Stereoselective oxidation of alkanes with m-CPBA as an oxidant and cobalt complex with isoindole-based ligands as catalysts†
Oksana V. Nesterova ,
Maximilian N. Kopylovich and
Dmytro S. Nesterov
,
Maximilian N. Kopylovich and
Dmytro S. Nesterov *
*
Centro de Química Estrutural, Instituto Superior Técnico, Universidade de Lisboa, Av. Rovisco Pais, 1049-001 Lisboa, Portugal. E-mail: dmytro.nesterov@tecnico.ulisboa.pt
First published on 26th September 2016
Abstract
Two complexes with isoindole-core ligands of general formula [M{C6H4C(NH2)NC(ONCMe2)2}2](NO3)2 (M = Co for 1 and M = Ni for 2) were studied as catalysts for the mild stereoselective alkane oxidation with m-chloroperbenzoic acid (m-CPBA) as an oxidant and cis-1,2-dimethylcyclohexane (cis-1,2-DMCH) as a main model substrate. Complex 1 disclosed a pronounced activity, with high retention of stereoconfiguration of substrates (>98% for cis-1,2-DMCH) and highest cis/trans ratio of tertiary alcohols (products) of 56, under mild conditions. The best achieved yields of tertiary cis-alcohols were of 13.7 and 50.5%, based on the substrate (cis-1,2-DMCH) and the oxidant (m-CPBA) respectively. Kinetic experiments, high bond and stereoselectivity parameters, kinetic isotope effect of 7.2(2) in the oxidation of cyclohexane, and incorporation of 18O from H218O support the involvement of CoIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O high-valent metal–oxo intermediates as main C–H attacking species.
O high-valent metal–oxo intermediates as main C–H attacking species.
Introduction
Alkanes are cheap and available raw materials for the synthesis of valuable chemicals.1 However, due to the inertness of C–H and C–C bonds, the modification of alkanes generally requires strong activators (e.g. oxidizing agents) and harsh conditions. This usually leads to low chemo-, regio- and/or stereoselectivities and tedious isolation and purification of desired isomers.2 Moreover, a synthetic approach may fail completely if the substrate contains impurities, as usually occurs in real raw materials. As a result, while some particular protocols for direct selective functionalization of C–H bond have already contributed to fine organic synthesis,3 more general approaches to the selective C–H activation in alkanes remain a challenge.Elaboration of effective catalytic systems is a common approach, and it has long been known that various alkanes can be selectively hydroxylated under relatively mild conditions using peroxides as oxidants and coordination compounds as catalysts.4 The studies in this field were inspired by impressive effectivity of such enzymes as cytochrome P450,5 Rieske dioxygenase6 or methane monooxygenase (MMO),7 able to insert oxygen into C–H bond with exciting stereoselectivity under ambient conditions.6 Active centres of the enzymes usually contain transition metals (for instance, iron in the case of P450 or Rieske dioxygenase), which facilitate the alkane oxidation via formation of e.g. metal–oxo high-valent species.8 Attempts to simulate the mechanism of enzymatic activation led to development of a number of catalytic systems,4a e.g. with iron and manganese complexes as catalysts and peroxides as terminal oxidants.6,9 Although some of the man-made catalytic systems exhibit high selectivity, only few of them are suitable for practical applications for fine organic synthesis,10 mainly due to low yields of products and instability of the catalysts. Hence, new effective catalytic systems for the direct selective transformation of alkanes continue to be actively searched for.
Apart from the catalyst per se, an oxidant and activation additives are essential components of the catalytic systems, and m-chloroperbenzoic acid (m-CPBA)11 is a renown terminal oxidant for the alkanes modification with enhanced selectivity, as was firstly demonstrated by using iron12 catalysts. Later on, an intriguing activity of cobalt complexes in a stereospecific hydroxylation of alkanes with m-CPBA has been also reported.13 However, all the above mentioned systems possess rather low yields of products based on alkane substrate (typically less than 2%), while this parameter is critical for most of practical applications.
We decided address this point, and herein we report on the catalytic activity of cobalt complex with isoindole-based ligands in the selective hydroxylation of various alkanes, mainly substituted cycloalkanes, with m-CPBA as an oxidant. The studied here cobalt-based catalytic system allowed to reach yields of products up to 16%, based on the alkane substrate, with high stereo and bond selectivity. A nickel complex with the same ligand was also tried for comparative purposes, but demonstrated much lesser activity.
Experimental
Materials and methods
The chemicals (Aldrich) were of AR reagent grade and used as received. All experiments were carried out in air. UV-vis spectra were recorded using Perkin-Elmer Lambda 35 spectrometer in a 200–800 nm spectral range. Mass spectra were obtained using LCQ Fleet mass spectrometer with an ESI source (Thermo Scientific). Coordination cobalt(II) and nickel(II) compounds with isoindole-core ligands [M{C6H4C(NH2)NC(ONCMe2)2}2](NO3)2 (M = Co for 1 and M = Ni for 2, Fig. 1) were prepared according to the published protocols,14 by refluxing the mixture of cobalt/nickel nitrate hexahydrate M(NO3)2·6H2O, phthalonitrile and acetoxime in acetone for 8 h. The complexes precipitate from reaction mixture, and their isolation involves simple filtration with subsequent washing. The Co(II) complex 1 was almost exclusively used in all the described here studies since the nickel catalyst 2 has not demonstrated appreciable activity in the studied transformations.Catalytic oxidation of alkanes
The reactions were typically carried out in air in thermostated cylindrical vials with vigorous stirring. Firstly, acetonitrile solution of CH3NO2 (used as GC internal standard of 0.2 M final concentration) was mixed with the solid complex 1, and then liquid substrate (alkane) was added. In the case of solid adamantane, it was placed into the vial prior the addition of acetonitrile solution. Solid m-CPBA oxidant was dissolved in acetonitrile (typically 30 mg in 1 mL of CH3CN) and added dropwise within 10 seconds to a warm (40 °C) solution of other components (CAUTION: the combination of m-CPBA with organic compounds at elevated temperatures may be explosive!). Samples were quenched at room temperature with excess of solid PPh3 according to developed methods15 and directly analysed by GC and GC-MS techniques.Gas chromatography
A Perkin-Elmer Clarus 600 gas chromatograph, equipped with two non-polar capillary columns (SGE BPX5; 30 m × 0.32 mm × 25 μm), one having an EI-MS (electron impact) detector and the other one with a FID detector, was used for analyses of the reaction mixtures. The following GC sequence has been used: 50 °C (3 min), 50–120 °C (8 degrees per minute), 120–300 °C (35 degrees per minute), 300 °C (3.11 min), 20 min total run time; 200 °C injector temperature. For analysis of oxidation products of highly-boiling compounds, such as adamantane or decahydronaphthalene, different conditioning was employed: 50 °C (3 min), 50–150 °C (30 degrees per minute), 150–300 °C (14 degrees per minute), 300 °C (2.95 min), 20 min total run time; 200 °C injector temperature. Helium was used as the carrier gas with a constant 1 mL per minute flow. All EI-MS spectra were recorded with 70 eV energy.Experiments with H218O
The 18O-labelled water (97% of 18O, Sigma-Aldrich) was added prior the addition of m-CPBA. The 16O/18O compositions of oxygenated products (tertiary cis- and trans-alcohols formed from the cis-1,2-dimethylcyclohexane substrate) were determined by the relative abundances of mass peaks at m/z = 128/130.Mass-spectrometry
The identification of peaks at chromatograms was made by comparison of respective EI mass-spectra with those from the NIST v.2.0f database (PerkinElmer TurboMass v.5.4.2.1617 software). The mass-spectra patterns of some products (tertiary cis- and trans-1,2-dimethylcyclohexanoles, tertiary cis- and trans-1,4-dimethylcyclohexanoles) were not found in the NIST database, and identification of these products was made by comparing with the reported previously mass-spectra.16Results and discussion
Synthesis and crystal structures of complexes 1 and 2
The preparation of analogous Co(II) and Ni(II) complexes 1 and 2 with two O,O′-(3-amino-1H-isoindole-1,1-diyl)bis(propan-2-one oxime) ligands (Fig. 1) is straightforward and involves cheap and available starting materials (phthalonitrile, acetoxime and metal nitrate).14The overall structural configurations of 1 and 2 are similar (Fig. 1 and 2) and will be presented on the example of the complex 1. The single crystal X-ray analysis showed that 1 consists of [Co{C6H4C(NH2)NC(ONCMe2)2}2]2+ (Fig. 2) and NO3− ions joined together by strong hydrogen bonds into extended supramolecular chains. The cobalt(II) atoms in 1 lie on the centre of symmetry and have slightly distorted octahedral coordination environments with a N6 donor sets formed by nitrogen atoms from isoindole-core ligands. The Co–N distances are in the range from 2.070(5) to 2.186(5) Å. The cis N–Co–N bond angles vary from 75.54(19) to 104.46(19)°. The strong H-bonds, N–H⋯O, involving nitrogen atoms from the amino groups of polydentate ligands of the cations and oxygen atoms from the uncoordinated nitrates [N4–H1⋯O4; D⋯A = 2.925(6) Å, D–H⋯A = 166.43(1)°; N4–H2⋯O3; D⋯A = 2.801(4) Å, D–H⋯A = 146.24(1)°] are responsible for the formation of the overall 1D structure (Fig. 3 and 4).
