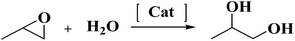Epoxide hydrolysis and alcoholysis reactions over crystalline Mo–V–O oxide†
Xiaochen Zhanga,
Min Wanga,
Chaofeng Zhangab,
Jianmin Lua,
Yehong Wanga and
Feng Wang*a
aState Key Laboratory of Catalysis, Dalian National Laboratory for Clean Energy, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian 116023, Liaoning, China. E-mail: wangfeng@dicp.ac.cn; Fax: +86-411-84379762; Tel: +86-411-84379762
bGraduate University of Chinese Academy of Sciences, Beijing 100049, China
First published on 13th July 2016
Abstract
Crystalline Mo–V–O oxides have been used as a catalyst for the hydrolysis and alcoholysis of propylene oxide to diols and ethers, respectively. Relationships between the active crystal facet, the acidity of Mo–V–O catalysts and the activity have been established. Our results indicate that the a–b plane is the active facet for the hydrolysis reaction.
Mo–V–O oxides represent rare zeolite-like oxides with crystalline structure but containing transition metals. These oxides have received increasing attention in recent years,1–8 focusing on the structure and properties of metal doped Mo–V–O oxides, such as Ni, Zn, Cu, Fe, Te, Ta, Ga, Sb, Zr, Sr, W, Bi, and Ln.9–20 These materials have a layered structure in the direction of the c-axis and consist of uniform five-, six-, and seven-membered rings on the a–b plane.21
In view of their unique structures, more interest has been paid to the catalytic applications of Mo–V–O materials in the selective oxidation of hydrocarbons. For example, the ammoxidation of propane to acrylonitrile and propane oxidation to acrylic acid,1–3,11,13,21–32 selective oxidation of isobutane14,19,33–37 and ethane,9,12,28,38–44 and the oxidation of acrolein to acrylic acid.5,28,45–47 Moreover, Guliants et al.31,48 investigated the catalytic behaviors of Mo–V–O in the vapor phase oxidation reaction through chemisorption method. The applications of Mo–V–O materials mostly used in vapour phase reactions. Recently, we reported the oxidation of alcohols in liquid phase.49–52 Sergio Castillón et al.53 reported epoxidation/alcoholysis and epoxidation/hydrolysis of glucal and galactal derivatives by Mo catalyst. These studies expand the application of Mo–V–O-based materials into broad catalytic processes.
As it is well known, epoxides are important organic chemicals and intermediates. Ring-opening of epoxides is an important route to dihydric alcohol derivatives which are the precursors for cosmetics, pharmaceuticals and other products.54–56 Generally, the procedure can be catalyzed by basic or acid conditions.
On the basis of the special performance of Mo–V–O materials, we here aim to correlate the feature with the ring-opening of epoxides. The Mo–V–O materials were employed as the catalyst in the hydrolysis and alcoholysis of propylene oxide and gave a good conversion and selectivity. The apparent activation energy was calculated to be 50.3 kJ mol−1. We verified the a–b plane possess the most amount of acid sites and is an active crystal facet. A wide range of epoxides were converted to the corresponding dihydric alcohol derivatives with moderate to excellent yields.
We synthesized the Mo–V–O materials by hydrothermal method according to literature38 firstly. The orthorhombic, trigonal and tetragonal Mo–V–O materials were synthesized from a reaction mixture of aqueous solution of (NH4)6Mo7O24, and VOSO4·nH2O under some conditions. By controlling the pH value of the precursor solution, the orthorhombic or trigonal Mo–V–O was selectively synthesized. The tetragonal material was prepared by heat-treatment of the orthorhombic material. The obtained crude materials was purified with aqueous oxalic acid. The crystalline structure of the materials was studied by XRD characterization (Fig. 1). The XRD peaks around 10° for all the materials are ascribed to the structure of a–b plane. Diffraction peaks at 22° and 45° indicates that the catalysts are a kind of layered-type material in the c axis. These two peaks were ascribed to (001) and (002) plane reflections.38
 | ||
| Fig. 1 XRD patterns of different crystalline Mo–V–O. A: orthorhombic, B: tetragonal, C: trigonal (milling time-10 min), D: trigonal (milling time-5 min), E: trigonal (milling time-3 min). | ||
Then the hydrolysis reaction (see the ESI†) of propylene oxide was used as the model reaction to investigate the catalytic performance of crystalline Mo–V–O catalyst. The reaction did not happen in the absence of catalyst (Table 1, entry 1). The Orthorhombic, trigonal and tetragonal Mo–V–O were used as catalyst for this reaction. Trigonal Mo–V–O showed highest activity in the propylene oxide hydrolysis reaction.
