Winchinines A and B, two unusual monoterpene indole alkaloids with a third nitrogen atom from Winchia calophylla†
Yu-Ting Li‡
a,
Xian-Da Gao‡a,
Wei Zhanga,
Xiao-Jun Huangab,
Dong-Mei Zhanga,
Ren-Wang Jianga,
Lei Wang*ab,
Xiao-Qi Zhang*ab and
Wen-Cai Ye*ab
aInstitute of Traditional Chinese Medicine & Natural Products, College of Pharmacy, Jinan University, Guangzhou 510632, P. R. China. E-mail: cpuwanglei@126.com; tzhxq01@jnu.edu.cn; chywc@aliyun.com; Tel: +86-20-85221559
bJNU-HKUST Joint Laboratory for Neuroscience & Innovative Drug Research, Jinan University, Guangzhou 510632, P. R. China
First published on 15th June 2016
Abstract
Two novel monoterpenoid indole alkaloids (MIAs) with a third nitrogen atom, winchinines A (1) and B (2), along with three known ones (3–5) were isolated from the twigs and leaves of Winchia calophylla. Compound 1 is the first natural MIA possessing a 6/5/6/5/6/5 hexacyclic ring system with an oxazolidinone unit. Compound 2 is an unprecedented cyano-substituted aspidosperma-type alkaloid. Their structures with absolute configurations were elucidated by means of NMR spectroscopy, single crystal X-ray diffraction, and electronic circular dichroism data analyses.
The plant Winchia calophylla is a species natively abundant in the Yunnan and Hainan provinces of China.1 This plant has been traditionally used as a folk medicine to treat chronic asthma and bronchitis.2 Previous phytochemical investigations of this plant have led to the isolation of more than 35 monoterpene indole alkaloids (MIAs)3–6 that showed antitumor, anti-inflammatory, and antiasthma activities.7–9 It has been proposed that MIAs arise from strictosidine, which originates from the coupling of tryptamine and secologanin by a series of biosynthetic processes.10,11 MIAs are characterized with two nitrogen atoms and those with three nitrogen atoms are uncommon.12–15 During our ongoing research on structurally and biologically interesting MIAs from medicinal plants,16–18 two novel MIAs with a third nitrogen atom, winchinines A (1) and B (2), and three known ones (3–5) were isolated from W. calophylla. Winchinine A (1) is the first natural MIA possessing an oxazolidinone unit with a unique 6/5/6/5/6/5 hexacyclic ring system. Winchinine B (2) is an unprecedented cyano-substituted aspidosperma-type alkaloid. In this paper, we describe the isolation, structural elucidation and cytotoxic activities of 1 and 2.
The powdered twigs and leaves of W. calophylla (16 kg) were extracted with 95% EtOH. The extract was suspended in H2O and acidified with 5% HCl to pH 3, which was further partitioned with CHCl3 to remove the neutral components. The aqueous layer was then basified with NH3·H2O to pH 9 and re-extracted with CHCl3 to obtain a total alkaloid fraction (215 g). The alkaloid fraction was separated continuously by column chromatography over silica gel, Sephadex LH-20, ODS (octadecylsiyl) and HPLC (High Performance Liquid Chromatography) to yield compounds 1 (10.8 mg, yield: 0.067%) and 2 (7.4 mg, yield: 0.046%) (Fig. 1).