 | ||
| Fig. 2 The crystal structure of [Co{C6H4C(NH2)NC(ONCMe2)2}2]2+ in 1, showing the atom numbering. H atoms are omitted for clarity. | ||
 | ||
| Fig. 4 The packing of the supramolecular chains in 1 viewed down the c axis. Colour scheme: Co, pink; O, red; N, blue; C, grey. | ||
The bulky ligand structure in 1 prevents the formation of the hydrogen bonds between adjacent chains making impossible the further increasing of the overall dimensionality of the supramolecular polymer (Fig. 3). The shortest Co⋯Co separation within the H-bonded chain is 11.168(2) Å, while the nearest Co⋯Co distance between chains is 9.335(2) Å.
The complexes are stable in solid state but slowly degrade upon their dissolution in some solvents, e.g. alcohols, transforming into more stable phthalocyanines and related products.17 The instability is an obvious drawback of a catalyst, which, however, can be avoided by immobilization or inclusion onto/into a support,18 if significant improvement of a catalytic process is recognized.
The electrospray mass-spectra (ESI-MS) of acetonitrile solutions of 1 revealed few peaks, the strongest of which were identified as dication of 1 and its derivatives having one coordinated nitrate anion (Fig. S1†). The latter suggests the possibility of partial decoordination of isoindole-based ligand to bind nitrate anion. The UV/vis spectrum of the solution of 1 in acetonitrile exhibits negligible absorption in visible region (λ > 400 nm), but showing three clear absorption peaks at 323, 299 and 276 nm, as well as one shoulder at 249 nm (Fig. 5), in the UV region. The isoindole19 and its derivatives20 are known to exhibit numerous characteristic absorption peaks in the 250–340 nm range, depending on substituents. Therefore, the bands observed for the complex 1 (Fig. 5) can be attributable to transitions within the electronic structure of the ligand, coordinated to the metal centre.
The UV absorption exhibits a slight intensity increase within the time (Fig. 5, inset). No isosbestic points and no shifts of absorption peaks were observed during the 50 min time period. Further, no peaks appeared in the visible range (400–800 nm) within the time period studied, while it is known that acetonitrile complexes of cobalt absorb at 613 and 679 nm.21 From these observations we suppose that the isoindole ligands remain coordinated to the cobalt centre and no “free” cobalt ions appeared in solution. However, one cannot exclude the presence of reversible decoordination–coordination of the oxime groups of the ligands.
Catalytic oxidation of alkanes with m-CPBA
The selective oxidation of C–H bond should avoid formation of long-living radicals, a subject of epimerization (Scheme 1), and hence the insertion of oxygen should occur shortly after abstraction of hydrogen atom (radical formation), as it happens in the oxygen-rebound mechanism of the P450 enzyme5 and many model complexes.22 In this view, oxidation of cis-1,2-dimethylcyclohexane (cis-1,2-DMCH, Scheme 1) is a useful model reaction to test both the stereo- and bond selectivities.23 Retention of stereoconfiguration of methyl groups in cis-1,2-DMCH indicates an absence of the long-living alkyl radicals,24 emerging from the hydrogen abstraction by poorly selective O-centred radicals (for example, hydroxyl, arylperoxyl or tert-butoxyl ones), which in their turn arise from the oxidants (H2O2, m-CPBA, tBuOOH, etc.).In accordance with the above considerations, we studied the oxidation of cis-1,2-DMCH with m-CPBA, catalysed by 1 (Scheme 2), and found the tertiary (3°) cis-alcohol to be the main product. The by-products are tertiary trans-alcohol and oxygenates (ketones) emerged through the activation of secondary (2°) C–H bonds of cis-1,2-DMCH. As known,13b in such a type of chemistry ketones usually appear as result of overoxidation of secondary alcohols, hence, we assume that the ketones are formed via the respective alcohols (Scheme 2), rather than by direct oxidation of C–H bonds of the alkane.
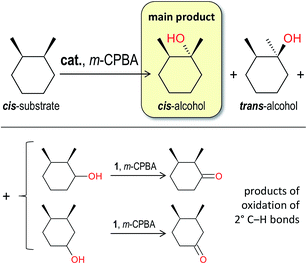 | ||
| Scheme 2 The oxidation of cis-1,2-DMCH substrate leads to tertiary cis-(retention) and trans-(inversion) alcohols (main products), as well as the products of 2° C–H bonds oxidation (mainly ketones). | ||
Accumulation of the tertiary alcohols and their cis/trans ratio in time upon the oxidation of cis-1,2-DMCH with m-CPBA, catalysed by 1, are shown in Fig. 6. As can be seen, lower initial concentration of 1 ([1]0) of 3.3 × 10−4 M (Fig. 6, blue squares) leads to better yields and the cis/trans selectivities (Fig. 6, bottom); in this case the retention of stereoconfiguration (RC) was found to be >98% (Fig. 6, bottom, inset). The best achieved yields of tertiary cis-alcohol were 13.7 and 50.5% based on the substrate and oxidant, respectively. The dependence of the initial reaction rate W0 on the concentration of 1 was found to be linear (Fig. 6, top, inset), pointing out that the mechanism of action of the complex 1 is the same within the studied concentration range. The maximum achieved initial reaction rate W0 of 1.2 × 10−4 M s−1 corresponds to turnover frequency, TOF, of 430 h−1.
