Efficient deep-blue non-doped organic light-emitting diode with improved roll-off of efficiency based on hybrid local and charge-transfer excited state†
Haichao Liua,
Qing Baia,
Weijun Lib,
Yachen Guoa,
Liang Yaoa,
Yu Gaoa,
Jinyu Lia,
Ping Lua,
Bing Yang*a and
Yuguang Mac
aState Key Laboratory of Supramolecular Structure and Materials, Jilin University, Changchun, 130012, P. R. China. E-mail: yangbing@jlu.edu.cn
bCollege of Chemical Engineering, Zhejiang University of Technology, Hangzhou, 310014, P. R. China
cState Key Laboratory of Luminescent Materials and Devices, South China University of Technology, Guangzhou, 510640, P. R. China
First published on 19th July 2016
Abstract
High-efficiency deep-blue light-emitting materials are of significance in the fields of commercial full-color displays and solid-state lightings. A hybrid local and charge-transfer (HLCT) excited state not only favors deep-blue emission avoiding a large redshift from a strong charge-transfer (CT) state, but also simultaneously harvests both high photoluminescence efficiency and high exciton-utilizing efficiency. Herein, we report a new V-shaped acceptor–donor–acceptor (A–D–A) type molecule TPA–2PPI with an HLCT emissive state, which is modified from D–A type TPA–PPI. The non-doped device based on the TPA–2PPI emitter still exhibits deep-blue emission peaking at 452 nm with a full width at half maximum (FWHM) of only 50 nm and Commission International de L'Eclairage (CIE) coordinates of (0.151, 0.108). Compared with TPA–PPI, the electroluminescence (EL) not only maintains high efficiency with a maximum external quantum efficiency (EQE) of 4.91%, but also the EL device displays a significantly slower roll-off of efficiency at high luminances with an EQE of 4.89% (or 4.56%) at 100 cd m−2 (or 1000 cd m−2), which is confidently beneficial for the operative stability of OLED devices.
Introduction
Efficient deep-blue light-emitting materials play an irreplaceable role in the field of full-color displays and solid-state lightings.1 In organic light-emitting diodes (OLEDs), deep-blue materials not only widen the color gamut and reduce power consumption, but also can be utilized as excitation sources to generate other visible and white emissions by an energy cascade.2 Through persistent efforts, a series of important progresses have been made in the efficiencies of OLEDs in recent years.3 However, high-efficiency deep-blue OLEDs are really scarce for both phosphorescent and fluorescent types.4 One of the reasons resulting in low efficiency of deep-blue OLEDs is ascribed to the intrinsic wide bandgap of deep-blue emitter, which is difficult to match the energy levels of functional layers or electrodes in device structure and further causes unbalanced charge injection and charge transport in the electroluminescence (EL) process.5In the past decades, organic donor–acceptor (D–A) type molecules have been gradually constructed to be one of the most commonly-used methods to improve charge injection and carrier transport in organic semiconductor materials.6 Owing to energy level difference between donor and acceptor, D–A molecule employs the highest occupied molecular orbital (HOMO) of donor and the lowest unoccupied molecular orbital (LUMO) of acceptor to reconstitute proper energy level,7 in favor of designing device structures with more balanced charge injection and transport. Simultaneously, D–A molecule usually possesses weakly-bound charge transfer (CT) exciton8 or sufficiently small singlet-triplet energy splitting,3b–d which facilitates the reverse intersystem crossing (RISC) process (T → S) to increase triplet exciton-utilizing efficiency of OLEDs. Nevertheless, as inherent CT properties in D–A molecules, the negative effects are also accompanied that CT-state molecule exhibits either a low-efficiency fluorescence due to the forbidden transition induced by the spatially separated hole and electron wavefunctions or a red-shifted emission because of narrowed bandgap, which is disadvantageous for high-efficiency deep-blue emissive materials. In contrast to CT state, locally-excited (LE) state molecule can produce high-efficiency fluorescence due to large oscillator strength from almost complete overlap of hole and electron wavefunctions, and possess non-redshift emission from the small dipole moment of excited state insensitive to the polar environment, which are actually complementary to those of CT state.7
