Enantioselective construction of novel chiral spirooxindoles incorporating a thiazole nucleus†
L.-Y. Cuia,
Y.-M. Wanga and
Z.-H. Zhou*ab
aInstitute and State Key Laboratory of Elemento-Organic Chemistry, Nankai University, Tianjin 300071, P. R. China. E-mail: z.h.zhou@nankai.edu.cn
bCollaborative Innovation Center of Chemical Science and Engineering (Tianjin), Nankai University, Tianjin 300071, P. R. China
First published on 1st July 2016
Abstract
Asymmetric cascade Michael/cyclization reaction between 2-substituted thiazol-4-ones and 2-(2-oxoindolin-3-ylidene)malononitriles was investigated using a series of chiral bifunctional hydrogen-bonding organocatalysts. Good yields (up to 91%) and excellent enantioselectivities (up to 98% ee) were achieved by using a (1R,2R)-1,2-diphenylethane-1,2-diamine derived thiourea catalyst. This method provides an elegant synthetic route to access novel thiazole-fused spirooxindoles with potential bioactivity.
Introduction
1,3-Thiazole, a five-membered heterocyclic ring with nitrogen and sulfur atoms, is one of the most important scaffolds in heterocyclic chemistry. The thiazole nucleus is recognized as a privileged pharmacophore in drug design and discovery.1 In recent years, numerous thiazole derivatives have been reported to exhibit diverse biological activities including antibacterial,2 antifungal,3 antitubercular,4 antiviral,5 anticancer,6 anticonvulsant,7 anti-inflammatory8 and antioxidant9 activity. Moreover, several thiazole-based enzyme inhibitors have proven to be useful for treatment of diabetes10 and Alzheimer's disease.11 As a subtype of indole derivatives, spirooxindole is a very important structural motif that make ups the core of many naturally occurring12 and synthetic products13 exhibiting a broad range of bioactivities. The considerable medicinal potential of both thiazole and spirooxindole has made them attractive synthetic targets. As such, a number of synthetic methods have been developed in pursuit of diversely functionalized chiral thiazole14 and spirooxindole derivatives.15,16 Despite the elegant methods noted in the literature to synthesize spirooxindoles, the development of novel and efficient synthetic methods to access functionalized spirooxindoles with structural diversity is still desirable. In contrast to the importance of thiazole and spirooxindole units in pharmaceutical science and as promising candidates for drug discovery, the direct catalytic asymmetric approach to access optically active thiazole-fused spirooxindoles still remain unexplored. Intrigued by the diverse pharmacological activities of spirooxindole and thiazole derivatives, we wish to synthesize molecules which comprise both of these pharmacophores. The hybrid molecule, thiazole-fused spirooxindole, may inherit biological activities of both and thiazole skeletons. In this context, we report the organocatalyzed asymmetric Michael/cyclization cascade reaction between 2-substituted thiazol-4-ones and 2-(2-oxoindolin-3-ylidene)malononitriles, which provide a convenient approach to access enantiomerically enriched thiazole-fused spirooxindoles in good yields, and good to excellent enantioselectivities.Results and discussion
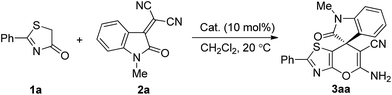
The initial studies commenced with a cascade Michael/cyclization between 2-phenylthiazol-4(5H)-one (1a) and 2-(1-methyl-2-oxoindolin-3-ylidene)malononitrile (2a) catalyzed by bifunctional squaramide I (10 mol%) in 1 mL of dichloromethane at room temperature (20 °C) (Table 1, entry 1). As anticipated, the reaction proceeded smoothly to generate the desired product, (S)-5′-amino-1-methyl-2-oxo-2′-phenylspiro[indoline-3,7′-pyrano[2,3-d]thiazole]-6′-carbonitrile (3aa) in an acceptable yield of 39%, but with a rather poor enantioselectivity of 7% ee (Table 1, entry 1). Subsequently, a variety of squaramide17- and thiourea-based18 bifunctional double hydrogen-bonding catalysts II–V (Fig. 1) were screened for the model reaction, and the results are summarized in Table 1. As shown, both the chiral diamine skeleton and the type of the hydrogen-bonding donor moiety played a crucial role on the