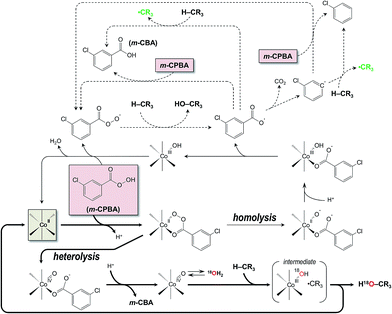
Chlorobenzene is a by-product formed by the homolytic splitting of the O–O bond of m-CPBA and decarboxylation of the m-chlorobenzoate radical (Scheme 3, top).25 The rate of decarboxylation (ca. 106 s−1) is known25a,26 to be comparable to that for H abstraction by the m-chlorobenzoate radical. Hence, one can assume that half of m-chlorobenzoate is finally transformed into chlorobenzene while another half reacts with the alkane substrate. As result, a simple estimation of the homolysis contribution to the overall catalytic process can be achieved. For example, the reported considerable amounts of chlorobenzene (more than 50% based on the products) formed upon the oxidation of cycloalkanes with m-CPBA, catalysed by pyridylalkylamine complexes of cobalt,27 nickel28 and ruthenium,29 reveal a homolytic mechanism in the respective process.
In contrast to the above cases, the oxidation catalysed by 1 reveals much lower amount of chlorobenzene, not exceeding 3.3% based on m-CPBA and 8% based on cis-1,2-DMCH. The accumulation curves of chlorobenzene for the tests with [1]0 = 3.3 × 10−4 and 1 × 10−3 M (Fig. 7) show that in both cases nearly the same concentrations of chlorobenzene are reached, ca. 8.7 × 10−4 M, but with slightly different accumulation rates WPhCl. The dependence of WPhCl on [1]0 is close to linear (Fig. 6, inset), figuring out the catalyst 1 as an origin of the chlorobenzene formation. From these data, as well as from the high retention index (cis/trans ratio > 50, RC > 98%, Fig. 6, bottom), we assume that the main reaction pathway proceeds via the heterolytic cleavage of the O–O bond of coordinated m-CPBA with formation of a high-valent cobalt–oxo species (Scheme 3, middle-bottom part). Coordination of m-CPBA and the following formation of cobalt–oxo species (Scheme 3) require the presence of two free sites in the coordination sphere of cobalt atom in 1. From the structure of the complex 1 (Fig. 1 and 2) one may suppose these could appear through the decoordination of two oxime groups from different ligand molecules (Fig. 2).
The introduction of 18O from H218O into hydroxylated products is an evidence of involvement of high-valent metal–oxo species, as they exchange oxygen with water.13a,30 To check this possibility, we performed the oxidation of cis-1,2-DMCH with m-CPBA catalysed by 1 in the presence of H218O (0.8 M). Incorporation of 18O into the tertiary cis- and trans-alcohols are depicted in Fig. 8, top. As can be seen, both tertiary alcohols reach 3.5% of 18O. The observed incorporation of the labelled oxygen in the final products (Fig. 8, top) is in accord with the proposed general mechanism with participation of high-valent cobalt–oxo species as oxidants (Scheme 3). The relatively low incorporation level of 3.5% can be explained by slow rate of the oxygen exchange between CoIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and H2O (Scheme 3), if compared to that of hydrogen abstraction from cis-1,2-DMCH. Cobalt high-valent metal–oxo intermediates are known to be hardly trapped (in contrast to, for example, iron–oxo species), what is, particularly, due to their high reactivity and, as a consequence, short lifetime.31 A similarly low incorporation of 18O into the cyclohexanol (5%) was observed earlier for oxidation of cyclohexane with m-CPBA, catalysed by cobalt porphyrin complex.13a
O and H2O (Scheme 3), if compared to that of hydrogen abstraction from cis-1,2-DMCH. Cobalt high-valent metal–oxo intermediates are known to be hardly trapped (in contrast to, for example, iron–oxo species), what is, particularly, due to their high reactivity and, as a consequence, short lifetime.31 A similarly low incorporation of 18O into the cyclohexanol (5%) was observed earlier for oxidation of cyclohexane with m-CPBA, catalysed by cobalt porphyrin complex.13a
When reaction is performed in the presence of D2O instead of H218O (other conditions are the same) the D-labelled alcohols are observed at ca. 20% level, nearly equal for cis- and trans-alcohols. This result is not unexpected as the alcohols normally undergo isotopic exchange with heavy water.
The accumulation of the main reaction product, tertiary cis-alcohol, was not affected by D2O. However, the overall cis/trans ratio was found to be twice lower (Fig. 9) due to enhanced amounts of the by-product – tertiary trans-alcohol. Similar effect was observed for H218O, but in that case both cis- and trans-products were affected, lowering the final stereopurity of the tertiary alcohols. Thus, the water has a suppression effect on the overall catalytic activity of the system, probably influencing the ratio of heterolytic/homolytic O–O splitting pathways towards the homolytic one (Scheme 3).
To distinguish the participation of the alkyl radicals in the oxidation of cis-1,2-DMCH (Scheme 3), we checked how the respective radical trap, CCl3Br, would influence the catalytic process. The radical trap concentration was equal to that of substrate (0.1 M), while [1]0 = 3.3 × 10−4 M and other conditions were as stated in Fig. 6 caption. When CCl3Br was added to reaction mixture before addition of the m-CPBA oxidant, the cis/trans ratio of tertiary alcohols after 1 h reaction time was lowered to 15, what is much lower comparing to that (56) without the addition of the radical trap (Fig. 6, bottom, blue squares). The yields of alcohols in the presence of CCl3Br were also lower (11% based on the m-CPBA; compare to 57% in the trap-free test). Formation of 2° products (Scheme 2) was almost suppressed, while a number of new products have been detected (Fig. 10).
The mass-spectra of the peaks I–IX (Fig. S2–S8†) were mainly not recognized by the NIST database, except of III and IV. Tentative considerations of the mass-spectra (Fig. S2 and S3†) allowed to assume that peaks I and II may be related to chlorinated C8 moieties, probably cis- and trans-1-chloro-1,2-dimethylcyclohexanes. The peak III, that overlaps with the weak group of 2° products (Fig. 10), was recognized as 1,2-dimethylcyclohexene. Formation of a double bond is possible from the tertiary alkyl radical, attacked at the second tertiary carbon.32 3-Bromochlorobenzene (IV) was also detected, as expected from interaction of aryl radical (Scheme 3) with CCl3Br. The strong peaks V–IX with similar mass-spectra (Fig. S4–S8†) were assigned to be the isomers of brominated dimethylcyclohexanes, where V and VI are tertiary cis- and trans-1-bromo-1,2-dimethylcylohexanes, respectively, and VII–IX are secondary cis-1-bromo-2,3- and cis-1-bromo-3,3-domethylcyclohexanes (Fig. 10).
The generally accepted mechanism for hydrogen abstraction with a high-valent metal–oxo species (as in some iron containing enzymes, such as P450) proceeds via formation of a short-living alkyl radical, ˙CR3.22 This radical immediately interacts with the same M–O centre to give the final hydroxylated product (Scheme 3). The high rate of this process is the origin of stereoselectivity of a whole process, because the radical collapses before its epimerization occurs. However, some catalysts operating via the high-valent metal–oxo species are known to form large quantities of brominated alkanes when reactions are performed in the presence of CCl3Br.22,33 This can be explained by interaction of CCl3Br either with alkyl radical inside the “transition state” (intermediate) or with a free alkyl radical in solution.
