Controlled growth of NiMoO4·H2O nanoflake and nanowire arrays on Ni foam for superior performance of asymmetric supercapacitors†
Chen Qing,
Yanan Liu,
Xiaodan Sun,
Xiaxia OuYang,
Hai Wang,
Daming Sun,
Bixiao Wang,
Qin Zhou,
Lifeng Xu and
Yiwen Tang*
Institute of Nano-science and Technology, College of Physical Science and Technology, Central China Normal University, Wuhan, 430079, China. E-mail: ywtang@phy.ccnu.edu.cn
First published on 8th July 2016
Abstract
A facile hydrothermal method is developed for fabrication of large-scale NiMoO4·H2O arrays with robust adhesion on Ni foam. Importantly, the morphology of NiMoO4·H2O can be easily controlled to be nanoflake (H-NF) or nanowire (H-NW) arrays by using NH4F as additive. The obtained nanoflake morphology delivers better electrochemical activity than that of nanowire. The electrochemical performance of anhydrous NiMoO4 arrays obtained by annealing the NiMoO4·H2O has also been investigated for comparison. It is believed that the presence of the structural water of NiMoO4 enhances the capacitive performance by making it a good ionic conductor. Furthermore, an asymmetric supercapacitor (ASC) is constructed using the as-prepared NiMoO4·H2O nanoflake arrays as the positive electrode and activated carbon (AC) as the negative electrode. The optimized ASC with an extended operating voltage range of 0–1.6 V displays excellent electrochemical performance with a high energy density of 53.8 W h kg−1 at a power density of 239 W kg−1 in addition to superior rate capability. Moreover, the H-NF//AC ASC device exhibits remarkable cycling stability with 73.4% specific capacitance retention after 4000 cycles. Our result shows that this unique NiMoO4·H2O nanoflake array is promising for electrochemical energy applications.
1. Introduction
With the increasing energy and power demands in portable electronic devices such as mobile telephones, notebooks and even electric automobiles, energy demands are fast growing and this has attracted much recent attention in the search for high performance materials for electrochemical energy storage. Supercapacitors, as a new class of electronics and electric devices, have attracted upgraded attention due to their fast recharge ability, high energy storage capability, good rate performance and long cycle stability.1,2 Pseudocapacitive-type electrode materials can provide energy density relative to that delivered by electrical double layer capacitors which use carbon-based active materials.3 Furthermore, transition metal oxides (NiCoO4, ZnCoO4, CoMoO4 and MnMoO4) were regarded as excellent candidates for application in pseudocapacitors in consideration of their multiple oxidation states.4–7 Moreover, it has been well established that the electrochemical performance of asymmetric supercapacitors (ASCs) is directly affected by the properties and structures of electrode materials.8–10 Therefore, development of high-energy density ASCs mainly depends on the selection and fabrication of advanced electrode materials.11–13In particular, encouraged by the high electrochemical activity of Ni, nickel molybdate (NiMoO4) has attracted great research interest because of its higher reversible capacity, better structural stability and electrochemical activity.14–16 Although various structures of NiMoO4, including bundles structures,17 1D nanowires structures18 and 2D high-ordered nanoporous structures19 have been developed for pseudocapacitive application, the practical application of NiMoO4 electrodes in ASCs is still hindered by its poor conductivity. As a solution, hierarchical NiMoO4 nanostructures which directly synthesized on conductive substrate (such as Ni foam) was expected to enhance electron conduction and a large open surface.20 Furthermore, the higher dimensional nanoflake structures on Ni foam manifest smaller sized microstructures, which reveal more electroactive sites and shorter diffusion distance.21 The large surface area and high electrical conductivity lead to higher specific capacitance and better cycling stability and rate capability.22–24
Hydrous RuO2 is the typical material with pseudo-capacitance and plentiful research have focused on the combined water of RuO2.25 It was found that the structural water in RuO2 enhanced the diffusion of cations inside the electrode layer which means that more internal hydrated oxide states of Ru atom served as redox active have participated in electrochemistry reaction.26–28 Coincidentally, the structure water entrapped in the layered graphene oxide structure has a dramatic effect on its ionic conductivity, the higher water contents means increasing in ionic conductivity.29
Hence, on the basis of the above ideas and the structure engineering of electrodes, we report in this work the fabrication of NiMoO4·H2O nanoflake (H-NF) and NiMoO4·H2O nanowire arrays (H-NW) directly on Ni foam via hydrothermal method for supercapacitor. The morphology of NiMoO4·H2O could be facilely controlled with or without NH4F in the process of hydrothermal synthesis. The presence of NH4F is considered to play an important role in reducing the nucleation rate and activating substrates.30 The H-NF shows enhanced specific surface, higher conductivity and more excellent cycle performance than that of H-NW. Moreover, the electrochemical ability of anhydrous NiMoO4 nanowire (NW) and nanoflake arrays (NF) are also investigated, which is a primary research on the electrochemical impact of combined water for the NiMoO4. The results show that the H-NF electrode reveals higher specific capacitance and rate ability. Furthermore, the H-NF regarded as an integrated positive electrode and activated carbon (AC) as the negative electrode have been assembled for asymmetric supercapacitor (ASC) device which delivers a high energy density of 53.824 W h kg−1 at a power density of 239.37 W kg−1 (56.389 mA h g−1 at 1 A g−1), approaching the lower end of LIBs. Even at a high power density of 8290.44 W kg−1, the device still has an energy density of 21.187 W h kg−1. The novel capacitive behaviours imply the H-NF are promising energy-storage materials.
