed areas, etc. can be achieved by using large size filters which also contain adsorbent material like activated alumina, impregnated carbon systems, transition metal oxide/nano metal oxides, etc. However, their protection efficiency is limited and the service life of the filters is also very short in both the case of individual and collective protection equipment.These draw backs can be addressed by utilizing Pressure Swing Adsorption (PSA) based filtration systems which are well explored in the industry for separation of gases mixtures as well as environmental cleanup applications.1–4 The main advantage of the PSA system is better protection against most of the harmful chemicals and enhanced service life of the filters. The adsorbent material utilized in PSA systems are zeolites and activated alumina.
Unfortunately, most of the CWA's have low vapor pressure and posses high affinity towards surfaces such as activated carbon, transition metal oxides, zeolite, etc. The adsorbed CWA's on activated carbon materials are often stable for hours to many days5–7 and leads to the long-term environmental risk due to these contaminated chemical protective equipments such as gas mask filters, canisters, and protective over garments etc. On the other hand, increasing concern for the environment as well as safe disposal of contaminated adsorbent dictates that techniques for the degradation of adsorbed CWAs on the activated carbon/zeolite materials is of great interest to the scientific community to decompose the adsorbed CWA's to harmless products. It is believed that the decontaminant used to destroy the sulfur mustard efficiently, can also be used to decontaminate the other chemical warfare agents. The kinetics of the degradation of various CWA's on impregnated carbon8 transition metal oxides/nano metal oxides9,10 and on bare zeolites11–13 is usually very slow and takes a longer reaction time for degradation (half life 2–3 days) through a series of oxidation, elimination and hydrolysis reactions there by rendering these toxic chemicals to harmless products by utilizing the high surface area, Lewis acidity and surface hydroxyl groups of the catalyst materials.
Osovsky et al.14 studied the hydrothermal treatment of sulfur mustard adsorbed on activated carbon spheres and they reported that complete degradation of HD adsorbed was achieved within 0.5 to 2.5 h at a temperature of 120–160 °C by using water/carbon ratio of 0.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 5
1 to 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. But, the bottleneck is that the reactive adsorbents often take longer reaction time for the degradation process. Furthermore, some of the degradation products formed in the process (mustard sulfone and divinyl sulfone etc.) is potentially toxic to the human health as well as to the environment.
1. But, the bottleneck is that the reactive adsorbents often take longer reaction time for the degradation process. Furthermore, some of the degradation products formed in the process (mustard sulfone and divinyl sulfone etc.) is potentially toxic to the human health as well as to the environment.
S. Popiel15 et al. reported the destruction of sulfur mustard in liquid phase using ozone, UV, hydrogen peroxide and their combination in buffered aqueous media. However, this method is applicable only for the laboratory study and did not find any practical importance. Furthermore, they did not report the degradation products identified in their study. Therefore, development of new methodology which has high practical importance to completely degrade/decompose the CWA's at ambient conditions in a shorter time is of immense important to the scientific community.
To the best of our knowledge, so far there are no reports on the complete degradation/mineralization of sulfur mustard on M/zeolite-13X (M = 5% Mn, Fe, Co) metal oxide materials using gaseous ozone. Therefore, we have chosen Mn, Fe, Co/zeolite-13X as the adsorbent materials for this study due to their excellent ozone decomposition ability at ambient conditions and are also proven as active catalysts for the removal of many organic pollutants as well as for waste water treatment under ozonation conditions.16–18 Furthermore, zeolite-13X support has high thermal stability, resistance to chemicals, inexpensive, environmental friendly and readily available. The high kinetic diameter (1.1 nm) is also the key factor for the adsorption of most of CWA's. In this study we reported the complete degradation of HD adsorbed at ambient conditions on M/zeolite-13X materials by the utilizing the strong oxidizing potential of ozone gas. We also focused on the influence of metal content, and catalyst quantity on the complete mineralization of HD adsorbed on M/zeolite-13X catalytic systems.
2. Experimental procedures
2.1. Materials
The chemicals Mn (NO3)2·4H2O (purity > 97%), Fe(NO3)3·9H2O (purity > 97%), Co(NO3)2·6H2O (purity > 97%), and acetonitrile AR grade (purity > 99.99) were purchased from M/s Sigma-Aldrich India Pvt. Ltd. Zeolite-13X materials (composition Na2O·Al2O3 (2.8 ± 0.2) SiO2·(6–7)H2O) were from M/s Sorbead India Ltd. Mass flow controller (MFC, precession ± 1%) were procured from M/s Sierra, Switzerland. CO (range 1–2000 ppm, ± 1 ppm), CO2 (range 10–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, ± 10 ppm) analyzers (from M/s Technovation Analytical Instruments Ltd India) were used to measure the concentrations of carbon oxides. O2 (Purity 99.9% from M/s Everest Kanto cylinders Ltd India) gas was used as the source for the production of gaseous ozone. He (99.999%) gas which was used in this study for GC-MS analysis was supplied by M/s Everest Kanto cylinders Ltd India. Ozone generator (M/s. Eltech Engineers Ind. Ltd) and ozone analyzers were used to generate and monitor the ozone gas respectively. Ozone analyzer (M/s. Eltech Eng. India, range 0–200 ng m−3) was used to estimate the generated ozone concentration.