Considering the above issues, if CT and LE states could be integrated into one D–A molecule, it would be an ideal strategy to greatly improve the efficiencies of OLEDs. Recently, hybrid local and charge-transfer (HLCT) excited state8,9 has been proposed to achieve high-efficiency OLEDs, particularly to meet the challenge between high efficiency and deep-blue emission of OLEDs. In addition, HLCT-state material intrinsically possesses short-lived exciton character, avoiding severe efficiency roll-off from long-lived exciton quenching.8b In terms of HLCT state, LE state is introduced to form a new type of excited state with both advantages of LE and CT states, alleviating largely red-shifted emission from strong CT state together with compatible high photoluminescence quantum yield (ηPL) and high exciton-utilizing efficiency (ηs). Using HLCT-state conception, high-efficiency deep-blue OLEDs have been obtained in our group, such as a maximum external quantum efficiency (EQE) of 5.02% with a Commission Internationale de L'Eclairage (CIE) coordinate of (0.153, 0.114),8a 6.80% with CIE (0.152, 0.077)4b and even near ultraviolet emission with EQE of 3.33% and CIE (0.161, 0.049).8i Su and Ge also reported HLCT-state molecules, one of whose devices showed violet-blue emission with a maximum EQE of 2.39% and a CIE of (0.16, 0.05).10 Herein, we report a deep-blue molecule with V-shaped structure, N-phenyl-4′-(1-phenyl-1H-phenanthro[9,10-d]imidazol-2-yl)-N-(4′-(1-phenyl-1H-phenanthro[9,10-d]imidazol-2-yl)-[1,1′-biphenyl]-4-yl)-[1,1′-biphenyl]-4-amine (TPA–2PPI), which is composed of one donor (triphenylamine, TPA) and two acceptors (1,2-diphenyl-1H-phenanthro[9,10-d]imidazole, PPI), which was derived from TPA–PPI ever reported. It has been demonstrated that TPA acts as donor and PPI as acceptor in TPA–PPI compound.8a,i,11 In this article, the new type A–D–A molecule is also demonstrated as a typical HLCT-state with two-section different HLCT states. Compared with TPA–PPI, the non-doped TPA–2PPI device also showed a good performance with a maximum EQE of 4.91%, a maximum current efficiency (CE) of 4.76 cd A−1 and CIE of (0.151, 0.108). Especially, a significantly slower roll-off of efficiency with EQE (or CE) of 4.89% (or 4.74 cd A−1) and 4.56% (or 4.42 cd A−1) at 100 and 1000 cd m−2, respectively.
Results and discussion
Synthetic procedures
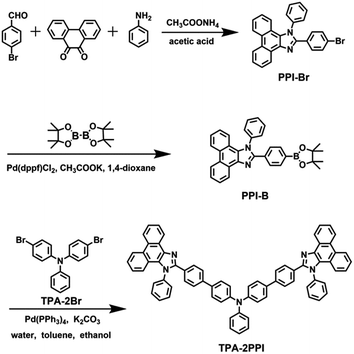
TPA–2PPI was prepared by facile synthesis routes as shown in Scheme 1. PPI–Br was synthesized according to literature 8a. TPA–2Br and PPI–B were coupled by Suzuki reaction, and crude mixture was purified by chromatography to get target compound. TPA–2PPI was fully characterized and confirmed by 1H NMR, 13C NMR, MS and elemental analysis (see ESI†).Theoretical calculation
To clearly view molecular conformation of TPA–2PPI, we optimized the ground-state geometry using the density functional theory (DFT) at the level of B3LYP/6-31G(d,p). TPA–2PPI presents a V-shaped conformation with a twist angle of 35° between TPA and PPI moieties for each branch of TPA–2PPI (Fig. S1†). Such moderate twist angle does not completely interrupt molecular conjugation between donor (TPA) and acceptor (PPI), and further it is reasonable to construct HLCT state for TPA–2PPI using a proper state coupling between LE and CT. To describe the nature of emission, the lowest singlet state (S1) was optimized using time-dependent DFT theory. As shown in Fig. 1, on the one hand, from the natural transition orbitals (NTO), both hole and particle are mainly delocalized over the molecular backbone and share large overlap. In principle, the NTO of TPA–2PPI well accords with the characteristic of HLCT state. On the other hand, the absorption NTO from hole to particle is mainly related to the transition from HOMO to LUMO, indicating the possible equality of optical and electrical bandgaps (Fig. S2†). | ||
