Preparation of a Sb/Cu2Sb/C composite as an anode material for lithium-ion batteries†
Abstract
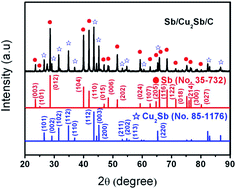
Sb/Cu2Sb/C composites are synthesized via a thermolysis approach with copper citrate and antimony acetate as precursors, respectively. The as-synthesized composites display an initial reversible capacity of 602 mA h g−1 and maintain 461 mA h g−1 after 60 cycles. The excellent electrochemical performance of the Sb/Cu2Sb/C composites can be ascribed to the presence of Sb/Cu2Sb and amorphous carbon layers. The amorphous carbon layers could prevent the damage of electrodes that resulted from large volume expansion. The Sb/Cu2Sb improves their electronic conductivity.

 Please wait while we load your content...
Please wait while we load your content...