| Catalyst | Conv./% | Sel./% | |
|---|---|---|---|
| a Reaction conditions: 0.01 g catalyst, 3 mmol propylene oxide, 1 mL H2O, 2 h, 40 °C. | |||
| 1 | None | 0 | 0 |
| 2 | Orthorhombic | 27.1 | >99 |
| 3 | Tetragonal | 41.5 | 97 |
| 4 | Trigonal | 53 | 97 |
Using the trigonal Mo–V–O as the catalyst, the optimization of propylene oxide hydrolysis condition was investigated (Fig. 2). The conversion of propylene increases with reaction time. 88% conversion was obtained at 25 °C for 12 h. When the reaction increases to 40 °C and 55 °C, 95% conversion were obtained at 8 h and 3 h, respectively. The selectivity is above 95% at the range of temperature.
 | ||
| Fig. 2 The reaction time profiles over trigonal Mo–V–O in propylene oxide hydrolysis. Reaction conditions: 0.01 g catalyst, 3 mmol propylene oxide, 1 mL H2O. | ||
Based on above results, the reaction kinetics was studied at 25–55 °C (Fig. 3). The hydrolysis reaction is shown as follows:
 | ||
| Fig. 3 (a) The kinetic analysis of propylene oxide hydrolysis in the presence of the trigonal Mo–V–O catalyst. (b) Arrhenius plot. The X-axis is multiplied by 1000. | ||
The total volume of the reaction mixture in all kinetic experiments was 1 mL. The concentration of water and the amount of catalyst remained constant through the reaction. The kinetic study indicates a pseudo-first-order reaction (Fig. 3a). The apparent reaction rate constants at 25, 40, and 55 °C were calculated from the slopes of plots to be 9.3 × 10−5, 1.82 × 10−4, 5.86 × 10−4 mol−1 L−1 s−1, respectively. From the Arrhenius plot shown in Fig. 3b, the activation energy Ea was evaluated to be 50.3 kJ mol−1.
In light of the results of the propylene oxide hydrolysis above, the catalytic properties of Mo–V–O catalyst for hydrolysis of other epoxides were investigated as listed in Table 2. During these studies, the catalyst exhibit 99% conversion and above 95% selectivity to corresponding ring-opening reaction for styrene oxides and cyclohexene oxides within 4 h at room temperature (Table 2, entries 1–2). Higher temperature and longer reaction time are needed for the hydrolysis of 2-butyloxirane and chloropropylene oxide (Table 2, entries 3–5). Besides, 1,3-dioxane show 42% conversion. And there is no obvious increase with the temperature rise from 150 °C to 180 °C which may due to the symmetrical structure (Table 2, entries 7–8). 4-Methyl-1,3-dioxane exhibit 55% conversion at 180 °C within more long time.
| Entry | Substrate | Time/h | Temp./°C | Product | Conv./% | Sel./% |
|---|---|---|---|---|---|---|
| a Reaction conditions: 0.01 g trigonal Mo–V–O, 0.2 mL substrate, 1 mL H2O. a: 0.02 g catalyst, 1.5 mmol substrate, 1 mL H2O. | ||||||
| 1 | 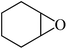 |
4 | 25 | 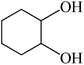 |
99 | >99 |
| 2 | 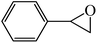 |
4 | 25 | 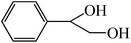 |
>99 | 95 |
| 3 | 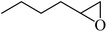 |
4 | 80 | 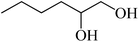 |
>99 | >99 |
| 4 | 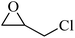 |
4 | 80 | 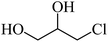 |
84 | 90 |
| 5 | 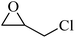 |
6 | 80 | 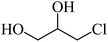 |
94 | 89 |
| 6 | 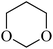 |
4 | 100 |  |
5 | >99 |
| 7 | 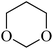 |
3 | 150 |  |
42 | >99 |
| 8 | 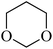 |
3 | 180 |  |
42 | >99 |
| 9a | 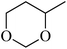 |
3 | 180 | 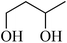 |
25 | >99 |
| 10a | 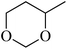 |
24 | 180 | 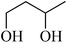 |
55 | >99 |
In addition, considering the similarity between the hydrolysis and the alcoholysis, the alcoholysis of propylene oxide with a series of alcohols over trigonal Mo–V–O were investigated (Table 3). We firstly optimized the reaction conditions (see Table S1†). Increase the catalyst loading, and reaction temperature and time can promote propylene oxide conversion. The optimized results were provided in Table 3. It is noted that the catalyst showed a conversion above 90% and a higher selectivity of two main products in the methanol and ethanol alcoholysis, respectively (Table 3, entries 1–2). But, the conversion decreased with increasing the carbon chain of the alcohols. The conversion for octanol is 64% (Table 3, entry 7). On the other hand, compared to the 1-butanol (94% conversion), a 34% conversion was obtained for t-butanol (Table 3, entry 4). The lower conversion may be due to the steric hindrance. For cyclohexanol and benzyl alcohol, 57% and 58% conversion of propylene oxide were obtained (Table 3, entries 8–9).
