Atomic layer deposition of ZnO/TiO2 multilayers: towards the understanding of Ti-doping in ZnO thin films
Abstract
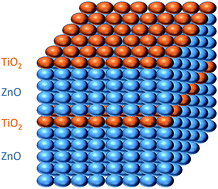
Undoped and Ti-doped ZnO (TZO) films were deposited by atomic layer deposition (ALD). Titanium was introduced by alternating consecutive ZnO cycles between each TiO2 single layer (Ti nominal concentration of 2%). The electrical, optical, morphological, and structural properties of the films (less than 80 nm thick) were tested as a function of the deposition temperature (from 120 to 240 °C). The TZO films deposited at 200 °C showed the lowest resistivity: 3.0 × 10−3 Ω cm. All the resulting TZO films were highly transparent, with an absorbance in the visible range of less than 3%. Thanks to an accurate investigation through transmission electron microscopy we ruled out the well-assessed explanation of an extrinsic doping mechanism of transparent conductive oxide (TCO), suggesting an alternative mechanism. These results demonstrate that the ALD is a promising technique to grow thin TZO films with remarkable electrical and optical properties.

 Please wait while we load your content...
Please wait while we load your content...