In case of the cis-1,2-DMCH/1/m-CPBA/CCl3Br catalytic system, the notable cis/trans ratio and yield of tertiary cis-alcohol retain even in the presence of CCl3Br. On the other hand, tertiary cis- and trans-brominated products V and VI are observed in a nearly equal quantities and the ratio of 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° brominated products (assuming V + VI as tertiary and VII + VIII + IX as secondary ones) is low, close to 4
2° brominated products (assuming V + VI as tertiary and VII + VIII + IX as secondary ones) is low, close to 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, while the system cis-1,2-DMCH/1/m-CPBA exhibits 3°
1, while the system cis-1,2-DMCH/1/m-CPBA exhibits 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° ratio of alcohols of 30
2° ratio of alcohols of 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 or higher. Moreover, the overall yield of products (sum of hydroxylated and brominated alkanes) as well as conversion of the cis-1,2-DMCH substrate are ca. 20% higher when reaction is performed in the presence of CCl3Br. From all these data we assume that reaction of cobalt-alkyl radical intermediate with CCl3Br (Scheme 4) results in release of a free ˙CCl3 radical. The latter can abstract hydrogen from alkane substrate,26 forming CHCl3 (detected by chromatography) and a free alkyl radical. This explains the absence of stereoselectivity in the brominated tertiary products as well as the elevated overall yields (sum of hydroxylated and brominated 1,2-DMCH) in the presence of CCl3Br. It also can be presupposed that alcohols are formed through the intermediates (ca. 20%), which did not react with CCl3Br.
1 or higher. Moreover, the overall yield of products (sum of hydroxylated and brominated alkanes) as well as conversion of the cis-1,2-DMCH substrate are ca. 20% higher when reaction is performed in the presence of CCl3Br. From all these data we assume that reaction of cobalt-alkyl radical intermediate with CCl3Br (Scheme 4) results in release of a free ˙CCl3 radical. The latter can abstract hydrogen from alkane substrate,26 forming CHCl3 (detected by chromatography) and a free alkyl radical. This explains the absence of stereoselectivity in the brominated tertiary products as well as the elevated overall yields (sum of hydroxylated and brominated 1,2-DMCH) in the presence of CCl3Br. It also can be presupposed that alcohols are formed through the intermediates (ca. 20%), which did not react with CCl3Br.
Apart from cis-1,2-DMCH, the selective oxidation of other alkane substrates was performed (Tables 1–3), and it was found that generally the stereospecificity and bond selectivity of the 1/m-CPBA catalytic system depends on the spatial configuration of substrate. The 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° bond selectivities vary from 11
2° bond selectivities vary from 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 35
1 to 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
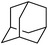
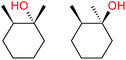
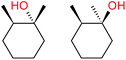
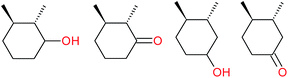
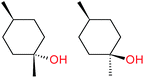
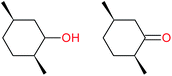
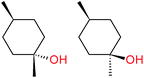
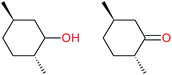
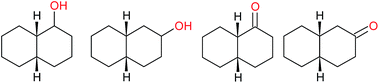
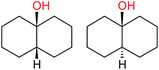
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, being also dependent on the configuration of alkane skeleton (Table 1). The highest cis/trans ratio of tertiary alcohols is observed for cis-1,2-DMCH (Table 1, a), while cis-1,4-DMCH was found to show the lowest retention of stereoconfiguration (Table 1, c). A tentative explanation is the close proximity of an electron rich tertiary carbons in cis-1,2-DMCH, that may facilitate hydrogen abstraction. A weak regioselectivity between different methylene groups of a linear alkane, n-heptane, was observed (Table 2): while the C–H bond dissociation energies of (2), (3) and (4) methylene groups of n-heptane (Table 2) are nearly equal,37 the 1/m-CPBA system was shown to be slightly more active towards oxidation of (2) and (3) methylene groups, rather than (4) ones (Table 2). Oxidation of adamantane under similar conditions (Table 3) revealed 21
1, being also dependent on the configuration of alkane skeleton (Table 1). The highest cis/trans ratio of tertiary alcohols is observed for cis-1,2-DMCH (Table 1, a), while cis-1,4-DMCH was found to show the lowest retention of stereoconfiguration (Table 1, c). A tentative explanation is the close proximity of an electron rich tertiary carbons in cis-1,2-DMCH, that may facilitate hydrogen abstraction. A weak regioselectivity between different methylene groups of a linear alkane, n-heptane, was observed (Table 2): while the C–H bond dissociation energies of (2), (3) and (4) methylene groups of n-heptane (Table 2) are nearly equal,37 the 1/m-CPBA system was shown to be slightly more active towards oxidation of (2) and (3) methylene groups, rather than (4) ones (Table 2). Oxidation of adamantane under similar conditions (Table 3) revealed 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 normalized 3°
1 normalized 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° bond selectivity. This value is slightly higher than that observed for oxidation of adamantane with m-CPBA catalysed by nickel and cobalt complexes (Table 4).
2° bond selectivity. This value is slightly higher than that observed for oxidation of adamantane with m-CPBA catalysed by nickel and cobalt complexes (Table 4).
| Substrate | Products 3° | Products 2° | cis/trans | |
|---|---|---|---|---|
3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° 2° |
||||
| Yieldb (%) | ||||
| a [Alkane]0 = 0.1 M, [m-CPBA]0 = 0.027 M, [1]0 = 3.3 × 10−4 M in CH3CN at 40 °C, 1 h reaction time.b Yields based on the substrate (sum of 3° and 2° products).c Secondary products peaks are overlapped by a large peak of m-chlorobenzoic acid (m-CBA). | ||||
| (a) |  |
 |
 |
56 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
| 16 | ||||
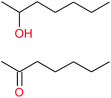
| (b) | 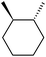 |
 |
 |
28−1 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
| 11 | ||||
| (c) | 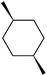 |
 |
 |
11 |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
| 9 | ||||
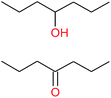
| (d) | 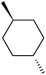 |
 |
 |
20−1 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
| 11 | ||||
| (e) |  |
 |
 |
46 |
| —c | ||||
| 19 | ||||
| (f) | 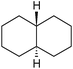 |
 |
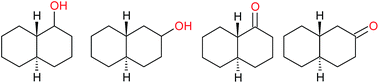 |
40−1 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
||||
| 12 | ||||
| Catalyst | Substrate | Yielda, % | TONb | cis/trans ratioc | 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° ratio 2° ratio |
Ref. | |
|---|---|---|---|---|---|---|---|
| a Yields based on the substrate (sum of 3° and 2° products).b Turnover numbers, moles of products (sum of 3° and 2° products) per mol of catalyst.c Moles of tertiary cis-alcohol per mol of trans-alcohol. H2L1 = salicylidene-2-ethanolamine. L2 = meso-tetrakis(pentafluorophenyl)porphinato dianion. L3 = meso-tetrakis(2,6-difluorophenyl)porphinato dianion. L4 = N,N-dimethyl-N′,N′-bis(quinolin-2-ylmethyl)ethane-1,2-diamine. L5 = N,N-dimethyl-N′,N′-bis(pyrid-2-ylmethyl)ethane-1,2-diamine. L6 = N-methyl-N,N′,N′-tris(pyrid-2-ylmethyl)-ethylenediamine. L7 = tris(2-pyridylmethyl)amine. | |||||||