enantioselectivity of the reaction. Thiourea Va containing a (1R,2R)-1,2-diphenylethane-1,2-diamine backbone gave the best results (62% yield, 89% ee) among these tested organocatalysts (Table 1, entry 6 vs. 1–5 and 7). By employing thiourea Va as the most promising catalyst, we then turned our attention to the solvent screening and found that chloroform was shown to be the ideal solvent as 3aa was generated with better yield and enantioselectivity (67% yield, 93% ee, Table 1, entry 8 vs. 6 and 7–14). Efforts to further improve the enantioselectivity by performing the reaction at lower reaction temperature did not bear any fruitful results (entries 15 and 16 vs. 9). Importantly, the cascade reaction proceeded smoothly with 5 mol% or even 2 mol% of catalyst Va with retained or slightly enhanced enantioselectivity (entries 17 and 18). Further lowering the amount of catalyst Va to 1 mol% resulted in an obviously decreased yield and enantioselectivity (entry 19).| Entry | Catalyst | Solvent | Time (h) | Yieldb % | Eec (%) |
|---|---|---|---|---|---|
| a Unless noted, the reactions were carried out with 1a (0.2 mmol), 2a (0.2 mmol), and catalyst (10 mol%) in 1.0 mL of dichloromethane at 20 °C.b Isolated yield.c Determined by HPLC analysis with a chiral stationary phase.d The reaction was performed at 0 °C.e The reaction was carried out at −20 °C.f The loading of Va is 5 mol%.g The amount of Va is 2 mol%.h In the presence of 1 mol% of Va. | |||||
| 1 | I | CH2Cl2 | 0.4 | 39 | 7 |
| 2 | II | CH2Cl2 | 0.15 | 91 | –6 |
| 3 | III | CH2Cl2 | 0.1 | 20 | –18 |
| 4 | IVa | CH2Cl2 | 0.25 | 38 | 69 |
| 5 | IVb | CH2Cl2 | 0.15 | 50 | 82 |
| 6 | Va | CH2Cl2 | 0.5 | 62 | 89 |
| 7 | Vb | CH2Cl2 | 0.15 | 75 | 81 |
| 8 | Va | ClCH2CH2Cl | 0.4 | 66 | 89 |
| 9 | Va | CHCl3 | 0.3 | 67 | 93 |
| 10 | Va | PhCH3 | 0.25 | 96 | 85 |
| 11 | Va | CH3CN | 0.15 | 76 | 68 |
| 12 | Va | Et2O | 0.3 | 72 | 85 |
| 13 | Va | EA | 0.5 | 92 | 85 |
| 14 | Va | THF | 4 | 92 | 71 |
| 15d | Va | CHCl3 | 0.5 | 83 | 92 |
| 16e | Va | CHCl3 | 2 | 78 | 92 |
| 17f | Va | CHCl3 | 0.4 | 87 | 91 |
| 18g | Va | CHCl3 | 0.8 | 74 | 94 |
| 19h | Va | CHCl3 | 1 | 69 | 84 |
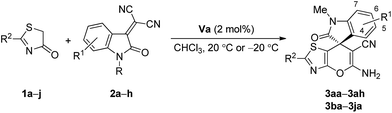
Having established the optimal reaction conditions (Table 1, entry 18), we then explored the substrate scope and the generality of the reaction. The results are summarized in Table 2. At first, a broad range of 3-ylideneoxindoles 2b–h with different substitution groups either on the nitrogen atom or the aromatic ring were explored in the reactions with thiazolone 1a (entries 2–8). All the reactions progressed smoothly to give the corresponding products 3 in moderate to good yields. N-substitution has an obvious influence on the stereoselectivity of the reaction. The replacement of methyl with benzyl group led to a sharply decreased enantioselectivity (entry 2 vs. 1). The electronic nature of the substituent on the aromatic ring has a minor impact on the stereochemical outcomes of the reaction. Generally, 3-ylideneoxindoles bearing neutral or electron-withdrawing groups (2a–f) provided higher ee values than those bearing electron-donating groups (2g–h). On the other hand, a number of thiazolones 1b–j with either electron-withdrawing (entries 9–14) or electron-donating groups (entries 15–17) on the aryl ring also were well tolerated in the reactions with 3-ylideneoxindole 2a. The position of the substituent on substrate 1 had a significant effect on the stereocontrol of the reaction. For instance, compared with the results obtained from 4-chloro and 3-bromo substituted thiazolones 1c and 1f, much better enantioselectivities were observed for 4-bromo and 2-chloro substituted thiazolones 1d and 1e (entry 11 vs. 10, entry 12 vs. 13). Since all the obtained products are solids with high melting point, in some cases, the optical purity of the products could be markedly improved via a single recrystallization (entries 2, 13 and 14). To demonstrate the potential of this method for preparative purposes, the reaction is also carried out in gram scale giving the isolated product with comparable ee value and chemical yield (entry 2 vs. 1).