000, ± 10 ppm) analyzers (from M/s Technovation Analytical Instruments Ltd India) were used to measure the concentrations of carbon oxides. O2 (Purity 99.9% from M/s Everest Kanto cylinders Ltd India) gas was used as the source for the production of gaseous ozone. He (99.999%) gas which was used in this study for GC-MS analysis was supplied by M/s Everest Kanto cylinders Ltd India. Ozone generator (M/s. Eltech Engineers Ind. Ltd) and ozone analyzers were used to generate and monitor the ozone gas respectively. Ozone analyzer (M/s. Eltech Eng. India, range 0–200 ng m−3) was used to estimate the generated ozone concentration.
2.2. Preparation and physicochemical characterization of catalysts
Zeolite-13X (particle size, sieved to BSS 18/25 mesh) supported Mn, Fe, Co metal oxide catalysts of different metal loadings were prepared by wet impregnation method and activated by calcinations in air for 4 h at 400 °C.19 The specific surface area and pore size distributions were obtained by using Brunauer Emmett Teller and Barrett Joyner Halenda methods respectively for both bare zeolite-13X as well as impregnated M/zeolite-13X (M = 5 wt% Mn, Fe, Co) catalysts. N2 adsorption–desorption experiments were performed using Micromeritics-ASAP-2010 unit at liquid nitrogen temperature (−196 °C). All the catalysts were degassed for 60 min at 120 °C prior to the analysis. Rigaku Miniflex (M/s. Rigaku corporation, Japan) was used for recording the XRD patterns of all the catalysts with Ni filtered Cu Kα radiation (λ = 1.5406 Å) source in the 2θ range from 10–80° with a scan speed of 2° min−1 at 30 kV and 50 mA. TPR/TPD Nuchrom Unit with a thermal conductivity detector was utilized to carry out the TPR and TPD-NH3 experiments. Approximately 100 mg of catalyst sample in a quartz reactor was used for the TPR studies. In a flow of 50 mL min containing 5% H2–Ar gas was used to study the reduction profile of the catalysts in the temperature range of 40–750 °C with 10 °C min−1 heating rate. NH3-TPD experiments were performed on the catalyst samples which were preheated for 1 h at 300 °C and then the samples were allowed to cool at 40 °C. The catalyst was saturated by ammonia gas by pulsing onto the catalyst reactor at 40 °C. After base line restoration, the samples were heated from 40–750 °C with a heating rate 10 °C min in helium flow of 50 mL min−1. Surface morphology of the catalysts were obtained by scanning electron microscope (SEM, JOEL, JAX-840) at 20 kV by coating the gold sputtering on the samples.2.3. Reaction studies for HD degradation
Complete degradation of sulfur mustard adsorbed on zeolite-13X as well as on metal oxides impregnated zeolite-13X materials with gaseous ozone was carried out at ambient conditions using a quartz reactor of (5 mm id, 150 mm length) interfaced with an online GC-MS. The schematics of experimental setup is represented in Fig. 1. Ozone generator and oxygen gas was used in this study to generate the gaseous ozone of required concentrations (ozone conc. 4 mg min−1 in 50 mL min−1 O2 flow) for HD degradation experiments. Hamilton 5μL syringe was used to introduce the HD onto the surface of the catalyst. 100 mg of catalyst (zeolite-13X and M/zeolite-13X) diluted with glass beads was packed in a quartz reactor with glass wool and mounted vertically in a laboratory stand with clamp for reaction studies. Prior to the experiments, the catalysts were activated at 400 °C in air for 1 h. 3μL of HD was adsorbed onto the catalysts bed with the help of Hamilton 5μL syringe. The quantity of HD adsorbed was far less the adsorption capacity of zeolite-13X materials and desorption of HD adsorbed in the 50 mL min−1 flow of O2 was not observed in the present study. | ||
| Fig. 1 Schematic of typical experimental set-up used for complete degradation of HD over M/zeolite-13X supported catalysts (M = 5 wt% Mn, Fe, Co). | ||
The operating conditions (ozone gas concentrations 4 mg min−1 and gas flow rates 50 mL min−1 oxygen flow) were selected based on our previous studies on decontamination of sulfur mustard and its simulants using ozone gas in liquid phase.20
The HD adsorbed was treated with gaseous ozone concentration of 4 mg min−1 in a 50 mL min−1 oxygen flow at temperature of 37 ± 3 °C for a fixed period of time. Later on the residual HD was extracted in 3 mL acetonitrile solvent and the solutions were subjected to gas chromatograph coupled with mass spectrometry. Bruker Scion MS-SQ gas chromatography coupled with Mass Spectrometry (GC-MS) was used for the monitoring of degradation of HD at selective intervals. Analysis was performed using HP-5 column of 30 m × 0.25 mm × 0.25 μm dimensions in Electron Impact (EI) mode. Ion source (70 eV electron energy) and transfer line temperatures were maintained at 200 °C, and 250 °C respectively. GC injection port was kept at 220 °C, split ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 and solvent delay of 2 minutes were maintained in all the experiments and a constant column flow of 1 mL min−1 He (>99.999% purity) was used as carrier gas. GC-MS was used in temperature programming mode from 60–230 °C at 15 °C min−1 heating rate. Ions were scanned in the range from 35 to 250 amu. GC-MS peak areas were used to estimate the % HD degradation with time using eqn (1). For reference experiments, the same quantity of HD was dissolved in acetonitrile solvent and standardized the HD concentration with chromatogram peak area. The gaseous CO and CO2 concentrations were analyzed by using calibrated CO, CO2 analyzers respectively. The product mixture of condensable phase was trapped in acetonitrile solvent at −5 °C and also analyzed by GC-MS. The exiting gaseous stream is passed through KI trap for the decomposition of residual ozone if present on the gaseous stream.