| Fig. 1 NTO for S1 → S0 transition in TPA–2PPI. Herein, f represents for the oscillator strength, and the percentage weights of hole–particle are given for the S1 → S0 emission. | ||
Photophysical properties
To understand basic photophysical properties of TPA–2PPI, ultraviolet-visible (UV) and photoluminescent (PL) spectra were recorded in both dilute tetrahydrofuran (THF) solution and vacuum-evaporated film (Fig. 2). In THF solution, TPA–2PPI has a long-wavelength absorption band peaked at 374 nm, corresponding to optical bandgap of 2.99 eV estimated from the onset of absorption spectrum. Vacuum-evaporated film of TPA–2PPI has a redshifted absorption peak at 380 nm relative to 374 nm of TPA–2PPI in THF solution, and also shows a level-off tail in long-wavelength region resulting from molecular aggregation.12 TPA–2PPI shows deep-blue emission (Fig. 2) in THF solution with a maximum peak at 437 nm and PL efficiency ηPL = 85%. As a contrast, its vacuum-evaporated film has deep-blue emission (Fig. 2) with a slight redshift to 450 nm and ηPL = 50%. | ||
| Fig. 2 UV and PL spectra of TPA–2PPI in both THF solution and evaporated film. Insets are the photographs of TPA–2PPI in THF solution and evaporated film, respectively. | ||
Solvatochromic effect is an inherent property for CT-state molecule,7 which is commonly used to examine the excited state properties in different solvents. From low-polarity hexane to high-polarity acetonitrile, TPA–2PPI gradually shows broadened PL spectra and vanished vibronic resolution with a redshift of 49 nm from deep blue to pure blue, indicating the existence of CT-state component (Fig. 3a). On the other hand, PL spectra significantly maintain vibrational structures without obvious redshift in low-polarity solvents, demonstrating LE-state component. We applied Lippert–Mataga relation to estimate the dipole moment of excited state (μe), describing the variation of excited state of TPA–2PPI. For this purpose, the linear relation of Stokes shift (νa − νf) against the orientation polarizability f(ε, n) was fitted for TPA–2PPI (Fig. 3b and Table S1†). For the vibronic PL spectrum, 0–0 band is chosen as the emission peak to calculate the Stokes shift. The ground-state dipole moment (μg) was estimated to be 5.42 D using DFT at the level of B3LYP/6-31G(d,p). As a result, two-section linear relations were well fitted for TPA–2PPI and accordingly two μes were estimated to be 10.01 D and 23.27 D, respectively. In low-polarity region, μe = 10.01 D shows a weak CT-state character while μe = 23.27 D indicates a strong CT character in high-polarity region. Most notably, high ηPL can be maintained and even slightly raised with the increasing solvent polarity (Fig. 3c), as well as easily distinguished vibronic resolution of PL spectrum in low-polarity solvent clearly demonstrates LE-state character. Thus, both LE and CT states of TPA–2PPI coexist in low-polarity solvents. Furthermore, time-resolved fluorescence reveals a mono-exponential decay of TPA–2PPI in hexane (0.80 ns) and ethyl ether (1.00 ns) (Fig. 3d), which exactly verifies HLCT-state character. In high-polarity region, μe = 23.27 D of TPA–2PPI is almost equal to that of 4-(N,N-dimethylamino)benzonitrile (DMABN), which is a typical CT molecule with a μe = 23 D.7 It can be inferred that TPA–2PPI possesses CT-dominated state character in high-polarity solvents. It is well-known that strong CT-state molecule suffers from the sharply declined ηPL as solvent polarity increases. However, the ηPLs of TPA–2PPI still remain at high level and mildly decline with the increasing solvent polarity, and even ηPL = 69% in high-polarity acetonitrile, which should be attributed to a certain degree of LE state component. Time-resolved fluorescence also reveals a mono-exponential decay of TPA–2PPI in THF (1.08 ns) and acetonitrile (1.68 ns), indicating that TPA–2PPI possesses the HLCT-state character in high-polarity solvents. Given this, the process from low to high polarity can be divided into two stages: LE-dominated low-polarity region and CT-dominated high-polarity region respectively for the HLCT state of TPA–2PPI. That is, TPA–2PPI maintains the HLCT-state character all the time from low-polarity hexane to high-polarity acetonitrile.