| Entry | 3 (R, R1, R2) | Time (h) | Yieldb (%) | Eec (%) |
|---|---|---|---|---|
| a Unless noted, all reactions were carried out with 1 (0.20 mmol), 2 (0.20 mmol) in the presence of 2 mol% of catalyst Va in chloroform (1 mL) at 20 °C.b Isolated yield.c Determined by HPLC analysis using a chiral stationary phase.d The reaction was carried out using a 4 mmol-scale.e Data in parentheses were obtained after a single recrystallization.f The reaction was performed at −20 °C. | ||||
| 1 | 3aa (Me, H, Ph) | 0.7 | 74 | 94 |
| 2d | 3aa (Me, H, Ph) | 0.7 | 70 | 92 |
| 3 | 3ab (Bn, H, Ph) | 0.3 | 77 (63)e | 78 (90)e |
| 3f | 3ac (Me, 5-F, Ph) | 1 | 75 | 90 |
| 4 | 3ad (Me, 5-Cl, Ph) | 0.3 | 90 | 98 |
| 5f | 3ae (Me, 4-Cl, Ph) | 0.7 | 93 | 89 |
| 6 | 3af (Me, 5-Br, Ph) | 0.4 | 54 | 98 |
| 7 | 3ag (Me, 5-Me, Ph) | 0.8 | 92 | 87 |
| 8f | 3ah (Me, 5-MeO, Ph) | 3 | 84 | 83 |
| 9 | 3ba (Me, H, 2,6-F2C6H3) | 0.3 | 51 | 89 |
| 10 | 3ca (Me, H, 4-ClC6H4) | 1.5 | 80 | 82 |
| 11 | 3da (Me, H, 2-ClC6H4) | 4.5 | 80 | 93 |
| 12 | 3ea (Me, H, 4-BrC6H4) | 0.3 | 64 | 97 |
| 13 | 3fa (Me, H, 3-BrC6H4) | 4.5 | 89 (60)e | 75 (90)e |
| 14 | 3ga (Me, H, 4-CF3C6H4) | 3 | 94 (63)e | 69 (93)e |
| 15 | 3ha (Me, H, 4-MeC6H4) | 10 | 64 | 88 |
| 16 | 3ia (Me, H, 4-MeOC6H4) | 0.3 | 61 | 95 |
| 17 | 3ja (Me, H, 3,5-(MeO)2C6H3) | 0.4 | 56 | 91 |
The absolute configuration of compound 3ad was determined unambiguously as (R) based on the X-ray structure of 3ad (Fig. 2) and the stereochemistry of other title compounds were assigned by analogy.19
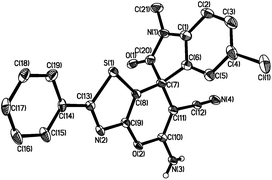 | ||
| Fig. 2 X-ray crystal structure of (S)-3ad. Most of the hydrogen atoms have been omitted for clarity. | ||
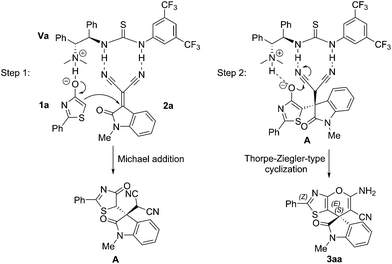
To account for the observed stereochemistry of this cascade transformation, a plausible mechanism is shown in Scheme 1. In light of the above results and the recent studies,20 thiourea Va is believed to act in a bifunctional fashion. An enolate is formed via the deprotonation of 1a by the tertiary amine group. Simultaneously, the isatylidenemalononitrile 2a is fixed and activated by the neighboring thiourea moiety through weak hydrogen bonding interaction. The enolate then predominantly attacks the si-face of the electron-deficient isatylidenemalononitrile 2a to generate the Michael addition intermediate A. Finally, subsequent intramolecular Thorpe–Ziegler-type cyclization of the intermediate A with the aid of the bifunctional thiourea catalyst Va via a similar dual activation fashion gave rise to the desired thiazole-fused spirooxindole 3aa.
Experimental
General methods
All reagents and solvents were commercial grade and purified prior to use when necessary. NMR spectra were acquired on Varian 400 MHz instrumental. Chemical shifts are measured relative to residual solvent peaks as an internal standard set to δ 7.26 and δ 77.0 (CDCl3), δ 2.50 and δ 39.5 (DMSO-d6). Specific rotations were measured on a Perkin-Elmer 341MC polarimeter. Enantiomeric excesses were determined on a Shimadzu LC-20A instrument (chiral column; mobile phase: hexane/i-PrOH). HRMS was performed on a Varian QFT-ESI instrumental. Melting points were determined on a Taike X-4 melting point apparatus. All temperatures were uncorrected.General procedure for Va catalyzed cascade Michael/cyclization reaction between thiazolones 1 and 2-(2-oxoindolin-3-ylidene)malononitriles 2
A solution of thiourea catalyst Va (2 mol %), thiazolones (1, 0.20 mmol) and 2-(2-oxoindolin-3-ylidene)malononitriles (2, 0.20 mmol) in chloroform (1 mL) was stirred at 20 °C. Upon completion of the reaction (monitored by TLC), the product was directly purified by column chromatography on silica gel (200–300 mesh, PE/EtOAc = 12/1) to afford the desired thiazole-fused spirooxindoles 3. The title compounds were fully characterized by 1H NMR, 13C NMR, HRMS and specific rotation data. The enantiomeric excess of the pure products was determined by HPLC analysis using a chiral stationary phase.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 14.30 (major) and 17.12 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 14.30 (major) and 17.12 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 24.17 (major) and 29.06 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 24.17 (major) and 29.06 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 9.48 (major) and 11.85 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 9.48 (major) and 11.85 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 10.40 (major) and 13.42 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 10.40 (major) and 13.42 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.46 (major) and 22.80 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.46 (major) and 22.80 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 75
2-propanol = 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.88 (major) and 17.44 min (minor).
25, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.88 (major) and 17.44 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.54 (major) and 15.64 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 12.54 (major) and 15.64 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 15.01 (major) and 18.40 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 15.01 (major) and 18.40 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 10.99 (major) and 15.28 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 10.99 (major) and 15.28 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 17.50 (major) and 21.28 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 17.50 (major) and 21.28 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 80
2-propanol = 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 32.93 (minor) and 38.94 min (major).
20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 32.93 (minor) and 38.94 min (major).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 80
2-propanol = 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 35.66 (minor) and 40.74 min (major).
20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 35.66 (minor) and 40.74 min (major).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 11.48 (major) and 14.36 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 11.48 (major) and 14.36 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 85
2-propanol = 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 50.50 (major) and 58.51 min (minor).
15, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 50.50 (major) and 58.51 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 15.85 (major) and 19.65 min (minor).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 15.85 (major) and 19.65 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 80
2-propanol = 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 49.94 (major) and 63.40 min (minor).
20, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 49.94 (major) and 63.40 min (minor).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2-propanol = 70
2-propanol = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 19.12 (minor) and 23.66 min (major).
30, flow rate = 1.0 mL min−1, wavelength = 254 nm): Rt = 19.12 (minor) and 23.66 min (major).Conclusions
In conclusion, we have demonstrated herein an efficient organocatalytic cascade Michael/cyclization reaction of 2-substituted thiazol-4-ones and 2-(2-oxoindolin-3-ylidene)-malononitriles. The present process enabled the formation of a variety of structurally diverse thiazole-fused spirooxindole derivatives in moderate to good yields and with high levels of enantioselectivity in a one-pot manner.Acknowledgements
We are grateful to the National Natural Science Foundation of China (No. 21421062), the Key laboratory of Elemento-Organic Chemistry and Collaborative Innovation Center of Chemical Science and Engineering for generous financial support for our programs.Notes and references
- N. Siddiqui, M. F. Arshad, W. Ahsan and M. S. Alam, Int. J. Pharm. Sci. Drug Res., 2009, 1, 136 Search PubMed; A. Ayati, S. Emami, A. Asadipour, A. Shafiee and A. Foroumadi, Eur. J. Med. Chem., 2015, 97, 699 CrossRef CAS PubMed.
- (a) H. Mohammad, A. S. Mayhoub, A. Ghafoor, M. Soofi, R. A. Alajlouni, M. Cushman and M. N. Seleem, J. Med. Chem., 2014, 57, 1609 CrossRef CAS PubMed; (b) K. Cheng, J.-Y. Xue and H.-L. Zhu, Bioorg. Med. Chem. Lett., 2013, 23, 4235 CrossRef CAS PubMed; (c) S. Bondock, T. Naser and Y. A. Ammar, Eur. J. Med. Chem., 2013, 62, 270 CrossRef CAS PubMed; (d) N. C. Desai, N. Bhatt, H. Somani and A. Trivedi, Eur. J. Med. Chem., 2013, 67, 54 CrossRef CAS PubMed; (e) Y.-S. Yang, F. Zhang, C. Gao, Y.-B. Zhang, X.-L. Wang, J.-F. Tang, J. Sun, H.-B. Gong and H.-L. Zhu, Bioorg. Med. Chem. Lett., 2012, 22, 4619 CrossRef CAS PubMed; (f) K. Liaras, A. Geronikaki, J. Glamočlija, A. Ćirić and M. Soković, Bioorg. Med. Chem., 2011, 19, 3135 CrossRef CAS PubMed.
- (a) L. T. Maillard, S. Bertout, O. Quinonéro, G. Akalin, G. T. Zitouni, P. Fulcrand, F. Demirci, J. Martinez and N. Masurier, Bioorg. Med. Chem. Lett., 2013, 23, 1803 CrossRef CAS PubMed; (b) F. Chimenti, B. Bizzarri, A. Bolasco, D. Secci, P. Chimenti, A. Granese, S. Carradori, M. D. Ascenzio, D. Lilli and D. Rivanera, Eur. J. Med. Chem., 2011, 46, 378 CrossRef CAS PubMed; (c) S. K. Bharti, G. Nath, R. Tilak and S. K. Singh, Eur. J. Med. Chem., 2010, 45, 651 CrossRef CAS PubMed; (d) P. Karegoudar, M. S. Karthikeyan, D. J. Prasad, M. Mahalinga, B. S. Holla and N. S. Kumari, Eur. J. Med. Chem., 2008, 43, 261 CrossRef CAS PubMed.