15 and solvent delay of 2 minutes were maintained in all the experiments and a constant column flow of 1 mL min−1 He (>99.999% purity) was used as carrier gas. GC-MS was used in temperature programming mode from 60–230 °C at 15 °C min−1 heating rate. Ions were scanned in the range from 35 to 250 amu. GC-MS peak areas were used to estimate the % HD degradation with time using eqn (1). For reference experiments, the same quantity of HD was dissolved in acetonitrile solvent and standardized the HD concentration with chromatogram peak area. The gaseous CO and CO2 concentrations were analyzed by using calibrated CO, CO2 analyzers respectively. The product mixture of condensable phase was trapped in acetonitrile solvent at −5 °C and also analyzed by GC-MS. The exiting gaseous stream is passed through KI trap for the decomposition of residual ozone if present on the gaseous stream.
| The conversion of HD = {([CHD]initial − [CHD]final)/([CHD]initial) × 100} | (1) |
Caution: HD is a carcinogen, vesicant, and cytotoxic agent; therefore this compound should be handled by the trained and authorized persons with proper personal protective measures in an efficient fuming hood only. To avoid any accident, sufficient amount of decontamination solution should be available at working place.
3. Results and discussions
3.1. Characterization of M/zeolite-13X catalysts (M = 5wt% Mn, Fe, Co)
| Catalyst | Mn content (wt%) | BET surface area (m2 g−1) | Pore diameter (Å) | Pore volume (cm3 g−1) | TPR | H2 uptake (mL g−1) | TPD-NH3 total acidity (mmol g−1) | ||
|---|---|---|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | |||||||
| Tmax (°C) | Tmax (°C) | Tmax (°C) | |||||||
| Zeolite-13X | 0 | 746 | 23 | 0.437 | — | — | — | 4.78 | |
| 5% Fe | 5 | 350 | 28.18 | 0.250 | 353 | 553 | 650 | 5.95 | 6.14 |
| 5% Co | 5 | 467 | 22.57 | 0.264 | 323 | 464 | 586 | 9.56 | 8.72 |
| 5% Mn | 5 | 547 | 24.44 | 0.334 | 344 | 515 | — | 8.42 | 9.12 |
 | ||
| Fig. 2 N2-adsorption–desorption isotherms of zeolite-13X and M/zeolite-13X catalysts (M = 5 wt% Mn, Fe, Co). | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnifications. SEM data clearly indicates that the Mn and Co impregnated metal oxides distribution is quite uniform on the surface of the zeolite-13X support materials and particle aggregation is also not observed on Mn, Co/zeolite-13X materials. On the other hand little aggregation was observed in case of iron oxide impregnated zeolite-13X system. The SEM data is in agreement with textural and physicochemical properties of M/zeolite-13X catalysts as shown in the Table 1.
000× magnifications. SEM data clearly indicates that the Mn and Co impregnated metal oxides distribution is quite uniform on the surface of the zeolite-13X support materials and particle aggregation is also not observed on Mn, Co/zeolite-13X materials. On the other hand little aggregation was observed in case of iron oxide impregnated zeolite-13X system. The SEM data is in agreement with textural and physicochemical properties of M/zeolite-13X catalysts as shown in the Table 1.
