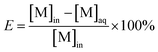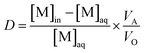A 2-mercaptobenzothiazole-functionalized ionic liquid for selective extraction of Pd(II) from a hydrochloric acid medium†
Hong Zhanga,
Chen Wanga,
Yan Zhenga,
Shubin Wanga,
Zhaohui Liub and
Yanzhao Yang*a
aKey Laboratory for Special Functional Aggregated Materials of Education Ministry, School of Chemistry and Chemical Engineering, Shandong University, Jinan 250100, China. E-mail: yzhyang@sdu.edu.cn
bShandong Academy of Agricultural Science, Jinan, 250100, P. R. China. E-mail: Liuzhaohuinky@163.com
First published on 27th June 2016
Abstract
A novel 2-mercaptobenzothiazole-functionalized ionic liquid (1,10-bis(3-methylimidazolium-1-yl)decane bis(triflic)imide two 2-mercaptobenzothiazolate, {[C10(mim)2]2+2[MBT]−}) with excellent selectivity for Pd(II) extraction was fabricated. Variable factors (e.g., functional ionic liquid concentration, hydrochloric acid concentration, sodium chloride concentration, sodium nitrate concentration and hydrogen ion concentration) were also investigated for extraction experiments. Accordingly, a special complexation extraction mechanism was proposed and confirmed by further experiments. Moreover, higher selectivity towards Pd(II) extraction was realized over other metal ions especially Pt(IV). Furthermore, 0.5 M thiourea/1.0 M HCl was recognized as an efficient stripping agent for the Pd(II) loaded organic phase. Consequently, together with high efficiency and selectivity, the 2-mercaptobenzothiazolate-functionalized ionic liquid can act as a high selective extractant for Pd(II) extraction.
Introduction
As an important noble metal, Pd(II) has received extensive research attention and has been widely used as a versatile material in automobile, pharmaceutical and petroleum industries.1 However, the scarcity of palladium resources in nature makes it valuable to recover palladium from waste liquids and spent catalysts by solvent extraction. Lately, encouraging advances have been made to remove/recover Pd(II) from aqueous phases relying on conventional extractants (e.g., sulfur-containing,2–4 azocalix[4]arene derivative extractant,5 2-hydroxy-5-nonylacetophenone oxime6 and benzoylmethylenetriphenylphosphorane7). However, the efficiency of these extractants has been found to be limited. Hence, looking for an efficient and environmentally-friendly extractant for the separation of Pd(II) from waste liquids has become a focus point.Ionic liquids (ILs), consisting of organic cations and either organic or inorganic anions, are characterized by the advantages of negligible volatility, non-flammability, high thermal stability, and controllable hydrophobic properties,7–13 which make them promising alternatives to conventional extractants. In comparison with ILs, recent advances on ammonium, phosphonium, imidazolium and pyridinium-based functional ionic liquids (FILs) with superior extractability and high selectivity for the extraction of Pd(II) have also been achieved.13–19 However, these poisonous organic solvents always involve in extraction processes acting as organic phase and betray the notion of green chemistry. On the basis of these considerations, it is favourable to bring in hydrophobic ILs as diluting agents in extraction systems. Hydrophobic ILs are often prepared by fluorinated anions, such as hexafluorophosphate (PF6−) or bis(trifluoromethylsulfonyl)−imide (NTf2−).20 In this content, [NTf2] with high solubility is a more appropriate anion to construct hydrophobic ILs as diluting agent. And introduction of a special chelating function group into the cations and/or anions of ILs to build FILs with high solubility/coordinating ability of metal ions could enhance metal affinity to the organic phase and ensure a superior extraction efficiency/yield.21,22 Among these FILs, anionic functional groups23–25 (e.g., dicyanamide, di(2-ethylhexyl)phosphate and (2-ethyhexyl)diglycolamate) substituted ILs in extraction of metals have been intensively studied. 2-Mercaptobenzothiazole (MBT), a bicyclic heteroatomic molecule, exhibits sulfur and nitrogen atoms acting as chelating agent. So, incorporating MBT as extractant with hydrophobic IL phase can strengthen its metal affinity and overcome its insolubility nature. In all cases, further extending the application of MBT-based FILs to the separation of noble metals and revealing the related mechanism involved26 especially when it comes to Pd(II) extraction are still needed.