- (a) P. Makam, R. Kankanala, A. Prakash and T. Kannan, Eur. J. Med. Chem., 2013, 69, 564 CrossRef CAS PubMed; (b) V. U. Jeankumar, J. Renuka, P. Santosh, V. S. Jonnalagadda, P. Sridevi, P. Suryadevara and P. Y. D. Sriram, Eur. J. Med. Chem., 2013, 70, 143 CrossRef CAS PubMed; (c) X. Lu, X. Liu, B. Wan, S. G. Franzblau, L. Chen, C. Zhou and Q. You, Eur. J. Med. Chem., 2012, 49, 164 CrossRef CAS PubMed; (d) G. Aridoss, S. Amirthaganesan, M. S. Kim, J. T. Kim and Y. T. Jeong, Eur. J. Med. Chem., 2009, 44, 4199 CrossRef CAS PubMed; (e) M. R. Shiradkar, K. K. Murahari, H. R. Gangadasu, T. Suresh, C. A. Kalyan, D. Panchal, R. Kaur, P. Burange, J. Ghogare, V. Mokalec and M. Raut, Bioorg. Med. Chem., 2007, 15, 3997 CrossRef CAS PubMed.
- (a) Z. Xu, M. Ba, H. Zhou, Y. Cao, C. Tang, Y. Yang, R. He, Y. Liang, X. Zhang, Z. Li, L. Zhu, Y. Guo and C. Guo, Eur. J. Med. Chem., 2014, 85, 27 CrossRef CAS PubMed; (b) F. Curreli, S. Choudhury, I. Pyatkin, V. P. Zagorodnikov, A. K. Bulay, A. Altieri, Y. D. Kwon, P. D. Kwong and A. K. Debnath, J. Med. Chem., 2012, 55, 4764 CrossRef CAS PubMed; (c) A. V. Stachulski, C. Pidathala, E. C. Row, R. Sharma, N. G. Berry, M. Iqbal, J. Bentley, S. A. Allman, G. Edwards, A. Helm, J. Hellier, B. E. Korba, J. E. Semple and J.-F. Rossignol, J. Med. Chem., 2011, 54, 4119 CrossRef CAS PubMed; (d) A. S. Mayhoub, M. Khaliq, R. J. Kuhn and M. Cushman, J. Med. Chem., 2011, 54, 1704 CrossRef CAS PubMed; (e) Y. Liu, F. Jing, Y. Xu, Y. Xie, F. Shi, H. Fang, M. Li and W. Xu, Bioorg. Med. Chem., 2011, 19, 2342 CrossRef CAS PubMed.
- (a) A. Reichelt, J. M. Bailis, M. D. Bartberger, G. Yao, H. Shu, M. R. Kaller, J. G. Allen, M. F. Weidner, K. S. Keegan and J. H. Dao, Eur. J. Med. Chem., 2014, 80, 364 CrossRef CAS PubMed; (b) A. Carbone, M. Pennati, B. Parrino, A. Lopergolo, P. Barraja, A. Montalbano, V. Spanò, S. Sbarra, V. Doldi, M. De Cesare, G. Cirrincione, P. Diana and N. Zaffaroni, J. Med. Chem., 2013, 56, 7060 CrossRef CAS PubMed; (c) M. Salehi, M. Amini, S. N. Ostad, G. H. Riazi, A. Assadieskandar, B. Shafiei and A. Shafiee, Bioorg. Med. Chem., 2013, 21, 7648 CrossRef CAS PubMed; (d) M. Banimustafa, A. Kheirollahi, M. Safavi, S. Kabudanian Ardestani, H. Aryapour, A. Foroumadi and S. Emami, Eur. J. Med. Chem., 2013, 70, 692 CrossRef CAS PubMed; (e) M. Azizmohammadi, M. Khoobi, A. Ramazani, S. Emami, A. Zarrin, O. Firuzi, R. Miri and A. Shafiee, Eur. J. Med. Chem., 2013, 59, 15 CrossRef CAS PubMed.
- (a) N. Ahangar, A. Ayati, E. Alipour, A. Pashapour, A. Foroumadi and S. Emami, Chem. Biol. Drug Des., 2011, 78, 844 CrossRef CAS PubMed; (b) N. Siddiqui and W. Ahsan, Eur. J. Med. Chem., 2010, 45, 1536 CrossRef CAS PubMed; (c) K. M. Amin, D. E. Abdel Rahman and Y. A. Al-Eryani, Bioorg. Med. Chem., 2008, 16, 5377 CrossRef CAS PubMed.
- (a) C. B. Rödl, D. Vogt, S. B. M. Kretschmer, K. Ihlefeld, S. Barzen, A. Brüggerhoff, J. Achenbach, E. Proschak, D. Steinhilber, H. Stark and B. Hofmann, Eur. J. Med. Chem., 2014, 84, 302 CrossRef PubMed; (b) S. Koppireddi, J. R. Komsani, S. Avula, S. Pombala, S. Vasamsetti, S. Kotamraju and R. Yadla, Eur. J. Med. Chem., 2013, 66, 305 CrossRef CAS PubMed; (c) M. H. M. Helal, M. A. Salem, M. S. A. El-Gaby and M. Aljahdali, Eur. J. Med. Chem., 2013, 65, 517 CrossRef CAS PubMed; (d) R. Aggarwal, S. Kumar, P. Kaushik, D. Kaushik and G. K. Gupta, Eur. J. Med. Chem., 2013, 62, 508 CrossRef CAS PubMed.