 | ||
| Fig. 6 Temperature-programmed reduction (H2-TPR) profiles of zeolite-13X and M/zeolite-13X (M = 5 wt% Mn, Fe, Co) supported catalysts. | ||
In the case of Fe/zeolite-13X catalyst, one broad peak (T2max) centered between 300 to 600 °C (corresponding to consumption of hydrogen) is attributed to the reduction of Fe2O3 to Fe3O4. The broadening of the peak indicating that iron oxide over zeolite-13X support is present as isolated iron oxide species which could interact strongly with the support. Besides broadening, a shoulder is observed on the low temperature side of the second peak at 353 °C (T1max) first peak. It could be attributed to the reduction of hydroxilated iron oxide species.26,27 The third high temperature peak (T3max) centered between 570–710 °C corresponding to the subsequent reduction of Fe3O4 to metallic iron. Whereas in case of Mn/zeolite-13X catalysts, the first reduction peak (T1max) (corresponding to consumption of hydrogen) could be assigned to the reduction of MnO2 to Mn2O3 and the second peak corresponds to the reduction of Mn2O3 to Mn3O4. The third and less intensity peak at 565 °C represents the reduction of Mn3O4 to MnO.28
In the similar fashion Co/zeolite-13X catalyst also exhibited a multi step reduction pattern (Fig. 6). The first reduction peak (T1max) at 323 °C could be assigned to the reduction of CoO2/Co2O3 to Co3O4 29,30 and the second reduction at 464 °C corresponded to the reduction of Co3O4 to CoO. The third reduction peak at 586 °C indicates the reduction of CoO to metallic cobalt or it might be due to strong interaction of Co-zeolite-13X support.31 H2-TPR studies helped us to conclude the presence of different metal oxide species and also their interactions with zeolite-13X support.
 | ||
| Fig. 7 Temperature programmed desorption profiles (TPD-NH3) of bare zeolite-13X and M/zeolite-13X catalysts (M = 5 wt% Mn, Fe, Co). | ||
The peak in the temperature region of 100–200 °C represents the ammonia desorbed from the weaker acidic sites (physisorbed ammonia). The high temperature peak in the temperature region of 200–400 °C represents the ammonia desorbed from Bronsted acidic sites present on the surface of the catalyst. Whereas the broad peak in the high temperature region of 400–700 °C represents the ammonia desorbed from Lewis acidic sites32 present on the surface of the catalysts. But, zeolite-13X support contains very little amount of stronger acidic sites in the high temperature region (Bronsted and Lewis acid sites). The increases of acidity values of all the M/zeolite-13X catalysts are attributed to the impregnation of metal oxides on the surface of the zeolite-13X support material.
The overall results (Table 1) indicate that the impregnated catalysts have more total acidic sites than that of bare zeolite-13X catalyst. The desorbed amount of ammonia per gram was in the order of acidity was Mn > Co > Fe > 13X.
3.2. Catalytic properties of M/zeolite-13X (M = 5 wt% Mn, Fe, Co) systems
On the other hand, in presence of ozone as the oxidizing agent, HD degradation is very fast and complete degradation is achieved within one minute of time at same reaction condition as shown in Fig. 8b. This remarkable tendency is attributed to the strong oxidizing potential of the gaseous ozone (Eo = 2.07 eV) and its ability to react with most of the organic compounds at room temperature. The influence of various impregnated M/zeolite-13X catalysts on HD adsorbed degradation is presented in the Fig. 8a. From the figure it is concluded that all the zeolite-13X catalysts are capable to completely degrade the HD adsorbed within one minute of time in the presence of ozone at ambient reaction conditions. This is due to the high ozone decomposition capacity of these catalysts materials at mild to moderate reactions temperature35,36 which produces highly reactive atomic oxygen species. It was also reported that, the O3 was decomposed to O2 on supported metal oxides catalysts according to the following reaction steps (eqn (2)–(4)), where * refers to the catalytic active sites.37,38 The active oxygen species (O* and O2*) formed on the catalyst surface by ozone decomposition were responsible for the HD degradation.
| O3 + * → O2 + O* | (2) |
| O* + O3 → O2 + O2* | (3) |
| O2* → O2 + * | (4) |
Fig. 9b describes the concentrations of carbon oxides formed during the degradation of HD adsorbed over metal oxides impregnated and bare zeolite-13X catalysts. The results inferred that with time the concentration of CO, CO2 are increased and reached a maximum concentration with all the impregnated and bare zeolite-13X catalysts in the presence of ozone gas. Thereafter, the COX concentrations are declined with time and approached to almost zero after 10–15 minutes of time. It indicated the complete degradation of HD adsorbed over the catalysts materials with ozone. From the results it is concluded that with increasing the reaction time total oxidation products TOPs (COX) are predominates in the degradation of HD adsorbed on M/zeolite-13X catalyst in the presence of ozone at ambient reaction conditions.