Winchinine A (1) was obtained as colourless crystal. The molecular formula C20H25N3O2 was deduced on the basis of its HRESIMS (m/z 340.2027 [M + H]+), which indicated the presence of ten degrees of unsaturation.§ The UV spectrum of 1 displayed the absorption maxima at 207 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, π → π*), 245 (C
C, π → π*), 245 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, π → π*), and 302 (C
C, π → π*), and 302 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, n → π*) nm. The IR spectrum showed characteristic absorptions for aromatic ring (1609, 1488 cm−1), amino (3290 cm−1), carbonyl (1754 cm−1) and methyl (2935 cm−1) groups. The 1H NMR (Nuclear Magnetic Resonance) spectrum of 1 showed signals for an ortho-disubstituted phenyl ring [δH 6.99 (1H, overlapped, H-9), 6.99 (1H, overlapped, H-11), 6.71 (1H, dd, J = 7.5, 7.5 Hz, H-10), 6.52 (1H, d, J = 7.5 Hz, H-12)],19 an ethyl group [δH 1.12 (1H, dd, J = 14.5, 7.4 Hz, H-19a), 0.79 (1H, dd, J = 14.5, 7.4 Hz, H-19b), 0.66 (3H, t, J = 7.4 Hz, H-18)], and an oxygenated methine proton [δH 4.52 (1H, dd, J = 11.5, 6.1 Hz, H-16)]. The 13C NMR and DEPT spectra of 1 exhibited twenty carbon signals including a methyl, seven methylenes, six methines and six quaternary carbons. The six aromatic carbon signals [δC 144.7 (C-13), 134.9 (C-8), 128.2 (C-11), 122.9 (C-9), 120.0 (C-10), and 109.2 (C-12)], a spiro quaternary carbon [δC 54.7 (C-7)] and an isolated ethyl group [δC 30.2 (C-19), 7.0 (C-8)] were characteristic for aspidosperma-type alkaloid. Comparison of the NMR data of 1 (Table 1) with those of the known compound aspidospermidine20 revealed that their NMR signals were similar except for the existence of signals for a carbonyl [δC 158.7 (C-22)], a methine [δC 85.0; δH 4.52 (1H, dd, J = 11.5, 6.1 Hz) (CH-16)] and an amino hydrogen [δH 6.56 (1H, s) (23-NH)], as well as the absence of signals for CH2-16 and CH-2 in 1. The 1H–1H COSY spectrum of 1 revealed the presence of five spin systems as shown in Fig. 2. In the HMBC spectrum, the correlations between H-16 (δH 4.52) and C-22 (δC 158.7), between H-23 (δH 6.56) and C-16 (δC 85.0), and between H-23 (δH 6.56) and C-22 (δC 158.7) suggested the presence of a –NH–C (O)–O– fragment which was adjacent to C-2 and C-16 positions to form an unusual oxazolidinone moiety. This conclusion was further confirmed by the ten degrees of unsaturation and by the downfield chemical shifts of C-2 (δC 83.7) and C-16 (δC 85.0) in 1. Based on the above evidence, the planar structure of 1 was established as shown in Fig. 2.
O, n → π*) nm. The IR spectrum showed characteristic absorptions for aromatic ring (1609, 1488 cm−1), amino (3290 cm−1), carbonyl (1754 cm−1) and methyl (2935 cm−1) groups. The 1H NMR (Nuclear Magnetic Resonance) spectrum of 1 showed signals for an ortho-disubstituted phenyl ring [δH 6.99 (1H, overlapped, H-9), 6.99 (1H, overlapped, H-11), 6.71 (1H, dd, J = 7.5, 7.5 Hz, H-10), 6.52 (1H, d, J = 7.5 Hz, H-12)],19 an ethyl group [δH 1.12 (1H, dd, J = 14.5, 7.4 Hz, H-19a), 0.79 (1H, dd, J = 14.5, 7.4 Hz, H-19b), 0.66 (3H, t, J = 7.4 Hz, H-18)], and an oxygenated methine proton [δH 4.52 (1H, dd, J = 11.5, 6.1 Hz, H-16)]. The 13C NMR and DEPT spectra of 1 exhibited twenty carbon signals including a methyl, seven methylenes, six methines and six quaternary carbons. The six aromatic carbon signals [δC 144.7 (C-13), 134.9 (C-8), 128.2 (C-11), 122.9 (C-9), 120.0 (C-10), and 109.2 (C-12)], a spiro quaternary carbon [δC 54.7 (C-7)] and an isolated ethyl group [δC 30.2 (C-19), 7.0 (C-8)] were characteristic for aspidosperma-type alkaloid. Comparison of the NMR data of 1 (Table 1) with those of the known compound aspidospermidine20 revealed that their NMR signals were similar except for the existence of signals for a carbonyl [δC 158.7 (C-22)], a methine [δC 85.0; δH 4.52 (1H, dd, J = 11.5, 6.1 Hz) (CH-16)] and an amino hydrogen [δH 6.56 (1H, s) (23-NH)], as well as the absence of signals for CH2-16 and CH-2 in 1. The 1H–1H COSY spectrum of 1 revealed the presence of five spin systems as shown in Fig. 2. In the HMBC spectrum, the correlations between H-16 (δH 4.52) and C-22 (δC 158.7), between H-23 (δH 6.56) and C-16 (δC 85.0), and between H-23 (δH 6.56) and C-22 (δC 158.7) suggested the presence of a –NH–C (O)–O– fragment which was adjacent to C-2 and C-16 positions to form an unusual oxazolidinone moiety. This conclusion was further confirmed by the ten degrees of unsaturation and by the downfield chemical shifts of C-2 (δC 83.7) and C-16 (δC 85.0) in 1. Based on the above evidence, the planar structure of 1 was established as shown in Fig. 2.