2. Experimental section
2.1. Synthesis of hierarchical NiMoO4 H-NW and H-NF
All chemicals were used as received without further purification. Firstly, a 2 × 5 cm2 Ni foam was cleaned by concentrated HCl solution, ethanol, and deionized water with assistance of ultrasonication for several minutes. In the synthesis of NiMoO4 H-NW, the substrate was transferred into a Teflon-lined stainless steel autoclave (with capacity of 90 mL), which contained a homogenous solution of Ni(NO3)2·6H2O (1.5 mmol) and Na2MoO4·2H2O (1.5 mmol) in a mixed solvent of 25 mL H2O and 25 mL ethanol. Afterwards, the autoclave was sealed and maintained at 160 °C for 4 h to synthesize H-NW. Then, the substrate was taken out and cleaned by ultrasonication for several minutes to remove the loosely attached products on the surface before being fully dried at 60 °C overnight under vacuum. The whole process can be easily adjusted to prepare NiMoO4 H-NF by simply adding 5 mmol NH4F into the reaction solution while keeping other conditions unchanged.2.2. Synthesis of hierarchical NiMoO4 NW and NF
After being calcined at 450 °C for 2 h with a temperature ramp rate of 2 °C min−1 in nitrogen atmosphere, the as-prepare NiMoO4 H-NW and H-NF could be converted into HW and HF.2.3. Materials characterization
The products were characterized using X-ray diffraction (XRD, PANalytical Empyrean, Cu Kα radiation; λ = 1.5418 Å), field emission scanning electron microscopy (FESEM, JEOL JSM-6700F, 10 kV), transmission electron microscopy (TEM, Titan G2 60-300), X-ray energy-dispersive spectroscope (EDS) attached to a TEM. X-ray photoelectron spectroscopy (XPS) was carried out using VG Multilab 2000 by setting the peak corresponding to carbon to 284.6 eV. The mass of electrode materials was measured on a BT 125D Balance (Sertorius, d = 0.01 mg). The thermal properties of the fabricated precursors were studied by a Thermo Gravimetric Analyzer (STA 449 F3, NETZSCH, Germany). The specific surface area analysis was conducted using an instrument of Belsorp-miniII (BEL, Japan).2.4. Electrochemical measurements
Electrochemical measurements were carried out in an aqueous KOH electrolyte (2 M) with a three-electrode cell where NiMoO4·H2O directly on Ni foam was used as the working electrode without any polymer binder or conductive additives, a platinum foil as the counter electrode, and a mercuric oxide electrode (Hg/HgO) as the reference electrode. The nominal area of NiMoO4–Ni foam immersed into the electrolyte was controlled to be around 2 cm−2. On average, the mass loading of NiMoO4·H2O nanostructures on Ni foam was around 0.666 mg cm2 for H-NF, 0.519 mg cm−2 for NF, 1.319 mg cm2 for H-NW and 1.124 mg cm−2 for NW, determined by careful weighing after calcination.2.5. Supercapacitor devices
The activated carbon (AC) electrode was first prepared by mixing 80% activated carbon, 10% polytetrafluoroethylene (PTFE) binder, and 10% carbon black (super P), which was coated onto Ni foam as negative electrodes and dried at 80 °C for 10 h. The ASCs device was fabricated using AC as the negative electrode and NiMoO4·H2O-Ni foam (H-NF) as the positive electrode, NKK MPF30AC-100 (Japan) as the separator, and 2 M KOH as electrolyte (denoted as H-NF//AC ASCs device). In order to obtain good electrochemical performance for ASCs, the mass ratio of H-NF and AC on Ni foam was optimized to be around 0.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.4, respectively, in order to balance the charge between positive and negative electrodes. All of the electrochemical measurements were performed on a PARSTAT 4000 test system (Princeton Applied Research).
2.4, respectively, in order to balance the charge between positive and negative electrodes. All of the electrochemical measurements were performed on a PARSTAT 4000 test system (Princeton Applied Research).