To check the photostability of TPA–2PPI material, we collected the 1H NMR spectra after the sample dissolving in deuterated dimethylsulphoxide (DMSO) solvent was irradiated for different time under 365 nm UV irradiation. All 1H NMR spectra are consistent (Fig. S3†), demonstrating the photostability of TPA–2PPI material, i.e., unchanged molecular structure of TPA–2PPI compound after UV irradiation.
Thermal and electrochemical properties
The thermal properties were investigated by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA), which demonstrates that A–D–A type TPA–2PPI is highly thermally stable. The glass transition temperature (Tg) and thermal decomposition temperature (Td) were measured as 201 °C and 576 °C respectively (Fig. S4†), which are very high probably due to the larger molecular dimension of TPA–2PPI. To further ensure the thermal stability of TPA–2PPI material, we annealed TPA–2PPI powder at 360 °C. Compared with the prepared solid powder after column chromatography, both 1H NMR spectra are identical (Fig. S5†), demonstrating the unchanged structure and the excellent thermal stability after annealing TPA–2PPI powder. Cyclic voltammetry (CV) were measured to calculate the HOMO and LUMO levels of −5.19 eV and −2.29 eV, respectively (Fig. S4†). Correspondingly, the electrical bandgap of TPA–2PPI was estimated to be 2.90 eV, which is almost the same as 2.99 eV of optical bandgap, as predicted by theoretical calculation.Electroluminescence properties
In order to investigate the EL property, TPA–2PPI serves as emitter to construct the multi-layer non-doped device (Fig. 4a): ITO/MoO3 (7 nm)/NPB (50 nm)/TCTA (10 nm)/TPA–2PPI (20 nm)/TPBi (40 nm)/LiF (1 nm)/Al (120 nm), in which NPB = N,N′-di-1-naphthyl-N,N′-diphenylbenzidine, TCTA = 4,4′,4′′-tri(N-carbazolyl)-triphenylamine, TPBi = 1,3,5-tri(phenyl-2-benzimidazolyl)-benzene. MoO3 was utilized as a hole-injecting layer, NPB and TCTA as hole-transporting and buffer layers, and TPBi as an electron-transporting and hole-blocking layer. Deep-blue emission was observed, and EL spectrum is peaked at 452 nm with a full width at half maximum (FWHM) of only 50 nm and CIE coordinate of (0.151, 0.108) (Fig. 4b). Narrow FWHM contributes to good CIE chromaticity of TPA–2PPI device. Fig. 4c shows the luminance–voltage–current density characteristics of the device. TPA–2PPI device has a very low turn-on voltage of 2.8 V at 1 cd m−2, which is ascribed to well-matched energy level alignment in the device. Moreover, the EL spectra of the TPA–2PPI device are surely independent of driving voltages from 3 V to 11 V, revealing that the recombination of electrons and holes is well confined within the emitter layer during the EL process (Fig. S6†).TPA–2PPI device shows a good performance with a maximum EQE, a maximum CE and a maximum power efficiency (PE) of 4.91% (at 4.2 V and 65 cd m−2), 4.76 cd A−1 and 3.61 lm W−1, respectively (Fig. 4d and 5). The maximum luminance reaches to 9277 cd m−2, and particularly, the device has a very low roll-off of EQE and CE with the enhanced luminance. In terms of the practical applications, between 100 and 1000 cd m−2, EQE (or CE) can be maintained at 4.89% (or 4.74 cd A−1) and 4.56% (or 4.42 cd A−1), as shown in Table 1. Furthermore, the ηs can be roughly estimated for TPA–2PPI devices on the basis of the known ηPL value, maximum EQE and the other two assumed parameters of the light out-coupling efficiency of 20% and the 100% hole–electron recombination in the OLEDs. Thus, the ηs = 49% in the TPA–2PPI device is obtained, which breaks through the upper-limit of ηs = 25% in the conventional fluorescent OLEDs, which can be attributed to the HLCT-state and hot exciton mechanism, indicating a harmony between high ηPL (50%) and high ηs (49%).