| Product name | Molecular formula | m/z values |
|---|---|---|
| Acetaldehyde | CH3CHO | 44, 43, 31, 29, 26, 15 |
| Carbon dioxide | CO2 | 44, 28, 22, 16 |
| 2-Chloro ethyl vinyl sulfide | C4H7SCl | 122, 94, 73, 65, 45, 27 |
| 1,4-Oxathian | C4H8OS | 104, 74, 61, 6, 34 |
| Di vinyl sulfide | C4H6S | 86, 85, 59, 58, 45 |
| Di vinyl sulfone | C4H6SO2 | 118, 75, 66, 47, 27, 26 |
| Ethanol-2-(2-chloroehyl thio) | C4H9OSCl | 140, 141, 111, 109, 104 |
| Half mustard | 73, 63, 45, 27, 29 | |
| Thiodiglycol | C4H10OSCl | 122, 104, 91, 61, 43 |
| 2-Hydroxy ethyl vinyl sulfide | C4H8OSl | 104, 91, 61, 43 |
| Bis (2-chloro ethyl) sulfone | C4H8SO2Cl2 | 127, 128, 92, 63, 27 |
| Bis (2-chloro ethyl) sulfoxide | C4H8SOCl2 | 174, 158, 127, 104, 63, 27 |
| Sulfur dioxide | SO2 | 64, 48, 32 |
A plausible degradation mechanism is proposed based on the products identified in the GC-MS analyses of HD adsorbed over M/zeolite-13X catalysts with and without treatment from ozone gas.
In absence of ozone. In the absence of ozone, HD reacts with surface hydroxyl groups of M/zeolite-13X catalysts very slowly by means of hydrolysis, elimination and cyclisation reactions to yield various products (Table 2). The possible reactions of HD degradation in absence of ozone were reflected in Scheme 1. According to the Scheme 1, HD molecules react with surface of the M/zeolite-13X catalysts in the following two ways.
 | ||
| Scheme 1 Elimination and hydrolysis reactions of sulfur mustard on the surface of the M/zeolite-13X catalyst. | ||
In first way they react with the physisorbed (the moisture adsorbed while transferring the samples into the reactor) or intercalated water molecules and surface hydroxyl groups that are present on the surface of the catalyst to form hemi mustard followed by thiodiglycol. This reaction is proposed to proceed through the formation of cyclic sulfonium ion intermediate.39 Cyclisation of thiodiglycol by elimination of water molecule leads to the formation of 1,4-oxathiane.
In the second way, HD molecules react with Lewis acid sites of metal oxide catalyst (Mn+4, Co+3, Fe+3) to form surface bound alkoxyl species followed by hydrolysis, elimination and surface complexation of HD which poison the active sites of the catalysts and leads the reduction of reactivity towards HD degradation.40 HCl elimination from HD molecule leads the formation of vinyl chloro ethyl sulfide and divinyl sulfide respectively. Hydrolysis of vinyl chloro ethyl sulfide results the formation of hydroxy vinyl ethyl sulfide (Scheme 1).
In the presence of ozone. As the decontamination reactions are found to be very slow on M/zeolite-13X catalyst (M = 5 wt% Mn, Co, Fe), ozone assisted catalytic degradation is opted for the faster and complete decontamination of HD adsorbed. The reaction mechanism proposed for the degradation of HD adsorbed in the presence of ozone over M/zeolite-13X catalysts based on the products identified by GC-MS analyses as displayed in (Scheme 2).
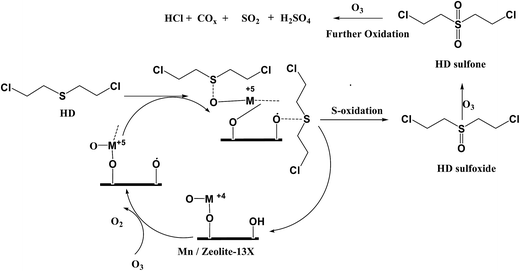 | ||
| Scheme 2 Reaction mechanism indicating the degradation of adsorbed HD over Mn/zeolite-13X in presence of ozone. | ||
It is proposed that presence of ozone significantly alters the catalytic mechanism from molecular oxygen by ozone decomposition in the presence of catalyst in gas phase reactions which proceeds mostly through surface mechanisms. Oyama et al.41 reported that adsorbed active oxygen species are formed on the surface of the MyOx catalysts during O3 decomposition and these adsorbed oxygen species play the major role while oxidizing the VOCs to complete mineralized products due to its remarkable oxidizing potential.42 Peroxide ion species formed during the ozone decomposition on manganese oxide catalyst was identified by in situ Raman spectroscopy.43 It is observed that addition of ozone significantly increases the oxidation efficiency of metal oxide catalysts and reduces the oxidation temperature. It might be due to the active oxygen species formed during the O3 decomposition on the metal oxide catalysts and its further reaction with HD molecules.