- (a) G. G. Mandawad, B. S. Dawane, S. D. Beedkar, C. N. Khobragade and O. S. Yemul, Bioorg. Med. Chem., 2013, 21, 365 CrossRef CAS PubMed; (b) B. F. Abdel-Wahab, G. E. A. Awad and F. A. Badria, Eur. J. Med. Chem., 2011, 46, 1505 CrossRef CAS PubMed; (c) K.-M. Kim, K.-H. Kim, T.-C. Kang, W.-Y. Kim, M.-R. Lee, H.-J. Jung, I. K. Hwang, S.-B. Ko, J.-Y. Koh, M. H. Won, E.-G. Oh and I. Shin, Bioorg. Med. Chem. Lett., 2003, 13, 2273 CrossRef CAS PubMed.
- (a) S. Y. Kang, K. S. Song, J. Lee, S. H. Lee and J. Lee, Bioorg. Med. Chem., 2010, 18, 6069 CrossRef CAS PubMed; (b) C. S. Li, L. Belair, J. Guay, R. Murgasva, W. Sturkenboom, Y. K. Ramtohul, L. Zhang and Z. Huang, Bioorg. Med. Chem. Lett., 2009, 19, 5214 CrossRef CAS PubMed; (c) R. Pereira, C. Gaudon, B. Iglesias, P. Germain, H. Gronemeyer and A. R. de Lera, Bioorg. Med. Chem. Lett., 2006, 16, 49 CrossRef CAS PubMed; (d) W. R. Oliver, J. L. Shenk, M. R. Snaith, C. S. Russell, K. D. Plunket, N. L. Bodkin, M. C. Lewis, D. A. Winegar, M. L. Sznaidman, M. H. Lambert, H. E. Xu, D. D. Sternbach, S. A. Kliewer, B. C. Hansen and T. M. Willson, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 5306 CrossRef CAS PubMed.
- (a) R. Raza, A. Saeed, M. Arif, S. Mahmood, M. Muddassar, A. Raza and J. Iqbal, Chem. Biol. Drug Des., 2012, 80, 605 CrossRef CAS PubMed; (b) Y. L. Chen, K. Cherry, M. L. Corman, C. F. Ebbinghaus, C. B. Gamlath, D. Liston, B. A. Martin, C. E. Oborskib and B. G. Sahagan, Bioorg. Med. Chem. Lett., 2007, 17, 5518 CrossRef CAS PubMed; (c) M. R. Shiradkar, K. C. Akula, V. Dasari, V. Baru, B. Chiningiri, S. Gandhid and R. Kaure, Bioorg. Med. Chem., 2007, 15, 2601 CrossRef CAS PubMed.
- For reviews on spirooxindole alkanoids, see: (a) Z. Poulos, Spirooxindole alkanoids, LAP LAMBERT Academic Publishing, 2014 Search PubMed; (b) A. Millemaggi and R. J. K. Taylor, Eur. J. Org. Chem., 2010, 4527 CrossRef CAS; (c) R. M. Williams and R. J. Cox, Acc. Chem. Res., 2003, 36, 127 CrossRef CAS PubMed. For selected examples of spirooxindole-containing natural products, see: (d) K. Guo, T. Fang, J. Wang, A. Wu, Y. Wang, J. Jiang, X. Wu, S. Song, W. Su, Q. Xu and X. Deng, Bioorg. Med. Chem. Lett., 2014, 24, 4995 CrossRef CAS PubMed; (e) S.-S. Ma, W.-L. Mei, Z.-K. Guo, S.-B. Liu, Y.-S. Zhao, D.-L. Yang, Y.-B. Zeng, B. Jiang and H.-F. Dai, Org. Lett., 2013, 15, 1492 CrossRef CAS PubMed; (f) K. Wang, X.-Y. Zhou, Y.-Y. Wang, M.-M. Li, Y.-S. Li, L.-Y. Peng, X. Cheng, Y. Li, Y.-P. Wang and Q.-S. Zhao, J. Nat. Prod., 2011, 74, 12 CrossRef CAS PubMed; (g) S. Tsukamoto, T. Kawabata, H. Kato, T. J. Greshock, H. Hota, T. Ohta and R. M. Williams, Org. Lett., 2009, 11, 1297 CrossRef CAS PubMed; (h) T. J. Greshock, A. W. Grubbs, P. Jiao, D. T. Wicklow, J. B. Gloer and R. M. Williams, Angew. Chem., Int. Ed., 2008, 47, 3573 CrossRef CAS PubMed; (i) H. Kato, T. Yoshida, T. Tokue, Y. Nojiri, H. Hirota, T. Ohta, R. M. Williams and S. Tsukamoto, Angew. Chem., Int. Ed., 2007, 46, 2254 CrossRef CAS PubMed; (j) Y.-K. Xu, S.-P. Yang, S.-G. Liao, H. Zhang, L.-P. Lin, J. Ding and J.-M. Yue, J. Nat. Prod., 2006, 69, 1347 CrossRef CAS PubMed; (k) M. Kitajima, T. Nakamura, N. Kogure, M. Ogawa, Y. Mitsuno, K. Ono, S. Yano, N. Aimi and H. Takayama, J. Nat. Prod., 2006, 69, 715 CrossRef CAS PubMed; (l) M. Tsuda, Y. Kasai, K. Komatsu, T. Sone, M. Tanaka, Y. Mikami and J. Kobayashi, Org. Lett., 2004, 6, 3087 CrossRef CAS PubMed; (m) A. C. Whyte, J. B. Gloer, D. T. Wicklow and P. F. Dowd, J. Nat. Prod., 1996, 59, 1093 CrossRef CAS PubMed; (n) C.-B. Cui, H. Kakeya and H. Osada, Tetrahedron, 1996, 52, 12651 CrossRef CAS; (o) A. Jossang, P. Jossang, H. A. Hadi, T. Sevenet and B. Bodo, J. Org. Chem., 1991, 56, 6527 CrossRef CAS; (p) J. Polonsky, M.-A. Merrien, T. Prange and C. Pascard, J. Chem. Soc., Chem. Commun., 1980, 601 RSC.