3. Results and discussion
Unique hierarchical H-NW and H-NF could be easily grown on Ni foam substrates through a facile hydrothermal route. The schematic formation process of H-NW and H-NF on Ni foam is presented in Fig. 1. During hydrothermal process, the Ni2+ react with MoO42− and form NiMoO4·H2O. H-NW is obtained through the reaction between Ni(NO3)2 and Na2MoO4 in a mixed solvent of H2O and ethanol. But when adding NH4F in the reaction system, the obtained NiMoO4·H2O nanocrystals present nanoflake structure. The NH4F plays an important role on the morphology evolution of the resulting nanocrystals.3.1. Structure and morphology of H-NW and H-NF
The H-NW and H-NF were directly synthesized on Ni foam substrates for one-step hydrothermal process. As displayed filed emission scanning electron microscopy (FE-SEM) images in Fig. 2a and b and its inset, clearly, the well-defined H-NW and H-NF with high density have a smooth surface and a homogeneous distribution of diameter, length and thickness. It is well acknowledged that supersaturation for nucleation and crystal growth is determined by the solvent polarity and solubility.31 When the mixed liquid of H2O and ethanol was employed as solvent, one dimensional H-NW was fully deposited on the surface of Ni foam. While the NH4F was added and keeping other conditions consistent, the growing direction of NiMoO4 transferred to two dimensionality. | ||
| Fig. 2 The high and low magnification SEM images of H-NW (a) and H-NF (b). (c) XRD pattern of H-NW and H-NF synthesized on Ni foam. | ||
The X-ray diffraction (XRD) patterns in Fig. 2c reveals that the as-prepared products are NiMoO4·H2O. The peaks in 26.87° and 29.63° agree well with the reported pattern for NiMoO4·H2O (JCPDS no. 13-0128).15 Obviously, the intensity of the H-NF peaks are lower than that of H-NW. It implies that H-NF has a weak crystallinity, which could be confirmed by the transmission electron microscopy (TEM) images (Fig. 4). Unfortunately, the detailed crystal structure of NiMoO4·H2O is not clear. It is still unknown which plane the obtained spacing corresponds to.14,32
In order to acquire the detail information about the formation of the H-NF and H-NW, we studied their temporal morphology evolution by taking SEM images obtained at different hydrothermal reaction time intervals. Fig. S1(a–d)† shows the SEM images of the Ni foam and morphological evolution of the H-NW obtained with different reaction time (0.5, 1, 2 and 4 h). Initially, a few distinct slender nanowires which were assembled small size particles were found in first 0.5 h (Fig. S1a†). By increasing the reaction time to 1 h, the thin nanowires become much bigger and smoother, and the nanoparticles attached in the surface of nanowires become smaller. A morphological change occurred when the incubation time extended to 2 h. While the reaction time was prolonged to 2 h, the nanowire has preliminarily formed with the nanoparticles disappearing gradually. Ultimately, uniform and strong-adhesion nanowire arrays have developed on the Ni foam after 4 h. The probable scheme process was shown in Fig. S1e.†
As contrast, the morphology evolution of H-NF has also been investigated which is revealed in Fig. S1(f–i).† It can be seen that large amount of uneven flake layer formed on the Ni foam after reacting for 0.5 h. After 1 h, tiny and sparse nanoflake has occupied on the surface of Ni foam. With the time extended to 2 h, uniform and high density flake arrays have formed. With further extension of the reaction time to 4 h, mass of highly ordered flakes became the prevalent products.
To shed light on the role of NH4F in tuning the morphology of NiMoO4·H2O, a series of experiments have been performed by changing the concentration of NH4F for the same reaction time of 4 h. The morphology evolution has been shown in Fig. S1(k–n).† Without the addition of NH4F, the Ni foam is completely covered by H-NW (Fig. S1k†). Under the condition of 2.5 mmol of NH4F, the H-NW were dominant products, only few H-NF has appeared as shown in Fig. S1l.† When the NH4F concentration was increased to 5 mmol, the Ni foam was completely covered by smooth and uniform H-NF (Fig. S1m†). Nevertheless, As the NH4F concentration was increased to 7.5 mmol, sparse and unsatisfied H-NF ware produced. It is apparent that the NH4F plays an essential role in constructing the nanostructure of H-NF. Firstly, NH4F adhere around the active sites of NiMoO4 to restrain the further nucleation.33 Meanwhile, the NH4F result in the formation of abundant grain-boundary of polycrystalline H-NF, which was shown in Fig. 5.34,35 Hence, an appropriate concentration of NH4F is necessary to achieve the high specific surface area and excellent electrochemical ability of H-NF.
3.2. Structure and morphology of NW and NF
The XRD patterns of as-decomposed products of the H-NW and H-NF obtained after 2 h treatment at 450 °C. After complete decomposition at 450 °C, the well-defined H-NW and H-NF were fully transformed to NW and NF, and almost preserved the corresponding morphologies with H-NW and H-NF as shown in Fig. 3a and b and its inset. It is worth to note when lose the crystal water, abundant micro hole appears on the surface of samples. The peaks in 25.554°, 26.579°, 28.470°, 32.140° and 36.905° are confirmed to the (201) (220) (31![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) (112) and (400) diffraction of monoclinic NiMoO4 (JCPDS 45-0142).36,37 Furthermore, the DTA-TG curves of the as-prepared H-NF and H-NW measured in N2 was shown in Fig. S2.† The slight weight loss up to 200 °C was associated with the loss of physisorbed water. When the temperature continued to be improved to almost 450 °C, the structural water released. The TGA–DSC investigation have also demonstrated that the formation of the pores were resulted from the release of the water.