| No. | 1a | 2a | ||
|---|---|---|---|---|
| δHb | δC | δHb | δC | |
| a Data were recorded in CDCl3 at 400 MHz for 1H NMR and 100 MHz for 13C NMR.b Overlapped signals are reported without designating multiplicity. | ||||
| 1-NH | 4.77 s | 3.93 s | ||
| 2 | — | 83.7 | — | 64.8 |
| 3 | α 3.07 br d (9.3) | 53.6 | α 3.03 br d (9.3) | 53.5 |
| β 1.96 td (12.1, 3.9) | β 1.97 | |||
| 5 | α 3.17 td (9.1, 4.3) | 53.2 | α 3.25 td (9.2, 4.3) | 51.8 |
| β 2.33 d (9.1) | β 2.24 d (9.2) | |||
| 6 | α 2.41 m | 36.1 | α 2.66 m | 36.7 |
| β 1.56 m | β 1.50 | |||
| 7 | — | 54.7 | — | 57.9 |
| 8 | — | 134.9 | — | 133.3 |
| 9 | 6.99 | 122.9 | 7.05 | 123.3 |
| 10 | 6.71 dd (7.5, 7.5) | 120.0 | 6.79 dd (7.6, 7.6) | 120.8 |
| 11 | 6.99 | 128.2 | 7.05 | 128.2 |
| 12 | 6.52 d (7.5) | 109.2 | 6.63 d (7.6) | 111.1 |
| 13 | — | 144.7 | — | 146.5 |
| 14 | α 1.69 m | 21.5 | α 1.50 | 21.6 |
| β 1.47 dd (13.3, 3.9) | β 1.07 dd (13.4, 3.7) | |||
| 15 | α 1.62 | 34.1 | α 1.62 d (13.7) | 34.3 |
| β 1.05 m | β 1.12 dd (13.7, 3.7) | |||
| 16 | 4.52 dd (11.5, 6.1) | 85.0 | α 1.97 | 32.7 |
| β 1.78 | ||||
| 17 | α 2.24 d (11.5) | 30.0 | α 2.28 dd (11.0, 6.0) | 22.1 |
| β 1.62 | β 1.78 | |||
| 18 | 0.66 t (7.4) | 7.0 | 0.63 t (7.5) | 7.1 |
| 19 | a 1.12 dd (14.5, 7.4) | 32.0 | a 1.38 dd (14.5, 7.5) | 29.9 |
| b 0.79 dd (14.5, 7.4) | b 0.91 dd (14.5, 7.5) | |||
| 20 | — | 36.0 | — | 36.0 |
| 21 | 2.29 s | 73.3 | 2.35 s | 69.7 |
| 22 | — | 158.7 | — | 122.2 |
| 23-NH | 6.56 s | — | ||
In the ROESY spectrum, the correlations between H-21 (δH 2.29) and H-9 (δH 6.99)/H-3β (δH 1.96)/H-15β (δH 1.05)/H-19b (δH 0.79), between H-18 (δH 0.66) and H-16 (δH 4.52), as well as between H-23 (δH 6.56) and H-6α (δH 2.41) indicated the relative configuration of 1 as shown in Fig. 3. Fortunately, crystals suitable for single crystal X-ray diffraction experiment were obtained from methanol solution. Thus, the structure and relative stereochemistry of 1 were unequivocally determined (Fig. 4).