| Entry | Substrate | Product | Conv./% | Sel./% |
|---|---|---|---|---|
| a Reaction conditions: 3 mmol propylene oxide, 1 mL alcohol, 0.015 g trigonal Mo–V–O, 100 °C, 12 h. | ||||
| 1 | MeOH | 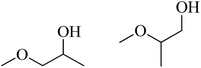 |
>99 | 39/54 |
| 2 | EtOH | 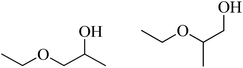 |
94 | 40/50 |
| 3 | n-BuOH | 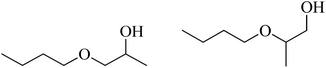 |
82 | 30/55 |
| 4 | t-BuOH | 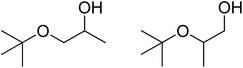 |
34 | 35/42 |
| 5 | n-Pentanol | 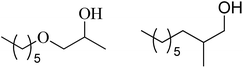 |
76 | 31/61 |
| 6 | n-Hexanol | 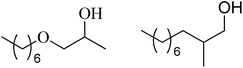 |
74 | 30/62 |
| 7 | n-Octanol | 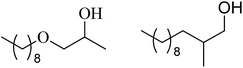 |
64 | 29/63 |
| 8 | Cyclohexanol | 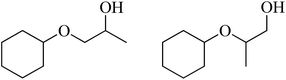 |
57 | 32/57 |
| 9 | Benzyl alcohol | 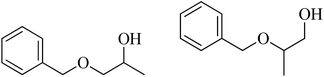 |
58 | 37/53 |
To verify the structure of the catalyst material after the reaction, the FT-IR spectrum and XRD characterization were carried out (Fig. 4 and Fig. S1†). The bands at 906 cm−1 ascribable for V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 870 cm−1 for Mo
O, 870 cm−1 for Mo![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 810, 712 and 646 cm−1 for Mo–O–Mo and 599 cm−1 for V–O–Mo were observed in both the fresh and used catalyst.18 And, there is no change in the diffraction peaks for the fresh and used catalysts. Besides, the catalyst after the reaction still keep the morphology of rod crystal, and there is no obvious change in the average particle size (Fig. S2†). This indicated that the catalyst crystal structure was not changed during the reaction.
O, 810, 712 and 646 cm−1 for Mo–O–Mo and 599 cm−1 for V–O–Mo were observed in both the fresh and used catalyst.18 And, there is no change in the diffraction peaks for the fresh and used catalysts. Besides, the catalyst after the reaction still keep the morphology of rod crystal, and there is no obvious change in the average particle size (Fig. S2†). This indicated that the catalyst crystal structure was not changed during the reaction.