) (112) and (400) diffraction of monoclinic NiMoO4 (JCPDS 45-0142).36,37 Furthermore, the DTA-TG curves of the as-prepared H-NF and H-NW measured in N2 was shown in Fig. S2.† The slight weight loss up to 200 °C was associated with the loss of physisorbed water. When the temperature continued to be improved to almost 450 °C, the structural water released. The TGA–DSC investigation have also demonstrated that the formation of the pores were resulted from the release of the water.
 | ||
| Fig. 3 The high and low magnification SEM images of NW (a) and NF (b). (c) XRD pattern of NW and NF synthesized on Ni foam. | ||
To further illustrate the morphology and microstructure of H-NW, H-HF, NW and NF, TEM and EDS analysis were performed. The TEM image in Fig. 4a shows a typical H-NW, which diameter is about 150 nm. Typically, from its HRTEM image in Fig. 4b, the spacing between two adjacent lattice fringes is 3.26 Å and 2.46 Å, clearly presenting a distinct lattice of NiMoO4·H2O (JCPDS no. 13-0128). Moreover, the corresponding SAED pattern in the inset of Fig. 4a reveals the single-crystalline nature of the H-NW. In comparison, the low and high magnification TEM images of H-NF are shown in Fig. 4c and d. Fig. 4c exhibits a continuous sheet-like structure of NiMoO4·H2O. The HRTEM image in Fig. 4d displays representative polycrystal which are composed by various round single crystal. The adjacent lattice fringes of 2.46 Å and 2.02 Å confirmed the existing of NiMoO4·H2O (JCPDS no. 13-0128). And the corresponding SAED pattern also displayed its polycrystal nature.
The TEM images and corresponding SAED patterns of NW and NF are shown in Fig. 5a–d. Fig. 5a shows the TEM image of a single nanowire with nanopores and the corresponding SAED pattern (the inset of Fig. 5a) implied that the nanowire exhibited single crystal. The HRTEM of NW were observed (Fig. 5b). It can be seen that the nanowire are well-crystallized with clearly resolved lattice fringe of NiMoO4. The adjacent lattice fringes of 3.48 Å confirmed well with the (201) of NiMoO4 (JCPDS 45-0142). On the other hand, the TEM image of NF as shown in Fig. 5c and d confirmed that the nanoflakes were composed of a large number of individual nanocrystals. The SAED pattern of NF (inset in Fig. 5c) shows a well-defined rings which implies that NF is manifested a polycrystalline structure. The adjacent lattice fringes of 2.13 Å in Fig. 5d also confirmed to (222) of NiMoO4 (JCPDS 45-0142) which demonstrates the crystal texture transformation of H-NF to NF. Additionally, the energy dispersive X-ray spectroscopy (EDS) of H-NW and H-NF corresponding to the TEM test was also investigated in Fig. S3a.† Both of H-NW and H-NF show the same result. The Ni, Mo, and O peaks are observed in this spectrum, suggesting that the sample is mainly composed of Ni, Mo, and O. The Cu and C peaks come from the copper sample holder.
In order to get more detailed elemental composition and valance state of the composite, X-ray photoelectron spectroscopy spectra (XPS) was recorded. The result of XPS spectra for H-NW and H-NF as shown in Fig. S3b–d† reveal a common character. The XPS of NiMoO4 are calibrated with reference to C 1s peak at 285 eV. There are three main elements (Ni, Mo and O) detected on the surface of the sample, which is consistent with the EDS result. Fig. S3b† represents the Ni 2p core level spectrum and is reasonably decomposed into four peaks. The binding energy peak at 855.9 eV and its satellite peak at 861.5 eV correspond to Ni 2p3/2, meanwhile, the peaks at 873.1 and 879.5 eV are due to the Ni 2p1/2 level. The gap between main binding energy peaks of Ni 2p3/2 and Ni 2p1/2 is 17.8 eV, indicating the Ni2+ oxidation state.9 In Fig. S3c,† the Mo 3d exhibits two peaks with binding energies of 232 eV and 235.1 eV, corresponding to Mo 3d5/2 and Mo 3d3/2. The separation of 3.1 eV between the two peaks signifies a Mo6+ oxidation state.38 The O 1s (Fig. S3d†) contains two peaks at the binding energies of 530.79 eV and 532.39 eV, representing the O 1s level in NiMoO4·H2O. The peak at 532.3 eV is likely attributed to the O–H group and the water attached to the NiMoO4.39 The results of XPS and EDS demonstrate that both of H-NF and H-NW are NiMoO4·H2O.
To investigate the pseudocapacitive properties of NiMoO4, we apply the H-NW, NW, H-NF and NF supported on Ni foam as an integrated electrode in a three-electrode configuration with 2 M KOH as the electrolyte. Fig. 6a shows the typical cyclic voltammetry (CV) curves of the H-NF, H-NW, NF and NW electrode at the scan rates of 5 mV s−1. The shapes of the CV curves clearly confirm the pseudocapacitive characteristics. Specifically, a distinct pair of redox peaks can be observed within the potential range from 0.1 to 0.7 V versus Hg/HgO at all scan rates, mainly originating from the faradaic redox reactions related to Ni(II) ↔ Ni(III) + e−.9,21 The H-NW and H-NF show higher redox peaks than that of NW and NF. Furthermore, the H-NF also reveal superior performance than H-NW, indicating the H-NF has a higher electrochemical reaction activity. In addition, the typical cyclic voltammetry (CV) curves of the H-NF electrode at different scan rates ranging from 1 to 10 mV s−1 are also shown in Fig. S4a.† At higher scan rates, the anodic/cathodic peaks shift to positive/negative potentials and redox current increases. It may imply that the charge transfer kinetics is the limiting step of the reaction. Fig. S4b† shows the galvanostatic discharge curves of H-NF at current densities ranging from 1 to 12 A g−1, and a distinct plateau region can be observed during the process which implies a typical pseudo-capacitive process.