Decomposition of ozone on the surface of the metal oxide catalyst results the formation of catalytic active sites O−M+5 as shown in the Scheme 2. Initially, the HD adsorbed interacts with the actives sites derived during the ozone decomposition and forms the HDO which on further oxidation yields the HDO2 by ‘S’ atom oxidation. Several studies on degradation of reduced sulfur compounds using transition metal oxide catalysts evidenced the formation of sulfoxide and sulfone on the surface of the catalyst material.44–46 The formed HDO and HDO2 on extended oxidation reactions with adsorbed active oxygen species results the formation of carbon oxides, sulfur dioxide, HCl, H2SO4 and water molecules. In the present investigation, initially HDO and HDO2 are observed as major products in GC-MS analyses. With time the concentrations of HDO and HDO2 are declined and the concentrations of carbon oxides increased. This is attributed to the higher ozone concentration which leads the extended oxidation of formed HDO and HDO2 to total oxidation products by breakage of C–S bond. As the time progresses the available surface adsorbed oxygen species are more and the HD adsorbed concentration is also decreases which favors the extended oxidation reactions to proceeds even at ambient reaction conditions. Literature reports support the cleavage of C–S bond on the metal oxide catalysts and the extended oxidation of partial oxidation products to completely mineralized products in the presence of ozone.44,47–51
In another way, formation of acetaldehyde, carbon oxide and small amounts of acetic acid, etc. can be attributed to the oxidation and cleavage of C–S bond of HD by superoxide anion radical eqn (5)–(10). It should be noted that, the mechanism is highly complex and can also follow combination of mechanisms such as oxidation of S-atom, C-atom, cleavage of C–S bond, hydrolysis of C–Cl bond or elimination of HCl and free radical mechanisms as well. However, based on the quantities of products observed, it is under stood that C–S bond cleavage and S atom oxidation contributes to a major extend for the degradation of HD adsorbed over M/zeolite-13X catalysts at ambient temperature and atmospheric pressure.
| (Cl-C2H4)2S + O2˙− + H2O → CH3CHO + CH3CH2SH | (5) |
| CH3CH2SH + O2˙− → CO2 + H2O + H2SO4 | (6) |
| CH3CHO + O2˙− → CH3COOH | (7) |
| CH3COOH + O2˙− → CO2 + H2O | (8) |
| ClCH2CH2OH + O2˙− → CO2 + H2O + HCl | (9) |
| HSCH2CH2OH + O2˙− → CO2 + H2O + H2SO4 | (10) |
It is observed that the selectivity of catalyst is governed by the nature of oxygen species; electrophilic oxygen species such as O2− and O− are responsible for total oxidation of hydrocarbons, whereas the nucleophilic lattice stabilized O2− is responsible for the formation of partial oxidation products.52 Here, the major products identified in the HD degradation with other M/zeolite-13X (M = 5 wt% Co, Fe) catalysts are also mainly carbon oxides, mustard sulfoxide and sulfone. Therefore, it is assumed that the HD degradation mechanism on Fe and Co catalyst might be proceeding through the formation of O–Fe+3 and O–Co+4 active sites respectively in the presence of ozone.38,41–43,53
3.3. Reusability of the catalysts for degradation of HD adsorbed
The catalysts spent for the degradation of HD adsorbed in presence of ozone were activated by calcination in the atmospheric air flow of 1 lpm for 1 h at 400 °C. After activation, the spent catalysts were tested by adsorbing the same quantity of sulfur mustard followed by degradation of HD adsorbed in presence of ozone. The degradation efficiency of the Co/zeolite-13X catalysts are tested with ozone for several times (Fig. 11) and the results reveal that the catalysts exhibit 100% efficiency for the degradation of HD adsorbed with ozone after using it for 4 times. Similar results are obtained in case of other (5 wt% Mn, Fe/zeolite-13X) catalysts also except with bare zeolite-13X catalyst. Based on the results it is concluded that the impregnated metal oxide catalysts on zeolite-13X support can be reused several times without loss of its efficiency for the degradation of HD adsorbed using ozone as the oxidizing agent. The reason for not getting 100% efficiency with zeolite-13X materials might be due to decline of ozone decomposition efficiency of bare zeolite-13X at ambient temperature which is the key factor for activity of the catalyst with ozone. The reusability results concludes that impregnated zeolite-13X materials are the better choice for HD degradation compared to bare zeolite-13X materials.4. Conclusions
The present study describes the complete degradation of HD adsorbed over bare zeolite-13X and M/zeolite-13X (M = 5 wt% Mn, Fe, Co) catalysts in presence of gaseous ozone. Depending on the reaction time the HD adsorbed can be degraded to its sulfoxide and sulfone as well as to total oxidation products using ozone gas. Little longer reaction times (>5 minutes) yields the complete oxidation products, whereas short reaction times (<1 minute) leads the formation of mustard sulfoxide and sulfone which are somewhat toxic compounds compared to HD. Nevertheless, 100% HD degradation is achieved in both the cases at ambient conditions. M/zeolite-13X materials exhibited 100% ozone decomposition efficiency even at ambient conditions for more than 15 minutes, but bare zeolite-13X materials exhibits the same efficiency only for 5 minutes. Furthermore, the metal oxides impregnated zeolite-13X materials are reusable for several times without loss of its efficiency. Therefore, M/zeolite-13X materials in presence of ozone can successfully be used for the complete degradation of adsorbed HD in Pressure Swing Adsorption (PSA) based NBC filtration systems for in situ degradation to non toxic compounds. This method can also applicable for the degradation of other CWA as well. In conclusion, the present method has more practical importance for the in situ degradation of CWA's and can be used for the decontamination of adsorbed CWA's in PSA based regenerable NBC filtration systems in combination with small quantity of gaseous ozone and metal oxides impregnated zeolite-13X materials.Acknowledgements
The authors would like to thank Dr Lokendra Singh, Director DRDE for his keen interest and encouragement to carry out this study and also to permit the work for publishing.References
- R. S. Thakur, N. Kaistha and D. P. Rao, Comput. Chem. Eng., 2011, 35, 973–983 CrossRef CAS.