To ascertain the active crystal facet, a series of experiment were carried. Milling the Mo–V–O materials was used to create more a–b plane. The morphology of trigonal with different milling time was shown in the SEM images (Fig. 5). The rod crystal in the c-axis was grinded into shorter rod, the average particle size decreased from 1300 nm to 280 nm, thus exposing more a–b planes. The diffraction peaks of the trigonal Mo–V–O corresponding to the crystal at 4.9°, 8.4°, and 9.6°, were ascribed to the plane of (100), (110), and (200), respectively (Fig. 1). It can be seen that the crystalline structure did not change significantly after grinding, which indicated that grinding did not destroy the crystal structure. As reported, the a–b crystal mainly consists of 6- or 7-member rings (6MRs and 7MRs) where highly dense unsaturated metal cation centers and oxygen anions may function as catalytic active sites for many reactions.50 The Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V atoms ratio of trigonal Mo–V–O was determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES) and was about 2.4
V atoms ratio of trigonal Mo–V–O was determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES) and was about 2.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The structure of trigonal Mo–V–O have been studied in detail by Ueda18 and provided in the ESI (Fig. S3†). Aiming to create more a–b plane, we then milled Mo–V–O material and test the activity in the epoxide hydrolysis reaction. As shown in SEM, the Mo–V–O became shorter with increasing the milling time. The milling of the Mo–V–O material to short rod can exposed more a–b plane. In the reaction, the yield increases with milling time. Considering that hydroloysis reaction is catalyzed by acid sites, we investigated the acidity of Mo–V–O by NH3-TPD. From the Fig. 6, the amount of acidity increase with the milling time, indicating that acid sites is mainly located on the a–b plane. Thus, we postulate that hydrolysis reaction takes place on the acid sites on the a–b plane.
1. The structure of trigonal Mo–V–O have been studied in detail by Ueda18 and provided in the ESI (Fig. S3†). Aiming to create more a–b plane, we then milled Mo–V–O material and test the activity in the epoxide hydrolysis reaction. As shown in SEM, the Mo–V–O became shorter with increasing the milling time. The milling of the Mo–V–O material to short rod can exposed more a–b plane. In the reaction, the yield increases with milling time. Considering that hydroloysis reaction is catalyzed by acid sites, we investigated the acidity of Mo–V–O by NH3-TPD. From the Fig. 6, the amount of acidity increase with the milling time, indicating that acid sites is mainly located on the a–b plane. Thus, we postulate that hydrolysis reaction takes place on the acid sites on the a–b plane.
 | ||
| Fig. 5 The SEM images (a–d) and the average particle size (e) of trigonal Mo–V–O with different milling time. | ||
Milling the Mo–V–O materials could create more a–b plane, and create more acid sites. This is the reason why the yield of 1,2-propylene glycol increased almost linearly with milling time.
Conclusions
In conclusion, the new application of Mo–V–O materials in the ring-opening of epoxide was developed. It was utilized as a catalyst for the hydrolysis and alcoholysis of propylene oxide and gave a good conversion and selectivity. A relationship between the active crystal facet, acidity of Mo–V–O and the yield of product was built up. The result verified the a–b plane is the active crystal facet. The hydrolysis reaction of propylene oxide was the pseudo-first-order reaction through the kinetic analysis. And, the apparent activation energy was evaluated to be 50.3 kJ mol−1. A wide range of epoxides were converted to the corresponding dihydric alcohol derivatives with moderate to excellent yields.Acknowledgements
This work was supported by the National Natural Science Foundation of China (21273231, 21403216), and Dalian Excellent Youth Foundation (2014J11JH126).Notes and references
- T. Katou, D. Vitry and W. Ueda, Chem. Lett., 2003, 32, 1028–1029 CrossRef CAS.
- T. Katou, D. Vitry and W. Ueda, Catal. Today, 2004, 91–2, 237–240 CrossRef.
- W. Ueda, D. Vitry and T. Katou, Catal. Today, 2004, 96, 235–240 CrossRef CAS.
- F. Wang and W. Ueda, Chem. Commun., 2009, 1079–1081 RSC.
- M. Sadakane, N. Watanabe, T. Katou, Y. Nodasaka and W. Ueda, Angew. Chem., Int. Ed., 2007, 46, 1493–1496 CrossRef CAS PubMed.
- S. Ishikawa, M. Tashiro, T. Murayama and W. Ueda, Cryst. Growth Des., 2014, 14, 4553–4561 CAS.
- S. Ishikawa, T. Murayama, S. Ohmura, M. Sadakane and W. Ueda, Chem. Mater., 2013, 25, 2211–2219 CrossRef CAS.
- C. C. Chiu, T. Vogt, L. L. Zhao, A. Genest and N. Rosch, Dalton Trans., 2015, 44, 13778–13795 RSC.
- J. S. Valente, H. Armendariz-Herrera, R. Quintana-Solorzano, P. del Angel, N. Nava, A. Masso and J. M. L. Nieto, ACS Catal., 2014, 4, 1292–1301 CrossRef CAS.