The electrochemical impedance spectrum (EIS) was further carried out to investigate the electrochemical behaviors of the H-NF, NF, H-NW and the NW electrodes. Fig. 6d shows the Nyquist plots of H-NF, NF, H-NW and the NW electrodes in the frequency range of 0.01–100 kHz after 2500 cycles. Obviously, the H-NF electrode exhibits a lower charge-transfer resistance at the high frequency region, indicating that the fast pseudo-capacitive process at the surface of H-NF. Meanwhile, the H-NF electrode also has a more ideal vertical line (compare with H-NW), which demonstrates it has lower diffusion resistance and high ionic conductivity, indicating the faster ion diffusion of electrolyte.40 When lose the combined water, the NW and NF manifest a lager semicircle which lead to a weak electrochemical behaviour. These results demonstrate the high specific capacitance and rate ability of H-NF, making it as a promising electrode material for actual applications.
Moreover, the calcined NiMoO4 while lose the structural water demonstrated degenerative electrochemical ability. These hydrous regions can allow appreciable good ionic conductor for high energy and high-power electrochemical capacitors.9 The lacking of chemically bound water inhibits proton intercalation and leads to a decrease in specific capacitance.28,41 Meanwhile, as a similar result for RuO2, the well crystallized NiMoO4 structure may have difficulty in hydroxyl expanding or contracting.
Furthermore, in consideration of the H-NW and H-NF show better capacitance ability than NW and NF, we only investigated the BET test of H-NW and H-NF as a support date to testify the reason why H-NF show higher capacitance than H-NW. The porous feature of H-NF guarantees a high specific surface area of around 14.107 m2 g−1 with pore size mostly below 10 nm, which is much larger than that of H-NW (only around 6.4375 m2 g−1) (Fig. S5, ESI†). The relatively high specific surface area is not only increase electrolyte/electrode contact areas but also provide more active sites for fast faradaic redox reactions, hence resulting in a high specific capacitance.
From the above results, one can see that H-NF exhibits more superior electrochemical performance compared with H-NW in term of both specific capacitance and electrochemical activity. This might be attributed to the following structural features. The relatively high specific surface area will not only increase electrolyte/electrode contact areas but also provide more active sites for fast faradaic redox reactions, hence resulting in a high specific capacitance. On the other hand, it can be clearly observed that the nanoflakes are much thinner than the nanowires, which will ensure faster ion and electron transport, and at the same time better accommodate the volume variation during the charge/discharge cycling process. As a result, H-NF exhibits higher specific capacitance and cycling stability.
To further evaluate the H-NF electrode for practical application, an asymmetric supercapacitors (ASC) was fabricated using H-NF and active carbon as the positive and negative electrodes. In order to obtain a capacitor operating in a 1.6 V potential window, it is crucial to control the experimental conditions for the H-NF to work in the potential range from 0 to 0.6 V and the active carbon electrode in the range from 0 to −1.0 V. As for a supercapacitor to satisfy the principle of conservation of electricity, the charge balance will follow the relationship q+ = q−. The charge stored by each electrode depends on the specific capacitance (C), the potential range for the charge/discharge process (ΔE) and the mass of the electrode (m) following the eqn (1):42
| q = C × ΔE × m | (1) |
 | (2) |
However, not all of their surface area contributes to the capacitance, and most of the activated carbons have the capacitance of 50–200 F g−1 in aqueous electrolytes.43 Meanwhile, according to the CV and CP studies in three electrode system with a platinum foil as auxiliary electrode and a mercuric oxide electrode (HgO) as reference electrode for the individual H-NF and AC electrodes. Additional, the capacitance of activated carbon is about 165.37 F g−1 under 5 mV s−1 (Fig. S6, ESI†). On the basis of the specific capacitance values and potential windows found for the H-NF and AC, the mass ratio of the positive electrode to the negative electrode is optimized to about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.075 to balance the charge between positive and negative electrodes. A series of CV measurements with different voltage windows at 5 mV s−1 was performed to estimate the best operating potential of ASC, as shown in Fig. 7a. The stable potential window of the ASC can be extended to as large as 1.6 V without obvious polarization curves. The higher polarization curve in 1.7 V, which imply the water electrolysis which is inadvisable for actual ASCs. The CV curves of the optimized ASC device at various scan rates (ranging from 1 to 20 mV s−1) between 0.0 and 1.6 V are displayed in Fig. 7b, which is noted that contribution from both electric double-layer capacitance and pseudocapacitance can be clearly observed at all scan rates. The further properties of the H-NF//AC ASC are illustrated in Fig. 7c, the galvanostatic charge/discharge curves at various current densities exhibit its excellent charge/discharge ability. The slight nonlinearity of the discharge curves, particularly at lower current densities, suggests some contribution from the redox reaction of metal oxides, which is in congruent with CV results. As a supplementary, the calculated specific capacitance as a function of the discharge current density is plotted in Fig. 7d (based on the total mass of active materials from both positive and negative electrodes). The H-NF//AC ASC reveals a maximum specific capacitance of 126.88 F g−1 at the current density of 1 A g−1. The Fig. 7e reveals the outstanding cycling life (at the current density of 5 A g−1) of our supercapacitor device up to 4000 times, which capacity retention is about 73.4%. We also assembled two supercapacitors in series, and after charging for only 50 s to ∼3.2 V, the device could power both of the 5 mm diameter red LED (2.5 V, 20 mA) and green LED (2.4 V, 20 mA) which shows in the inset of Fig. 7e.