- Examples of bioactive synthetic spirooxindoles, see: (a) M. Rottmann, C. McNamara, B. K. Yeung, M. C. Lee, B. Zou, B. Russell, P. Seitz, D. M. Plouffe, N. V. Dharia, J. Tan, S. B. Cohen, K. R. Spencer, G. E. Gonzalez-Paez, S. B. Lakshminarayana, A. Goh, R. Suwanarusk, T. Jegla, E. K. Schmitt, H. P. Beck, R. Brun, F. Nosten, L. Renia, V. Dartois, T. H. Keller, D. A. Fidock, E. A. Winzeler and T. T. Diagana, Science, 2010, 329, 1175 CrossRef CAS PubMed; (b) B. K. Yeung, B. Zou, M. Rottmann, S. B. Lakshminarayana, S. H. Ang, S. Y. Leong, J. Tan, J. Wong, S. Keller-Maerki, C. Fischli, A. Goh, E. K. Schmitt, P. Krastel, E. Francotte, K. Kuhen, D. Plouffe, K. Henson, T. Wagner, E. A. Winzeler, F. Petersen, R. Brun, V. Dartois, T. T. Diagana and T. H. Keller, J. Med. Chem., 2010, 53, 5155 CrossRef CAS PubMed; (c) S. Shangary, D. Qin, D. McEachern, M. Liu, R. S. Miller, S. Qiu, Z. Nikolovska-Coleska, K. Ding, G. Wang, J. Chen, D. Bernard, J. Zhang, Y. Lu, Q. Gu, R. B. Shah, K. J. Pienta, X. Ling, S. Kang, M. Guo, Y. Sun, D. Yang and S. Wang, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 3933 CrossRef CAS PubMed; (d) Q. Ding, J.-J. Liu and Z. Zhang, WO 2007104714, 2007Chem. Abstr., 2007, 147, 385857; (e) A. Fensome, R. Bender, J. Cohen, M. A. Collins, V. A. Mackner, L. L. Miller, J. W. Ullrich, R. Winneker, J. Wrobel, P. Zhang, Z. Zhang and Y. Zhu, Bioorg. Med. Chem. Lett., 2002, 12, 3487 CrossRef CAS PubMed.
- (a) L. Lin, Y. Yang, M. Wang, L. Lai, Y. Guo and R. Wang, Chem. Commun., 2015, 51, 8134 RSC; (b) Z.-Q. Liang, L. Yi, K.-Q. Chen and S. Ye, J. Org. Chem., 2016, 81, 4841 CrossRef CAS PubMed.