In our work, {[C10(mim)2]2+2[MBT]−} (Scheme 1) was synthesized and then mixed with [C8mim][NTf2] for Pd(II) extraction. The performance of the system for Pd(II) extraction was studied and the extraction mechanism was also discussed in detail. As a further investigation, the selectivity of this extraction system for other metal ions such as nickel, aluminium, cobalt, platinum, rhodium and ruthenium ions was assessed. According to the results, 2-mercaptobenzothiazolate-functionalized ionic liquid extractant, {[C10(mim)2]2+2[MBT]−}, showed excellent extractability and high selectivity for Pd(II).
Experimental
Materials
PdCl2, H2PtCl6, RuCl3 and RhCl3 were purchased from Guangfu Institute of Fine Chemical (Tianjin, China). 1-Octyl-3-methyl imidazolium bis(trifluoromethylsulfonyl)imide, [C8mim][NTf2], (99%) were obtained from Lanzhou Greenchem ILS, LICP, CAS, China. 1,10-Dichlorodecane was supplied by Dixiai Synthetic Chemistry (Shanghai, China). CoCl2·6H2O, NiCl2·6H2O and AlCl3·6H2O were procured from Kermel Chemical Reagent Tianjin Co., Ltd. (Tianjin, China). All the other reagents (hydrochloric acid, sulfuric acid, sodium chloride, sodium nitrate, ethanol, acetonitrile, acetone, chloroform, etc.) were used without additional purification. Distilled water was used to prepare the aqueous solutions in all experiments.Analysis methods
To confirm the structure and purity of FIL and FIL–Pd complexes, 1H NMR spectra was obtained in DMSO with an AV-300 NMR spectrometer (Bruker, Fällanden, Switzerland). Thermal analysis melting point of FIL was determined by WRR melting point Apparatus (WRR, Shanghai manufacturing physical optics, Instruments Co., Ltd., Shanghai, China). In order to further investigate the mechanism of extraction Pd(II) system, UV spectrophotometry (UV-9000, Shanghai Metash, Instruments Co., Ltd., Shanghai, China) and Fourier transform infrared (FT-IR) spectrophotometry (Tensor27, Bruker corporation, Karlsruhe, Germany) with the scanning wave number ranged from 4000 to 400 cm−1 were involved. The concentration of Pd(II) in the aqueous phase was determined by an atomic absorption spectrophotometer (3150, Precision & Scientific Instrument Shanghai Co., Ltd., Shanghai, China). The organic phase of Pd(II) concentration was calculated by mass balance. Other metals concentrations were tested by inductively coupled plasma atomic emission spectrometer (IRIS Intrepid II XSP, Thermo electron corporation, Boston, US).Synthesis of 2-mecaptobenzothiazolate FIL extractant, {[C10(mim)2]2+2[MBT]−}
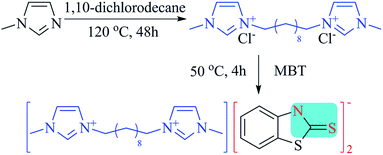
The synthesis of {[C10(mim)2]2+2[MBT]−} was carried out according to the literature procedure.27–29 Briefly, C10(mim)2Cl2 was prepared by adding equal amount of 1,10-dichlorodecane and 1-methylimdazole into 30 mL acetonitrile in a 250 mL round-bottom flask under nitrogen protection at 120 °C for 48 h. The as-formed golden viscous liquid was washed by acetone for six times and dried under vacuum. Brown viscous liquid was obtained at a yield of 90%. 