| 1 | 1 | cis-1,2-DMCH | 15.5 | 46 | 56 | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— |
| 2 | [Fe(L2)](ClO4) | cis-1,2-DMCH | 1.5 | 15 | >55 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 3 | [Mn(L2)](ClO4) | cis-1,2-DMCH | 1.1 | 11 | >40 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 4 | [Co(L2)](ClO4) | cis-1,2-DMCH | 1.3 | 13 | >60 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 5 | Co(ClO4)2 | cis-1,2-DMCH | 1.8 | 18 | >81 | 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 6 | Fe(ClO4)2 | cis-1,2-DMCH | 0.3 | 3 | >10 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 7 | Mn(ClO4)2 | cis-1,2-DMCH | 0.7 | 7 | >25 | 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13b |
| 8 | [Fe(L3)Cl] | cis-1,2-DMCH | 0.3 | 1 | >72 | 58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
12a |
| 9 | 1 | Adamantane | 18.9 | 57 | — | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— |
| 10 | [Co(L2)](SF3CO3) | Adamantane | <1 | 3.1 | — | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
13a |
| 11 | [Ni(L4)(CH3CN)2](BPh4)2 | Adamantane | 29.2 | 584 | — | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
34 |
| 12 | [Ni(L5)(CH3CN)2](BPh4)2 | Adamantane | 32.0 | 639 | — | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
34 |
| 13 | [Ni(L6)(CH3CN)](BPh4)2 | Adamantane | 29.8 | 595 | — | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
35 |
| 14 | [Ni(L7)(OAc)(H2O)]BPh4 | Adamantane | 4.2 | 320 | — | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
36 |
The parameters of the stereoselective oxidation of cis-1,2-DMCH (cis/trans ratio and yields of products) exhibited by the catalyst 1 are among the highest ones reported to date (Table 4). Additionally, the complex 1 is able to perform stereoselective oxidation of alkanes without any promoters. A series of porphyrin complexes (2–4, 8, Table 4) reveal pronounced retention of stereoconfiguration, but the reported yields of products are close to 1%, much lower to that reported here. The same yields of products were observed if metal salts (5–7, Table 4) were tested as catalysts. Although cobalt perchlorate exhibited the bond selectivity and cis/trans ratio better than those for the complex 1, the overall yields of products did not exceed 2%. In the oxidation of adamantine, the yields provided by the catalyst 1 are moderate but comparable to those with nickel catalysts with tripodal N-donor ligands (11–14, Table 4), although showing better 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° selectivity. Apart from that, the catalytic systems based on complexes 11–14 (Table 4) produce large amounts of undesirable chlorobenzene by-product (comparable to those of main products), while the 1/m-CPBA catalytic system allows to eliminate this drawback.
2° selectivity. Apart from that, the catalytic systems based on complexes 11–14 (Table 4) produce large amounts of undesirable chlorobenzene by-product (comparable to those of main products), while the 1/m-CPBA catalytic system allows to eliminate this drawback.
Competitive oxidation of equimolar mixture of normal and deuterated cyclohexane was performed to determine the kinetic isotope effect (KIE) value ([C6H12]0 = [C6D12]0 = 0.1 M, [1]0 = 3.3 × 10−4 M, other conditions were as in stated in Fig. 6 caption). The accumulations of both normal and deuterated products (sums of respective alcohol and ketone) exhibit linear character up to 4 min reaction time (Fig. 11, inset). The ratio of their slopes, after linearization, gives KIE value of 7.0(3). However, non-linear dependences of cyclohexanols and cyclohexanones accumulation (Fig. 11) suggest that the consecutive reaction Cy → Cy–OH → Cy![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O takes place (where Cy means cyclohexane). In this model, reaction rate constants k1 and k2 describe oxidation of cyclohexane and cyclohexanol, respectively. The data in the time region from 0 to 4 min (Fig. 11) were fitted to analytical expression,40 derived for such kinetic process (see the ESI†). The fitting procedure gave k1H = 3.14(6) × 10−4, k2H = 7.9(3) × 10−3, k1D = 4.4(1) × 10−5 and k2D = 2.6(2) × 10−3 s−1. From these values one can see that oxidation of the alcohols (k2 constants) occurs 25 and 58 times faster than oxidation of the alkanes (k1 constants), for C6H12 and C6D12, respectively, leading to a large range of alcohol/ketone (A/K) ratios (Fig. 12). From these data the KIE kH/kD values for oxidation of cyclohexane and cyclohexanol can be estimated as 7.2(2) and 3.1(2), respectively. Such a KIE values suggest that (1) hydrogen abstraction is a rate-determining step and (2) cyclohexanol is oxidized via abstraction of hydrogen atom of carbon rather than hydroxyl group. The KIE k1H/k1D of 7.2(2) supports the proposed reaction pathway that involves high-valent cobalt–oxo species (Scheme 3). However, at the same time, such a high KIE value does not rule out the possibility of radicals as attacking species, particularly metal–oxo radicals or selective O-centred radicals, such as aroylperoxyl ones.26
O takes place (where Cy means cyclohexane). In this model, reaction rate constants k1 and k2 describe oxidation of cyclohexane and cyclohexanol, respectively. The data in the time region from 0 to 4 min (Fig. 11) were fitted to analytical expression,40 derived for such kinetic process (see the ESI†). The fitting procedure gave k1H = 3.14(6) × 10−4, k2H = 7.9(3) × 10−3, k1D = 4.4(1) × 10−5 and k2D = 2.6(2) × 10−3 s−1. From these values one can see that oxidation of the alcohols (k2 constants) occurs 25 and 58 times faster than oxidation of the alkanes (k1 constants), for C6H12 and C6D12, respectively, leading to a large range of alcohol/ketone (A/K) ratios (Fig. 12). From these data the KIE kH/kD values for oxidation of cyclohexane and cyclohexanol can be estimated as 7.2(2) and 3.1(2), respectively. Such a KIE values suggest that (1) hydrogen abstraction is a rate-determining step and (2) cyclohexanol is oxidized via abstraction of hydrogen atom of carbon rather than hydroxyl group. The KIE k1H/k1D of 7.2(2) supports the proposed reaction pathway that involves high-valent cobalt–oxo species (Scheme 3). However, at the same time, such a high KIE value does not rule out the possibility of radicals as attacking species, particularly metal–oxo radicals or selective O-centred radicals, such as aroylperoxyl ones.26
Similar KIE value of 6.8(4) was obtained when twice lower concentrations of C6H12 and C6D12 were used (0.05 M each): k1H = 1.31(5) × 10−4, k2H = 7.0(4) × 10−3, k1D = 1.93(9) × 10−5 and k2D = 2.3(3) × 10−3 s−1 (Fig. S9 and S10†). The dependences of k1 rate constants values on the cyclohexane concentration are linear for both normal and deuterated cyclohexane (Fig. 12, inset). In contrast, the k2 constants (oxidation of cyclohexanol to cyclohexanone) are nearly the same as for 0.1 M test (see above). These results are in agreement with the model where oxidation of alkane is a limiting step and ketone is formed through the alcohol oxidation.