To determine the absolute configuration of winchinine A (1), ECD curves for the two possible enantiomers (2R,7S,16S,20R,21R-1 and 2S,7R,16R,20S,21S-1) were calculated using the TD-DFT theory method (see the ESI†). As shown in Fig. 5, the experimental ECD spectrum of 1 showed a negative Cotton effect at 300 nm and positive Cotton effects at 245 and 207 nm, which were similar to the calculated one for the isomer with 2R, 7S, 16S, 20R, and 21R configurations. Therefore, the absolute stereochemistry of 1 was assigned as 2R, 7S, 16S, 20R, and 21R.
Winchinine B (2), a white amorphous powder, possessed the molecular formula C20H25N3 according to its HRESIMS at m/z 308.2128 [M + H]+ (calcd for C20H26N3+ 308.2121). The UV spectrum showed absorption maxima at 206 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, π → π*) and 296 (C
C, π → π*) and 296 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, n → π*) nm. The IR spectrum of 2 suggested the presence of NH (3323 cm−1), CN (2362 cm−1), methyl (2937 cm−1) and benzene ring (1680, 1540, 1457 cm−1). The 1H and 13C NMR data of 2 were similar to those of 1 (Table 1), except for the signals for CH-16 (δH 4.52, δC 85.0) and C-22 (δC 158.7) in 1 were replaced by a methylene (δH 1.97, 1.78; δC 32.7) and a cyano group (δC 122.2) in 2, respectively. Based on the ten degrees of unsaturation and the 13C NMR data of 2, a cyano group was proposed to connect at C-2 position, which was further confirmed by the HMBC correlation between H-16 (δH 1.78) and C-22 (δC 122.2) (Fig. 2) and by the fragment ion peak at m/z 281.2033 in the HRESIMS that originated by the loss of cyano unit (see the ESI†).
C, n → π*) nm. The IR spectrum of 2 suggested the presence of NH (3323 cm−1), CN (2362 cm−1), methyl (2937 cm−1) and benzene ring (1680, 1540, 1457 cm−1). The 1H and 13C NMR data of 2 were similar to those of 1 (Table 1), except for the signals for CH-16 (δH 4.52, δC 85.0) and C-22 (δC 158.7) in 1 were replaced by a methylene (δH 1.97, 1.78; δC 32.7) and a cyano group (δC 122.2) in 2, respectively. Based on the ten degrees of unsaturation and the 13C NMR data of 2, a cyano group was proposed to connect at C-2 position, which was further confirmed by the HMBC correlation between H-16 (δH 1.78) and C-22 (δC 122.2) (Fig. 2) and by the fragment ion peak at m/z 281.2033 in the HRESIMS that originated by the loss of cyano unit (see the ESI†).
The relative configurations of C-7, C-20 and C-21 of 2 were deduced to be the same as 1 according to the ROESY experiment (Fig. 3). However, the stereochemistry of the cyano group cannot be determined by ROESY spectrum. To determine the absolute configuration of 2, a comparison between the experimental and calculated CD spectra using the time-dependent DFT method was performed (see the ESI†).21 ECD curves for the two possible isomers (2R,7S,20R,21R-2 and 2S,7S,20R,21R-2) were calculated. The measured CD spectrum of 2 (Fig. 6) exhibited a negative Cotton effect at λmax 302 nm and positive Cotton effects at λmax 205 and 248 nm, which were similar with the calculated curves of 2R,7S,20R,21R-2. Thus, the structure of 2 was determined.
The known compounds Nb-demethylechitamine (3),22 Nb-demethylechitamine-Nb-oxide (4),23 and eburenine (5)24 were identified by comparison of their physical and spectroscopic data with those reported in the literature.
In comparison with the known MIAs, the structures of 1 and 2 incorporated a third nitrogen atom to form unusual oxazolidinone unit and cyano group. Compounds 1 and 2 represent the first natural MIA with an oxazolidinone unit and unusual cyano-substituted aspidosperma-type alkaloid, respectively.