1H NMR (300 M, DMSO-d6, δ/ppm): 9.46 (s, 2H), 7.87 (s, 2H), 7.78 (s, 2H), 4.21 (t, 4H), 3.90 (s, 6H), 1.80 (t, 4H), 1.26 (s, 12H).2-Mercaptobenzothiazole was dissolved in ethyl alcohol and deprotonated with sodium hydroxide in a round-bottom flask. An equimolar amount of C10(mim)2Cl2 was dissolved into the mixture under magnetic stirring at 50 °C for 4 h, and the precipitation was separated by filtration. After removal of ethyl alcohol by rotary evaporation, the obtained product was purified by anhydrous acetone. Yellow solid was dried under vacuum at 0.1 Pa for 24 h at 40 °C (85%, yields). The final product was characterized with 1H NMR (300 M, DMSO-d6, δ/ppm): 1.23 (s, 12H), 1.73 (t, 4H), 3.85 (s, 6H), 4.12 (t, 4H), 6.85 (t, 2H), 7.02 (t, 2H), 7.21 (d, 2H), 7.35 (d, 2H), 7.65 (s, 1H), 7.76 (s, 1H),9.13 (s, 1H). FT-IR (KBr, cm−1) 3412(w), 3145(s), 3061(s), 2929(s), 2859(s), 1666(w), 1567(s), 1453(s), 1378(s), 1239(s), 1166(s), 1128(w), 1076(s), 969(s), 852(w), 756(s). Melting point: 79–81 °C.
Process of Pd(II) precipitation or extraction
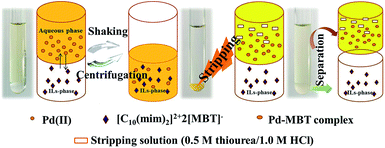
The precipitation process of Pd(II) was performed by mixing a required amount of {[C10(mim)2]2+2[MBT]−} and Pd(II) solution, equilibrating in an orbital shaker. The vibration time was 15 minutes to reach the balance. After the precipitation reaction, an orange suspension was obtained and then centrifuged, washed by water. The FIL–Pd complexes transformed to orange powder after drying.The extraction process was performed as follows: [C8mim][NTf2] mixed with a certain amount of {[C10(mim)2]2+2[MBT]−} was used as organic phase (expressed as {[C10(mim)2]2+2[MBT]−}/[C8mim][NTf2]) and the solution containing Pd(II) or other metal ions (Ni(II), Co(II), Al(III), Pt(IV), Ru(III) and Rh(III)) was employed as aqueous phase. A shaking time of 15 min after the mixture of two phases would be enough to reach balance. The mixture was then centrifuged (1500 rpm, 5 min) and the two phases were separated quickly (Scheme 2). All the experiments were carried out at 298 ± 1 K. Each experiment was performed three times and the standard deviations did not exceed 5%.
The percentage of extraction (E) and distribution ratio (D) were calculated by the following relations:
where VA and VO are the volumes of aqueous phase and organic phase, the volume ratio of VA/VO (RO/A) used in the extraction experiments is 10.
Crystal growth and analysis
Single crystals were grown in a mixture of n-hexane and chloroform (the volume ratio was 1.0) by solvent evaporation. The FIL–Pd complexes were dissolved in the mixed solution. Subsequently, the solution was filtered with a funnel and volatilized in a serum bottle at 298 K for about two weeks. Single crystal X-ray diffraction was performed on an area detecting system (Bruker-Nonius SMART APEX II CCD) and graphite monochromated Mo-Kα radiation (λ = 0.71000 Å). Data collection, integration and absorption corrections were conducted using SMART software and refined with SAINT on all observed reflections.30 Data reduction was implemented with the SAINT software; the data were also revised by Lorentz and polarization effects. Absorption corrections were used by the program SADABS.31 The CCD data were integrated and scaled with the assistance of the Bruker SAINT software package, and the structure was obtained and refined using SHEXTL V6.12.2.32 Non-hydrogen atoms in structure were located from repeating examination of different F-maps following least squares refinement of earlier models. Hydrogen atoms were placed in calculated positions and allowed to ride on the carrier atoms. Crystal data for Pd2[MBT]4: Mr = 949.89, triclinic, space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 7.7864(3) Å, b = 10.872(5) Å, c = 11.188(5) Å, T = 298 K, α = 75.96(5)°, β = 83.81(4)°, V = 868.5(7) Å3, Z = 1, R = 0.0792(1983), wR2 = 0.1750(3456).