A comparable value of 8(1) of KIE k1H/k1D was observed earlier for oxidation of alkanes with m-CPBA, catalysed by cobalt salts and cobalt porphyrins (2 and 3, Table 5) with the yields of products up to 3%. The yields of oxygenates (sum of alcohol and ketone) exhibited by the system 1/m-CPBA in the oxidation of C6H12 (0.1 M) did not reach 5%. This is comparable to the yields of secondary products (up to 1.6%), observed in the course of cis-1,2-DMCH oxidation (Tables 1 and 4) and other substrates containing tertiary C–H bonds, showing that the present system is not much active towards activation of secondary C–H bonds.
| Catalyst | KIEa | A/Kb | ε-Cc | Ref. | |
|---|---|---|---|---|---|
| a Kinetic isotope effect (kH/kD) in the oxidation of equimolar mixture of normal and deuterated cyclohexane.b Cyclohexanol/cyclohexanone ratio.c Detection of ε-caprolactone.d Not reported. L2 = meso-tetrakis(pentafluorophenyl)porphinato dianion; L3 = N,N-bis(2-pyridylmethyl)-N-[(1-tBu-1H-1,2,3-triazol-4-yl)methyl]amine; L4 = bis(4-ethoxycarbonyl-2-pyridylmethyl)(2-pyridylmethyl)amine; L5 = tris(2-pyridylmethyl)amine; L6 = tris[2-(2-pyridyl)ethyl]amine. L7 = N-benzyl-bis(2-pyridylmethyl)amine; L8 = N-methyl-N,N′,N′-tris(pyrid-2-ylmethyl)-ethylenediamine. L9 = 1,2-bis[2-di(2-pyridyl)-methyl-6-pyridyl]ethane. H2L10 = N,N′-o-phenylenebis(3,5-di-tert-butylsalicylideneimine). | |||||
| 1 | 1 | 7.2(2) | 0.4–2.8 | — | — |
| 2 | [Co(L2)](SF3CO3) | 8(1) | 4.9 | n.r.d | 13a |
| 3 | Co(ClO4)2 | 8(1) | 1.0 | n.r. | 13b |
| 4 | [Co(L2)](ClO4) | n.r. | 1.2 | n.r. | 13b |
| 5 | [Fe(L2)](ClO4) | n.r. | 13.0 | n.r | 13b |
| 6 | [Mn(L2)](ClO4) | n.r. | 6.3 | n.r. | 13b |
| 8 | [Co(L3)(OAc)](BPh4) | n.r. | 5.4 | + | 27 |
| 9 | [Ru(L4)Cl2](ClO4) | 4.3 | 0.2–2.6 | — | 29 |
| 10 | [Ru(L5)Cl2](ClO4) | 3.1 | 0.8–2.1 | n.r. | 29 |
| 11 | [Ni(L6)(NO3)](BPh4) | n.r. | 46.0 | + | 28 |
| 12 | [Ni(L7)(OAc)2(H2O)] | n.r. | 6.6 | + | 28 |
| 13 | [Ni(L5)(OAc)(H2O)](BPh4) | 2.8 | 8.5 | + | 36 |
| 14 | [Ni(L8)(CH3CN)](BPh4)2 | n.r. | 10.6 | + | 35 |
| 15 | [Fe2O(L9)(OAc)2](ClO4)2 | 3.2 | 2.4 | + | 38 |
| 16 | [Fe(L10)Cl] | n.r. | 1.7 | n.r. | 39 |
The alcohol/ketone (A/K) parameter, often used to characterize oxidative catalytic systems, ranges from 0.4 to 2.8 for C6H12 and from 1.5 to 7.1 for C6D12, depending on the reaction time (Fig. 12). Obviously, the A/K ratio tends to decrease with the time and, therefore, for the present catalytic system no definite A/K value can be derived. This behaviour differs from most of the reported catalytic systems for cyclohexane oxidation with m-CPBA, for which constant A/K values are reported (Table 5). Oxidation of pure cyclohexane (0.1 M) gave the same A/K ratios and yields of products, as the oxidation of C6H12/C6D12 mixture.
Formation of ε-caprolactone, appearing through the Baeyer–Villiger reaction of cyclohexanone with m-CPBA, was reported for many catalytic systems oxidizing cyclohexane with m-CPBA (Table 5), particularly for some cobalt complexes with pyridyl ligands (8, Table 5). In the case of 1/m-CPBA catalytic system no ε-caprolactone was detected (Fig. S12†).
Recently it was shown that cyclohexyl hydroperoxide, Cy–OOH, can be directly detected in a catalytic mixture using a non-polar GC column and characterized by mass-spectroscopy.41 Cyclohexyl hydroperoxide is formed by the reaction of a free cyclohexyl radical with O2 and is a marker for a free-radical reaction.15 In the present case, no Cy–OOH was detected in GC-MS chromatograms, even at trace levels (Fig. S12†), for both normal and deuterated cyclohexane oxidation (samples were injected without quenching by PPh3). The absence of Cy–OOH rules out the presence of free alkyl radicals in the 1/m-CPBA catalytic system. Therefore, brominated products, observed as a result of reaction in the presence of CCl3Br (see above, Fig. 10, Scheme 4), appear from the “caged” alkyl radicals, but not free ones. These results are in conformity with the proposed reaction mechanism, operating via a short-lived alkyl radicals (Schemes 3 and 4).
An uncommon feature of the catalytic system 1/m-CPBA was formation of chlorocyclohexane by-product as a result of the oxidation of C6H12 (Fig. S12†), C6D12 or equimolar mixture thereof. From the +11 m/z isotopic shifts of molecular ions in mass-spectra of normal and deuterated chlorocyclohexanes (Fig. S13†) one may conclude that the chlorocyclohexane is formed from the alkane substrate, but not via hydrogenation of chlorobenzene (Scheme 3). From the selected catalytic investigations (Table 5) there are two reporting formation of chlorocyclohexane (15 and 16) during cyclohexane oxidation with m-CPBA. However, in both cases the catalytic systems contain a source of chloride: the system 15 (Table 5) operates in a CH3CN/CH2Cl2 medium, while the catalyst 16 contains chloride anion in its structure. The latter was postulated to produce chlorinated product by oxidative ligand transfer mechanism,42 with long-lived alkyl radicals as intermediates. In the case of the catalyst 1 the only source of chlorine is m-CBPA or chlorobenzene by-product (Scheme 3, Fig. S11†).
In general, the combination of KIE and A/K values exhibited by the 1/m-CPBA catalytic system are closest to complexes of cobalt (2 and 3, Table 5) and ruthenium (9). For the cobalt catalysts 2 and 3 participation of high-valent cobalt–oxo intermediates was proposed, after 18O studies.13b Complex 9 was also presumed to oxidize via RuIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O/RuIII–O˙ species, on the basis of 18O labelling and Raman spectroscopy;29 this complex also did not produce ε-caprolactone. Coordination compound of ruthenium with slightly different structure (10, Table 5) was concluded to oxidize cyclohexane via a free-radical pathway.29 A series of nickel complexes 11–14 (Table 5) show common features, such as formation of ε-caprolactone. For the complex 14 participation of high-spin NiII–O˙ attacking species was postulated on the basis of DFT calculations.35
O/RuIII–O˙ species, on the basis of 18O labelling and Raman spectroscopy;29 this complex also did not produce ε-caprolactone. Coordination compound of ruthenium with slightly different structure (10, Table 5) was concluded to oxidize cyclohexane via a free-radical pathway.29 A series of nickel complexes 11–14 (Table 5) show common features, such as formation of ε-caprolactone. For the complex 14 participation of high-spin NiII–O˙ attacking species was postulated on the basis of DFT calculations.35
As mentioned above (11–14, Table 4; 11–14, Table 5), some complexes of nickel are active catalysts in the oxidation of alkanes with m-CPBA, thus, we also studied the catalytic activity of the isostructural to 1 nickel complex 2. The accumulation of tertiary cis-alcohol in the course of cis-1,2-DMCH (0.1 M) oxidation with m-CPBA (0.027 M) catalysed by the complex 2 (3.3 × 10−4 M) revealed a nearly linear dependence with a low reaction rate W = 1 × 10−7 M s−1 (Fig. 13, top). Interestingly, both tertiary alcohols exhibit significant rise of concentrations in the first minutes of reaction; then the concentration of trans-alcohol remains unchanged within the reaction time studied (Fig. 13, top). This may point to the presence of some rapid processes at initial stage with the mechanism different from the described above. Interestingly, although the reaction is slow, the reaction rate remains almost unchanged within 24 h (Fig. 13, bottom, inset). After 24 h the cis/trans ratio of 29.3 (Fig. 13, bottom) was observed with negligible amounts of chlorobenzene formed (ca. 1.8% based on the m-CPBA). The yields of products were 11.2 and 41.4% based on the substrate and oxidant, respectively, and 3°![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2° was 30
2° was 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. This indicates that the reaction mechanism with the catalyst 2 differs from that with nickel complexes 11–14 (Table 4), which produces considerable amounts of chlorobenzene.