| λEL,max (nm) | CIE (x, y) | CEmax (cd A−1) | PEmax (lm W−1) | EQEmax (%) | Lmax (cd m−2) | @100 cd m−2 | @1000 cd m−2 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CE (cd A−1) | PE (lm W−1) | EQE (%) | CE (cd A−1) | PE (lm W−1) | EQE (%) | |||||||
| TPA–2PPI | 452 | (0.151, 0.108) | 4.76 | 3.61 | 4.91 | 9277 | 4.74 | 3.30 | 4.89 | 4.42 | 2.04 | 4.56 |
| TPA–PPI | 434 | (0.153, 0.114) | 5.66 | 6.13 | 5.02 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 675 675 |
4.25 | 2.70 | 3.76 | 3.50 | 1.39 | 3.13 |
As a comparison, TPA–PPI as a typical deep-blue molecule whose device exhibited excellent performance with a maximum EQE of 5.02%, a maximum CE of 5.66 cd A−1 and CIE coordinate of (0.153, 0.114), has been reported by our group in 2012.8a Although A–D–A type TPA–2PPI and D–A type TPA–PPI share almost the same device performance, TPA–2PPI device exhibits the slower roll-off of efficiency (Table 1 and Fig. 5) and the more stable thermal property than those of TPA–PPI, which may jointly contribute to the better final device operative stability. As a whole, TPA–2PPI device is still the one among the best deep-blue fluorescent OLEDs.
Conclusions
A V-shaped molecule TPA–2PPI was synthesized with one donor and two acceptors, which was modified from D–A type TPA–PPI. TPA–2PPI shows HLCT-state property from low-polarity hexane to high-polarity acetonitrile, although two-section linear relations were well fitted by Lippert–Mataga equation. Moreover, its thermal stability is greatly improved relative to TPA–PPI. The non-doped device based on TPA–2PPI emitter exhibits deep-blue EL spectrum peaked at 452 nm with a FWHM of only 50 nm and CIE coordinates of (0.151, 0.108). The device shows excellent performance with a maximum EQE of 4.91% and a maximum CE of 4.76 cd A−1. Moreover, the device also presents very slow roll-off of EQE (or CE), which can be maintained at 4.89% (or 4.74 cd A−1) and 4.56% (or 4.42 cd A−1) at a practical range between 100 and 1000 cd m−2. This work not only proposes an idea to design high-efficiency deep-blue OLED materials using HLCT mechanism, but also provides a feasible way to enhance the OLED device stability with the increased thermal stability and the slower roll-off of efficiency and without the sacrifice of efficiency and CIE coordinate using the modified ratio of D and A.Acknowledgements
This work was supported by the National Natural Science Foundation of China (51273078, 51473063 and 91233113), the Ministry of Science and Technology of China (2013CB834801 and 2015CB655003) and the Open Project Foundation of the State Key Laboratory of Luminescence and Applications (Changchun Institute of Optics, Fine Mechanics and Physics of the Chinese Academy of Science, SKLA-2016-04).Notes and references
- (a) X. Yang, X. Xu and G. Zhou, J. Mater. Chem. C, 2015, 3, 913 RSC; (b) M. Zhu and C. Yang, Chem. Soc. Rev., 2013, 42, 4963 RSC; (c) J. Kwak, J. Lim, M. Park, S. Lee, K. Char and C. Lee, Nano Lett., 2015, 15, 3793 CrossRef CAS PubMed; (d) H. Shen, X. Bai, A. Wang, H. Wang, L. Qian, Y. Yang, A. Titov, J. Hyvonen, Y. Zheng and L. S. Li, Adv. Funct. Mater., 2014, 24, 2367 CrossRef CAS; (e) Z. Yuan, Y. Shu, Y. Tian, Y. Xin and B. Ma, Chem. Commun., 2015, 51, 16385 RSC; (f) K.-H. Lee, J.-H. Lee, W.-S. Song, H. Ko, C. Lee, J.-H. Lee and H. Yang, ACS Nano, 2013, 7, 7295 CrossRef CAS PubMed.