- B. Chattopadhyay, A. Poddar, S. Das, C. Majumder and R. Ranganathan, J. Alloys Compd., 2004, 366, 28–33 CrossRef CAS.
- X. D. Yi, H. Lin, Y. M. He, C. J. Huang, W. Z. Weng and H. L. Wan, Acta Phys.–Chim. Sin., 2004, 20, 309–312 CAS.
- M. Roussel, M. Bouchard, E. Bordes-Richard, K. Karim and S. Al-Sayari, Catal. Today, 2005, 99, 77–87 CrossRef CAS.
- N. R. Shiju and V. V. Guliants, Catal. Commun., 2008, 9, 2253–2256 CrossRef CAS.
- G. Mitran, A. Urda, I. Sandulescu and I. C. Marcu, React. Kinet., Mech. Catal., 2010, 99, 135–142 CAS.
- H. El Moll, B. Nohra, P. Mialane, J. Marrot, N. Dupre, B. Riflade, M. Malacria, S. Thorimbert, B. Hasenknopf, E. Lacote, P. A. Aparicio, X. Lopez, J. M. Poblet and A. Dolbecq, Chem.–Eur. J., 2011, 17, 14129–14138 CrossRef PubMed.
- B. Nohra, H. El Moll, L. M. R. Albelo, P. Mialane, J. Marrot, C. Mellot-Draznieks, M. O'Keeffe, R. N. Biboum, J. Lemaire, B. Keita, L. Nadjo and A. Dolbecq, J. Am. Chem. Soc., 2011, 133, 13363–13374 CrossRef CAS PubMed.
- L. S. Wang, P. C. Yin, J. Zhang, J. Hao, C. L. Lv, F. P. Xiao and Y. G. Wei, Chem.–Eur. J., 2011, 17, 4796–4801 CrossRef CAS PubMed.
- W. Ueda, J. Jpn. Pet. Inst., 2013, 56, 122–132 CrossRef CAS.
- X. T. Wang, F. Wang, S. Jiang and S. H. Zhong, Chin. J. Catal., 2011, 32, 352–356 CAS.
- G. Mitran, I. C. Marcu, M. Florea and I. Sandulescu, Rev. Roum. Chim., 2008, 53, 391–397 CAS.
- V. V. Guliants, R. Bhandari, B. Swaminathan, V. K. Vasudevan, H. H. Brongersma, A. Knoester, A. M. Gaffney and S. Han, J. Phys. Chem. B, 2005, 109, 24046–24055 CrossRef CAS PubMed.
- W. Ueda, D. Vitry and T. Katou, Catal. Today, 2005, 99, 43–49 CrossRef CAS.
- W. Ueda, Y. Endo and N. Watanabe, Top. Catal., 2006, 38, 261–268 CrossRef CAS.
- W. Ueda, D. Vitry, T. Kato, N. Watanabe and Y. Endo, Res. Chem. Intermed., 2006, 32, 217–233 CrossRef CAS.
- X. J. Yang, W. F. Zhang, R. M. Feng, W. J. Ji and C. T. Au, Catal. Lett., 2008, 124, 288–296 CrossRef CAS.
- R. Lopez-Medina, H. Golinska, M. Ziolek, M. O. Guerrero-Perez and M. A. Banares, Catal. Today, 2010, 158, 139–145 CrossRef CAS.
- T. H. K. Oshihara and W. Ueda, Top. Catal., 2001, 15, 2–4 CrossRef.
- S. Ishikawa, X. D. Yi, T. Murayama and W. Ueda, Catal. Today, 2014, 238, 35–40 CrossRef CAS.
- V. V. Guliants, R. Bhandari, J. N. Al-Saeedi, V. K. Vasudevan, R. S. Soman, O. Guerrero-Perez and M. A. Banares, Appl. Catal., A, 2004, 274, 123–132 CrossRef CAS.
- F. Ivars, B. Solsona, E. Rodriguez-Castellon and J. M. L. Nieto, J. Catal., 2009, 262, 35–43 CrossRef CAS.
- V. V. Guliants, R. Bhandari, A. R. Hughett, S. Bhatt, B. D. Schuler, H. H. Brongersma, A. Knoester, A. M. Gaffney and S. Han, J. Phys. Chem. B, 2006, 110, 6129–6140 CrossRef CAS PubMed.
- M. A. Banares and S. J. Khatib, Catal. Today, 2004, 96, 251–257 CrossRef CAS.