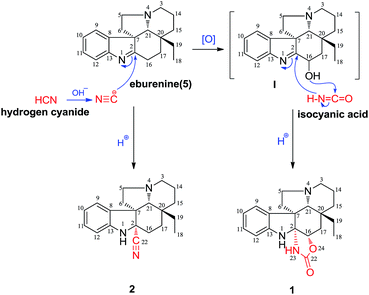
On the basis of the literature and our research results, a plausible biosynthetic pathway to compounds 1–2 is proposed (Scheme 1). The alkaloid eburenine (5) is a major component of W. calophylla.4 The nucleophilic attack from the cyanide ion to the imine group of 5 could afford 2. In addition, oxidation of eburenine (5) could afford an intermediate I, which could incorporate with an isocyanic acid moiety by nucleophilic addition to give compound 1.
The inhibitory effects of compounds 1–5 on the viability of HepG2, MDA-MB-231, PC3 and A549 cells were determined using the MTT method as reported previously.16 Doxorubicin (Dox, Sigma, USA) was used as the positive control. Compound 2 showed moderate cytotoxicity against PC3, MDA-MB-231, and A549 cells with IC50 values of 31.51 ± 8.06, 43.43 ± 4.26 and 40.77 ± 2.93 μM, respectively. The other compounds were inactive (IC50 values > 50 μM) (Table 2).
| Compounds | aIC50 (x ± SD) μM | |||
|---|---|---|---|---|
| HepG2 | MDA-MB-231 | A549 | PC-3 | |
| a IC50: concentration of the tested compound inhibits 50% of cell growth.b Doxorubicin is the positive control in the test. | ||||
| 1 | >50 | >50 | >50 | >50 |
| 2 | >50 | 43.43 ± 4.26 | 40.77 ± 2.93 | 31.51 ± 8.06 |
| 3 | >50 | >50 | >50 | >50 |
| 4 | >50 | >50 | >50 | >50 |
| 5 | >50 | >50 | >50 | >50 |
| Doxb | 0.25 ± 0.03 | 0.32 ± 0.04 | 0.18 ± 0.06 | 0.40 ± 0.08 |
Acknowledgements
This work was financially supported by the Program for National Natural Science Foundation of China (No. U1401225, 81273391, 81573307), the Ministry of Science and Technology of China (No. 2013DFM30080, 2013BAI11B05, 2012ZX09103201-056), the Guangdong Natural Science Foundation for Distinguished Young Scholar (No. 2015A030306022).Notes and references
- Guangdong Farming and Forestry College, Chinese Flora (Zhongguo Zhiwu Zhi), 1980, 63, 95–97.
- State Administration of Traditional Chinese Medicine, Chinese Herb (Zhonghua Bencao), 1999, 6, 320.
- L. M. Li, T. Yang, Y. Liu, J. Liu, M. H. Li, Y. T. Wang, S. X. Yang, Q. Zou and G. Y. Li, Org. Lett., 2012, 14, 3450–3453 CrossRef CAS PubMed.
- L. S. Gan, S. P. Yang, Y. Wu, J. Ding and J. M. Yue, J. Nat. Prod., 2006, 69, 18–22 CrossRef CAS PubMed.
- W. M. Zhu, H. P. He, L. M. Fan, Y. M. Shen, J. Zhou and X. J. Hao, J. Integr. Plant Biol., 2005, 47, 892–896 CrossRef CAS.
- C. W. Ming, Z. P. Ling and G. Rucker, Planta Med., 1988, 54, 480–481 CrossRef CAS PubMed.
- Q. H. Sun, Sci. Tech., 2014, 12, 89–90 Search PubMed.
- F. Guo, J. P. Yu, Y. Zhang, C. M. Yuan and H. P. He, Nat. Prod. Res. Dev., 2014, 26, 361–363 Search PubMed.
- C. M. Li, X. M. Zhang, Y. L. Zhou, L. Y. Huang and G. D. Tao, Acta Pharmacol. Sin., 1993, 28, 512–515 CAS.