4.075 to balance the charge between positive and negative electrodes. A series of CV measurements with different voltage windows at 5 mV s−1 was performed to estimate the best operating potential of ASC, as shown in Fig. 7a. The stable potential window of the ASC can be extended to as large as 1.6 V without obvious polarization curves. The higher polarization curve in 1.7 V, which imply the water electrolysis which is inadvisable for actual ASCs. The CV curves of the optimized ASC device at various scan rates (ranging from 1 to 20 mV s−1) between 0.0 and 1.6 V are displayed in Fig. 7b, which is noted that contribution from both electric double-layer capacitance and pseudocapacitance can be clearly observed at all scan rates. The further properties of the H-NF//AC ASC are illustrated in Fig. 7c, the galvanostatic charge/discharge curves at various current densities exhibit its excellent charge/discharge ability. The slight nonlinearity of the discharge curves, particularly at lower current densities, suggests some contribution from the redox reaction of metal oxides, which is in congruent with CV results. As a supplementary, the calculated specific capacitance as a function of the discharge current density is plotted in Fig. 7d (based on the total mass of active materials from both positive and negative electrodes). The H-NF//AC ASC reveals a maximum specific capacitance of 126.88 F g−1 at the current density of 1 A g−1. The Fig. 7e reveals the outstanding cycling life (at the current density of 5 A g−1) of our supercapacitor device up to 4000 times, which capacity retention is about 73.4%. We also assembled two supercapacitors in series, and after charging for only 50 s to ∼3.2 V, the device could power both of the 5 mm diameter red LED (2.5 V, 20 mA) and green LED (2.4 V, 20 mA) which shows in the inset of Fig. 7e.
The energy and power densities (E and P) were calculated using equations 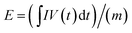 and P = E/Δt, where I is the discharging current, V(t) is discharging voltage excluding the IR drop, dt is time differential, m is the total mass of the two active electrode materials, and Δt is the discharging time. Our asymmetric supercapacitor displays a high energy density of 53.824 W h kg−1 at a power density of 239.37 W kg−1 (56.389 mA h g−1 at 1 A g−1), approaching the lower end of LIBs. Even at a high power density of 8290.44 W kg−1, the device still has an energy density of 21.187 W h kg−1, much superior to that of EDLC at the same power level. The energy and power densities of our supercapacitor are also higher than those of CoMoO4–NiMoO4 nanotube//AC (33 W h kg−1 at 375 W kg−1),44 β-NiMoO4–CoMoO4·xH2O composites//AC (28 W h kg−1 at 100 W kg−1)45 and comparable to those of Co3O4@NiMoO4 nanosheets//AC (37.8 W h kg−1 at 482 W kg−1).46 The volumetric energy and powder densities of our supercapacitor are further shown in Fig. 7f and compared with other data. The exceptional performance of H-NF//AC ASC could be ascribed to the unique structural features. Specifically, the integrated smart architecture could ensure good mechanical adhesion and excellent electrical contact with the conductive substrate, thus leading to enhanced rate performance. Moreover, due to the structure of water and weak crystallized structure, H-NF shows a higher ionic conductor and electrochemical activity resulting in superior specific capacitance and cycling stability.
and P = E/Δt, where I is the discharging current, V(t) is discharging voltage excluding the IR drop, dt is time differential, m is the total mass of the two active electrode materials, and Δt is the discharging time. Our asymmetric supercapacitor displays a high energy density of 53.824 W h kg−1 at a power density of 239.37 W kg−1 (56.389 mA h g−1 at 1 A g−1), approaching the lower end of LIBs. Even at a high power density of 8290.44 W kg−1, the device still has an energy density of 21.187 W h kg−1, much superior to that of EDLC at the same power level. The energy and power densities of our supercapacitor are also higher than those of CoMoO4–NiMoO4 nanotube//AC (33 W h kg−1 at 375 W kg−1),44 β-NiMoO4–CoMoO4·xH2O composites//AC (28 W h kg−1 at 100 W kg−1)45 and comparable to those of Co3O4@NiMoO4 nanosheets//AC (37.8 W h kg−1 at 482 W kg−1).46 The volumetric energy and powder densities of our supercapacitor are further shown in Fig. 7f and compared with other data. The exceptional performance of H-NF//AC ASC could be ascribed to the unique structural features. Specifically, the integrated smart architecture could ensure good mechanical adhesion and excellent electrical contact with the conductive substrate, thus leading to enhanced rate performance. Moreover, due to the structure of water and weak crystallized structure, H-NF shows a higher ionic conductor and electrochemical activity resulting in superior specific capacitance and cycling stability.