- J. Q. Guan, C. Xu, B. Liu, Y. Yang, Y. Y. Ma and Q. B. Kan, Catal. Lett., 2008, 126, 301–307 CrossRef CAS.
- J. Q. Guan, H. Y. Xu, K. Song, B. Liu, F. P. Shang, X. F. Yu and Q. B. Kan, Catal. Lett., 2008, 126, 293–300 CrossRef CAS.
- J. Q. Guan, S. J. Wu, S. B. Jing, C. Xu, Z. Q. Wang, F. P. Shang and Q. B. Kan, Pol. J. Chem., 2009, 83, 1831–1836 CAS.
- X. T. Wang, M. Li, F. Wang, S. J. Zhong, S. Jiang and S. H. Wang, J. Nat. Gas Chem., 2012, 21, 165–169 CrossRef CAS.
- G. Mitran, I. C. Marcu, A. Urda and L. Sandulescu, Rev. Roum. Chim., 2008, 53, 383–390 CAS.
- T. Konya, T. Katou, T. Murayama, S. Ishikawa, M. Sadakane, D. Buttrey and W. Ueda, Catal. Sci. Technol., 2013, 3, 380–387 CAS.
- B. Solsona, A. Dejoz, T. Garcia, P. Concepcion, J. M. L. Nieto, M. I. Vazquez and M. T. Navarro, Catal. Today, 2006, 117, 228–233 CrossRef CAS.
- C. A. Gartner, A. C. van Veen and J. A. Lercher, ChemCatChem, 2013, 5, 3196–3217 CrossRef.
- S. Ishikawa, D. Kobayashi, T. Konya, S. Ohmura, T. Murayama, N. Yasuda, M. Sadakane and W. Ueda, J. Phys. Chem. C, 2015, 119, 7195–7206 CAS.
- S. Ishikawa, X. D. Yi, T. Murayama and W. Ueda, Appl. Catal., A, 2014, 474, 10–17 CrossRef CAS.
- M. Roussel, S. Barama, A. Lofberg, S. Al-Sayari, K. Karim and E. Bordes-Richard, Catal. Today, 2009, 141, 288–293 CrossRef CAS.
- M. Roussel, M. Bouchard, K. Karim, S. Al-Sayari and E. Bordes-Richard, Appl. Catal., A, 2006, 308, 62–74 CrossRef CAS.
- C. T. Qiu, C. Chen, S. Ishikawa, T. Murayama and W. Ueda, Top. Catal., 2014, 57, 1163–1170 CrossRef CAS.
- C. Chen, N. Kosuke, T. Murayama and W. Ueda, ChemCatChem, 2013, 5, 2869–2873 CrossRef CAS.
- J. Tichy, Appl. Catal., A, 1997, 157, 363–385 CrossRef CAS.
- V. V. Guliants, H. H. Brongersma, A. Knoester, A. M. Gaffney and S. Han, Top. Catal., 2006, 38, 41–50 CrossRef CAS.
- F. Wang and W. Ueda, Top. Catal., 2008, 50, 90–97 CrossRef CAS.
- F. Wang and W. Ueda, Appl. Catal., A, 2008, 346, 155–163 CrossRef CAS.
- F. Wang and W. Ueda, Chem. Lett., 2008, 37, 184–185 CrossRef CAS.
- F. Wang and W. Ueda, Catal. Today, 2009, 144, 358–361 CrossRef CAS.
- I. Marin, M. I. Matheu, Y. Diaz and S. Castillón, Adv. Synth. Catal., 2010, 352, 3407–3418 CrossRef CAS.
- A. Dhakshinamoorthy, M. Alvaro and H. Garcia, Chem.–Eur. J., 2010, 16, 8530–8536 CrossRef CAS PubMed.
- S. Das and T. Asefa, ACS Catal., 2011, 1, 502–510 CrossRef CAS.
- D. B. Williams and M. Lawton, Org. Biomol. Chem., 2005, 3, 3269–3272 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Details of the materials, synthesis procedure, characterization techniques, catalytic experimental methods, GC-MS spectra of some products, 1H NMR and 13C NMR spectra of some products, alcoholysis of propylene oxide in different conditions, XRD patterns of fresh and used trigonal catalyst, the scheme with the structure of the trigonal material and the morphology of the used catalyst. See DOI: 10.1039/c6ra10212c |
| This journal is © The Royal Society of Chemistry 2016 |