- S. E. O'Connor and J. J. Maresh, Nat. Prod. Rep., 2006, 23, 532–547 RSC.
- A. Patthy-Lukats, L. Karolyhazy, L. F. Szabo and B. Podanyi, J. Nat. Prod., 1997, 60, 69–75 CrossRef CAS.
- G. Y. Zhu, X. J. Yao, L. Liu, L. P. Bai and Z. H. Jiang, Org. Lett., 2014, 16, 1080–1083 CrossRef CAS PubMed.
- K. H. Lim and T. S. Kam, Org. Lett., 2006, 8, 1733–1735 CrossRef CAS PubMed.
- Z. W. Liu, T. T. Yang, W. J. Wang, G. Q. Li, B. Q. Tang, W. Zhang, C. L. Fan, D. M. Zhang, X. Q. Zhang and W. C. Ye, Tetrahedron Lett., 2013, 54, 6498–6500 CrossRef CAS.
- L. L. Liu, Y. T. Di, Q. Zhang, X. Fang, F. Zhu, D. L. Chen, X. J. Hao and H. P. He, Tetrahedron Lett., 2010, 51, 5670–5673 CrossRef CAS.
- W. Zhang, X. J. Huang, S. Y. Zhang, D. M. Zhang, R. W. Jiang, J. Y. Hu, X. Q. Zhang, L. Wang and W. C. Ye, J. Nat. Prod., 2015, 78, 2036–2044 CrossRef CAS PubMed.
- S. Ouyang, L. Wang, Q. W. Zhang, G. C. Wang, Y. Wang, X. J. Huang, X. Q. Zhang, R. W. Jiang, X. S. Yao, C. T. Che and W. C. Ye, Tetrahedron, 2011, 67, 4807–4813 CrossRef CAS.
- B. Q. Tang, W. J. Wang, X. J. Huang, G. Q. Li, L. Wang, R. W. Jiang, T. T. Yang, L. Shi, X. Q. Zhang and W. C. Ye, J. Nat. Prod., 2014, 77, 1839–1846 CrossRef CAS PubMed.
- H. A. Guimarães, R. Braz-Filho and I. J. C. Vieira, Molecules, 2012, 17, 3025–3043 CrossRef PubMed.
- T. Ishikawa, K. Kudo, K. Kuroyabu, S. Uchida, T. Kudoh and S. Saito, J. Org. Chem., 2008, 73, 7498–7508 CrossRef CAS PubMed.
- J. G. Dong, W. Bornmann, K. Nakanishi and N. Berova, Phytochemistry, 1995, 40, 1821–1824 CrossRef CAS PubMed.
- N. Keawpradub, H. Takayama, N. Aimi and S. I. Sakai, Phytochemistry, 1994, 37, 1745–1749 CrossRef CAS.
- W. M. Chen, P. L. Zhang and G. Ruecker, Planta Med., 1988, 54, 480–481 CrossRef CAS PubMed.
- Y. Q. Wu, M. Kitajima, N. Kogure, Y. S. Wang, R. P. Zhang and H. Takayama, Chem. Pharm. Bull., 2010, 58, 961–963 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: The general experimental procedure, extraction and isolation, spectroscopic data of 1 and 2, single-crystal X-ray data of 1, NMR spectra and quantum chemical CD calculation of compounds 1 and 2. CCDC 1451513. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6ra10137b |
| ‡ These authors have contributed equally to this work. |
| § Crystal data for winchinine A (1): C20H25N3O2, triclinic, P1, a = 7.8960(10) Å, b = 8.4207(7) Å, c = 14.3158(8) Å, β = 81.536(10)°, γ = 89.868(8)°, V = 929.27(17) Å3, T = 173(2) K, Z = 2, Dc = 1.328 mg mm−3, F(000) = 400. The final refinement gave R = 0.0556, Rw = 0.1540, S = 1.116 and Flack parameter = −0.4(3). Crystal data of 1 were deposited in the Cambridge Crystallographic Data Centre (CCDC 1451513). |
| This journal is © The Royal Society of Chemistry 2016 |