4. Conclusion
In summary, we have facilely synthesized NiMoO4 H-NW, H-NF, NW and NF directly on Ni foam as additive-free electrodes for supercapacitors. Among these electrodes, H-NF exhibits the highest specific capacitance, rate capability and cycle performance. With the addition of NH4F in the hydrothermal process, the morphology of NiMoO4·H2O was controlled to be nanoflake with abundant grain-boundary, which exhibited higher specific surface area and electrochemical activity than that of H-NW. Moreover, the structural water in NiMoO4 was demonstrated to be an essential role for its significant electrochemical performance, due its enhanced ionic conductivity. After assembling H-NF as positive electrode and activated carbon as the negative electrode, the aqueous asymmetric supercapacitor exhibits a high energy density of 53.8 W h kg−1 at a power density of 239 W kg−1, and reveal favorable cycling stability with 73.4% specific capacitance retention after 4000 cycles. It is believe that this strategy might be readily extended for preparing other nanostructured electrode materials for advanced supercapacitor applications.Acknowledgements
The authors acknowledge the financial support of the Natural Science Foundation of China (Grant No. 51572102) and Self-determined Research Funds of CCNU from the Colleges' basic Research and Operation of MOE (CCNU15GF006).Notes and references
- P. Simon and Y. Gogotsi, Nat. Mater., 2008, 7, 845–854 CrossRef CAS PubMed.
- J. R. Miller and P. Simon, Science, 2008, 321, 651–652 CrossRef CAS PubMed.
- Y. Zhang, H. Feng, X. Wu, L. Wang, A. Zhang, T. Xia, H. Dong, X. Li and L. Zhang, Int. J. Hydrogen Energ., 2009, 34, 4889–4899 CrossRef CAS.
- Z. Wu, Y. Zhu and X. Ji, J. Mater. Chem. A, 2014, 2, 14759 CAS.
- K. Karthikeyan, D. Kalpana and N. Renganathan, Ionics, 2009, 15, 107–110 CrossRef CAS.
- B. Wang, K. Zhou, S. Jiang, Y. Hu and Z. Gui, Polym. Adv. Technol., 2014, 25, 1419–1425 CrossRef CAS.
- Y. Cao, W. Li, K. Xu, Y. Zhang, T. Ji, R. Zou, J. Yang, Z. Qin and J. Hu, J. Mater. Chem. A, 2014, 2, 20723–20728 CAS.
- X. Zhang, W. Shi, J. Zhu, D. J. Kharistal, W. Zhao, B. S. Lalia, H. H. Hng and Q. Yan, ACS Nano, 2011, 5, 2013–2019 CrossRef CAS PubMed.
- J. Yan, Z. Fan, W. Sun, G. Ning, T. Wei, Q. Zhang, R. Zhang, L. Zhi and F. Wei, Adv. Funct. Mater., 2012, 22, 2632–2641 CrossRef CAS.
- T. Brezesinski, J. Wang, S. H. Tolbert and B. Dunn, Nat. Mater., 2010, 9, 146–151 CrossRef CAS PubMed.
- Y. Lu, X. Liu, W. Wang, J. Cheng, H. Yan, C. Tang, J.-K. Kim and Y. Luo, Sci. Rep., 2015, 5, 16584 CrossRef CAS PubMed.
- Y. Lu, X. Liu, K. Qiu, J. Cheng, W. Wang, H. Yan, C. Tang, J.-K. Kim and Y. Luo, ACS Appl. Mater. Interfaces, 2015, 7, 9682–9690 CAS.
- Y. Lu, H. Yan, K. Qiu, J. Cheng, W. Wang, X. Liu, C. Tang, J.-K. Kim and Y. Luo, RSC Adv., 2015, 5, 10773–10781 RSC.
- M.-C. Liu, L. Kang, L.-B. Kong, C. Lu, X.-J. Ma, X.-M. Li and Y.-C. Luo, RSC Adv., 2013, 3, 6472 RSC.
- H. Wan, J. Jiang, X. Ji, L. Miao, L. Zhang, K. Xu, H. Chen and Y. Ruan, Mater. Lett., 2013, 108, 164–167 CrossRef CAS.
- C. Wang, Y. Xi, C. Hu, S. Dai, M. Wang, L. Cheng, W. Xu, G. Wang and W. Li, RSC Adv., 2015, 5, 107098–107104 RSC.
- M.-C. Liu, L.-B. Kong, C. Lu, X.-J. Ma, X.-M. Li, Y.-C. Luo and L. Kang, J. Mater. Chem. A, 2013, 1, 1380 CAS.