- (a) H. Xiao, L. Ding, D. Ruan, B. Li, N. Ding and D. Ma, Dyes Pigm., 2015, 121, 7 CrossRef CAS; (b) G. Schwartz, M. Pfeiffer, S. Reineke, K. Walzer and K. Leo, Adv. Mater., 2007, 19, 3672 CrossRef CAS; (c) S. J. Lee, J. S. Park, K.-J. Yoon, Y.-I. Kim, S.-H. Jin, S. K. Kang, Y.-S. Gal, S. Kang, J. Y. Lee, J.-W. Kang, S.-H. Lee, H.-D. Park and J.-J. Kim, Adv. Funct. Mater., 2008, 18, 3922 CrossRef CAS; (d) L. S. Hung and C. H. Chen, Mater. Sci. Eng., R, 2002, 39, 143 CrossRef; (e) Z. Zhang, W. Jiang, X. Ban, M. Yang, S. Ye, B. Huang and Y. Sun, RSC Adv., 2015, 5, 29708 RSC.
- (a) M. A. Baldo, D. F. O'Brien, Y. You, A. Shoustikov, S. Sibley, M. E. Thompson and S. R. Forrest, Nature, 1998, 395, 151 CrossRef CAS; (b) H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Nature, 2012, 492, 234 CrossRef CAS PubMed; (c) Q. Zhang, B. Li, S. Huang, H. Nomura, H. Tanaka and C. Adachi, Nat. Photonics, 2014, 8, 326 CrossRef CAS; (d) Y. Tao, K. Yuan, T. Chen, P. Xu, H. H. Li, R. F. Chen, C. Zheng, L. Zhang and W. Huang, Adv. Mater., 2014, 26, 7931 CrossRef CAS PubMed.
- (a) W. Qin, Z. Yang, Y. Jiang, J. W. Y. Lam, G. Liang, H. S. Kwok and B. Z. Tang, Chem. Mater., 2015, 27, 3892 CrossRef CAS; (b) X. Tang, Q. Bai, Q. Peng, Y. Gao, J. Li, Y. Liu, L. Yao, P. Lu, B. Yang and Y. Ma, Chem. Mater., 2015, 27, 7050 CrossRef CAS; (c) L. Li, B. Jiao, Y. Yu, L. Ma, X. Hou and Z. Wu, RSC Adv., 2015, 5, 59027 RSC.
- (a) J.-Y. Hu, Y.-J. Pu, F. Satoh, S. Kawata, H. Katagiri, H. Sasabe and J. Kido, Adv. Funct. Mater., 2014, 24, 2064 CrossRef CAS; (b) M.-J. Kim, C.-W. Lee and M.-S. Gong, Org. Electron., 2014, 15, 2922 CrossRef CAS; (c) C.-H. Chien, C.-K. Chen, F.-M. Hsu, C.-F. Shu, P.-T. Chou and C.-H. Lai, Adv. Funct. Mater., 2009, 19, 560 CrossRef CAS; (d) G. Mallesham, C. Swetha, S. Niveditha, M. E. Mohanty, N. J. Babu, A. Kumar, K. Bhanuprakash and V. J. Rao, J. Mater. Chem. C, 2015, 3, 1208 RSC; (e) W. Hua, Z. Liu, L. Duan, G. Dong, Y. Qiu, B. Zhang, D. Cui, X. Tao, N. Cheng and Y. Liu, RSC Adv., 2015, 5, 75 RSC; (f) W.-C. Chen, G.-F. Wu, Y. Yuan, H.-X. Wei, F.-L. Wong, Q.-X. Tong and C.-S. Lee, RSC Adv., 2015, 5, 18067 RSC.