- Z. Yuwen, W. Yuyuan, G. Jianying and Z. Jilin, Sep. Purif. Technol., 2005, 42, 123–127 CrossRef CAS.
- J. Beck, D. Friedrich, S. Brandani, S. Guillas and E. S. Fraga, Comput.–Aided Chem. Eng., 2012, 30, 1217–1221 CAS.
- A. K. Beeyani, K. Singh, R. K. Vyas, S. Kumar and S. Kumar, Pol. J. Chem. Technol., 2010, 12, 18–28 Search PubMed.
- C. J. Karwacki, J. H. Buchanan, J. J. Mahle, L. C. Buettner and G. W. Wagner, Langmuir, 1999, 15, 8645–8650 CrossRef CAS.
- D. Kaplan, L. Shmueli, I. Nir, D. Waysbort and I. Columbus, Clean: Soil, Air, Water, 2007, 35(2), 172–177 CrossRef CAS.
- I. Columbus, D. Waysbort, L. Shmueli, I. Nir and I. Kaplan, Environ. Sci. Technol., 2006, 40, 3952–3958 CrossRef CAS PubMed.
- A. Sharma, A. Saxena, B. Singh, M. Sharma, M. V. S. Suryanarayana, R. P. Semwal, K. Ganeshan and K. Sekhar, J. Hazard. Mater., 2006, B133, 106–112 CrossRef PubMed.
- B. Singh, T. H. Mahato, A. K. Srivastava, G. K. Prasad, K. Ganesan, R. Vijayaraghavan and R. Jain, J. Hazard. Mater., 2011, 190, 1053–1057 CrossRef CAS PubMed.
- G. K. Prasad, T. H. Mahato, B. Singh, K. Ganesan, P. Pandey and K. Sekhar, J. Hazard. Mater., 2007, 149, 460–464 CrossRef CAS PubMed.
- A. Bellamy, J. Chem. Soc., Perkin Trans. 2, 1994, 2325–2328 RSC.
- A. Bellamy, React. Polym., 1994, 23, 101–106 CrossRef CAS.
- A. Bellamy, React. Funct. Polym., 1997, 32, 293–297 CrossRef CAS.
- R. Osovsky, D. Kaplan, H. Rottera, S. Kendler, M. Goldvaser, Y. Zafrani and I. Columbus, Carbon, 2011, 49, 3899–3906 CrossRef CAS.
- S. Popiel, Z. Witkiewicz and M. Chrzanowski, J. Hazard. Mater., 2008, 153, 37–43 CrossRef CAS PubMed.
- P. C. C. Faria, D. C. M. Monteiro, J. J. M. Órfão and M. F. R. Pereira, Chemosphere, 2009, 74, 818–824 CrossRef CAS PubMed.
- R. C. Martins and R. M. Quinta-Ferreira, Desalination, 2011, 270, 90–97 CrossRef CAS.
- J. Wu, L. Ma, Y. Chen, Y. Cheng, Y. Liu and X. Zha, Water Res., 2016, 92, 140–148 CrossRef CAS PubMed.
- C. Ramakrishna, R. Krishna, T. Gopi, G. Swetha, B. Saini, S. Chandra Shekar and A. Srivastava, Chin. J. Catal., 2016, 37, 240–249 CrossRef CAS.
- C. Ramakrishna, R. Krishna, B. Saini, T. Gopi, G. Swetha and S. Chandra Shekar, Phosphorus, Sulfur Silicon Relat. Elem., 2016, 191(7), 965–970 CrossRef CAS.
- S. Lowell, J. E. Shields, M. A. Thomas and M. Thommes, Characterization of Porous Solids and Powders: Surface Area, Pore Size and Density, Springer, 2004, ISBN 1-4020-2302-2 Search PubMed.
- J. Okal, M. Zawadzki, L. Kepinski, L. Krajczyki and W. Tylus, Appl. Catal., A, 2007, 319, 202–209 CrossRef CAS.
- M. M. Treacy and J. B. Higgins, Collection of Simulated XRD Powder Patterns for Zeolites, 4th edn, 2001, pp. 152–153 Search PubMed.
- W. Yu, B. Wu, J. Xu, Z. Tao, H. Xiang and Y. Li, Catal. Lett., 2008, 125, 116–122 CrossRef CAS.
- J. Ma, B. Fan, R. Li and J. Cao, Catal. Lett., 1994, 23, 189–194 CrossRef CAS.
- G. Munteanu, L. Ilieva and D. Andreeva, Thermochim. Acta, 1997, 291, 171–177 CrossRef CAS.