- For most recent examples for the enantioselective synthesis of spirooxindoles, see: (a) P. Cheng, W. Guo, P. Chen, Y. Liu, X. Du and C. Li, Chem. Commun., 2016, 52, 3418 RSC; (b) J.-L. Han and C.-H. Chang, Chem. Commun., 2016, 52, 2322 RSC; (c) T.-R. Li, B.-Y. Cheng, S.-Q. Fan, Y.-N. Wang, L.-Q. Lu and W.-J. Xiao, Chem.–Eur. J., 2016, 22, 6243 CrossRef CAS PubMed; (d) N. Kumarswamyreddy and V. Kesavan, Org. Lett., 2016, 18, 1354 CrossRef CAS PubMed; (e) Z. Zhang, W. Sun, G. Zhu, J. Yang, M. Zhang, L. Hong and R. Wang, Chem. Commun., 2016, 52, 1377 RSC; (f) L.-J. Zhang, Y. Wang, X.-Q. Hu and P.-F. Xu, Chem.–Asian J., 2016, 11, 834 CrossRef CAS PubMed; (g) X. Zhao, X. Liu, Q. Xiong, H. Mei, B. Ma, L. Lin and X. Feng, Chem. Commun., 2015, 51, 16076 RSC; (h) R. Zhou, Q. Wu, M. Guo, W. Huang, X. He, L. Yang, F. Peng, G. He and B. Han, Chem. Commun., 2015, 51, 13113 RSC; (i) D. Jiang, S. Dong, W. Tang, T. Lu and D. Du, J. Org. Chem., 2015, 80, 11593 CrossRef CAS PubMed; (j) D.-Y. Zhang, L. Xu, H. Wu and L.-Z. Gong, Chem.–Eur. J., 2015, 21, 10314 CrossRef CAS PubMed; (k) C. Yin, L. Lin, D. Zhang, J. Feng, X. Liu and X. Feng, J. Org. Chem., 2015, 80, 9691 CrossRef CAS PubMed; (l) M.-Y. Wu, W.-W. He, X.-Y. Liu and B. Tan, Angew. Chem., Int. Ed., 2015, 54, 9409 CrossRef CAS PubMed; (m) W. Dai, X.-L. Jiang, Q. Wu, F. Shi and S.-J. Tu, J. Org. Chem., 2015, 80, 5737 CrossRef CAS PubMed; (n) S. Kayal and S. Mukherjee, Org. Lett., 2015, 17, 5508 CrossRef CAS PubMed; (o) L. Wang, D. Yang, D. Li, X. Liu, Q. Zhao, R. Zhu, B. Zhang and R. Wang, Org. Lett., 2015, 17, 4260 CrossRef CAS PubMed; (p) X. Huang, K. Pham, W. Yi, X. Zhang, C. Clamens, J. H. Hyatt, J. P. Jasinsk, U. Tayvah and W. Zhang, Adv. Synth. Catal., 2015, 357, 3820 CrossRef CAS; (q) S. Abbaraju, N. Ramireddy, N. K. Rana, H. Arman and J. C.-G. Zhao, Adv. Synth. Catal., 2015, 357, 2633 CrossRef CAS; (r) J.-Q. Zhao, M.-Q. Zhou, Z.-J. Wu, Z.-H. Wang, D.-F. Yue, X.-Y. Xu, X.-M. Zhang and W.-C. Yuan, Org. Lett., 2015, 17, 2238 CrossRef CAS PubMed; (s) F. Wei, H.-Y. Huang, N.-J. Zhong, C.-L. Gu, D. Wang and L. Liu, Org. Lett., 2015, 17, 1688 CrossRef CAS PubMed; (t) D. Du, Y. Jiang, Q. Xu, X.-Y. Tang and M. Shi, ChemCatChem, 2015, 7, 1366 CrossRef CAS.
- For reviews for the synthesis of enantiomerically enriched spirooxindoles, see: (a) D. Cheng, Y. Ishihara, B. Tan and C. F. Barbas, ACS Catal., 2014, 4, 743 CrossRef CAS; (b) R. Narayan, M. Potowski, Z.-J. Jia, A. P. Antonchick and H. Waldmann, Acc. Chem. Res., 2014, 47, 1296 CrossRef CAS PubMed; (c) L. Hong and R. Wang, Adv. Synth. Catal., 2013, 355, 1023 CrossRef CAS; (d) A. K. Franz, N. V. Hanhan and N. R. Ball-Jones, ACS Catal., 2013, 3, 540 CrossRef CAS; (e) N. R. Ball-Jones, J. J. Badillo and A. K. Franz, Org. Biomol. Chem., 2012, 10, 5165 RSC.
- For reviews of squaramide-based organocatalysis, see: (a) P. Chauhan, S. Mahajan, U. Kaya, D. Hack and D. Enders, Adv. Synth. Catal., 2015, 357, 253 CrossRef CAS; (b) J. Alemán, A. Parra, H. Jiang and K. A. Jørgensen, Chem.–Eur. J., 2011, 17, 6890 CrossRef PubMed.
- For reviews on thiourea-based organocatalysis, see: (a) X. Fang and C.-J. Wang, Chem. Commun., 2015, 51, 1185 RSC; (b) O. V. Serdyuk, C. M. Heckel and S. B. Tsogoeva, Org. Biomol. Chem., 2013, 11, 7051 RSC; (c) W.-Y. Siau and J. Wang, Catal. Sci. Technol., 2011, 1, 1298 RSC; (d) B. Han, J.-L. Li, Y.-C. Xiao, S.-L. Zhou and Y.-C. Chen, Curr. Org. Chem., 2011, 15, 4128 CrossRef CAS; (e) Y. Takemoto, Chem. Pharm. Bull., 2010, 58, 593 CrossRef CAS PubMed; (f) S. J. Connon, Chem.–Eur. J., 2006, 12, 5418 CrossRef PubMed.
- CCDC 1481047 contains the supplementary crystallographic data for this paper.
- (a) S. J. Zuend and E. N. Jacobsen, J. Am. Chem. Soc., 2009, 131, 15358 CrossRef CAS PubMed; (b) S. J. Zuend and E. N. Jacobsen, J. Am. Chem. Soc., 2007, 129, 15872 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1481047. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra14178a |
| This journal is © The Royal Society of Chemistry 2016 |