, a = 7.7864(3) Å, b = 10.872(5) Å, c = 11.188(5) Å, T = 298 K, α = 75.96(5)°, β = 83.81(4)°, V = 868.5(7) Å3, Z = 1, R = 0.0792(1983), wR2 = 0.1750(3456).
Results and discussion
Influence of {[C10(mim)2]2+2[MBT]−} concentration and initial Pd(II) concentration on Pd(II) extraction
In this study, the influence of {[C10(mim)2]2+2[MBT]−} concentration on the Pd(II) extraction percentage (E) and distribution ratio (D) was investigated. As we could see from Fig. 1a, in the absence of {[C10(mim)2]2+2[MBT]−}, the partitioning of Pd(II) into [C8mim][NTf2] was negligible (E = 6.0%). Therefore, [C8mim][NTf2] just acted as diluting agent. However, as the {[C10(mim)2]2+2[MBT]−} concentration increased, Pd(II) were more inclined to transfer into the hydrophobic IL phase. When the concentration of {[C10(mim)2]2+2[MBT]−} reached 2.1 mM, the extraction percentage of Pd(II) was almost 100%. Further increase of the {[C10(mim)2]2+2[MBT]−} concentration only led to slight increase of extraction percentage. Hence, the concentration of {[C10(mim)2]2+2[MBT]−} (2.1 mM) was chosen for later experiments. Meanwhile, the initial Pd(II) concentration was also investigated. As shown in Fig. 1b, the extraction percentage of Pd(II) was almost 100% as Pd(II) concentration was less than 0.2 mM. On the contrary, the extraction percentage (E) and distribution ratio (D) of Pd(II) decreased sharply when initial Pd(II) concentration surpassed 0.2 mM. Therefore, the initial Pd(II) concentration of 0.2 mM was optimum.Influence of hydrochloric acid concentration, sodium chloride and sodium nitrate concentration on Pd(II) extraction
The influence of hydrochloric acid concentration varying from 0.1 M to 3.0 M on Pd(II) extraction was studied. As was shown in Fig. 2, the extraction percentage of Pd(II) decreased with the increasing concentration of hydrochloric acid. It is evident that at relatively high concentration level of {[C10(mim)2]2+2[MBT]−}, hydrochloric acid concentration between the range from 0.1 M to 1.0 M exerted a minor effect on Pd(II) extraction. However, when the concentration was above 1.0 M, extraction percentage witnessed a clear decrease. Moreover, the {[C10(mim)2]2+2[MBT]−}/[C8mim][NTf2] system for Pd(II) extraction are capable of resisting hydrochloric acid influence, which makes it superior to other extraction systems. However, it is hard to define whether H+ or Cl− affects the extraction behavior of Pd(II). In order to examine the effect of H+ and Cl− separately, the influence of H+ was studied at a fixed 3.0 M Cl− concentration, and the H+ ions concentrations were varied between 0.1 M and 3.0 M using hydrochloride acid, the intermediate Cl− concentrations were adjusted by sodium chloride contents. As seen from Fig. 2, the extraction percentage of palladium did not change with the increase in H+ concentration, suggesting that the participation of protonated extractant in Pd(II) extraction is negligible. Subsequently, the effect of Cl− concentration varied from 0.1 M to 3.0 M on Pd(II) extraction was investigated (presented in Fig. 