1. This indicates that the reaction mechanism with the catalyst 2 differs from that with nickel complexes 11–14 (Table 4), which produces considerable amounts of chlorobenzene.
The catalytic system comprising 2 was found to possess more complex behaviour than the one with catalyst 1. Curiously, on the initial stages of data collection, it was observed that some experiments reveal no reaction products, whereas their repetition gave good yields of products and notable stereoselectivity. Further investigation revealed that the catalytic properties of 2 strongly depend on the number of factors, first of all on the time between dissolution of the solid sample of 2 and reaction initiation (addition of oxidant). This could point to a spontaneous transformation of 2 upon dissolution into another form, probably a dimeric Ni2 unit with much higher catalytic activity than that of 2. For the moment, we did not manage to assess reproducibility, and more detailed investigation with elaboration of the conditions (and respective factors) under which the catalytic system cis-1,2-DMCH/2/m-CPBA would give reproducible results is underway.
Conclusions
We have studied the catalytic behaviour of the isoindole-based complexes of cobalt (1) and nickel (2) in the reaction of mild stereospecific oxidation of alkanes with m-CPBA. We demonstrated that the combination of m-CPBA with the cobalt catalyst may stereoselectively oxidize saturated tertiary C–H bonds with the high yields of alcohols, based on substrate. The kinetic data, isotopic labelling and reaction with radical trap let us assume that the main reaction pathway proceeds without the formation of free alkyl radicals, most probably involving the high-valent metal–oxo species CoIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, which abstract the hydrogen from the alkane C–H bond in a mechanism, similar to the oxygen rebound one. This assumption is in accord with a high kinetic isotope effect of 7.2(2) observed in the oxidation of cyclohexane, while no cyclohexyl hydroperoxide (typical product in a free radical cyclohexane oxidation) was detected. Isostructural to 1 nickel catalyst 2 exhibits lower than 1 and somehow fluctuating activity. As continuation of this study, we plan to immobilize the catalysts with their possible stabilization and recovery.
O, which abstract the hydrogen from the alkane C–H bond in a mechanism, similar to the oxygen rebound one. This assumption is in accord with a high kinetic isotope effect of 7.2(2) observed in the oxidation of cyclohexane, while no cyclohexyl hydroperoxide (typical product in a free radical cyclohexane oxidation) was detected. Isostructural to 1 nickel catalyst 2 exhibits lower than 1 and somehow fluctuating activity. As continuation of this study, we plan to immobilize the catalysts with their possible stabilization and recovery.
Acknowledgements
This work was supported by Fundação para a Ciência e Tecnologia (FCT), Portugal, projects PTDC/QEQ-QIN/3967/2014, UID/QUI/00100/2013 and “Investigador 2013” (IF/01270/2013/CP1163/CT0007) and fellowships SFRH/BPD/63710/2009 (O.V.N.) and SFRH/BPD/99533/2014 (D.S.N.). The authors acknowledge the IST Node of the Portuguese Network of Mass-spectrometry for the ESI-MS measurements.References
- (a) J. A. Labinger and J. E. Bercaw, Nature, 2002, 417, 507–514 CrossRef CAS PubMed; (b) T. Newhouse and P. S. Baran, Angew. Chem., Int. Ed., 2011, 50, 3362–3374 CrossRef CAS PubMed.
- M. C. White, Science, 2012, 335, 807–809 CrossRef CAS PubMed.
- (a) J. Yamaguchi, A. D. Yamaguchi and K. Itami, Angew. Chem., Int. Ed., 2012, 51, 8960–9009 CrossRef CAS PubMed; (b) W. R. Gutekunst and P. S. Baran, Chem. Soc. Rev., 2011, 40, 1976–1991 RSC.
- (a) L. Que and W. B. Tolman, Nature, 2008, 455, 333–340 CrossRef CAS PubMed; (b) E. P. Talsi and K. P. Bryliakov, Coord. Chem. Rev., 2012, 256, 1418–1434 CrossRef CAS; (c) W. N. Oloo, K. K. Meier, Y. Wang, S. Shaik, E. Munck and L. Que, Nat. Commun., 2014, 5, 3046 Search PubMed.
- (a) E. O'Reilly, V. Kohler, S. L. Flitsch and N. J. Turner, Chem. Commun., 2011, 47, 2490–2501 RSC; (b) P. R. O. de Montellano, Chem. Rev., 2010, 110, 932–948 CrossRef PubMed.
- K. P. Bryliakov and E. P. Talsi, Coord. Chem. Rev., 2014, 276, 73–96 CrossRef CAS.
- S. Sirajuddin and A. C. Rosenzweig, Biochemistry, 2015, 54, 2283–2294 CrossRef CAS PubMed.
- (a) A. R. McDonald and L. Que, Coord. Chem. Rev., 2013, 257, 414–428 CrossRef CAS; (b) A. Gunay and K. H. Theopold, Chem. Rev., 2010, 110, 1060–1081 CrossRef CAS PubMed.
- E. P. Talsi, R. V. Ottenbacher and K. P. Bryliakov, J. Organomet. Chem., 2015, 793, 102–107 CrossRef CAS.
- (a) L. Gomez, I. Garcia-Bosch, A. Company, J. Benet-Buchholz, A. Polo, X. Sala, X. Ribas and M. Costas, Angew. Chem., Int. Ed., 2009, 48, 5720–5723 CrossRef CAS PubMed; (b) V. A. Yazerski, P. Spannring, D. Gatineau, C. H. M. Woerde, S. M. Wieclawska, M. Lutz, H. Kleijn and R. J. M. K. Gebbink, Org. Biomol. Chem., 2014, 12, 2062–2070 RSC; (c) P. E. Gormisky and M. C. White, J. Am. Chem. Soc., 2013, 135, 14052–14055 CrossRef CAS PubMed; (d) M. A. Bigi, S. A. Reed and M. C. White, J. Am. Chem. Soc., 2012, 134, 9721–9726 CrossRef CAS PubMed.
- H. Hussain, A. Al-Harrasi, I. R. Green, I. Ahmed, G. Abbas and N. U. Rehman, RSC Adv., 2014, 4, 12882–12917 RSC.