- D. Ghosh, S. Giri and C. K. Das, Nanoscale, 2013, 5, 10428 RSC.
- S. E. Moosavifard, J. Shamsi and M. Ayazpour, Ceram. Int., 2015, 41, 1831–1837 CrossRef CAS.
- D. Guo, P. Zhang, H. Zhang, X. Yu, J. Zhu, Q. Li and T. Wang, J. Mater. Chem. A, 2013, 1, 9024 CAS.
- S. Peng, L. Li, H. B. Wu, S. Madhavi and X. W. D. Lou, Adv. Energy Mater., 2015, 5(2) DOI:10.1002/aenm.201401172.
- D. Cai, D. Wang, B. Liu, Y. Wang, Y. Liu, L. Wang, H. Li, H. Huang, Q. Li and T. Wang, ACS Appl. Mater. Interfaces, 2013, 5, 12905–12910 CAS.
- J. Jiang, Y. Li, J. Liu, X. Huang, C. Yuan and X. W. D. Lou, Adv. Mater., 2012, 24, 5166–5180 CrossRef CAS PubMed.
- C. Zhou, Y. Zhang, Y. Li and J. Liu, Nano Lett., 2013, 13, 2078–2085 CrossRef CAS PubMed.
- J. P. Zheng and Y. Xin, J. Power Sources, 2002, 110, 86–90 CrossRef CAS.
- J. Zheng, P. Cygan and T. Jow, J. Electrochem. Soc., 1995, 142, 2699–2703 CrossRef CAS.
- R. Fu, Z. Ma and J. P. Zheng, J. Phys. Chem. B, 2002, 106, 3592–3596 CrossRef CAS.
- A. Foelske, O. Barbieri, M. Hahn and R. Kötz, Electrochem. Solid-State Lett., 2006, 9, A268 CrossRef CAS.
- W. Gao, N. Singh, L. Song, Z. Liu, A. L. Reddy, L. Ci, R. Vajtai, Q. Zhang, B. Wei and P. M. Ajayan, Nat. Nanotechnol., 2011, 6, 496–500 CrossRef CAS PubMed.
- J. Liu and Z. Huang, J. Phys. Chem. C, 2014, 118, 17870–17877 CAS.
- L. Yu, L. Zhang, H. B. Wu, G. Zhang and X. W. D. Lou, Energy Environ. Sci., 2013, 6, 2664–2671 CAS.
- J. n. L. Brito and A. L. Barbosa, J. Catal., 1997, 171, 467–475 CrossRef CAS.
- Y. Chen, B. Qu, L. Hu, Z. Xu, Q. Li and T. Wang, Nanoscale, 2013, 5, 9812 RSC.
- H. Amani Hamedani, J. A. Khaleel, K.-H. Dahmen and H. Garmestani, Cryst. Growth Des., 2014, 14, 4911–4919 CAS.
- H. Amani Hamedani, S. W. Lee, A. Al-Sammarraie, Z. R. Hesabi, A. Bhatti, F. M. Alamgir, H. Garmestani and M. A. Khaleel, ACS Appl. Mater. Interfaces, 2013, 5, 9026–9033 CAS.
- D. Cai, B. Liu, D. Wang, Y. Liu, L. Wang, H. Li, Y. Wang, C. Wang, Q. Li and T. Wang, Electrochim. Acta, 2014, 115, 358–363 CrossRef CAS.
- Z. Yin, S. Zhang, Y. Chen, P. Gao, C. Zhu, P. Yang and L. Qi, J. Mater. Chem. A, 2015, 3, 739–745 CAS.
- X. Xia, W. Lei, Q. Hao, W. Wang and X. Wang, Electrochim. Acta, 2013, 99, 253–261 CrossRef CAS.
- J. Haetge, I. Djerdj and T. Brezesinski, Chem. Commun., 2012, 48, 6726 RSC.
- C. Guan, J. Liu, C. Cheng, H. Li, X. Li, W. Zhou, H. Zhang and H. J. Fan, Energy Environ. Sci., 2011, 4, 4496 CAS.
- J. Wen, X. Ruan and Z. Zhou, J. Phys. Chem. Solids, 2009, 70, 816–820 CrossRef CAS.
- V. Khomenko, E. Raymundo-Pinero and F. Béguin, J. Power Sources, 2006, 153, 183–190 CrossRef CAS.
- Z. Fan, J. Yan, T. Wei, L. Zhi, G. Ning, T. Li and F. Wei, Adv. Funct. Mater., 2011, 21, 2366–2375 CrossRef CAS.
- Z. Yin, Y. Chen, Y. Zhao, C. Li, C. Zhu and X. Zhang, J. Mater. Chem. A, 2015, 3, 22750–22758 CAS.
- B. Senthilkumar, D. Meyrick, Y.-S. Lee and R. K. Selvan, RSC Adv., 2013, 3, 16542 RSC.
- W. Hong, J. Wang, P. Gong, J. Sun, L. Niu, Z. Yang, Z. Wang and S. Yang, J. Power Sources, 2014, 270, 516–525 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c6ra13483a |
| This journal is © The Royal Society of Chemistry 2016 |