2). The result showed that the extraction percentage of Pd(II) decreased with increasing Cl− concentration. It is indicated that Cl− prevents Pd(II) extraction from aqueous phase to organic phase in [{[C10(mim)2]2+2[MBT]−}/[C8mim][NTf2] system. On the other hand, the influence of sodium nitritate was also studied on Pd(II) extraction. However, the percentage extraction of Pd(II) increased with the increasing concentration of sodium nitritate, suggesting that ionic strength may be the main factor promoting the extraction percentage of Pd(II).Extraction mechanism
 | ||
| Fig. 3 (a) UV-vis spectra of {[C10(mim)2]2+2[MBT]−}, crystal and IL-phase after extraction. (b) UV-vis spectra of the aqueous phase after extraction and C10(mim)2Cl2 solution. | ||
To further investigate the mechanism of Pd(II) extraction by the {[C10(mim)2]2+2[MBT]−}/[C8mim][NTf2] system, the FT-IR spectra and 1H NMR spectra of Pd–MBT complexes were recorded in order to confirm any structural change in the FIL. The results were shown in Fig. 4a and b. The characteristic peaks of the chemical bonds which belong to {[C10(mim)2]2+2[MBT]−} were listed as follows. The peaks observed at ∼3145, 2929, 2859, 1666 and 1567 cm−1 could be assigned to imazole cation. The peaks at ∼1453, 1378 cm−1 were associated with the vibrations of the C–N–H group, while the peaks at ∼1239, 1166 and 1076 cm−1 were typical of the N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S group vibrations.33 However, after extraction, imidazole ring vibration peaks disappeared and many peaks which belong to [MBT]− have changed clearly. In comparison with the pure {[C10(mim)2]2+2[MBT]−}, the peaks of N–C–H group shifted from 1453 cm−1 and 1378 cm−1 to 1449 cm−1 and 1395 cm−1, respectively; the peaks of N–C
S group vibrations.33 However, after extraction, imidazole ring vibration peaks disappeared and many peaks which belong to [MBT]− have changed clearly. In comparison with the pure {[C10(mim)2]2+2[MBT]−}, the peaks of N–C–H group shifted from 1453 cm−1 and 1378 cm−1 to 1449 cm−1 and 1395 cm−1, respectively; the peaks of N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S also shifted from 1239 cm−1 and 1166 cm−1 to 1245 cm−1 and 1082 cm−1, suggesting that Pd(II) has been coordinated with nitrogen and sulfur of the [MBT]− anion.
S also shifted from 1239 cm−1 and 1166 cm−1 to 1245 cm−1 and 1082 cm−1, suggesting that Pd(II) has been coordinated with nitrogen and sulfur of the [MBT]− anion.
1H NMR spectra also supplied ESI† of FIL and Pd–MBT complexes. As seen in Fig. 4b, the peaks of cation of FIL have vanished which were consistent with the FT-IR spectra, the chemical shifts of hydrogen atoms related to benzene ring (E and H) altered as follows: F–F′, 6.854 → 7.272; G–G′, 7.019 → 7.474; H–H′, 7.204 → 7.709; E–E′, 7.351 → 8.948. It is estimated that the coordination of Pd(II) with nitrogen atom or sulfur atom resulted in the hydrogen atoms change in their chemical shifts. Taken together, the results suggested that Pd(II) entered into the IL phase via the formation of Pd–MBT complexes.