- (a) W. Nam, M. H. Lim, S. K. Moon and C. Kim, J. Am. Chem. Soc., 2000, 122, 10805–10809 CrossRef CAS; (b) G. B. Shul'pin, H. Stoeckli-Evans, D. Mandelli, Y. N. Kozlov, A. T. Vallina, C. B. Woitiski, R. S. Jimenez and W. A. Carvalhoc, J. Mol. Catal. A: Chem., 2004, 219, 255–264 CrossRef.
- (a) W. Nam, I. Kim, Y. Kim and C. Kim, Chem. Commun., 2001, 1262–1263 RSC; (b) W. Nam, J. Y. Ryu, I. Kim and C. Kim, Tetrahedron Lett., 2002, 43, 5487–5490 CrossRef CAS.
- (a) M. N. Kopylovich, M. Haukka and K. T. Mahmudov, Tetrahedron, 2015, 71, 8622–8627 CrossRef CAS; (b) M. N. Kopylovich, K. V. Luzyanin, M. Haukka, A. J. L. Pombeiro and V. Y. Kukushkin, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2006, 62, M2846–M2848 Search PubMed.
- G. B. Shul'pin, J. Mol. Catal. A: Chem., 2002, 189, 39–66 CrossRef.
- R. V. Ottenbacher, E. P. Talsi and K. P. Bryliakov, ACS Catal., 2015, 5, 39–44 CrossRef CAS.
- (a) M. N. Kopylovich, V. Y. Kukushkin, M. Haukka, K. V. Luzyanin and A. J. L. Pombeiro, J. Am. Chem. Soc., 2004, 126, 15040–15041 CrossRef CAS PubMed; (b) M. N. Kopylovich, M. Haukka, A. M. Kirillov, V. Y. Kukushkin and A. I. L. Pombeiro, Chem.–Eur. J., 2007, 13, 786–791 CrossRef CAS PubMed.
- S. Hubner, J. G. de Vries and V. Farina, Adv. Synth. Catal., 2016, 358, 3–25 CrossRef.
- R. Bonnett and R. F. C. Brown, J. Chem. Soc., Chem. Commun., 1972, 393–395 RSC.
- S. S. Simons, H. L. Ammon, R. Doherty and D. F. Johnsont, J. Org. Chem., 1981, 46, 4739–4744 CrossRef CAS.
- A. K. Hijazi, A. Al Hmaideen, S. Syukri, N. Radhakrishnan, E. Herdtweck, B. Voit and F. E. Kühn, Eur. J. Inorg. Chem., 2008, 2892–2898 CrossRef CAS.
- K. B. Cho, X. Wu, Y. M. Lee, Y. H. Kwon, S. Shaik and W. Nam, J. Am. Chem. Soc., 2012, 134, 20222–20225 CrossRef CAS PubMed.
- J. Serrano-Plana, W. N. Oloo, L. Acosta-Rueda, K. K. Meier, B. Verdejo, E. Garcia-Espana, M. G. Basallote, E. Munck, L. Que, A. Company and M. Costas, J. Am. Chem. Soc., 2015, 137, 15833–15842 CrossRef CAS PubMed.
- G. Olivo, O. Lanzalunga and S. Di Stefano, Adv. Synth. Catal., 2016, 358, 843–863 CrossRef CAS.
- (a) A. Bravo, H. R. Bjorsvik, F. Fontana, F. Minisci and A. Serri, J. Org. Chem., 1996, 61, 9409–9416 CrossRef CAS; (b) A. Bravo, F. Fontana, F. Minisci and A. Serri, Chem. Commun., 1996, 1843–1844 RSC.
- A. A. Fokin and P. R. Schreiner, Chem. Rev., 2002, 102, 1551–1593 CrossRef CAS PubMed.
- J. Nakazawa, A. Yata, T. Hori, T. D. P. Stack, Y. Naruta and S. Hikichi, Chem. Lett., 2013, 42, 1197–1199 CrossRef CAS.
- T. Nagataki, K. Ishii, Y. Tachi and S. Itoh, Dalton Trans., 2007, 1120–1128 RSC.
- T. Kojima, K. I. Hayashi, S. Y. Iizuka, F. Tani, Y. Naruta, M. Kawano, Y. Ohashi, Y. Hirai, K. Ohkubold, Y. Matsuda and S. Fukuzumi, Chem.–Eur. J., 2007, 13, 8212–8222 CrossRef CAS PubMed.
- (a) K. A. Lee and W. Nam, J. Am. Chem. Soc., 1997, 119, 1916–1922 CrossRef CAS; (b) K. Chen and L. Que, J. Am. Chem. Soc., 2001, 123, 6327–6337 CrossRef CAS PubMed.
- (a) S. Hong, F. F. Pfaff, E. Kwon, Y. Wang, M.-S. Seo, E. Bill, K. Ray and W. Nam, Angew. Chem., Int. Ed., 2014, 53, 10403–10407 CrossRef CAS PubMed; (b) F. F. Pfaff, S. Kundu, M. Risch, S. Pandian, F. Heims, I. Pryjomska-Ray, P. Haack, R. Metzinger, E. Bill, H. Dau, P. Comba and K. Ray, Angew. Chem., Int. Ed., 2011, 50, 1711–1715 CrossRef CAS PubMed.
- (a) S. K. Rout, S. Guin, W. Ali, A. Gogoi and B. K. Patel, Org. Lett., 2014, 16, 3086–3089 CrossRef CAS PubMed; (b) D. Wang, E. R. Farquhar, A. Stubna, E. Munck and L. Que, Nat. Chem., 2009, 1, 145–150 CrossRef CAS PubMed.
- J. T. Groves and T. E. Nemo, J. Am. Chem. Soc., 1983, 105, 6243–6248 CrossRef CAS.
- M. Balamurugan, R. Mayilmurugan, E. Suresh and M. Palaniandavar, Dalton Trans., 2011, 40, 9413–9424 RSC.
- M. Sankaralingam, M. Balamurugan, M. Palaniandavar, P. Vadivelu and C. H. Suresh, Chem.–Eur. J., 2014, 20, 11346–11361 CrossRef CAS PubMed.
- T. Nagataki, Y. Tachi and S. Itoh, Chem. Commun., 2006, 4016–4018 RSC.
- J. M. Hudzik, J. W. Bozzelli and J. M. Simmie, J. Phys. Chem. A, 2014, 118, 9364–9379 CrossRef CAS PubMed.
- M. Kodera, H. Shimakoshi and K. Kano, Chem. Commun., 1996, 1737–1738 RSC.
- R. Mayilmurugan, H. Stoeckli-Evans, E. Suresh and M. Palaniandavar, Dalton Trans., 2009, 5101–5114 RSC.
- V. Korobov and V. Ochkov, Chemical Kinetics with Mathcad and Maple, Springer, 2011 Search PubMed.
- G. B. Shul'pin, D. S. Nesterov, L. S. Shul'pin and A. J. L. Pombeiro, Inorg. Chim. Acta, 2016 DOI:10.1016/j.ica.2016.04.035 , in press.
- T. Kojima, R. A. Leising, S. Yan and L. Que, J. Am. Chem. Soc., 1993, 115, 11328–11335 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: ESI-MS spectrum of 1, EI mass spectra of reaction products from the oxidation of cis-1,2-DMCH in the presence of CCl3Br, accumulations of products in the course of oxidation of normal and deuterated cyclohexane, EI mass spectra of normal and deuterated chlorocyclohexanes, kinetic fitting details. See DOI: 10.1039/c6ra14382b |
| This journal is © The Royal Society of Chemistry 2016 |