- K. C. Wu, Y. L. Tung, Y. L. Chen and Y. W. Chen, Appl. Catal., B, 2004, 53, 111–116 CrossRef CAS.
- P. R. Ettireddy, N. Ettireddy, S. Mamedov, P. Boolchand and P. G. Smirniotis, Appl. Catal., B, 2007, 76, 123–134 CrossRef CAS.
- N. Fischer, E. Van Steen and M. Claeys, Catal. Today, 2011, 171, 174–179 CrossRef CAS.
- C. B. Wang, H. K. Lin and C. W. Tang, Catal. Lett., 2004, 94, 69–74 CrossRef CAS.
- A. Sirijaruphan, A. Horvath, J. G. Goodwin Jr and R. Oukaci, Catal. Lett., 2003, 91, 89–94 CrossRef CAS.
- F. T. T. Ng, A. Rahman, T. Ohasi and M. Jiang, Appl. Catal., B, 2005, 56(1–2), 127–136 CrossRef CAS.
- G. K. Prasad, B. Singh, K. Ganesana, A. Batra, T. Kumeria, P. K. Gutch and R. Vijayaraghavan, J. Hazard. Mater., 2009, 167, 1192–1197 CrossRef CAS PubMed.
- G. K. Prasad, P. V. R. K. Ramacharyulu, K. Batra, B. Singh, A. R. Srivastava, K. Ganesan and R. Vijayaraghavan, J. Hazard. Mater., 2010, 183, 847–852 CrossRef CAS PubMed.
- H. Einaga, M. Harada and A. Ogata, Catal. Lett., 2009, 129, 422–427 CrossRef CAS.
- E. Rezaei, J. Soltan and N. Chen, Appl. Catal., B, 2013, 136–137, 239–247 CrossRef CAS.
- H. Huang, X. Ye, W. Huang, J. Chen, Y. Xu, M. Wu, Q. Shao, Z. Peng, G. Ou, J. Shi, X. Feng, Q. Feng, H. Huang, P. Hu and D. Y. C. Leung, Chem. Eng. J., 2015, 264, 24–31 CrossRef CAS.
- B. Dhandapani and S. T. Oyama, Appl. Catal., B, 1997, 11, 129–166 CrossRef CAS.
- G. K. Prasad, P. V. R. K. Ramacharyulu, B. Singh, K. Batra, A. R. Srivastava, K. Ganesan and R. Vijayaraghavan, J. Mol. Catal. A: Chem., 2011, 340, 55–62 CrossRef.
- G. K. Prasad, T. H. Mahato, B. Singh, K. Ganesan, P. Pandey and K. Sekhar, J. Hazard. Mater., 2007, 149, 460–464 CrossRef CAS PubMed.
- S. T. Oyama, Catal. Rev.: Sci. Eng., 2000, 42(3), 279–322 CAS.
- A. Naydenov, R. Stoyanova and D. Mehandjiev, J. Mol. Catal. A: Chem., 1995, 98, 9–14 CrossRef CAS.
- C. Reed, Y. Xi and S. T. Oyama, J. Catal., 2005, 235, 378–392 CrossRef CAS.
- A. V. Vorontsov, E. V. Savinov, L. Davydov and P. G. Smirniotis, Appl. Catal., B, 2001, 32, 11–24 CrossRef CAS.
- G. K. Prasad, B. Singh, K. Ganesan, A. Batra, T. Kumeria, P. K. Gutch and R. Vijayaraghavan, J. Hazard. Mater., 2009, 167, 1192–1197 CrossRef CAS PubMed.
- T. H. Mahato, G. K. Prasad, B. Singh, A. R. Srivastava, K. Ganesan, J. Acharya and R. Vijayaraghavan, J. Hazard. Mater., 2009, 166, 1545–1549 CrossRef CAS PubMed.
- L. Wang and J. Zhang, J. Mol. Struct.: THEOCHEM, 2001, 543, 167–175 CrossRef CAS.
- W. S. Nip, D. L. Singleton and R. J. Cvetanovic, J. Am. Chem. Soc., 1981, 103, 3526–3530 CrossRef CAS.
- C. Wilson and D. M. Hirst, Prog. React. Kinet., 1996, 21, 69–132 CAS.
- H. Sievering, J. Boatman, E. Gorman, Y. Kim, L. Anderson, G. Ennis, M. Luria and S. Pandis, Nature, 1992, 360, 571–573 CrossRef CAS.
- I. Barnes, J. Hjorth and N. Mihalopoulos, Chem. Rev., 2006, 106, 940–975 CrossRef CAS PubMed.
- J. Sloczynski, R. Grabowski, R. T. Sobieraj and M. Witko, J. Mol. Catal. A: Chem., 2007, 277, 27–34 CrossRef CAS.
- P. Kanova, M. Stoyanova, A. Naydenov, S. Christoskova and D. Mehandjiev, Appl. Catal., A, 2006, 298, 109–114 CrossRef.
| This journal is © The Royal Society of Chemistry 2016 |