In order to get accurate information about the mode of coordination and calculate the complexing number, we employed the method of single crystal X-ray diffraction to analyse the structure of FIL–Pd complexes (shown in Fig. 5). The result showed that the stoichiometric number of FIL and Pd was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 during the extraction procedure. Two Pd(II) atoms each shares two bonding nitrogens and two bonding sulfurs which were respectively provided by four isolated MBT molecules, constructing a lantern-typed in the triclinic space group P
1 during the extraction procedure. Two Pd(II) atoms each shares two bonding nitrogens and two bonding sulfurs which were respectively provided by four isolated MBT molecules, constructing a lantern-typed in the triclinic space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . It is obviously revealed that Pd(II) simultaneously coordinated with nitrogen atom and sulfur atom. The lantern-typed complexes had an inversion center at the mid-point of the Pd(II)⋯Pd(II) bond, the Pd(II)⋯Pd(II) bond length (2.759 Å) completely agreed well with the value of 2.75 Å observed in metallic Pd.34 The four MBT ligands were in a syn–anti–syn–anti conformation. In addition, N1–C7 bond length (1.3266 Å) was appreciably shorter than the C–N single bond length (1.47 Å),35 yet longer than C
. It is obviously revealed that Pd(II) simultaneously coordinated with nitrogen atom and sulfur atom. The lantern-typed complexes had an inversion center at the mid-point of the Pd(II)⋯Pd(II) bond, the Pd(II)⋯Pd(II) bond length (2.759 Å) completely agreed well with the value of 2.75 Å observed in metallic Pd.34 The four MBT ligands were in a syn–anti–syn–anti conformation. In addition, N1–C7 bond length (1.3266 Å) was appreciably shorter than the C–N single bond length (1.47 Å),35 yet longer than C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond length (1.29 Å). The difference in bond length might resulted from charge delocalization of two sulphur atoms to nitrogen atom, presenting the resonating structure of 2-mercaptobenzothiazole. The S1–C7 single bond length (1.7660 Å) agreed well with those correlative compound, which is slightly longer than S2–C7 (1.7202 Å) double bond length, as the long pairs on the sulfur atom present in the skeleton of the ring which have very weak coordinating ability. Therefore, palladium was coordinated with the double bond sulfur instead of the single bond sulfur. As seen from the structure of the molecular, the N2–Pd1–S3 angle (179.255°) and the N1–Pd1–S2a angle (178.613°) suggested that the Pd1 atom and the N1, N2, S2a, S3a atoms were coplanar. All the aforementioned data was determined by X-ray diffraction (displayed in Table 1). According to the discussion, the extraction mechanism of Pd(II) using {[C10(mim)2]2+2[MBT]−} system can be represented by the following equation:
N double bond length (1.29 Å). The difference in bond length might resulted from charge delocalization of two sulphur atoms to nitrogen atom, presenting the resonating structure of 2-mercaptobenzothiazole. The S1–C7 single bond length (1.7660 Å) agreed well with those correlative compound, which is slightly longer than S2–C7 (1.7202 Å) double bond length, as the long pairs on the sulfur atom present in the skeleton of the ring which have very weak coordinating ability. Therefore, palladium was coordinated with the double bond sulfur instead of the single bond sulfur. As seen from the structure of the molecular, the N2–Pd1–S3 angle (179.255°) and the N1–Pd1–S2a angle (178.613°) suggested that the Pd1 atom and the N1, N2, S2a, S3a atoms were coplanar. All the aforementioned data was determined by X-ray diffraction (displayed in Table 1). According to the discussion, the extraction mechanism of Pd(II) using {[C10(mim)2]2+2[MBT]−} system can be represented by the following equation:
| 2PdCl42−(aq) + 2{[C10(mim)2]2+2[MBT]−}(IL) ⇔ [Pd(MBT)2]2(IL) + 2[C10(mim)2Cl2](aq) + 2Cl−(aq) | (1) |
| A1 | A2 | Bond A1–A2 | A1 A2 A3 | Angle A2 A1 A3 |
|---|---|---|---|---|
| Pd1 | –N1 | 2.0830(7) | Pd1–N1–N2 | 90.994(16) |
| Pd1 | –N2 | 2.0878(6) | Pd1a–N1a–S3 | 89.614(14) |
| Pd1 | –S3 | 2.2963(7) | Pd1–N1–S2a | 178.613(22) |
| Pd1 | –S2 | 2.3002(8) | Pd1–N2–S2a | 90.359(14) |
| S1 | –C6 | 1.7323(6) | Pd1–N2–S3a | 179.255(14) |
| S1 | –C7 | 1.7660(6) | Pd1–S3–S2 | 89.031(14) |
| S2 | –C7 | 1.7202(4) | S1–C6–C7 | 90.080(19) |
| N1 | –C7 | 1.3266(5) | N1–C7–C5 | 112.220(20) |
Conclusions
The extraction behavior of Pd(II) was investigated using {[C10(mim)2]2+2[MBT]−} for the first time. The extraction efficiency of palladium was found to be nearly 100%. Low concentration of hydrochloric acid was in favor of palladium extraction to the organic phase. The palladium extraction is independent of H+ concentration, while chloride ion concentration exerts a negative effect on palladium extraction, nitrate ion produce a positive effect on palladium extraction. The mechanism of the palladium extraction system is a special complexation mechanism in which two palladium atoms are coordinated with four anions of the FLI. Under the optimum condition, the {[C10(mim)2]2+2[MBT]−}/[C8mim][NTf2] system shows higher selectivity for Pd(II) extraction than other metals. Stripping of Pd(II) from loaded organic phase was successful using the combination of 0.5 M thiourea in 1.0 M hydrochloride acid with recovery percentage about 99%. Hence, the 2-mercaptobenzothiazolate functional ionic liquid has excellent extraction efficiency and high selectivity for Pd(II) extraction.Acknowledgements
This work was supported by the Natural Science Foundation of China (21276142, 21476129), the Science & Technology Development Projects of Shandong Province (grant no. 2014GSF117024) and Special Fund for “Taishan Scholar” construction engineering “agricultural nonpoint source pollution prevention and control” position.Notes and references
- F. L. Bernardis, R. A. Grant and D. C. Sherrington, React. Funct. Polym., 2005, 65, 205–217 CrossRef CAS.
- H. Narita, K. Morisaku, K. Tamura, M. Tanaka, H. Shiwaku, Y. Okamoto, S. Suzuki and T. Yaita, Ind. Eng. Chem. Res., 2014, 53, 3636–3640 CrossRef CAS.
- M. Rajiv Gandhi, M. Yamada, Y. Kondo, R. Sato and F. Hamada, Ind. Eng. Chem. Res., 2014, 53, 2559–2565 CrossRef CAS.
- J. Traeger, J. Koenig, A. Staedtke and H.-J. Holdt, Hydrometallurgy, 2012, 127, 30–38 CrossRef.
- R. Tayeb, A. Zaghbani, S. B. Maamar, F. Meganem, F. Vocanson and M. Dhahbi, C. R. Chim., 2009, 12, 1275–1279 CrossRef CAS.
- M. Mohan Raj, A. Dharmaraja, K. Panchanatheswaran, K. A. Venkatesan, T. G. Srinivasan and P. R. VasudevaRao, Hydrometallurgy, 2006, 84, 118–124 CrossRef.
- X. Sun, H. Luo and S. Dai, Chem. Rev., 2011, 112, 2100–2128 CrossRef PubMed.
- X. Han and D. W. Armstrong, Acc. Chem. Res., 2007, 40, 1079–1086 CrossRef CAS PubMed.
- M. P. Jensen, J. Neuefeind, J. V. Beitz, S. Skanthakumar and L. Soderholm, J. Am. Chem. Soc., 2003, 125, 15466–15473 CrossRef CAS PubMed.
- J. G. Huddleston, A. E. Visser, W. M. Reichert, H. D. Willauer, G. A. Broker and R. D. Rogers, Green Chem., 2001, 3, 156–164 RSC.
- K. N. Marsh, A. Deev, A. C. Wu, E. Tran and A. Klamt, Korean J. Chem. Eng., 2002, 19, 357–362 CrossRef CAS.
- H. Zhao, S. Xia and P. Ma, J. Chem. Technol. Biotechnol., 2005, 80, 1089–1096 CrossRef CAS.