- (a) Y. Shirota, M. Kinoshita, T. Noda, K. Okumoto and T. Ohara, J. Am. Chem. Soc., 2000, 122, 11021 CrossRef CAS; (b) H. Doi, M. Kinoshita, K. Okumoto and Y. Shirota, Chem. Mater., 2003, 15, 1080 CrossRef CAS.
- Z. R. Grabowski, K. Rotkiewicz and W. Rettig, Chem. Rev., 2003, 103, 3899 CrossRef PubMed.
- (a) W. Li, D. Liu, F. Shen, D. Ma, Z. Wang, T. Feng, Y. Xu, B. Yang and Y. Ma, Adv. Funct. Mater., 2012, 22, 2797 CrossRef CAS; (b) S. Zhang, W. Li, L. Yao, Y. Pan, F. Shen, R. Xiao, B. Yang and Y. Ma, Chem. Commun., 2013, 49, 11302 RSC; (c) W. Li, Y. Pan, R. Xiao, Q. Peng, S. Zhang, D. Ma, F. Li, F. Shen, Y. Wang, B. Yang and Y. Ma, Adv. Funct. Mater., 2014, 24, 1609 CrossRef CAS; (d) Y. Pan, W. Li, S. Zhang, L. Yao, C. Gu, H. Xu, B. Yang and Y. Ma, Adv. Opt. Mater., 2014, 2, 510 CrossRef CAS; (e) W. Li, Y. Pan, L. Yao, H. Liu, S. Zhang, C. Wang, F. Shen, P. Lu, B. Yang and Y. Ma, Adv. Opt. Mater., 2014, 2, 892 CrossRef CAS; (f) L. Yao, S. Zhang, R. Wang, W. Li, F. Shen, B. Yang and Y. Ma, Angew. Chem., Int. Ed., 2014, 53, 2119 CrossRef CAS PubMed; (g) S. Zhang, L. Yao, Q. Peng, W. Li, Y. Pan, R. Xiao, Y. Gao, C. Gu, Z. Wang, P. Lu, F. Li, S. Su, B. Yang and Y. Ma, Adv. Funct. Mater., 2015, 25, 1755 CrossRef CAS; (h) L. Yao, Y. Pan, X. Tang, Q. Bai, F. Shen, F. Li, P. Lu, B. Yang and Y. Ma, J. Phys. Chem. C, 2015, 119, 17800 CrossRef CAS; (i) H. Liu, Q. Bai, L. Yao, H. Zhang, H. Xu, S. Zhang, W. Li, Y. Gao, J. Li, P. Lu, H. Wang, B. Yang and Y. Ma, Chem. Sci., 2015, 6, 3797 RSC.
- D. Fan, Y. Yi, Z. Li, W. Liu, Q. Peng and Z. Shuai, J. Phys. Chem. A, 2015, 119, 5233 CrossRef CAS PubMed.
- X. Ouyang, X.-L. Li, X. Zhang, A. Islam, Z. Ge and S.-J. Su, Dyes Pigm., 2015, 122, 264 CrossRef CAS.
- Y. Zhang, S.-L. Lai, Q.-X. Tong, M.-F. Lo, T.-W. Ng, M.-Y. Chan, Z.-C. Wen, J. He, K.-S. Jeff, X.-L. Tang, W.-M. Liu, C.-C. Ko, P.-F. Wang and C.-S. Lee, Chem. Mater., 2012, 24, 61 CrossRef.
- J. Luo, Z. Xie, J. W. Y. Lam, L. Cheng, B. Z. Tang, H. Chen, C. Qiu, H. S. Kwok, X. Zhan, Y. Liu and D. Zhu, Chem. Commun., 2001, 1740 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra15079a |
| This journal is © The Royal Society of Chemistry 2016 |