- Y. Tong, C. Wang, J. Li and Y. Yang, Hydrometallurgy, 2014, 147–148, 164–169 CrossRef CAS.
- A. Cieszynska and M. Wiśniewski, Hydrometallurgy, 2012, 113, 79–85 CrossRef.
- A. Cieszynska and M. Wisniewski, Sep. Purif. Technol., 2011, 80, 385–389 CrossRef CAS.
- P. Giridhar, K. Venkatesan, T. Srinivasan and P. V. Rao, Hydrometallurgy, 2006, 81, 30–39 CrossRef CAS.
- J. Yang, F. Kubota, Y. Baba, N. Kamiya and M. Goto, Solvent Extr. Res. Dev., Jpn., 2014, 21, 129–135 CrossRef CAS.
- R. Kumaresan, R. Jain, K. A. Venkatesan, M. P. Antony and B. M. Bhanage, J. Radioanal. Nucl. Chem., 2014, 303, 1047–1052 CrossRef.
- J.-M. Lee, Fluid Phase Equilib., 2012, 319, 30–36 CrossRef CAS.
- R. P. Swatloski, J. D. Holbrey and R. D. Rogers, Green Chem., 2003, 5, 361–363 RSC.
- J. H. Davis Jr, Chem. Lett., 2004, 33, 1072–1077 CrossRef.
- A. E. Visser, R. P. Swatloski, W. M. Reichert, R. Mayton, S. Sheff, A. Wierzbicki, J. H. Davis Jr and R. D. Rogers, Chem. Commun., 2001, 135–136 RSC.
- A. Rout and K. Binnemans, Ind. Eng. Chem. Res., 2014, 53, 6500–6508 CrossRef CAS.
- A. Messadi, A. Mohamadou, S. Boudesocque, L. Dupont and E. Guillon, Sep. Purif. Technol., 2013, 107, 172–178 CrossRef CAS.
- X. Sun and K. E. Waters, ACS Sustainable Chem. Eng., 2014, 2, 1910–1917 CrossRef CAS.
- M. L. Dietz, J. A. Dzielawa, I. Laszak, B. A. Young and M. P. Jensen, Green Chem., 2003, 5, 682 RSC.
- J. Pernak, F. Walkiewicz, M. Maciejewska and M. Zaborski, Ind. Eng. Chem. Res., 2010, 49, 5012–5017 CrossRef CAS.
- Q. Liu, F. van Rantwijk and R. A. Sheldon, J. Chem. Technol. Biotechnol., 2006, 81, 401–405 CrossRef CAS.
- J. L. Anderson, R. Ding, A. Ellern and D. W. Armstrong, J. Am. Chem. Soc., 2005, 127, 593–604 CrossRef CAS PubMed.
- Bruker, SMART, SAINT and SADABS, Bruker AXS Inc., Madison, Wisconsin, USA, 1998 Search PubMed.
- G. M. Sheldrick, SHELXS-97, Program for X-ray Crystal Structure Determination, University of Gottingen, Germany, 1997 Search PubMed.
- SHELXTL/PC, Version 6.12 for Windows XP, Bruker AXS Inc., Madison, Wisconsin, USA, 2001 Search PubMed.
- A. K. Rai, R. Singh, K. Singh and V. Singh, Spectrochim. Acta, Part A, 2006, 63, 483–490 CrossRef PubMed.
- E. J. Gao, K. H. Wang, X. F. Gu, Y. Yu, Y. G. Sun, W. Z. Zhang, H. X. Yin, Q. Wu, M. C. Zhu and X. M. Yan, J. Inorg. Biochem., 2007, 101, 1404–1409 CrossRef CAS PubMed.
- Z. H. Liu, C. Y. Duan, J. Hu and X. Z. You, Inorg. Chem., 1999, 38, 1719–1724 CrossRef CAS PubMed.
Footnote |
| † CCDC 1450377. For crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra12002d |
| This journal is © The Royal Society of Chemistry 2016 |